Abstract
Background:
The observation of upper body movement is gaining interest in the gait analysis community. Recent studies involved the use of body-worn motion sensors, allowing translation of laboratory measurements to real-life settings in the context of patient monitoring and fall prevention.
Objectives:
It was shown that amputee persons demonstrate altered acceleration patterns due to the presence of prosthetic components, while no information is available on how accelerations propagate upwards to the head during level walking. This descriptive study aims to fill this gap.
Study design:
Original research report.
Methods:
Twenty definitive prosthesis users with transtibial amputation and 20 age-matched able-bodied individuals participated in the study. Three magneto-inertial measurement units were placed at head, sternum and pelvis level to assess acceleration root mean square. Three repetitions of the 10-m walking test were performed at a self-selected speed.
Results:
Acceleration root mean square was significantly larger at pelvis and head level in individuals with amputation than in able-bodied participants, mainly in the transverse plane (
Conclusion:
The obtained parameters allow an objective mobility assessment of amputee persons that can integrate with the traditional clinical approach.
Clinical relevance
Transtibial amputees exhibit asymmetries due to the sound limb’s support prevalence during gait: this is evidenced by amplified accelerations on the transverse plane and by related differences in upper body movement control. Assessing these accelerations and their attenuations upwards may be helpful to understand amputee’s motor strategies and to improve prosthetic training.
Keywords
Background
Recently, there has been a growing interest among the scientific community towards upper body mechanics during walking, both in healthy1–5 and in pathological subjects.6–8 Recent studies in this context have used body-worn inertial sensors to measure upper body accelerations in different groups, healthy adults and children, neurological patients or in the elderly, that is, to reduce risk of falling.9–12 Indeed, the accelerometric technique allows the translation of this type of evaluation to clinical settings, where it is essential to perform analysis in a simple and inexpensive way, without complex laboratory assessments and with minimum encumbrance for the patient. Acceleration data measured at different body levels in the three anatomical directions can provide insightful information about gait stability, 6 which has been referred to as the capacity to minimize oscillations during walking from the lower to the upper levels of the human body. 13
Regarding the role of the upper body mechanics in gait stability, there is a focus in characterizing how accelerations due to repeated foot impact during locomotion are transmitted upwards, where the vestibular receptors (primarily responsible for the ability to maintain the static and dynamic balance) are located. Indeed, observation of upper body movement allows greater understanding of the motor strategy adopted during locomotion.13,14 Based on the literature,15,16 linear accelerations of points distributed on the cranio-caudal axis, at the level of the lumbo-sacral/cervical-thoracic hinges and the head, are considered the most effective descriptors of the ambulatory task.
Recent studies have shown how these accelerations are transmitted in healthy people 5 and in some pathological subjects.11,17,18 Using either root mean square (RMS) values 10 or frequency domain measures, 19 upper body accelerations have been described in healthy subjects. Specifically, healthy subjects typically present a progressive reduction of acceleration from pelvis to sternum and from sternum to head, which reflects the adoption of postural control strategies. Conversely, it is conceivable that different acceleration patterns could be a symptom of difficulties in controlling upper body movement, which has been associated with risk of falling. 20
In particular, the lack of a limb and functional asymmetry introduced through the use of prosthetic elements may affect the stability of movement during walking in persons affected by lower-limb amputation. It is possible that individuals with amputations exhibit different locomotor strategies compared with able-bodied people, developing asymmetric compensation movements and altering the normal transmission of acceleration among upper anatomical body segments. In fact, a recent study 11 demonstrated that upper body accelerations and gait stability change with different variations of prosthetic components, depending also on amputation levels. Therefore, the particular sensory feedback and locomotive schema are not only conditioned by the impairment, but also by the presence of the prosthetic device. Using the same instrumental approach, Howcroft et al. 21 identified how these accelerations relate to clinical scores for balance and mobility, stating the need to explore upper anatomical locations for accelerometers. At the state-of-the-art level, no studies describe how accelerations propagate through the upper body from the pelvis to the head in amputee persons. In fact, only the quantification of the acceleration at a single body level is performed through the adoption of a mono-sensor approach,8,11,22 which does not allow for measurements of acceleration distribution among the upper body hinges.
Therefore, the aim of the present study is to investigate upper body accelerations in individuals with transtibial amputation during gait and to describe how these accelerations propagate from the pelvis to the head. A multi-sensor approach was adopted in order to investigate the trend of accelerations acquired at different levels of the upper body. The importance of this analysis is represented by the possibility of finding suitable quantitative indices to assess amputee persons’ mobility and gait stability, which could be related to the risk of falling and quality of life.
Methods
Subjects
A group of 20 people (amputation group-AG: 19 males, 1 female; age: 51.1 ± 12.7 years old; height: 1.73 ± 0.06 m) with post-traumatic unilateral transtibial amputation (7 right side, 13 left side), definitive prosthesis users and able to walk without aids, and another group of 20 able-bodied (control group-CG: 19 males, 1 female; age: 48.3 ± 8.7 years old; height: 178.7 ± 0.06 m) were recruited for the study. The groups were matched for gender and age. Leg length for each subject enrolled was defined as the distance between the greater trochanter and lateral malleolus, as obtained through manual bony landmark palpation by one experienced researcher. All participants to the AG were prosthesis users of at least 18 months for a minimum of 4 h per day, were physically active and had no comorbidities. The relevant prosthetic components (foot, socket and suspension system) are also listed (Table 1).
Prosthetic components of the 20 individuals with transtibial amputation enrolled in the study.
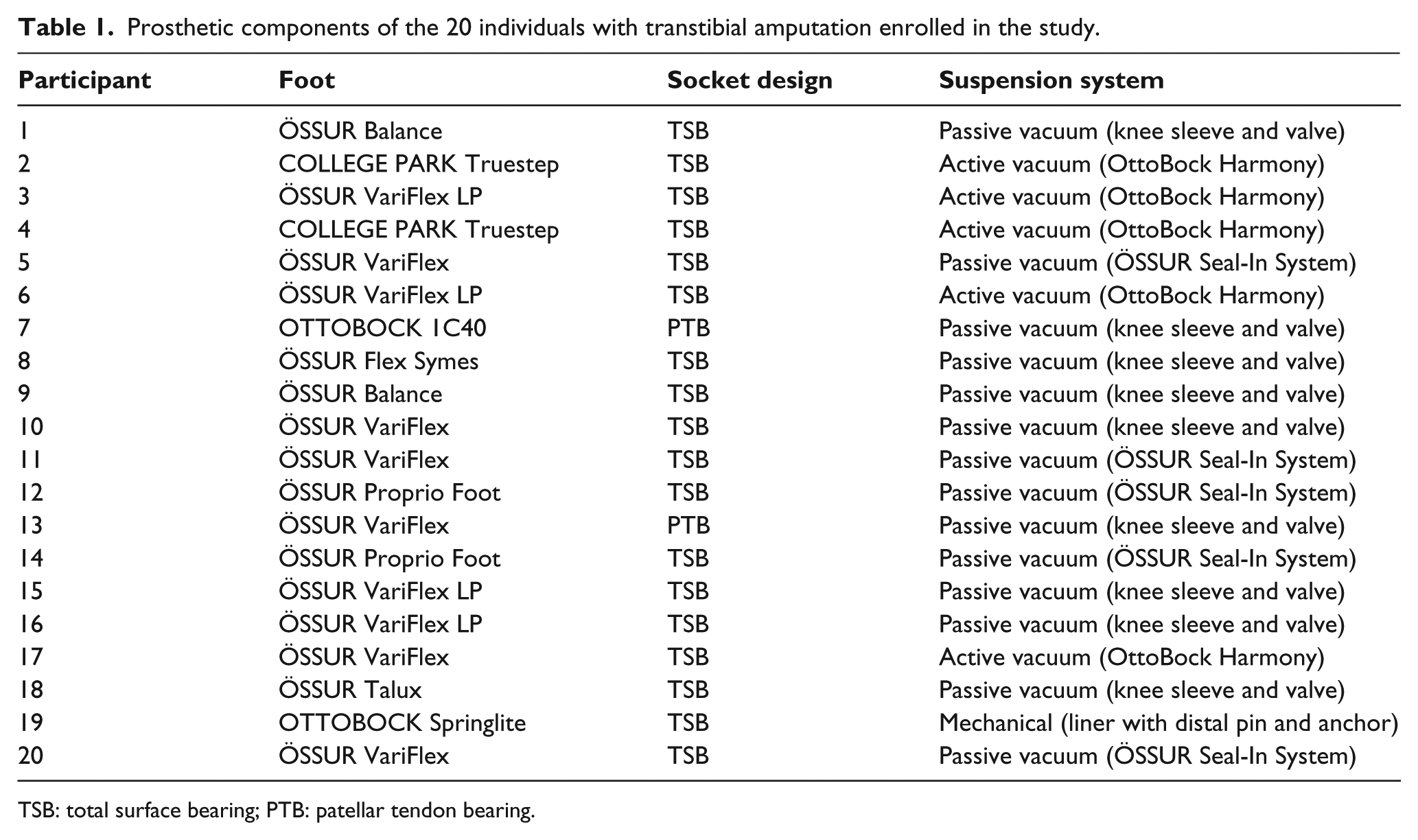
TSB: total surface bearing; PTB: patellar tendon bearing.
The Independent Ethics Committee at Santa Lucia Foundation (IRCCS) approved all experiments, adhering to the Declaration of Helsinki for medical research involving human subjects. Informed consent was obtained from all participants prior to data acquisition.
Equipment and data acquisition
Three magnetic inertial measurement units (Opal, APDM Inc., Portland, OR, USA) were used to collect 3D data of acceleration, angular velocity and magnetic field. Sample rate was set at 128 Hz, and full-range scale was set at ±2 g, with g = 9.81 m/s, ±1500°/s and ±600 mT, respectively. A robust synchronized streaming mode configuration was used to synchronize the units and to avoid any loss of data resulting from interruptions in the wireless transmission.
Participants were equipped with swim caps, stretch tops and stretch shorts, with specific pockets for sensor housing, in order to position the devices at the following body levels: (1) occipital cranium bone close to the lambdoid suture of the head (H); (2) centre of the sternum body (S); (3) L4-L5 level, slightly above the pelvis (P). Each sensor was identified by an inertial sensor frame formed of three orthogonal axes
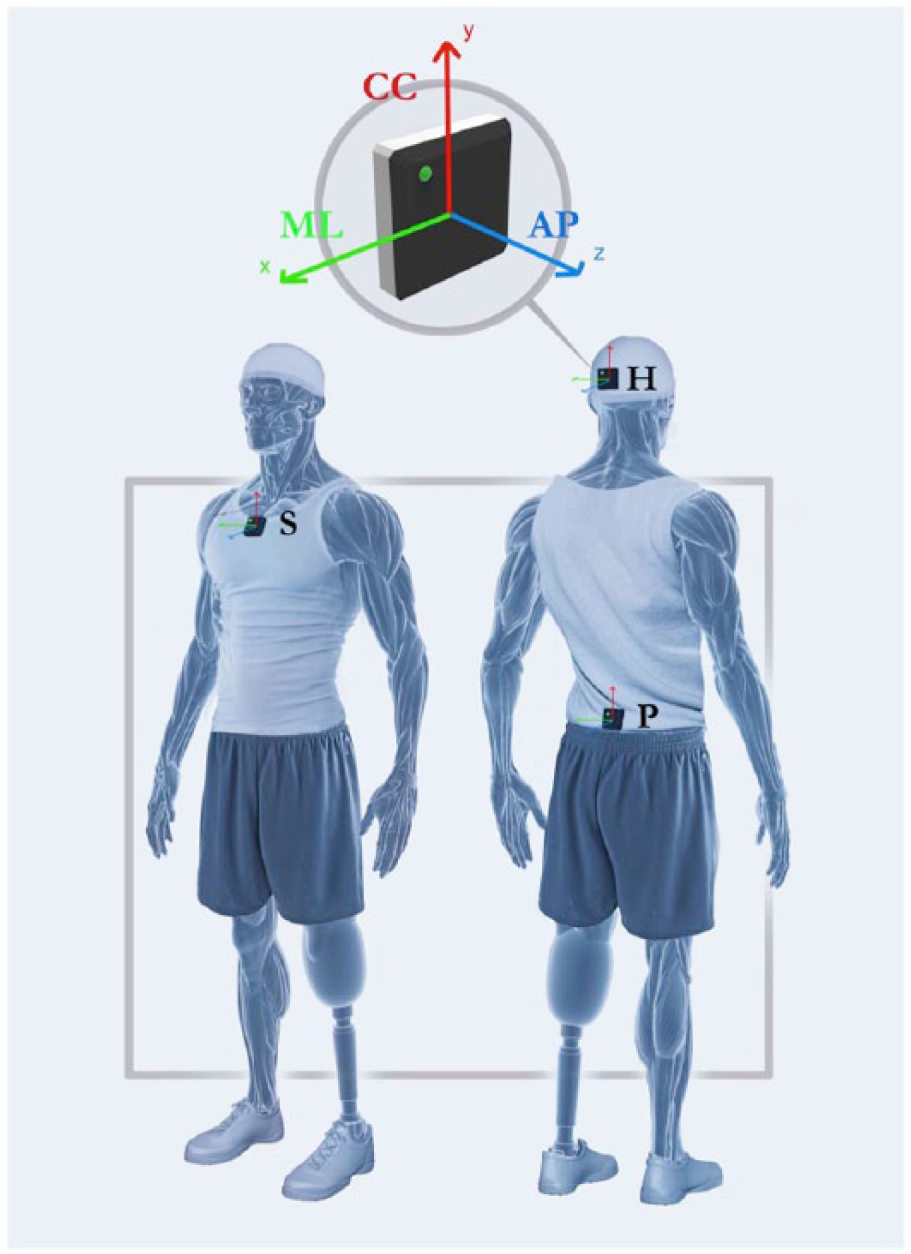
Sensor location and axes orientation of the device attached on the subject’s body segments: head (H), sternum (S), pelvis (P). The three local axes represent the cranio-caudal (CC), medio-lateral (ML), antero-posterior (AP) directions in the inertial references system.
Procedures
Each participant was asked to maintain the standing position for 10 s and then to walk in a 10 m linear pathway at his or her self-selected walking speed. The start and stop lines of the pathway were marked by two visible strips. Each participant performed three trials.
Data analysis
The data were processed using custom software written in MATLAB (The MathWorks Inc., Natick, MA, USA). A local anatomical (LA) frame was defined by correcting the inertial reference system (I) with the orientation data obtained in a 3s window of the initial static part of the acquisition (Figure 2). The inclination of the (I) reference frame with respect to gravity was computed from acceleration measurements when the participants were stationary in a standing position. Using these data, each unit reference frame (I) was rotated, through a rigid transformation, following the pitch–roll–yaw rotation sequence. The resultant LA frame was, therefore, characterized by a CC axis aligned with gravity, while the subject stood erect during the stationary phase, an AP axis aligned in the direction of progression and a ML axis obtained by vector product in a right-handed reference frame. The whole procedure is detailed in Bergamini et al. 23
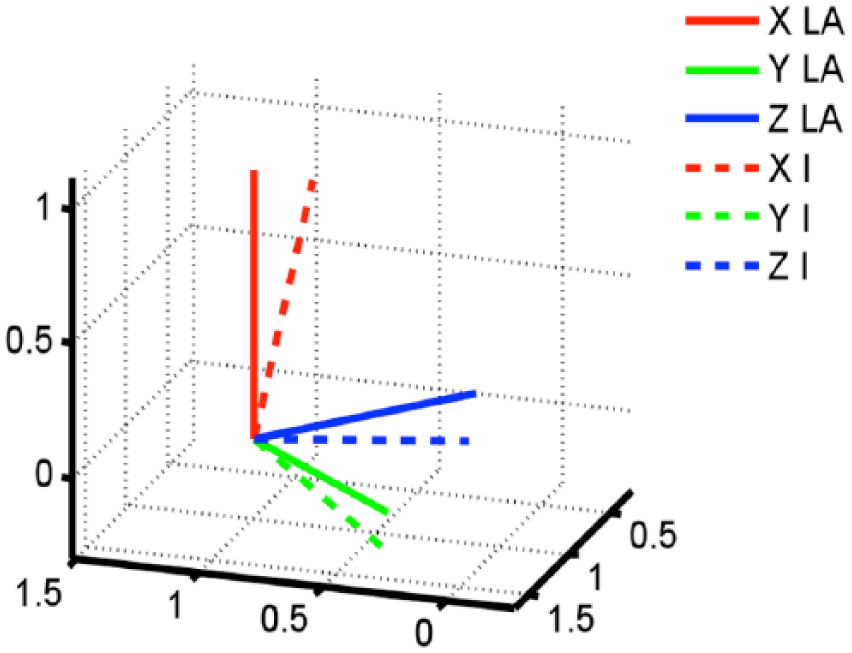
Local anatomical (LA) frame definition from the inertial unit frame (I) reference system through MATLAB software. This is obtained through a rigid transformation of (I), following the pitch–roll–yaw rotation sequence. The relevant rotation matrix is obtained by comparing gravity vector with measured acceleration when the participants were stationary in a standing position.
The acceleration components were low-pass filtered using a fourth-order Butterworth filter with a cut-off frequency of 20 Hz 24 and then expressed in the LA frame. The gravitation acceleration component was subtracted from the recorded signal before data processing. As an example, the CC component of the acceleration at pelvis level is reported for a representative walking trial performed by an AG participant (Figure 3).
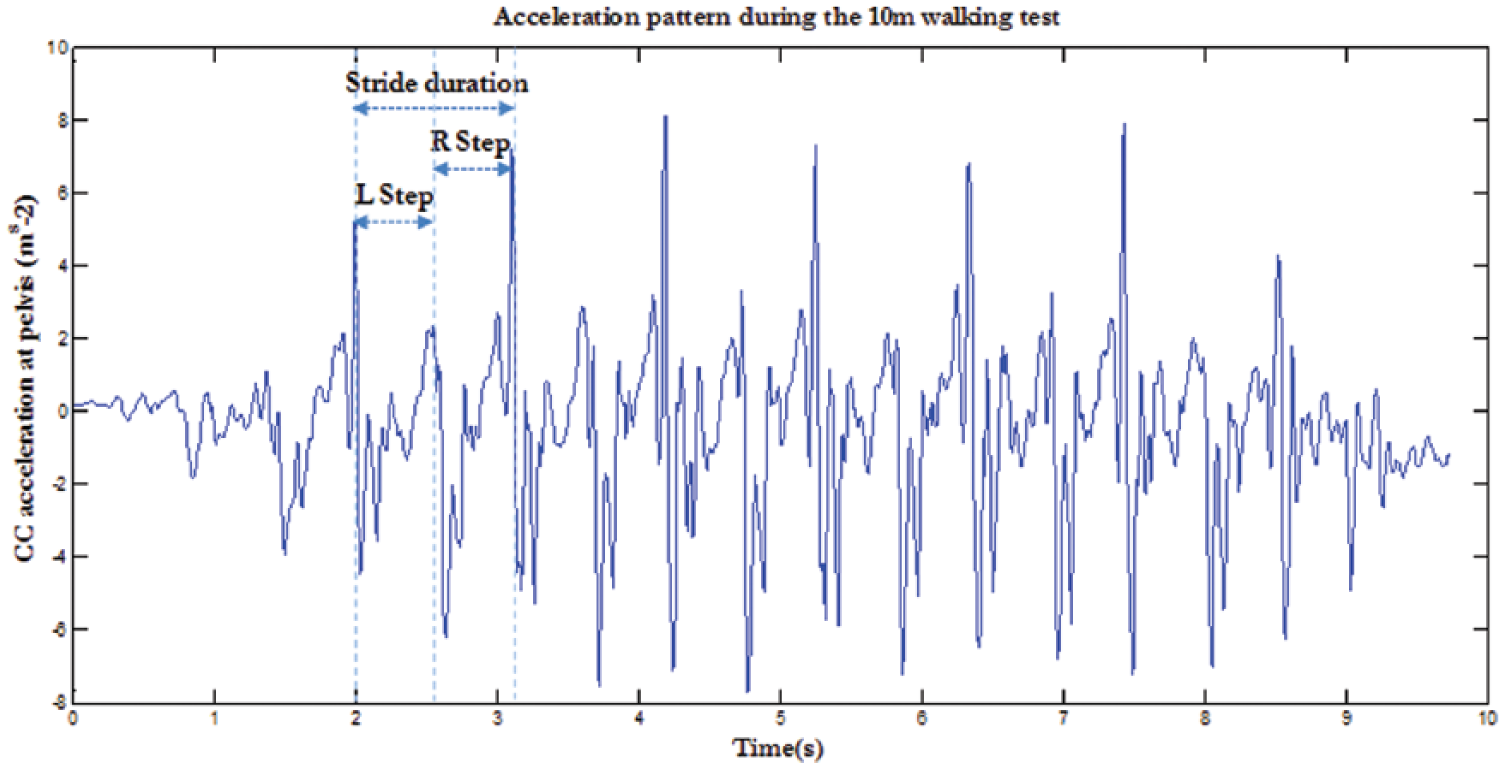
Cranio-caudal (CC) component of the acceleration measured at pelvis level for a representative walking trial performed by a participant with amputation. Acceleration peaks identify initial contacts of both amputee and sound limbs, indicated as L and R, respectively. These time instants delimit both steps of each gait cycle.
In accordance with previous literature,25,26 the steady-state phase was identified and then four consecutive steps (two strides) for each trial were selected for analysis. After bias removal of the acceleration signals, 23 root mean square of acceleration (RMSa) at the three aforementioned levels of the upper body were computed in the relevant LA frame in each stride, using the component accelerations (RMSa_CC, RMSa_ML and RMSa_AP). High RMSa values indicate large body segment accelerations and are associated with poor stability.
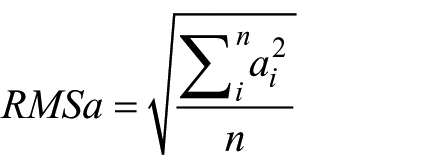
Variation of RMSa from a level
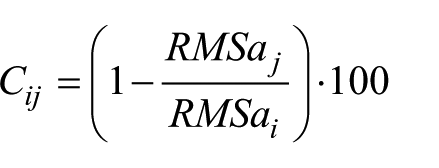
Positive percentage values refer to a reduction of upper body accelerations with respect to lower levels, while a negative value represents a percentage increase of the acceleration. Attenuation coefficients proved to be effective when characterizing how subjects attenuate upper body accelerations from the pelvis to the sternum and the head10,27 and, interestingly, how individuals with movement disorders exhibit alterations of the same patterns. 18 The average of the values obtained from the two selected strides was considered for each parameter. Moreover, the total duration and number of steps taken during the 10-m walking task were estimated through the procedure proposed by Brandes et al., 28 which makes use of the peaks of the acceleration time histories and confirmed using video data. From these measurements, step frequency (SF = total number of steps/time to complete the test), average step length (SL = 10 m/total number of steps) and average walking speed (WS = 10 m/time to complete the test) were estimated.
In particular, to take into account for possible differences in the WS values due to stature differences, the Froude number (Fr) 29 was also computed
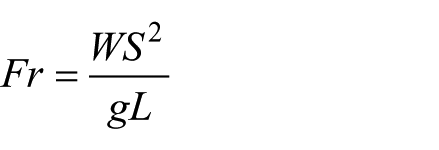
where
Both WS and Fr were only assessed to prevent potential influences of speed on the RMSa calculation. 30 In order to obtain stability indicators, two intra-subject RMSa component percentage ratios were defined: 11 RMSa_CC/RMSa_ML and RMSa_CC/RMSa_AP. Therefore, high ratio values indicate good stability and vice versa.
For all spatio-temporal parameters, RMSa values and attenuation coefficients were computed for the three trials. Since no outliers were detected after the completion of a Z-score procedure, mean values were further considered for statistical analysis. Inferential statistical analysis was then performed using the IBM SPSS Statistics software (v23, IBM Corp., Armonk, NY, USA;
The distribution of the above-mentioned biomechanical parameters was verified using the Shapiro–Wilk test of normality (
Results
Walking speed was significantly lower in AG than CG (
Mean ± standard deviation for spatio-temporal parameters in the individuals with amputation group (AG) and in the control group (CG).

Significant values of
MANOVA revealed a significant multivariate main effect for the group factor (Wilk’s
RMSa parameters computed along the CC, ML and AP directions at the head, sternum and pelvis level are reported (Figure 4, left panels). RMSa was significantly larger in AG than in CG at the head level along AP and ML directions (
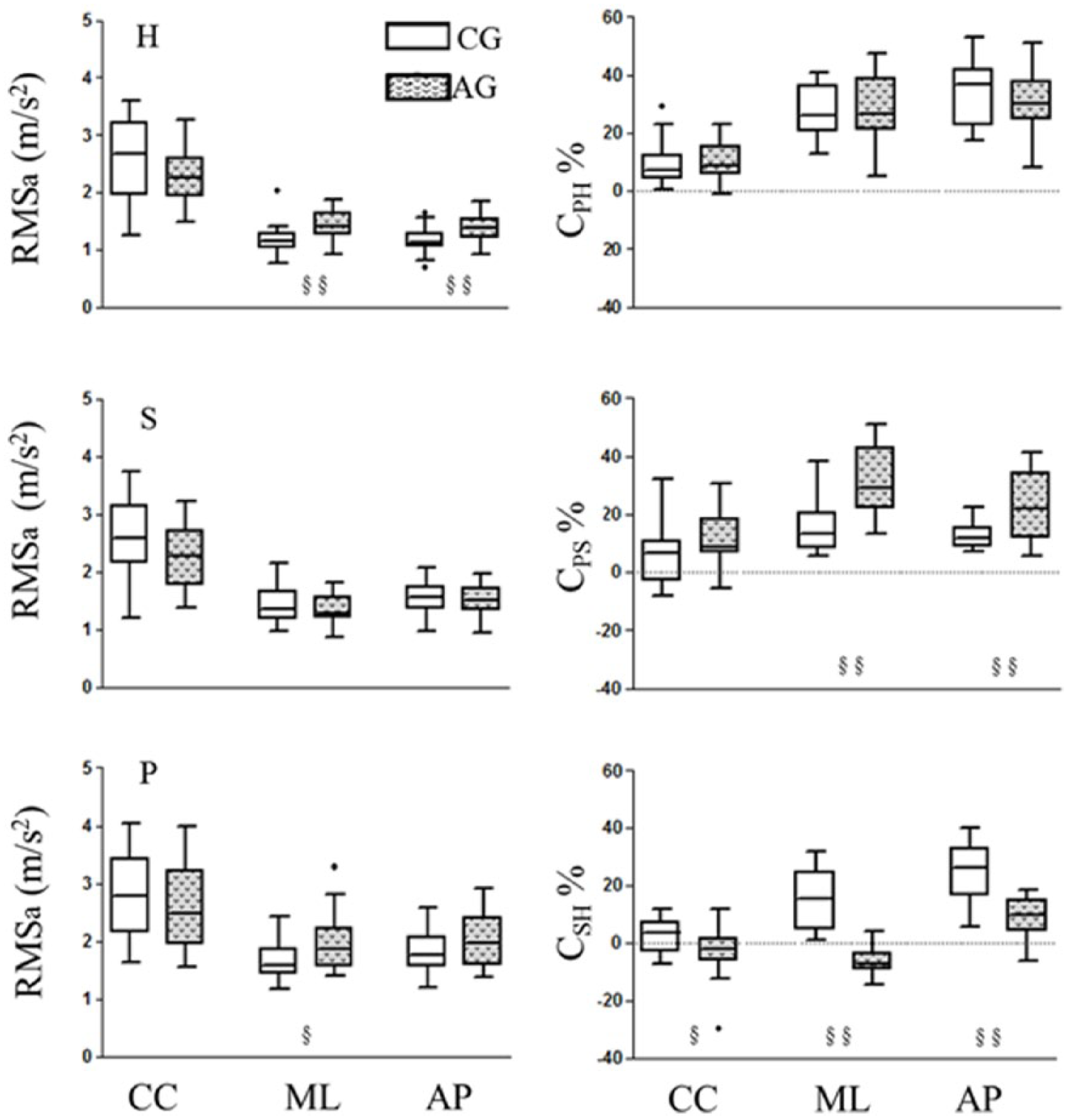
On the left, RMS of the acceleration components at the three levels: head (H), sternum (S) and pelvis (P). On the right, attenuation coefficients from pelvis to head (CPH), from pelvis to sternum (CPS) and from sternum to head (CSH) along the three anatomical axes. Parameters computed for the CG and AG are represented with empty and filled box-plots, respectively. Significant between-group differences (
Regarding the attenuation coefficients (Figure 4, right panel), CPS values were larger in AG than in CG along AP and ML directions (CPS_AP:
Finally, both AP- and ML-stability indicators were lower in individuals with amputation than in able-bodied subjects both at the pelvis and head level. The same was slightly, but not significantly, lower in AG than CG at the sternum level (Table 3).
Mean ± standard deviation of the percentage stability indicators (RMS_CC/RMS_AP, RMS_CC/RMS_ML) at the three anatomical levels for both AG and CG.
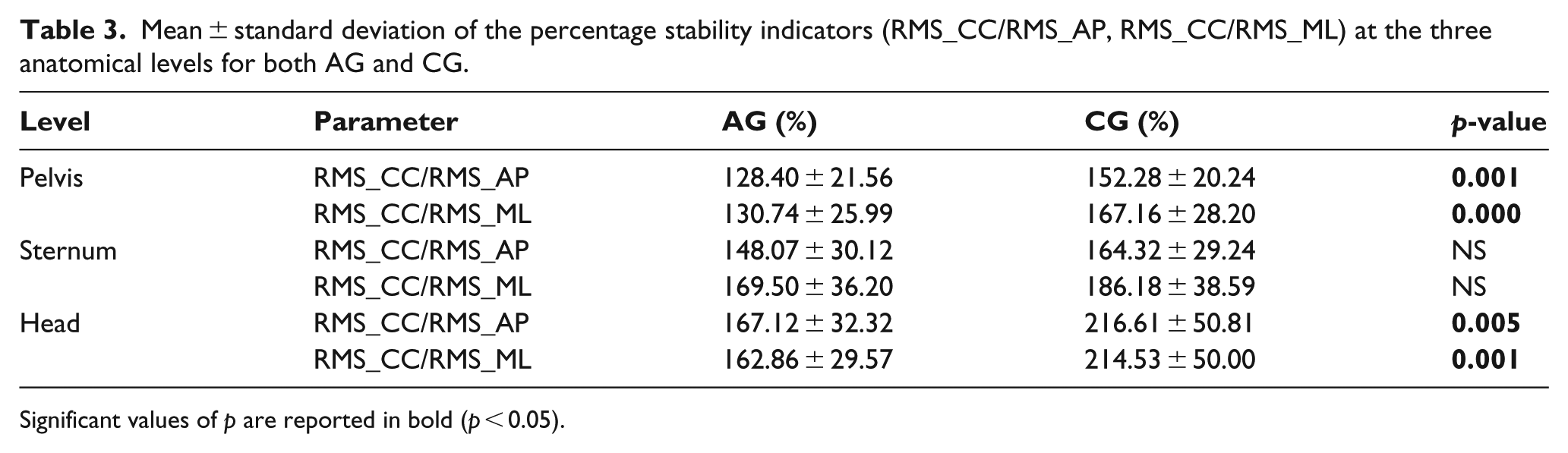
Significant values of
Discussion
In the present study, a multilevel inertial sensor set-up was proposed to characterize segmental accelerations of the pelvis, sternum and head during gait. This approach was used to investigate how accelerations are attenuated from the bottom to the top in individuals with transtibial amputation when compared to age-matched able-bodied subjects. Investigating accelerations and how they propagate through the upper body has a physiological significance and quantifies the role of upper body structures in reducing head accelerations. In fact, this role can be active, when looking at both trunk and arm movements during gait, but also passive, when taking into account the contribution of the underlying anatomical structures (bones, muscles, tendons, cartilages). 18
The AG and CG were homogeneous in terms of average step length, step frequency and normalized walking speed. This last circumstance justified the direct use of RMSa for between-group comparison. The relevant analysis of the CC, ML and AP components of acceleration measured at the pelvis, sternum and head level confirmed differences between the two groups.
A first consideration is related to the differences in RMSa along the ML direction, at the pelvis level, mainly related to the functional asymmetry caused by the prosthetic leg. To accommodate for this, persons with amputation implement compensation strategies related to the inability to perform ankle plantar/dorsiflexion with the prosthetic foot. As a consequence, typical adjustments at the proximal joints, pronounced anterior pelvic tilt and, thus, hyperlordosis of the lumbar spine during foot stance are observed.31,32 These pronounced movements in the sagittal plane, due also to the greater rigidity of the prosthetic joint with respect to the physiological one, may explain the higher attenuations between the pelvis–sternum anatomical districts with respect to the CG, in all directions.
The larger pelvic accelerations on the ML axis observed in individuals with amputation, if on one hand it influences pelvis-to-sternum attenuation, on the other hand it has to be linked with the increased head accelerations, taking into account that sternum RMSa is similar in the two groups. The latter circumstance could indicate that the increased ML head acceleration is not directly justified by increased ML instability at the lower body level due to the prosthesis, but rather by the adoption of particular compensation strategies implemented by the AG. Pelvic RMS accelerations on the AP axis were more amplified in the amputee persons. This is potentially linked to the existing asymmetries due to the sound limb’s support prevalence during gait.
The larger accelerations recorded in AG at the pelvis and head level, as it happens for gait stability issues in gait pathologies,11,18,33 might interfere with the processing of visual, vestibular and somatosensory information regarding body position and orientation. This circumstance could induce a vicious circle that potentially weakens the control of the upper body movement during gait. 18
The RMSa values obtained at the lower trunk were slightly higher than those previously reported in persons with transtibial amputation. 11 In the cited study, a sample of eight subjects performed a walking test in the last week before dismissal from a rehabilitation hospital. It is conceivable that the mobility level of these subjects was lower than that of the sample enrolled in the present study, as shown by a lower WS. This study confirms that the high RMSa at the pelvis level is an important clinical parameter, explaining the reduced gait stability in the pathological group. This paradigm has promising developments of spreading into the clinical community, proved that this can be associated with clinical scales about fall risk assessment, as the Berg Balance Scale. 33
Regarding the stability index, both ML- and AP-stability were lower in persons with amputation than in healthy subjects. In particular, RMS_CC/RMS_AP and RMS_CC/RMS_ML were significantly lower in AG at the pelvis and head level, likely a sign of control issues and precarious balance. These stability parameters were shown to be suitable for assessing trunk stability during walking in persons with amputation, 11 with lower values representing impaired stability.
Concerning the transmission of accelerations, it is revealed that subjects with a transtibial amputation apply an increased pelvis-to-sternum attenuation with respect to CG, compensating for higher RMSa at the pelvis level, thus exhibiting an RMSa at the sternum similar to healthy subjects.
Specifically, in AG, CPS is positive and significantly larger than in the CG, thus indicating that individuals with transtibial amputation attenuate more than able-bodied people from pelvis to sternum. The difference between pelvis-trunk AP accelerations is likely due to a different pattern of locomotion developed by amputee persons, as suggested by Iosa et al. 11 This circumstance could not be attributed to a deficit of the CG, but potentially to the fact that for able-bodied persons, large attenuations are not strictly required to guarantee safe walking and adequate dynamic stability of the trunk.
The analysis of sternum-to-head attenuation (CSH) presented a behaviour in the AG that was the opposite to that of the CG. While the latter group reduced accelerations from the sternum to the head, guaranteeing protection of the head, the former presented low and even negative CSH values, meaning that acceleration may increase from the sternum to the head. In a recent study performed on sub-acute stroke individuals, 33 this pattern was associated with a decreased walking ability and exhibited a correlation with clinical scales for fall risk assessment, representing a potential tool to support clinicians in the identification of patients exposed to a high risk of falling. In this respect, based also on the known similar correlations reported for people with amputation, 21 further work should be done to verify the feasibility of this approach in an adequate sample of persons with amputation.
The aforementioned inefficiency in attenuating upper body accelerations that are significantly higher in AG than in CG both in AP and ML directions at the head level could be related to rigidity issues of the head-trunk system for this particular population. It is conceivable that compensating for these higher upper body accelerations could be a potential cause of the typical back and neck pains associated with the lower-limb amputee population. 34
As overall consideration, the AG present CPH values similar to CG, showing a comparable behaviour with respect to the latter group. Therefore, regarding the increased pelvis acceleration observed in AG, it is likely that a physiotherapeutic intervention to stabilize the pelvis may also contribute to reducing accelerations at higher body levels.
Further research should be performed on larger cohorts to confirm and expand upon the results of this study. In this respect, different amputation levels such as transfemoral amputation should be considered to allow for a generalization of the above-mentioned results. Moreover, inertial sensor units positioned in sewn pockets on the study garments might not be rigid with the chosen anatomical districts, thus introducing possible errors. In this respect, a compromise should be reached between this need and the clinical practice which suggests to reduce patient time and discomfort. In the context of this work, this error is considered negligible due to the low accelerations associated with level walking. As for the use of the 10-m walking test, it is a well-reliable and validated clinical test on functional mobility for people with impairment, but a limited number of steps were analysed over a relatively short walking distance. Future analysis in this respect could imply longer pathways and, eventually, the analysis of walking in outdoor environments.
In summary, this is the first study proposing a multi-sensor approach to characterize upper body accelerations during gait in persons with lower-limb amputation. The adopted biomechanical parameters could represent a useful tool for both clinicians (physicians, physiotherapists, etc.) and technicians (prosthetists) involved in the amputee rehabilitation. Indices like the attenuation coefficients could be validated as functional indices to assess the outcomes of different therapeutic rehabilitation interventions. This approach could also be useful to test the efficacy of prosthesis components as ‘shock absorbers’ (i.e. carbon fibre feet, type of suspension system) directly affecting accelerations experienced during foot strike, which propagate through the upper body. In this respect, future research could investigate how the proposed indices change with the adoption of different prosthetic components.
Conclusion
The present study focused on the assessment of accelerations at different upper body levels during gait, and of their attenuation upwards, in persons with transtibial amputation using wearable inertial sensors. In this respect, the potential role of the upper body in compensating lower-limb asymmetry was highlighted, specifically involving decreasing/increasing accelerations at the different body levels in the transverse plane. Interestingly, the attenuation coefficients may represent a useful synthetic index for the objective assessment of the mobility of persons with amputations, when associated with the traditional clinical approach. Overall, the adopted biomechanical parameters were successful in highlighting gait differences between individuals with amputations and able-bodied persons. These results can therefore be used to support therapists and physicians in the design of innovative intervention protocols or in prescribing prosthetic devices, and to monitor their efficacy in terms of gait stability. This process could be greatly assisted by increased availability and accessibility of inertial measurement equipment for home-based monitoring applications.
Footnotes
Author contribution
Francesco Paradisi and Giuseppe Vannozzi contributed to: study conception and design; data acquisition; data analysis and interpretation; statistics; manuscript drafting and revision. Eugenio Di Stanislao contributed to: data acquisition; data analysis and interpretation; manuscript critical revision. Aurora Summa contributed to: data analysis and interpretation; statistics; manuscript critical revision. Stefano Brunelli and Marco Traballesi contributed to: data interpretation; manuscript critical revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Italian Ministry of Economic Development (grant/award number: ‘ICE-CRUI agreement, SIVAM project 4/2010’).
