Abstract
Background:
While home-based exergames help overcome accessibility barriers to rehabilitation, it is unclear what constitutes effective intervention design in using exergames to support self-efficacy and engagement.
Objective:
Examine usage of an in-home exergame, compared to control, unsupervised after supervised training by older persons with lower-limb amputation.
Study design:
Secondary analysis of a multi-site parallel evaluator-masked randomized control trial.
Methods:
WiiNWalk uses the WiiFit and teleconferencing for in-home group-based exergame therapy with clinical supervision. Participants engaged in a 4-week supervised training phase followed by a 4-week unsupervised phase in experimental (WiiNWalk) and attention control groups. Usage between phases and between groups was compared using unsupervised/supervised ratio of session count (over 4 weeks) and session time (mean min/session over 4 weeks) for each phase.
Results:
Participants: n=36 experimental, n=28 control, unilateral lower-limb amputation, age > 50 years, prosthesis usage ≥ 2 hours/day. Session count ratio unsupervised/supervised, median and interquartile range (IQR), was less than parity (p<0.01) for experimental (0.25, IQR 0.00 -0.68) and control (0.18, IQR 0.00 -0.67) groups, with no different between groups (p=0.92). Experimental session time unsupervised/supervised showed consistency (1.12, IQR 0.80 -1.41) between phases (p=0.24); control showed lower (0.76, IQR 0.57 -1.08) ratios compared to experimental (p=0.027).
Conclusions:
Unsupervised exercise duration remained consistent with supervised, but frequency was reduced. Social and clinical guidance features may remain necessary for sustained lower-limb amputation exergame engagement at home.
Clinical relevance
This study provides context regarding when prosthesis users are more likely to use exergames such as Wii Fit for exercise therapy. Clinicians may consider our results when applying exergames in their practice or when developing new exergame intervention strategies.
Background
Exercise therapy is crucial to rehabilitation for a variety of clinical populations including stroke, rheumatoid arthritis, and cancer. 1 However, geographic location and program costs remain common barriers to access of rehabilitation services. 2 While home-based exercise therapy is broadly used by clinicians to overcome such limitations, non-adherence to self-directed exercise programs has been a long-standing challenge. 3 Non-adherence has been associated with limitations in factors such as feedback, 4 intrinsic motivation, 1 patient perceptions, and social support. 5 With these considerations, technology-augmented approaches to exercise therapy offer unique opportunities to support exercise engagement and adherence, improve cost-effectiveness, and improve accessibility of rehabilitation.
Telerehabilitation represents a technology-based extension of rehabilitation into the home-setting and proposes several potential benefits 6 : it supports further structured and clinician-supervised therapy, remote monitoring of patients’ progress, and remote personalization of exercises by clinicians. Applications such as exergames, which can be implemented as telerehabilitation systems, have also shown promise in encouraging engagement with home exercise therapy and self-care. 7 Multiple reviews have also indicated that these interventions can have clinically meaningful effects across several populations.7–9 Overall, such exergames use performance monitoring and feedback, among other game elements, to engage users in a fun and motivating experience.10,11 User experience factors such as learning curve, negative user perception, and technology burden of exergames remain important considerations to overcome.12,13 However, older adults have indicated exergames can be both usable, acceptable, and beneficial in increasing their exercise motivation. 14
The Wii Fit (Nintendo, Kyoto, Japan) in particular has been a popular application for rehabilitation, consisting of a compilation of strength training, yoga, aerobics, and balance games. The most common outcomes targeted in clinical trials using the Wii Fit have been balance and walking.8,15,16 However, it has also been considered for treating depression, 17 improving cognitive function, 18 and improving other psychosocial functions. 19 Moreover, a recent survey of Canadian prosthetic rehabilitation facilities showed that 46% of the therapists already use commercial games in their practice, with 95% using the Nintendo Wii Fit. 20 As such, older adults with major lower-limb amputations (LLAs) may benefit from extension of this practice into a home-based exergame intervention. LLA has a profound impact on functional mobility, 21 mental and emotional health, 22 and quality of life. 23 With high incidence in at risk populations such as people with diabetes mellitus, 24 LLA remains a common occurrence and has a high economic cost. 25
However, in the field of telerehabilitation, it remains unclear what constitutes effective design for a home-based exergame intervention to support self-efficacy and improve engagement with exercise therapy. Therefore, we examined the usage of an in-home intervention for LLA rehabilitation based on the Wii Fit called “WiiNWalk.” 26 This intervention was developed according to Social Cognitive Theory (SCT) to support exercise motivation through enhanced self-efficacy. SCT is the most common theory underpinning behavior change interventions for increasing older adults’ self-efficacy and physical activity. 27 According to SCT, key motivational processes include goals and self-evaluations of progress, outcome expectations, values, social comparisons, and self-efficacy. The four sources of self-efficacy include performance mastery, vicarious learning, verbal persuasion, and reinterpretation of physiological responses. 28 As such, the intervention leverages the gaming experience of the Wii Fit coupled with a clinically directed social environment to improve self-efficacy and exercise motivation.
Previous clinic-based pilot testing of the WiiNWalk intervention for individuals with LLA demonstrated improvements in walking capacity. 16 However, there remains a gap in knowledge of whether end-users remain sufficiently motivated to continue engaging in self-directed exercise therapy using an exergame after such an intervention. To address this gap, we asked the following question:
How often and for how long do older adults with LLA use an in-home exergame unsupervised after a supervised training period compared to an attention control?
Methods
This study was a secondary analysis of data collected for two time points according to a previously published randomized control trial protocol by Imam and colleagues. 26 The trial was approved by the University of British Columbia Clinical Research Ethics Board (Approval #: H13-01858), University of Western Ontario Health Sciences Research Ethics Board (Approval #: 104688), and University of Alberta Health Research Ethics Board (Approval #: Pro00061142).
Participants
Individuals with unilateral transtibial or transfemoral amputation were recruited to the trial with the inclusion criteria as follows:
⩾50 years of age;
Prosthesis use for ⩾2 h/day on average for 6 months;
Be cognitively able to engage in the program (Modified Mini-Mental Status Exam score >23);
Have a television compatible with the Nintendo Wii.
The exclusion criteria are as follows:
Could not communicate in English or could not provide informed consent;
Had conditions limiting their exercise participation (defined by the American College of Sports Medicine Exercise Guidelines for Older Adults);
Had a Prosthetic Socket Fit Comfort Scale <6;
Were participating in another supervised exercise or training program.
These criteria aimed to capture a large proportion of people with LLA who regularly used their prosthesis. 26 Eligible participants required clearance for physical activity participation by a physician if they had positive responses in their Physical Activity Readiness Ques-tionnaire or were over 70 years old (n = 20).
Trial design
These data were collected from a multi-site parallel evaluator-masked randomized control trial (Clinicaltrials.gov NCT01942798) with two treatment groups and two temporal phases. Eligible participants who were successfully screened were stratified by site and block randomized to either the WiiNWalk experimental group or the Wii Big Brain Academy: Wii Degree (Nintendo, Kyoto, Japan) attention control group (BBA).
The WiiNWalk intervention uses the Wii Fit, which involves a pressure sensing “balance board” that detects the user’s center of mass. The intervention includes selected and modified Wii Fit activities. Conversely, BBA is a videogame for the Nintendo Wii consisting of activities (identify, memorize, analyze, or compute) aimed at improving cognitive function.
Participants in both treatment groups completed two home-based training phases. The 4-week supervised phase consisted of three sessions per week. These sessions involved telerehabilitation in the form of group therapy using the videoconferencing app VidyoMobile (Vidyo Inc, Hackensack, United States) on tablets. Three participants trained together in this format with a clinician supervising via videoconference. For each supervised session, participants were prescribed 40 min of either Wii Fit exercise or BBA activity. We planned sessions to take approximately 60 min to complete, including interposed activities: clinician instructions, rests, and discussion. However, clinicians were given flexibility to adjust session times according to their judgment on the day. Participants were instructed not to use their respective training devices between phases for 1 week while other assessments were made. 26
In the 4-week individual unsupervised phase, participants were encouraged to use their training devices as much as possible, continuing with the same exercises or games as they did during the supervised phase and progressing to more challenging exercises or game levels as they felt comfortable. Participants were not directed to adhere to a particular regimen.
Outcomes and data extraction
Usage was measured using session count and mean session time (min) over each 4-week phase for each participant. For session times, exercise time in Wii Fit may be considered the primary active component of the experimental intervention while interposed activities such as rest and discussion play a supportive role; conversely, the attention control was based on cognitive engagement, which may be involved in both playing the puzzle games and in the interposed activities. Accordingly, session time for the WiiNWalk group was extracted from the Wii Fit graphical user interface, representing total exercise time, not including interposed activities. For BBA, application run-time (from launching to closing) was extracted from the Wii Console and represented total session time, including interposed activities. All data were extracted from Wii consoles once returned from the participants.
Analyses
We summarized session counts and session time using descriptive statistics (mean ± standard deviation). Adher-ence to the 12 supervised sessions was also described. Supervised session time adherence was summarized according to the targeted 40-min exercise criteria for experimental and 60-min session criteria for control.
Our interest was in participants’ unsupervised device usage relative to supervised training. Therefore, we compared the ratios of unsupervised and supervised session counts to parity (unsupervised/supervised = 1.0). Similarly, we compared the ratios of mean session times between unsupervised and supervised phases to parity. These unsupervised/supervised ratios were also compared between experimental and control groups. While exercise time and application run-time are not directly comparable, we considered the relative session times between phases to be comparable between groups.
Preliminary analyses demonstrated ratio data were non-normal (p < 0.05, Kolmogorov–Smirnov). Therefore, we used non-parametric tests: Wilcoxon Sign-Rank test for single samples and Mann–Whitney U test for independent samples. Moreover, we summarized ratios in terms of median and interquartile range (IQR) to remain consistent with the statistical tests. For this study, we used α = 0.05 for the statistical tests, treating unsupervised/supervised comparisons to parity as primary and between-group comparisons as secondary outcomes.
Results
In total, 72 community-dwelling lower-limb prosthesis users from three sites in Canada provided written informed consent (Vancouver: 23; London: 36; Edmonton: 13). Four participants discontinued before completing the supervised phase. Therefore, usage data were collected for 68 participants. Of these, four participants in the experimental group and two participants in the control group completed less than 50% of the supervised sessions and did not continue with the unsupervised phase; these participants were excluded from analyses. We included 62 participants in the analyses, n = 20, 34, and 8 were from Vancouver, London, and Edmonton, respectively (Table 1). Ages ranged from 51 to 82 years of age (mean = 65); 16% of the participants were female; 48% of the amputations were left side, and months since rehabilitation ranged from 2 to 207 months.
Summary of participant demographics.

In the experimental group, 56% completed all 12 supervised sessions while 21% completed 11 sessions, and 24% completed 7–10 sessions (Figure 1). In the control group, 71%, 18%, and 15% completed 12, 11, and 9 or 10 supervised sessions, respectively.
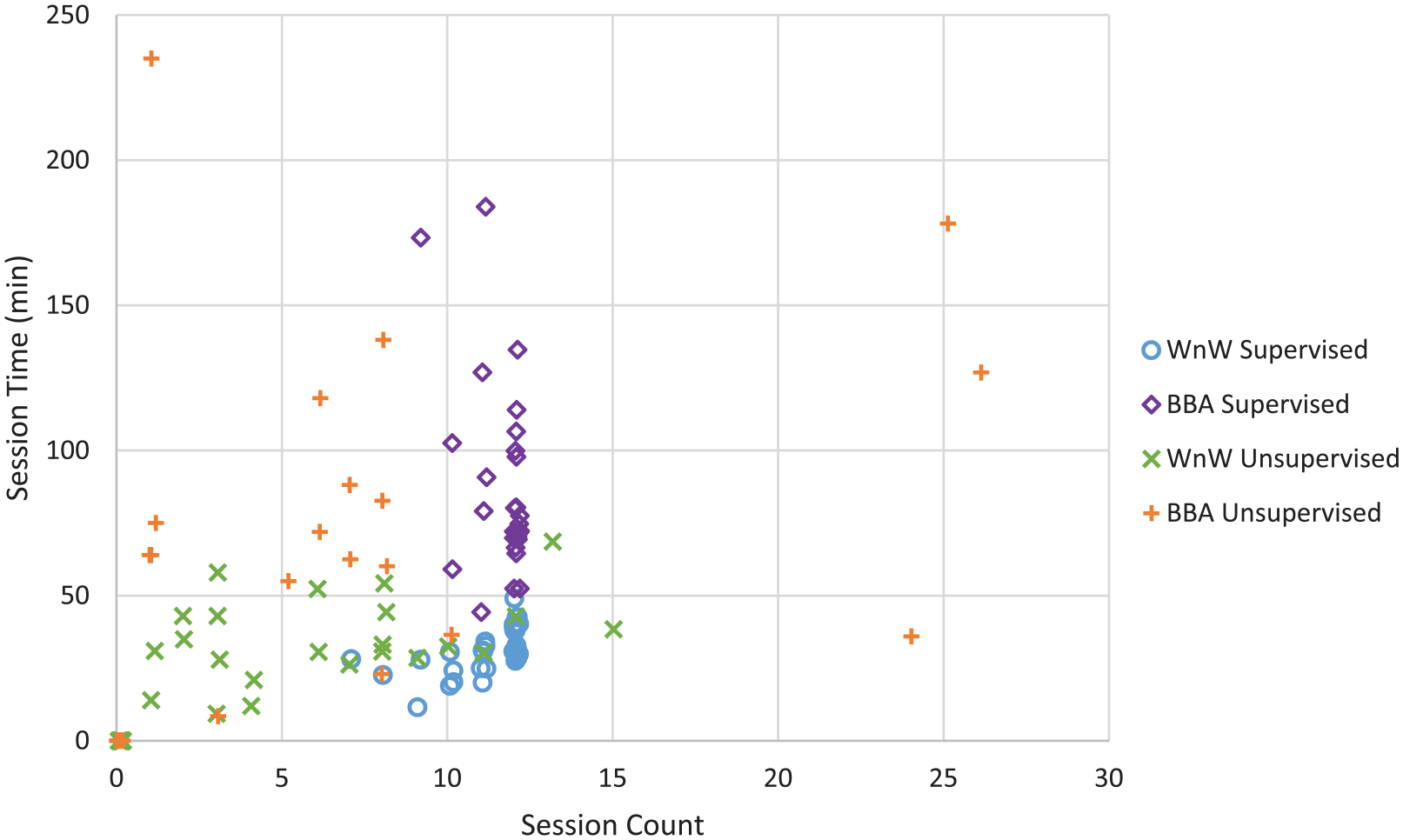
Plot of session time versus session count according to experimental (WiiNWalk) and control (BBA) as well as SP and UP.
Some participants, 15% in the experimental group and 50% in the control group, showed usage of their training devices outside scheduled sessions despite instructions not to do so. These “off-schedule” session counts ranged from 1 to 8 across both groups with one control participant showing 14 off-schedule sessions during the supervised phase.
During the unsupervised phase, 68% participants in the experimental group used their training device at least once (Figure 1). Similarly, 64% in the control group used their training device unsupervised. Including those who did not use their devices, the mean session count over 4 weeks was 4.3 ± 4.5 for the experimental group and 5.5 ± 7.7 for the control group. However, four participants in the experimental group appeared to follow a similar schedule as the supervised phase with 2–4 sessions per week. While no participants in the control group showed such similar behavior, three participants used their device almost daily with 24–26 sessions over the four unsupervised weeks.
To compare usage between unsupervised and supervised phases, we calculated the ratio of unsupervised session counts to supervised session counts. Both groups showed ratios statistically less than 1.0 (p < 0.001 for experimental, p = 0.005 for control) with median ratio 0.25 (IQR: 0.00–0.68) in the experimental group and 0.18 (IQR: 0.00–0.67) in the control group (Figure 2). The session count ratio unsupervised/supervised was not statistically different between experimental and control groups (p = 0.92).
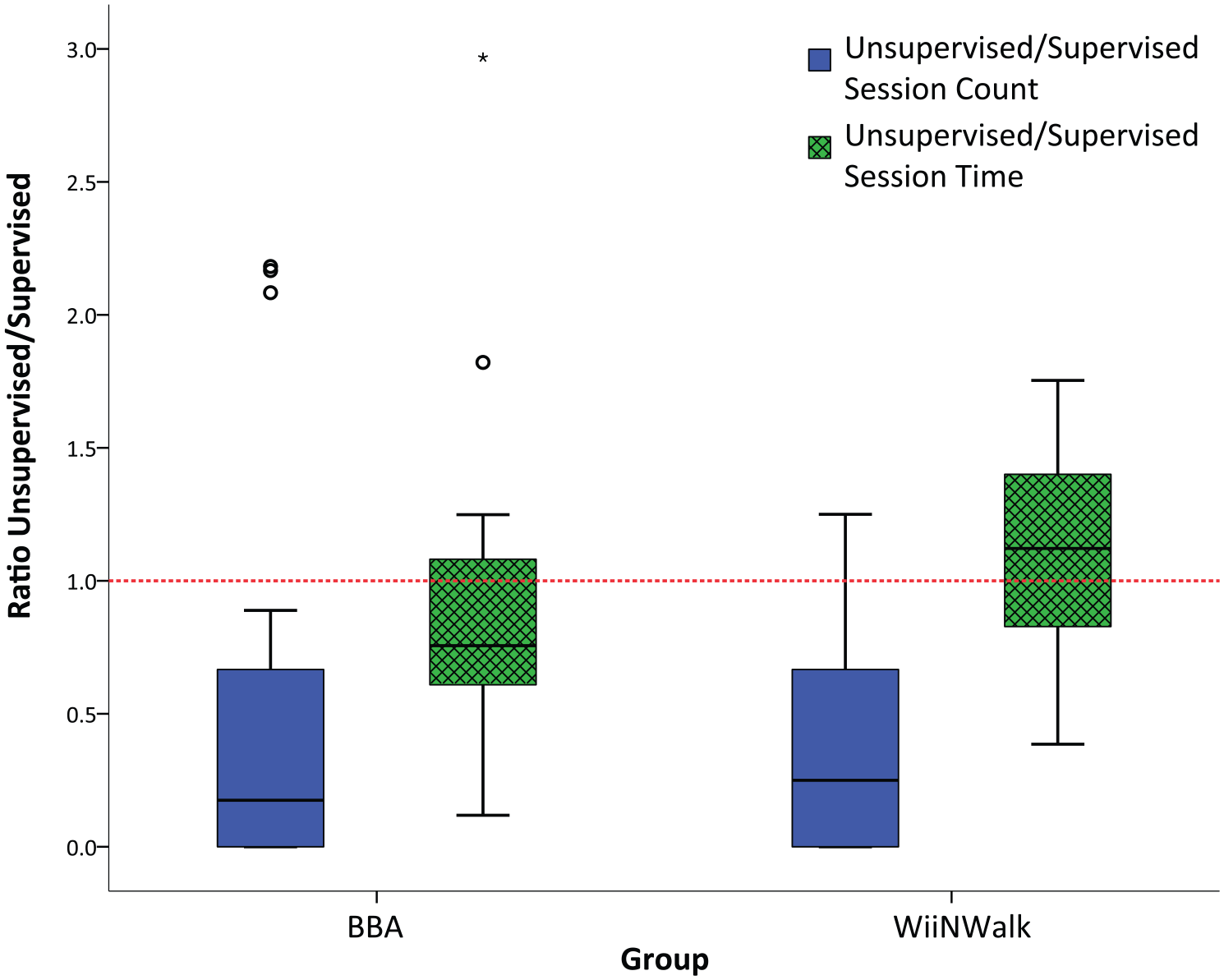
Box plot for ratio of unsupervised/supervised session counts and session times; boxes delineate 25th, 50th, and 75th percentiles while whiskers are set to 1.5 times the interquartile range. Ratio of 1.0 (red dashed line) represents parity. Ratios for participants who had zero usage during UP were included for session count and not included for session time ratios.
Mean ± standard deviation exercise time per session across participants was 31.5 ± 8.0 min in the experimental group, and the mean application run-time was 87.9 ± 33.6 min in the control group during the supervised phase. The control group showed four participants with individual unsupervised sessions of over 5 h: 1/9, 1/24, 7/26, and 8/25 sessions. We attributed this to accidentally leaving BBA running and removed them from mean time calculations. For the 23 participants who did use their training devices unsupervised, mean exercise times in the experimental group, 35.1 ± 14.6 min, were statistically shorter than the prescribed 40 min of exercise while the mean total session times in the control group, 84.6 ± 56.6 min, were statistically longer than the prescribed 60 min (p < 0.001 for both).
Comparing unsupervised session times with supervised times (Figure 2), the median ratio of unsupervised/supervised session times was 1.12 (IQR: 0.80–1.41) for the WiiNWalk group and not statistically different from 1.00 (p = 0.236). For the BBA group, the median ratio unsupervised/supervised was 0.76 (IQR: 0.57–1.08) and was not statistically different than 1.00 (p = 0.085). Finally, median ratio unsupervised/supervised of session times was statistically different between WiiNWalk and BBA groups (p = 0.027).
Discussion
Overall, participants demonstrated good adherence to the supervised regimen. Some participants in both groups used their training device independently, against instructions, during the supervised phase, suggesting these participants were particularly motivated to use their devices. Moreover, higher occurrence in the control group suggests BBA may be more motivating or enjoyable than WiiNWalk. Similarly, mean exercise time for the WiiNWalk group was less than prescribed time, while session time for the BBA group was greater than prescribed time in the supervised phase. A possible contributing factor to these differences is the inherent disparity in physical effort required for a prosthesis user to stand on the Wii Fit balance board compared to sitting and playing BBA.
Approximately two-thirds of participants used their training devices unsupervised at least once, suggesting some degree of desire for independent usage. Indeed, both groups demonstrated similar proportions of participants in this respect. Ultimately however, participants in both groups used their training devices substantially fewer times overall in the unsupervised phase. These results suggest that motivation for self-directed engagement with both WiiNWalk and BBA after supervised training was limited. Moreover, few participants appeared inclined to replicate the three sessions per week regimen of the supervised phase.
According to the SCT framework, two sources of self-efficacy were reduced moving from supervised to unsupervised: clinician direction and peer dynamics. In the supervised phase, the clinician directed and facilitated group therapy with participants through teleconferencing on tablets. This was anticipated to form a social dynamic that promoted vicarious learning and a sense of belonging to a group with a common goal. Furthermore, clinicians also helped end-users interpret their physiological responses in ways that supported self-efficacy, for example, “It’s normal to feel tired here.” Without these components, the gaming experiences (Wii Fit and BBA) still provided a means to support performance mastery and some degree of verbal persuasion. However, our results suggest vicarious experiences and social influences remain crucial to supporting self-efficacy in the long term, even after an initial training program.
Taken together, these results suggest an overall lack of acceptance of the training devices on their own. From the lens of the Unified Theory of Acceptance and Use of Technology, 29 the reduced usage may be explained by several reasons. Reduced performance expectancy may explain reduced usage, if participants stopped seeing the Wii games as therapeutically useful without clinicians’ coaching. Their attitude toward the games may change with loss of social influence whereby participants found the games less fun or felt less confident on their own. As such, we may expect more consistent engagement with the WiiNWalk intervention (and similar exergames) only if some therapeutic relevance, remote clinician involvement, or social component (e.g. online community) were maintained. For example, Nicholson and colleagues demonstrated that older adult participants could use the Wii Fit unsupervised in a group setting for 30 min, three times per week for 6 weeks, with statistical benefits in balance and mobility as well as excellent adherence. 30 While the Wii Fit lacks the capacity to facilitate this in home settings, future design and development of more patient-centered exergames may focus on these considerations and satisfy the need for social environments and clinical relevance in the long term.
When participants did use their WiiNWalk system unsupervised, they appeared to exercise for statistically equivalent durations as in supervised sessions. The mean exercise time of 31.5 min in the supervised and 35.1 min in the unsupervised phases fell short of the prescribed 40 min. However, a half hour of exercise using the Wii Fit can still improve balance. While older adults may tolerate Wii Fit training for up to an hour,31,32 the added strain of prosthesis use may have limited this tolerance. Similarly, other studies involving LLA exercises have limited exercise to 30 min.33,34 Nonetheless, our results indicate participants were willing to use the Wii Fit for therapeutically sufficient durations in both study phases.
Descriptively, control group participants showed a possible trend toward decreasing session time moving from supervised to unsupervised. However, this trend was not statistically significant. For control participants with unsupervised/supervised session times substantially lower than 1.0, a number of factors may explain the reduction: reduced novelty, increased puzzle difficulty, or simply lack of group discussion. Notably, the session time ratios were statistically different between experimental and control groups, suggesting exercise gaming may be more consistently motivating than non-exercise gaming in terms of session duration. Perhaps, once participants started exercising, the combination of gaming feedback and exercise-related endorphin release 35 maintained participants’ motivation up to their fatigue limits.
Limitations
While we used multiple Canadian sites for generalizability, sampling was ultimately insufficient to determine any site-dependent effects. Data were used for statistical tests both within and between groups. Therefore, between-group comparisons should be interpreted with caution due to multiplicity bias. For usage, session times lacked information on breaks. Therefore, we made a reasonable assumption of consistent proportions of breaks across phases. While we excluded outlier cases where the Wii was obviously left running, we could not determine if this occurred for shorter periods, for example, 10–30 min after actual completion. Furthermore, we were unable to confirm the user of the Wii for each unsupervised session, for example, if a participant decided to let someone else try the games.
Finally, while the data presented in this article provides some insight into the unsupervised usage of WiiNWalk, we were limited by purely quantitative measures. Personal factors such as decreased perceived benefit, memory, or other perceived barriers3,4 may also contribute to changes in usage. Design of home-based exergame interventions may benefit from user perspectives to contextualize such usage data, for example, what aspects of WiiNWalk are most motivating or enjoyable and what factors prevent more regular use when unsupervised.
Conclusion
Compared to a supervised clinician-guided training phase, participants in both WiiNWalk exercise gaming and BBA non-exercise gaming groups used their training devices less frequently in the unstructured unsupervised phase. While participants in the experimental group demonstrated consistent exercise times between supervised and unsupervised phases, inclusion of videogaming elements alone may be insufficient for motivating self-directed regimens of exercise in the unsupervised home-setting. Therefore, future designs of home-based exergame interventions may benefit by inclusion of sustainable clinician input or social involvement.
Footnotes
Author contributions
All authors contributed equally in the preparation of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Canadian Institutes of Health Research (CIHR).
