Abstract
Background: Riboflavin seems to have a promising effect on migraine in adults. The present study examines whether riboflavin has a prophylactic effect on migraine in children.
Objective: To investigate whether riboflavin in a dosage of 50 mg/day has a prophylactic effect on migraine attacks in young children.
Subjects and methods: This randomised, placebo-controlled, double-blind, cross-over trial included 42 children (aged 6–13 years) with migraine of whom 14 children were also suffering from tension-type headache. Following a 4-week baseline period, all children received placebo for 16 weeks then riboflavin for 16 weeks (or vice versa) with a washout period of 4 weeks in between. The primary outcome measure was reduction in mean frequency of migraine attacks and tension-type headache in the last 4 weeks at the end of the riboflavin and placebo phase, compared with the preceding baseline or wash-out period. Secondary outcome measures were mean severity and mean duration of migraine and tension-type headaches in the last 4 weeks at the end of the riboflavin and placebo phase, compared with the preceding baseline or wash-out period.
Results: No significant difference in the reduction of mean frequency of migraine attacks in the last month of treatment was found between placebo and riboflavin (P = 0.44). However, a significant difference in reduction of mean frequency of headaches with a tension-type phenotype was found in favour of the riboflavin treatment (P = 0.04).
Conclusions: In this group of children with migraine, there is no evidence that 50 mg riboflavin has a prophylactic effect on migraine attacks. We found some evidence that 50 mg riboflavin may have a prophylactic effect on interval headaches that may correspond to mild migraine attacks or tension-type headache attacks in children with migraine.
Introduction
Migraine is a common disorder in adolescents and children. The prevalence of headache and migraine is age and gender dependent. Sillanpää (1) reported a prevalence of migraine for boys and girls at 7 years of age of 2.9% and 2.5%, respectively; at age 14 years, the prevalence of migraine was 6.4% and 14.8% for boys and girls, respectively.
The efficacy of both pharmacological and non-pharmacological interventions in children and adolescents with migraine has been studied extensively (2–5). Some interventions in the field of symptomatic treatment of migraine in children have been proven effective, such as sumatriptan nasal spray, ibuprofen and acetaminophen (2,5). In the prophylactic treatment of migraine in children and adolescents, until now only flunarizine is an evidence-based effective drug, and probably also topiramate (4–6). Adverse effects have been reported in both treatments. In addition, flunarizine is not available in the US and both flunarizine and topiramate cannot be prescribed to children or adolescents in most European countries (4–6). Other treatment modalities in this field (which are not evidence based) are antihypertensive medications, antidepressants, serotonergic drugs, 5-HT2-antagonists (such as pizotifen or methysergide) and anti-epileptic drugs. However, in all of these agents, adverse effects have been described (4,5). Thus, there is a need for high-quality research to evaluate pharmacological prophylactic treatment that has minimal or no adverse effects in children and in adolescents with migraine.
From this perspective we were interested in examining riboflavin (vitamin B2). Riboflavin is a co-factor for mitochondrial oxidative phosphorylation as it is the precursor for the flavin compounds necessary for the transfer of electrons in the mitochondrial respiratory chain. In vivo studies have detected an impairment of mitochondrial oxidative phosphorylation in adult migraineurs between attacks (7–9). Supraphysiological doses of riboflavin might be helpful in reducing this impairment. In patients with mitochondrial encephalopathy with lactic acidosis and stroke-like episodes, a subgroup had a reduced frequency of migraine attacks during treatment with riboflavin (10). Therefore, riboflavin has been used as a prophylactic agent in studies on adults with migraine, including two open-label studies (11,14) and two randomised controlled trials (12,13). In these latter trials, riboflavin was proven effective with minimal adverse effects. These adverse effects were diarrhoea (1 of 43 adults with migraine treated with riboflavin) and polyuria (1 of 43 adults with migraine treated with riboflavin) (12,13). A common and harmless side effect of riboflavin is a bright yellow/orange discolouration of the urine.
At the time of the present study, no other trials were registered to investigate the effect of riboflavin in children with migraine. Therefore, we decided to perform a placebo-controlled, randomised trial in children with migraine using riboflavin as the active agent.
Subjects and methods
A 40-week, randomised, double-blind, cross-over design was used to examine the effect of riboflavin compared with placebo in young children with migraine. The study was conducted in two hospitals in The Netherlands. The children were prospectively recruited between October 2005 and March 2008, and the trial was completed in December 2008. The children were referred by general practitioners (in the region of Schiedam/Vlaardingen) and by paediatricians, neurologists and paediatric neurologists in the greater Rijnmond region in The Netherlands.
All potentially referring physicians received an information letter about this trial. The children were assessed at the out-patient paediatric department of Vlietland Hospital (in Schiedam/Vlaardingen) by the principal investigator (JB), and at the department of paediatric neurology of the Erasmus Medical Centre (EMC, Rotterdam) by the principal investigator (JB) and the co-investigators (ND and WFA).
Parental informed consent, and informed consent from children aged 12 years, was obtained. Headache diaries were composed in advance of the trial. The study was approved by the Medical Ethics committees of the EMC and Vlietland Hospital.
Subjects
The inclusion criteria were: (i) age 6–13 years – we deliberately chose this age group because only a limited number of evidence-based interventions on this group are available (2–5); (ii) migraine with or without aura according to the ICHD II criteria (15); and (iii) a frequency of two or more headache attacks per month.
The exclusion criteria were: (i) epilepsy or other serious neurological disease; (ii) diseases of the liver or kidneys, and gastrointestinal, metabolic or cardiovascular disease; (iii) use of other prophylactic medication for migraine within 1 month of the trial; (iv) use of other prophylactic treatment for migraine during the trial; and (v) inability to comply with the requirements of the trial, and/or inability to speak and read Dutch.
Procedure and intervention
The pharmacy department of the EMC manufactured placebo and riboflavin capsules, the latter containing 50 mg riboflavin. To ensure the double-blind design, carotene 100 mg was used as placebo. Both carotene and riboflavin give an orange discolouration of the urine. A PubMed search confirmed the absence of evidence that carotene has any effect on headache frequency or intensity in children.
After a baseline period of 4 weeks, children were randomised in phase 1 to receive either one capsule containing placebo or 50 mg riboflavin daily for 16 weeks. Children were instructed to swallow the capsule at breakfast. Following a wash-out period of 4 weeks, in phase 2 the children received either one capsule containing placebo daily for 16 weeks if they had received riboflavin in phase 1, or one capsule of 50 mg riboflavin daily for 16 weeks if they had received placebo in phase 1. Treatment allocation was concealed from the participants and investigators for the duration of the study. The hospital pharmacists guarded the randomisation key. To evaluate blinding, at the last visit parents were asked which treatment they believed that their child had received in which phase, and the reasons for their assumptions.
All children were primarily diagnosed with migraine by paediatric neurologists (JB or WFA). A thorough history was taken, and a complete neurological and physical examination was performed including measurement of blood pressure, body weight, height and skull circumference.
If the child was eligible for inclusion, the child and their parents were informed about all aspects of the trial and asked for their consent. They were informed that they could end their participation at any time during the trial without having to give any reason. No rewards or other response-enhancing policies were applied. A 2-week period was allowed for the child/parents to consider possible participation in the study.
The parents of the children were asked to keep a detailed headache diary during all stages of the study. This headache diary documented every headache attack, including: the date, the severity of the headache on a 4-point Likert scale, its location(s), duration and nature (pulsating or non-pulsating), possible aggravation by routine physical activity, associated symptoms such as nausea or vomiting, and the presence or absence of photophobia or phonophobia or fever. The diaries were based on the ICHD-II criteria in order to enable classification of each headache attack as a migraine attack, tension-type headache, headache probably due to an (ear-nose-throat) infection, or a headache attack not fulfilling the ICHD-II criteria of any of these. Parents also completed questionnaires on behavioural problems and life dimensions of the children for a different research focus (not reported here).
All parents and children received information regarding childhood migraine, and all children were treated symptomatically according to state-of-the-art evidence-based guidelines (5,16).
This meant that, in case of a migraine attack, they received acetaminophen and ibuprofen orally or rectally, and sumatriptan nasal spray if they were 12 years old.
Children were given simple instructions on how to improve sleep duration and quality, i.e. to stop the intake of caffeine, and decrease the amount of time spent watching television or computer activity to a maximum of 2–3 h per day.
Appointments with parents were made at the end of each stage. At each appointment, the headache diary of the preceding stage was given by the parents to the paediatric neurologists (JB or WFA), and, in return, a new headache diary for the following stage was given to the parents. During the trial, halfway in both phase 1 and phase 2, paediatric neurologist JB or co-investigator ND, would hold a telephone conversation with the parents.
At each visit and each telephone appointment, the parents were consistently asked about any possible adverse effects of the medication. This was documented in the personal file of the child. To improve compliance during both phases of the study, parents and child were asked to bring the remaining tablets with them at each visit.
Three months after the conclusion of the trial, all parents and children received a letter informing them when their child had received riboflavin or placebo, and the preliminary conclusions of the trial.
Outcome measures
The outcome measures were reduction in the mean frequency, mean severity and mean duration of migraine and tension-type headache in the last 4 weeks at the end of phase 1 and of phase 2 compared with the baseline and washout period, respectively. Any adverse effects were documented and compared.
Statistical analysis
Sample size calculations were based on a clinically relevant difference of 0.60 SD between the riboflavin and placebo condition in favour of riboflavin with regard to migraine frequency in accordance with the data of the randomised controlled trial by Schoenen et al (12). In addition, based on a cross-over design, an α of 0.05 (two-sided) and a statistical power of 0.80, a minimum number of 20 patients was required to participate in the trial. To allow for drop-outs, 30 patients were initially enrolled, which was later extended to 42 to allow also for exploratory analysis.
Statistical analysis was performed according to the intention-to-treat principle. As measures of central tendency, the mean values (for continuous data) and percentages (for categorical data) were estimated. In case of continuous data, the SD was used as measure of dispersion. To evaluate the effect of riboflavin on the outcome variables, the t-test for independent observations was applied (17). All statistical testing took place at the 0.05 level of significance.
We planned an exploratory analysis with regard to the effect of riboflavin versus placebo on tension-type headache. We also planned an exploratory analysis in children with a relatively high baseline headache attack frequency versus children with a relatively low baseline headache attack frequency.
Results
Demographics
A total of 57 children were assessed for eligibility; 15 patients were not enrolled because they did not meet the inclusion criteria, met one or more exclusion criteria, or because the parents or child refused to participate in the trial (Figure. 1).
The Consolidated Standard of Reporting Trials (CONSORT) flow diagram showing the flow of participants.
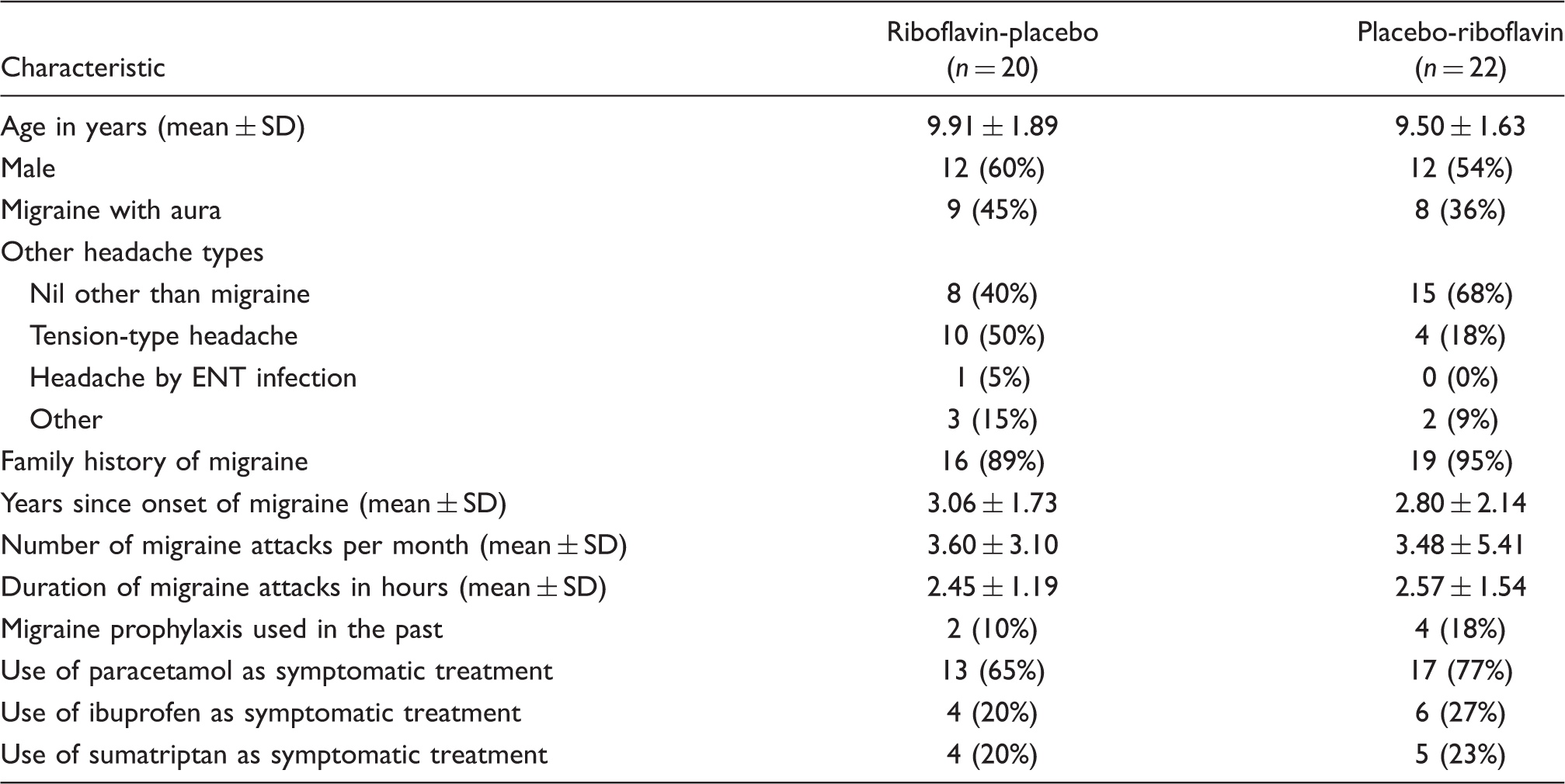
Baseline demographic and clinical characteristics of the study group
Outcome data
Data on headache frequency, intensity and headache duration in the last 4 weeks by riboflavin or placebo treatment

n, number of patients; TTH, tension-type headache; RP, order riboflavin-placebo; PR, order placebo-riboflavin; bold numbers = after riboflavin treatment; italic numbers = after placebo treatment.
Analysis of headache frequency, intensity and duration in the last 4 weeks by riboflavin or placebo treatment

n, number of patients; TTH, tension-type headache; dna, did not apply; RP, order riboflavin-placebo; PR, order placebo-riboflavin; *significant (P < 0.05); bold numbers, difference between start and end of riboflavin treatment; italic numbers, difference between start and end of placebo treatment; t-statistic and P-value for difference between difference a−b for order of RP and PR, respectively.
No significant difference was found in change or reduction of mean intensity of migraine attacks and tension-type headache attacks in the fourth and last month of treatment between placebo and riboflavin (P = 0.18 and did not apply, respectively). Also, no significant difference was found in change or reduction of mean duration of migraine attacks and tension-type headache attacks in the fourth and last month of treatment between placebo and riboflavin (P = 0.15 and did not apply, respectively).
Analysis for a period or carry-over effect was performed on all outcome measures but proved to be inconclusive.
An exploratory analysis of attack frequency in children with a relatively high baseline frequency of five or more attacks per month, and in children with four or less attacks per month at baseline, revealed no significant differences in both of these categories between riboflavin and placebo with regard to migraine prophylaxis (P = 0.36 and P = 0.29, respectively).
During the trial, no adverse effects were reported by parents or children; this was recorded in the patient files in both the riboflavin-placebo and the placebo-riboflavin groups.
Discussion
With regard to prophylaxis of migraine attacks, there was no significant difference in the mean migraine frequency per 4 weeks (P = 0.44) or mean intensity of migraine attacks (P = 0.18) or mean duration of migraine attacks (P = 0.15) in the last 4 weeks of treatment between riboflavin and placebo. It could be argued that this result might be due to the fact that we had no upper limit for attack frequency as an exclusion criterion. In children, a high frequency of migraine or headache attacks, especially with an attack frequency of 15 headache attacks per month or more (the main diagnostic criterion for chronic migraine/headache), is often indicative of resistance to pharmacological prophylactic treatment (18). Therefore, we also performed a separate exploratory analysis of attack frequency in children with a relatively high baseline frequency (five or more attacks per month), and in children with a relatively low baseline frequency (four or less attacks per month). In both of these categories, no significant differences were found between riboflavin and placebo with regard to migraine prophylaxis.
Our conclusions are in line with those of MacLennan et al. (19), who investigated 48 children with migraine aged 5–15 years (mean, 11.1 ± 2.1 years) with a higher dosage of 200 mg riboflavin per day in a placebo-controlled, double-blind, randomised trial with a parallel group design. This latter trial also reported no differences between riboflavin and placebo for primary or secondary outcome variables with regard to migraine prophylaxis. The trial of MacLennan et al. had an upper limit exclusion criterion of eight migraine attacks per month. They recommended performing future studies with a cross-over design, or with larger sample sizes. The fact that both our crossover study and that of MacLennan et al. showed no proof of effectiveness of riboflavin as a prophylactic agent in children with migraine is in contrast to the riboflavin studies conducted among adults with migraine (11–14).
The strength of our study is the double-blind, randomised, placebo-controlled, cross-over design, which gives more statistical power than a parallel design. Of the four drop-outs during the study, all were in the placebo-riboflavin group and dropped out during the placebo phase. It can be argued that this might be due to the lower effect of placebo compared with riboflavin. However, analysis of the other children in the riboflavin-placebo group showed a slightly better (non-significant) response to the placebo phase compared with the riboflavin phase.
A weakness of our study, on the other hand, could be the 50 mg per day dosage of riboflavin. Riboflavin has been used as a therapeutic agent in various mitochondrial diseases in children and adolescents, such as the NADH–CoQ reductase deficient myopathy and the mitochondrial encephalopathy and stroke studies (10,20). In these latter studies, riboflavin was given in doses approximately 100-fold higher than the normal dietary intake, e.g. between 100 and 300 mg per day. These dosages were well tolerated without adverse effects (except for nausea) and sometimes yielded striking improvement (20).
Before the start of our study, three trials in adults with migraine (all using 400 mg riboflavin per day) had suggested a dosage of 100–200 mg per day for children (12–14). However, shortly before the start of our trial, Maizels et al. (21) demonstrated no significant difference between a combination of high-dose riboflavin, magnesium and feverfew in comparison with a low dose (25 mg) of riboflavin acting as placebo in adults with migraine, suggesting an equivalent effect of low-dose riboflavin versus high-dose riboflavin. Therefore, we decided to treat our 6–13-year-old patients with a dosage of 50 mg riboflavin, which is relatively low compared with the dosage used in mitochondrial diseases and in riboflavin studies in adults with migraine.
On the other hand, if one assumes that children have a much higher metabolic rate than adults, the maximum dosage of riboflavin in children should be even higher than in adults to obtain a similar effect. From this perspective, our attention was drawn to the recent publication of Condò et al. (24), which describes a retrospective open-label study in 41 children or adolescents with migraine who were treated with 200 mg or 400 mg riboflavin on a daily basis for 3–6 months. In that study, 68.4% of the included children had a reduction of 50% or more in the frequency of all headache attacks. Statistical analysis showed no significant differences between frequency/intensity responders for a 200 mg or 400 mg/day dose. However, in that study, no placebo group was included and treatment was not concealed, which are essential factors for assessing study quality.
Another point for discussion could be the duration of 16 weeks of each treatment period, and the analysis that was limited to the last 4 weeks of both phases of the study. This set-up was based on the original riboflavin trial of Schoenen et al. (12) among adults with migraine. In that study, the maximal effect of riboflavin was seen in the fourth month of treatment (12).
In addition, the relatively small number of participants might be a factor explaining the negative results of our trial. However, our power estimation was according to standard criteria. Future studies using a randomised, controlled, parallel group design according to IHS guidelines, should employ a larger sample size.
One can also argue that, based on the low percentage of children receiving migraine prophylaxis in both the riboflavin-placebo and the placebo-riboflavin group (10% and 18%, respectively), the effectiveness of riboflavin was less significant due to the limited severity of the headache symptoms in the included children. However, in our opinion, these low percentages of migraine prophylaxis at inclusion do not mean that the severity of symptoms was less significant. First, the included children were relatively young (6–13 years) and migraine has a relatively low prevalence in pre-pubertal children compared with adolescents and adults. Therefore, most children in our study had a relatively short history of migraine. Second, most children were included after their first consultation with a specialist to whom they were referred by the general practitioner (GP) because of their headache; in the Dutch healthcare system, the GP is the key person for referral to a specialist. Referral to, or consultation with, a specialist is advised in the national Dutch guidelines for GPs if there is a need for prescribing symptomatic or prophylactic treatment in children with migraine. Finally, an earlier study from our group showed that the quality of life is very poor in children with migraine and tension-type headache at their first visit to one of the two hospitals which were also used for inclusion and follow-up procedures in the present study (25); this suggests that GPs in The Netherlands tend to refer only children with a high burden of headache or migraine. It is for these reasons that most of the children in the present study did not receive prophylactic treatment, and not because of limited severity of their headache symptoms.
A final drawback is that, in all trials among children with migraine (both for acute treatment and migraine prophylaxis), a high placebo response made it difficult to prove the efficacy of a verum drug (22). This will also apply to our study, especially since all children were given simple instructions about improving sleep quality and duration, stopping intake of caffeine, and decreasing the amount of time spent watching television or using a computer (besides receiving state-of-the-art, evidence-based, symptomatic treatment), which undoubtedly increased the placebo response in all the included children.
In addition, an interesting hypothesis that might explain our negative findings is related to the mitochondrial DNA make-up of our patients.
A recent pharmacogenetic study in adults with migraine showed that patients with non-H mitochondrial DNA haplotype respond better to treatment with riboflavin than patients with H-mitochondrial DNA haplotype (23). To investigate if this is a significant factor, mitochondrial DNA analysis should be performed and the presence of non-H or H-haplotype mitochondrial DNA should be taken into account in the analysis of our data.
In summary, with regard to effectiveness, based on our present study and those of Condò et al. (24) and MacLennan et al. (19), we conclude that, at this moment, there is inconclusive evidence for the effectiveness of riboflavin on migraine attacks in children or adolescents with migraine.
With regard to safety, in our present study none of the included children experienced adverse effects. In the study of Condò et al. (24), two patients experienced vomiting and increased appetite, respectively, during riboflavin treatment. This was most likely unrelated to the use of riboflavin. In the study of MacLennan et al. (19), none of the children on riboflavin experienced adverse effects. Therefore, one can now conclude that riboflavin in dosages up to 400 mg daily for a period of several months can be safely used in children with migraine.
Finally, we found some evidence that riboflavin may have a prophylactic effect on tension-type headache in children with migraine.
Unfortunately, due to the low numbers of patients involved, calculation of significant differences between placebo and riboflavin as a prophylactic agent in tension-type headache with regard to the secondary outcome variables (mean intensity of headache attacks and mean duration of headache attacks) was not possible.
A prophylactic effect of riboflavin on tension-type headache has not yet been described either in adults or in children (11–14,19,24). In these latter studies, headache frequency or migraine frequency was used as outcome variable and there was no separate analysis regarding tension-type headache or (other) non-migraine headache attacks.
Based on our study, one might argue that a medium dose of riboflavin has a prophylactic effect on tension-type headache in children. From that perspective, one has to bear in mind that the tension-type headaches in our study were an accompanying phenomenon in children with migraine as their main primary headache. Also, at baseline, the total number of children with both migraine and tension-type headache in both groups (riboflavin-placebo and placebo-riboflavin) was low, especially in the placebo-riboflavin group (50% and 18%, respectively).
In addition, one can speculate about the nature of accompanying tension-type headaches in children with migraine, especially since the Spectrum Study of Lipton et al. (26) provides evidence that all headache attacks in adults with migraine more or less represent a spectrum of migraine headaches. If one assumes that this is also the case in children, based on the present study one might conclude that riboflavin at a low dose reduces mild, but not severe, headaches in migrainous children. Therefore, caution is required when drawing conclusions about the prophylactic effect of riboflavin on tension-type headache in children. However, this is the first study giving indications that riboflavin can diminish tension-type headache in children with migraine compared with a placebo intervention. To investigate if riboflavin indeed has a primary effect on tension-type headache in children or adults, more placebo-controlled, randomised, double-blind trials need to be performed in children or adults with primary tension-type headache with and without migraine.
Conclusions
The present study shows that riboflavin in a dosage of 50 mg/day has no effect on migraine prophylaxis in young children with migraine compared with placebo. Currently, there is inconclusive evidence that riboflavin has a prophylactic effect on migraine attacks in children. However, in this study, we give evidence that riboflavin may have a prophylactic effect on interval headaches that may correspond to mild migraine attacks or tension-type headaches in children with migraine. More studies are needed to investigate whether the same prophylactic effect of riboflavin on migraine as seen in adults can be achieved in children. More studies are also needed to investigate the effect of riboflavin on tension-type headache, both in children and adults with and without migraine.
Footnotes
Acknowledgements
The authors thank all the children and their parents who participated in this study.
Special thanks go to the paediatricians in the Vlietland Hospital for their support and referral of patients: Coby Langendoen MD, Lia van Wijk MD, Bert den Exter MD, Arjan Pijning MD, Esther Hentzen MD, and Evelyne van Boeckel MD. Also, special thanks to Sjaan Scholten, Physician’s Assistant at the Paediatric Out-patient Department of the Vlietland Hospital.
The authors thank the management of the Vlietland Hospital for their support, especially Gerrit-Jan van Zoelen.
The authors also thank the referring paediatricians, neurologist and paediatric neurologists, especially P de Laat, J Samijn, A van Rossum, R van der Lee, D Kamphuis, B Koop and C de Korte. In addition, special thanks to Loes Visser and Hong San Lau, chief pharmacist of the pharmacy department of the Erasmus Medical Centre and the Vlietland Hospital, respectively.
This study was sponsored by the Nuts-Ohra Foundation, the Nolet Foundation and Van Der Linden Structon.
