Abstract
Background:
Thermal discomfort among lower-limb prosthesis wearers is prevalent with social and medical consequences.
Objectives:
This study aimed to verify the feasibility of out-of-laboratory thermal comfort studies.
Study design:
Repeated measures pilot study.
Methods:
Thermistors were placed on participants’ residual limbs during two experimental phases. In phase 1, mean limb temperature was calculated over a controlled 55-min rest-exercise-rest protocol. In phase 2, participants conducted activities of their choosing wherever they wanted away from the lab, while limb temperature data were collected. Descriptive statistics and statistical differences between phases are presented.
Results:
Five male amputees participated with an average age ±standard deviation of 30 ± 9 years. In phase 1, mean limb temperature change ranged between 1.6°C and 3.7°C. In phase 2, mean limb temperature change ranged between 1.8°C and 5.1°C. Limb temperature was significantly higher in out-of-lab studies (+1.9°C, p = 0.043) compared to in-lab studies.
Conclusion:
Independent multiple-hour temperature studies are shown to be feasible. Results also indicate that out-of-lab residual limb temperature can be significantly higher than in-lab temperatures.
Clinical relevance
Thermal discomfort and sweating may lead to skin conditions and reduce quality of life among prosthesis wearers. Out-of-lab, long-term temperature studies are needed to comprehensively characterize thermal discomfort to create preventive solutions.
Background
More than 50% of prosthesis wearers will suffer from overheating, excess sweating and thermal discomfort due to their prosthesis. 1 Amputees are susceptible to overheating and hyperhidrosis as thermoregulatory responses are less efficacious due to reduced body surface area. 2 Thermoregulation is also inhibited by prosthetic devices, as sockets and liners retain heat and sweat due to their low thermal conductivity2–4 and impermeability. 5 Ultimately, this creates a hot and humid skin–prosthesis interface6–11 which may reduce prosthesis wear 12 and as the interface is an ideal environment for bacteria, damaged skin may rapidly become infected.8,13,14 Skin damage can take up to 177.6 ± 113 days to heal, 15 during which amputees may not be able to wear their prosthesis until the skin has fully recovered. 16 Bacteria can also cause unpleasant odours, which can lead to increased social anxiety and isolation. 17
Previous prosthesis thermal comfort studies have been predominantly exploratory in nature and collected residual limb temperature data from male transtibial amputees in a laboratory setting.6,7,18–20 All prior works have consisted of a ‘rest-exercise-rest’ protocol, with rest ranging from 15 to 60 min and treadmill ambulation ranging from 10 to 30 min. Only one study explicitly controlled for environmental temperature, conducting testing at ambient temperatures of 10°C, 15°C, 20°C and 25°C. 19 Reported changes in mean residual limb temperature between the beginning and end of experiments have ranged from 1.7°C to 3.1°C.6,7,19,20 These studies present small datasets collected from one participant,7,19 five participants 6 or up to nine participants. 20 The findings show that wearing a prosthesis and physical activity increases mean residual skin temperature. A study by Segal and Klute 21 progressed the literature by conducting an out-of-lab study in a cold environment while doing physical exercise (snow-shoeing). Participants rested for 5 min, exercised for 30 min and rested for 5 min. A further exercise and rest period followed. Mean residual skin temperature increased by 3.9°C between the start and end of the experiment. Segal and Klute also manually recorded perceived thermal comfort (PTC), ultimately finding that PTC was significantly associated with activity and rest. Their study is a commendable contribution to prosthesis thermal comfort; however, the environment and exercise studied are unlikely to reflect everyday activities for individuals living in more temperate climates.
In light of the literature, we present a study which demonstrates a multi-hour research protocol conducted in a real-world environment, with full participant autonomy. We hypothesized that realism and participant freedom would uncover novel residual limb skin temperature behaviour as a result of daily prosthesis wear. We highlight the importance of out-of-lab studies by conducting a controlled laboratory study and comparing data with out-of-lab data. Experiments were conducted in Japan during the month of August, where ambient temperatures were on average 30°C ± 3°C. 22 Transfemoral amputees were predominantly recruited to provide insights from a previously untested participant group. 23 The combination of autonomous sensing, a real-world scenario and a rarely studied participant population mean that this study progresses lower-limb amputee thermal discomfort research. As research moves towards real-world, long-term studies, prosthetic devices that are thermally comfortable in a multitude of environmental and situational contexts becomes a realistic proposition.
Methods
Participants
Ethical approval was obtained from the ethics boards of University College London and the National Rehabilitation Center for Persons with Disabilities, Tokorozawa, Japan. A convenience sampling approach was adopted to recruit male lower-limb amputees who wore a prosthesis daily. As the research team was all males, females were not recruited to reduce potential embarrassment while participants were in a state of undress when sensors were applied. Diabetic amputees were not recruited due to perceived additional skin-damage risks.
Protocol
Phase 1
The study comprised two phases. The first phase obtained a standardized dataset for each participant and was conducted in a controlled air-conditioned laboratory environment. Participants were asked to remove their prosthesis and 12 thermistors were attached to the residuum using medical tape. Thermistors were placed within the lower, middle and top third of the residual limb, excluding the knee joint in transtibial amputees (Figure 1). Multiple thermistors provided insurance against thermistor failure. Thermistors were placed on fleshy parts of the residuum, avoiding bony prominences. Sensor attachment typically took 5 min and at most 10 min.
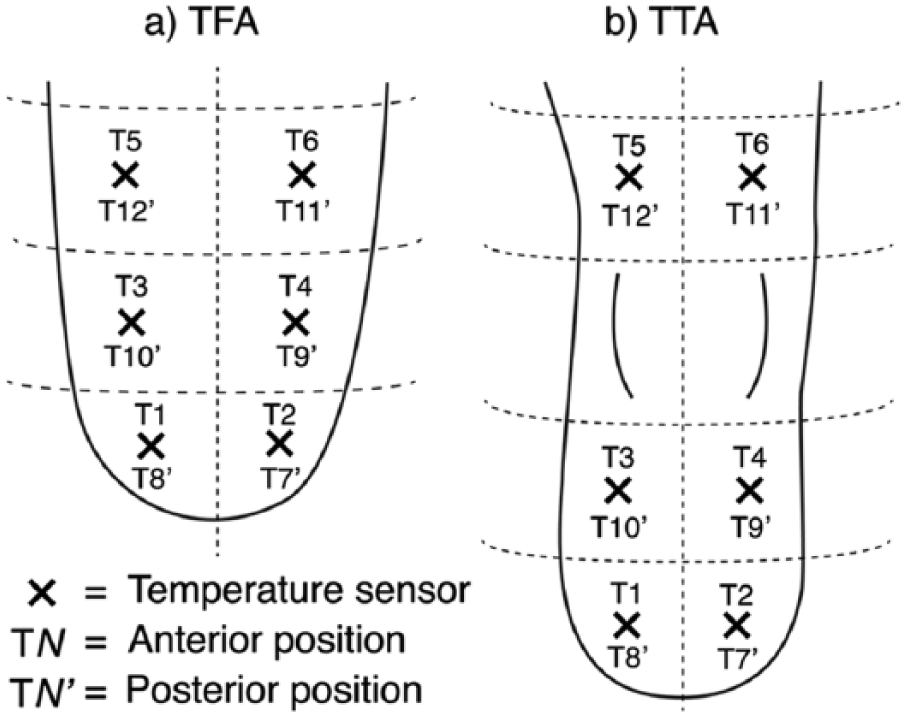
Temperature sensors were placed in similar positions as shown in the above diagram on both the anterior and posterior of the participants’ (a) transfemoral or (b) transtibial residual limb. For the TTA participant, sensors were not applied to the knee area to maintain mobility of the joint.
After attaching the sensors, participants donned their prosthesis and took a few supervised steps around the laboratory. Discomfort, pain and reductions in suspension were communicated and if present, the offending thermistor was repositioned. This process was repeated until no issues were present. The data logging system was then initiated. Participants were asked to sit and rest for 15 min, which allowed participants to relax and reduced limb heating effects caused by movement during thermistor application. Participants were then asked to walk for 10 min at a self-selected pace on a flat treadmill. Finally, participants were asked to sit for a further 30 min.
Phase 2
On a different day to phase 1, participants returned to the laboratory between 9 am and 2 pm to begin phase 2. The same sensor application procedure was used as in phase 1. Once sensors were applied, the sensor system was turned on and participants could leave the laboratory and return when convenient. No contact was made with the participants until they returned to the lab and participants could conduct activities of their choosing. However, researchers were contactable if needed and it was made clear that sensors should be removed if they caused pain or discomfort. This autonomous phase empowered participants to take part in the experiment while going about natural activities in real-world environments without interruption.
Temperature sensing system
To collect temperature data, an Arduino MEGA 2560 microcontroller (Arduino, Italy) was connected to a DS1307 real-time clock (Maxim Integrated, USA) and an SD card reader. A total of 16 B57863S103F40 NTC thermistors (Epcos, Germany) were connected to a linear potential divider (10 kΩ ± 1% tolerance resistors) and the system was powered by 6 AA 1.5 V batteries. New batteries were used for each study. The microcontroller was programmed using Arduino’s integrated development environment (IDE) to acquire and save data at 1 Hz. Thermistors were calibrated using the Steinhart–Hart equation and provided an absolute measurement accuracy of ±0.2°C in a range between 0°C and 70°C. The data logger was placed into a waist bag and cables were routed under clothing so as to not be visible. Four thermistors positioned on the outside of the waist bag recorded ambient temperature.
Statistical analysis
MATLAB 2016a (Mathworks, USA) was used for analysis. Mean limb temperature over all thermistor locations provided a descriptive summary of residuum temperature behaviour. If a thermistor broke during the experiment, data from that thermistor location were excluded from mean limb calculations. In phase 1, switching between segments took no more than 30 s for all participants. To standardize timings, intermediate switching data were removed. During both phases, residual limb temperature averages of the first and last 10 s of data were calculated to represent start and end temperatures. Finally, a Wilcoxon signed-rank test was implemented to assess differences between average limb temperatures in phase 1 and average limb temperatures in phase 2, with a two-tailed p-value being reported and 95% confidence interval (CI), with significance determined when p < 0.05.
Results
In total, five male participants were recruited, with an average age ±standard deviation of 30 ± 9 years. The time since amputation varied from 11 months to 37 years (congenital amputee). Table 1 describes the basic participant characteristics. P2 used a walking aid and sensors were only applied to his transfemoral residual limb.
Demographic and amputation information of the five participants who were recruited.
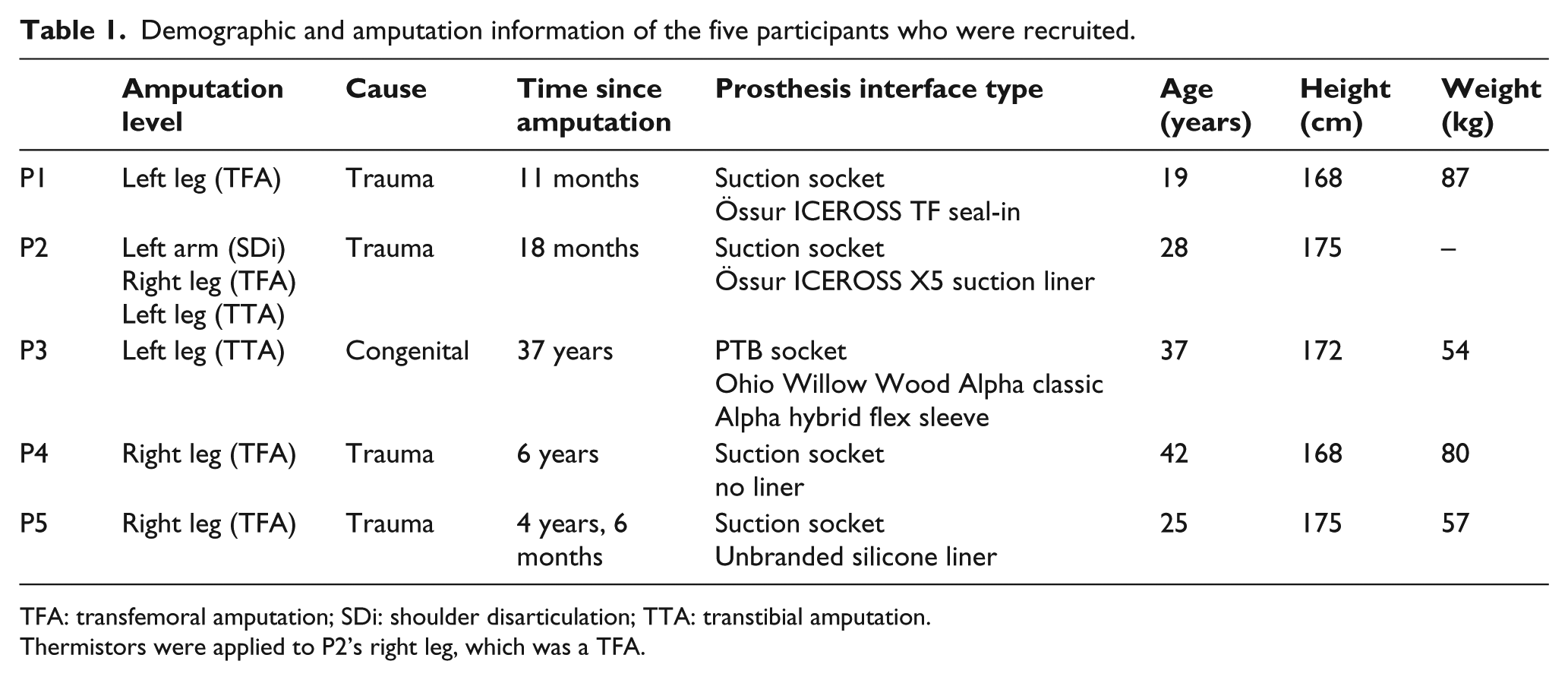
TFA: transfemoral amputation; SDi: shoulder disarticulation; TTA: transtibial amputation.
Thermistors were applied to P2’s right leg, which was a TFA.
Segment-by-segment limb temperature changes from phase 1 data are presented in Table 2 and limb temperature data are presented as a time series in Figure 2. Some thermistor wires broke during the experiments (Table 2). The mean laboratory temperature was 26.7°C.
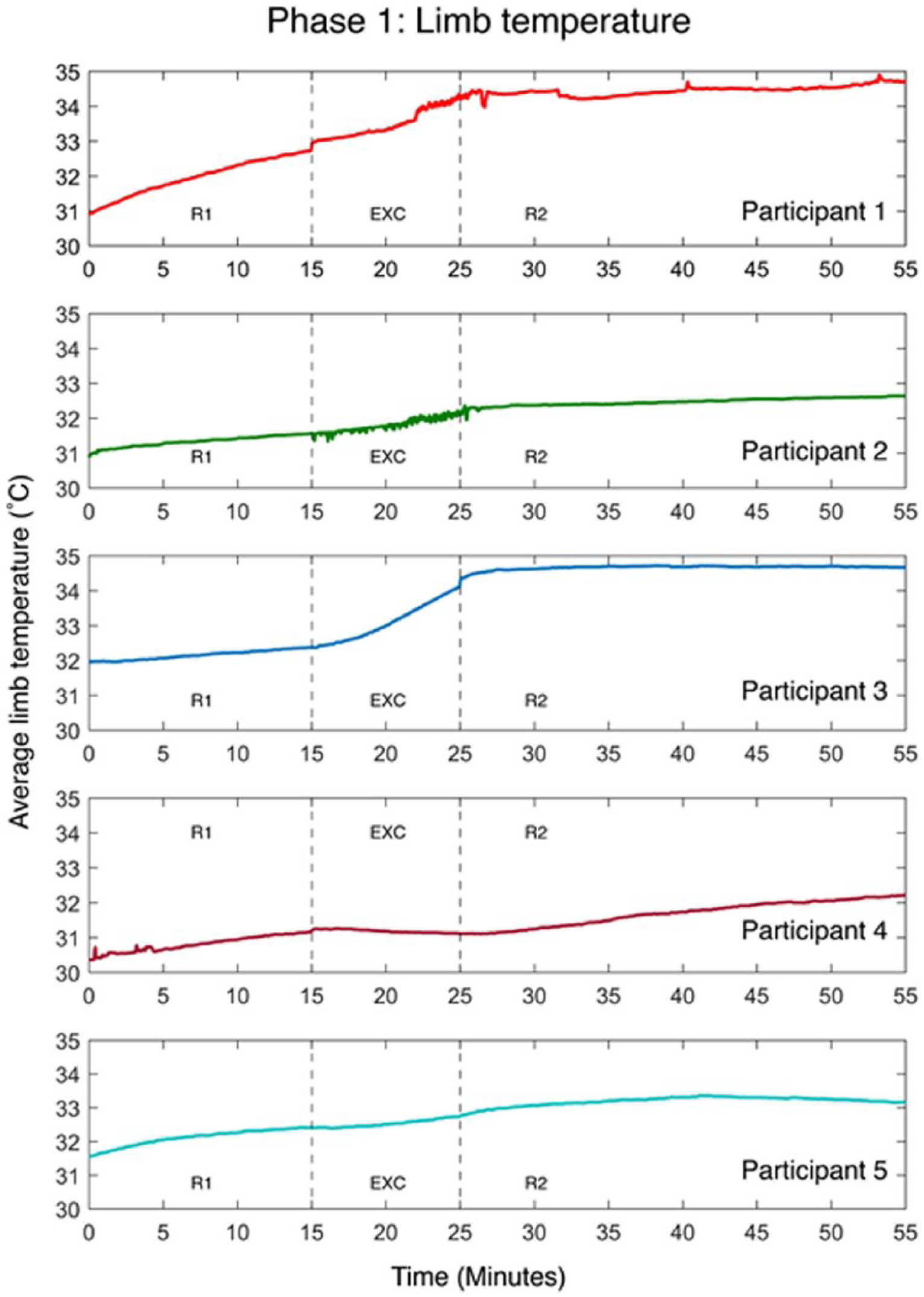
Residual limb temperature for each participant during phase 1 is shown as a time series. Each section of the protocol is labelled as rest 1 (R1), exercise (EXC) and rest 2 (R2).
Mean temperature data from the different parts of the first experimental phase are shown here ±standard deviation for all participants.
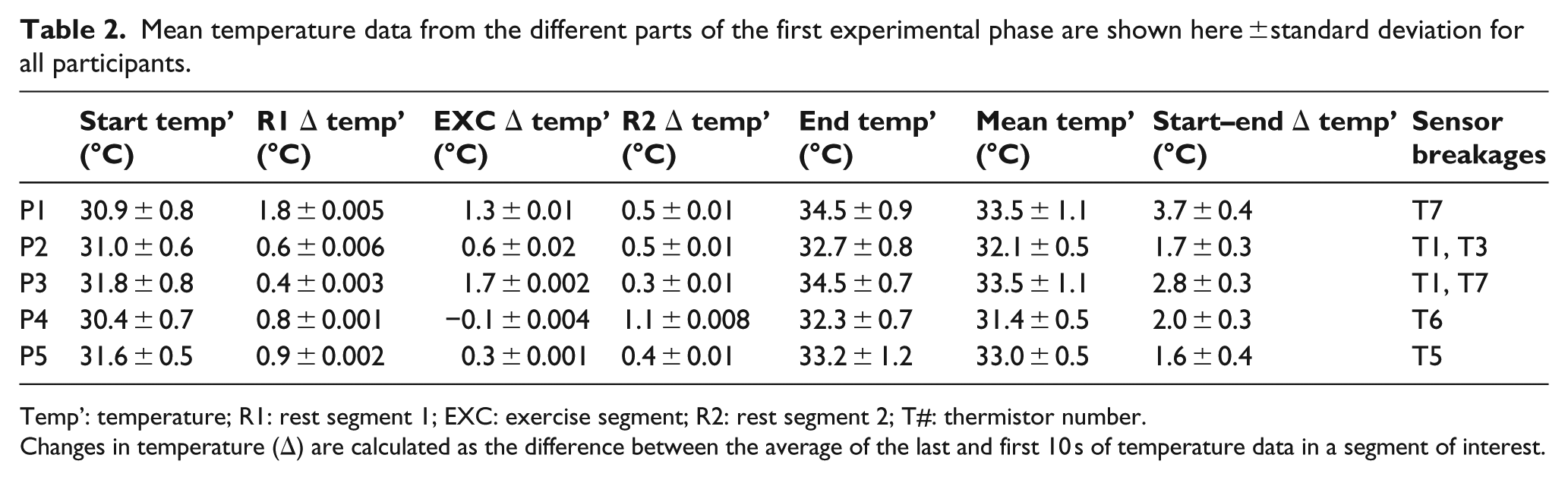
Temp’: temperature; R1: rest segment 1; EXC: exercise segment; R2: rest segment 2; T#: thermistor number.
Changes in temperature (Δ) are calculated as the difference between the average of the last and first 10 s of temperature data in a segment of interest.
Data from phase 2 are summarized in Table 3. The smallest change in limb temperature from the start to end of the experiment was P4, with a change of only 1.8°C and P5 presented the largest change in limb temperature at 5.1°C. Again, some thermistor wires broke during the experiments (Table 3).
Residual limb temperature and ambient temperature data were recorded for each participant during phase 2 of the experiment.
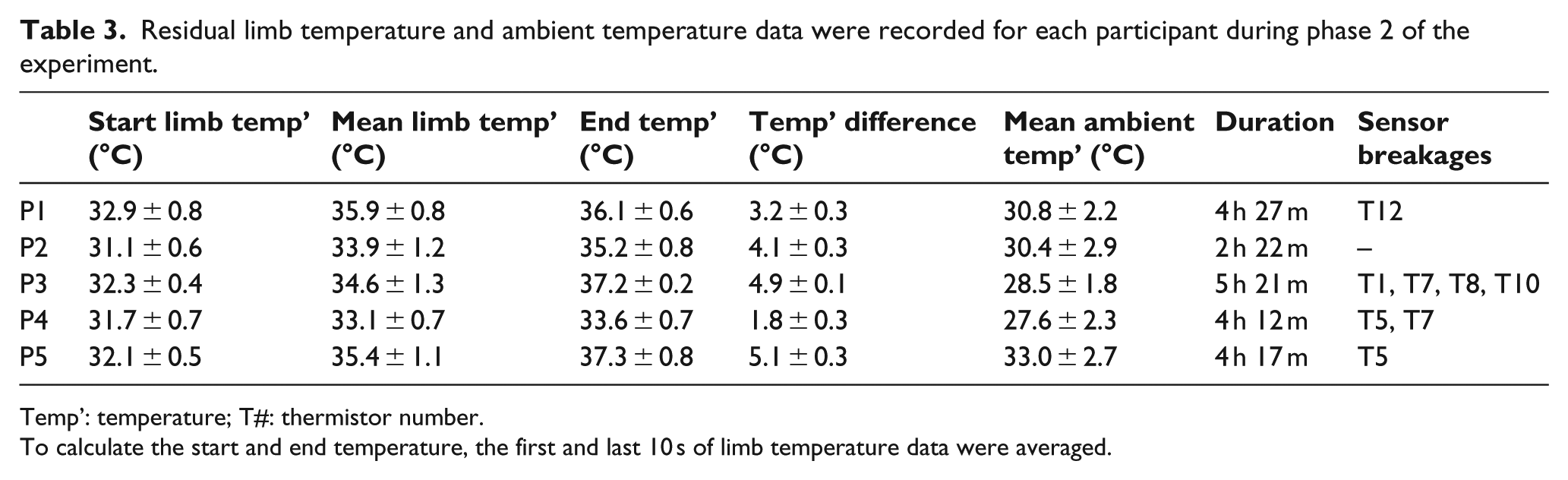
Temp’: temperature; T#: thermistor number.
To calculate the start and end temperature, the first and last 10 s of limb temperature data were averaged.
To better understand the data corpus, temperature distributions of limb temperature during phases 1 and 2, as well as ambient temperature during phase 2, were plotted as a boxplot (Figure 3). A Wilcoxon signed-rank test revealed that experiments conducted away from the lab yielded significantly hotter mean residual limb temperatures than in-lab tests (Z = –2.023; p = 0.043), with an increase of 1.9°C (95% CI = 1.2°C–2.6°C).
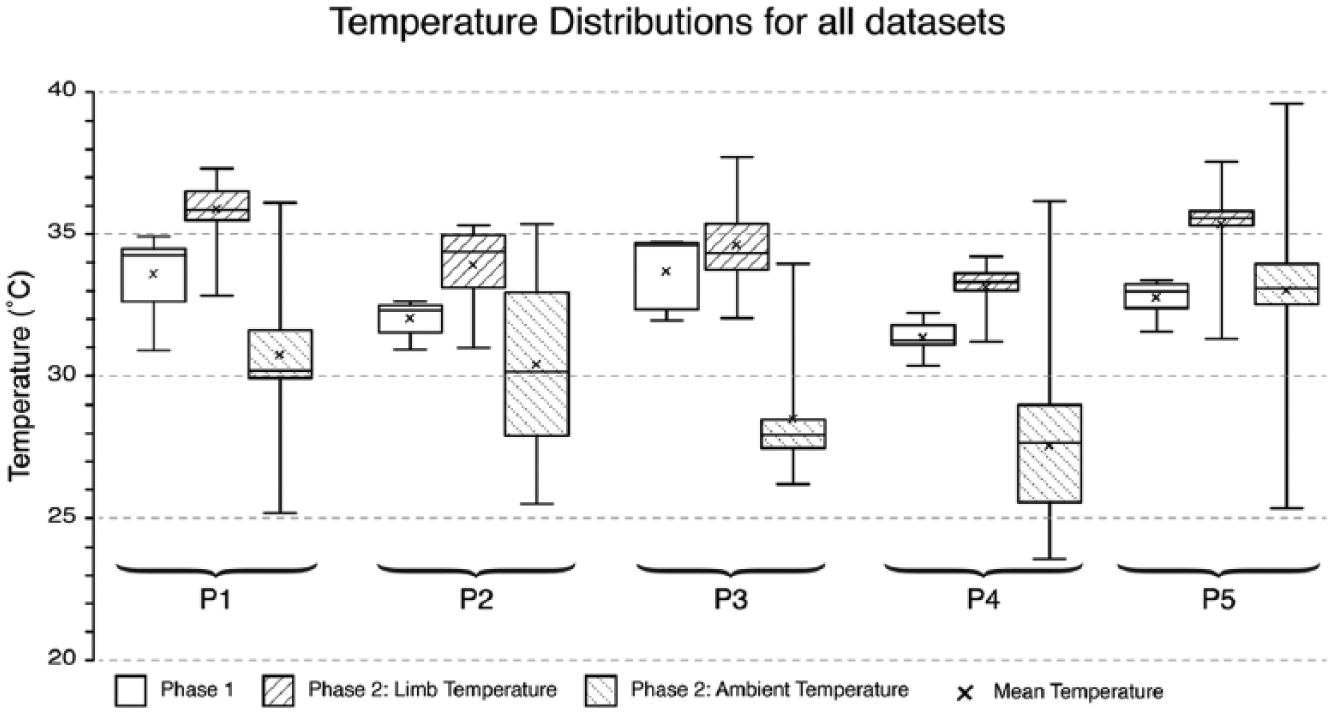
Residual limb temperature data from phases 1 and 2, as well as the ambient temperature data from phase 2 are displayed as a boxplot distribution for all participant datasets.
Discussion
Heat and sweat discomfort in lower-limb prosthesis wearers is a prevalent and serious problem that must be addressed and although some solutions have been created,24–27 their efficacy in the real world remains untested,26,27 or they require further development.24–26 Thermal discomfort studies have previously reported temperature changes seen in transtibial prosthesis wearers in laboratory settings, while performing controlled activities. A notable exception was the study by Segal and Klute, who investigated residual limb temperature in a cold non-laboratory environment. 21
Our study collected data in a laboratory-based research scenario and an out-of-lab scenario and proves that it is possible to conduct multiple hour autonomous studies away from the lab and generate unique insights. In-lab data were collected over a 55-min research protocol and out-of-laboratory data were collected between 2 h, 22 min and 5 h, 21 min dependent on participant schedules.
When reviewing phase 1 data, increases in residuum temperature are notable after donning the prosthesis – an observation first reported by Peery. 6 During the first rest phase, limb temperatures of all participants appear to steadily increase. It is likely that limb temperature would continue increasing up to a steady-state plateau if a long-enough rest period was implemented. Most participants also displayed an increase in temperature during the exercise phase – something that was clearly demonstrated in P1 and P3. P4 did not display a clear change in limb temperature, which could be due to not wearing a liner. In future work, it may be interesting to explore if sustained rest while wearing a prosthesis incites similar elevated skin temperature levels as periods of exercise. In the final rest phase, all participants demonstrated a further increase in limb temperature with P1, P2, P3 and P5 approaching a plateau. Only P4 appeared as though their limb would continue increasing in temperature if the rest period continued. None of the participants’ residual limbs returned to pre-exercise temperatures which demonstrates that heat is retained at the prosthesis–skin interface. Previous studies observed residual limb temperature changes ranging from 1.7°C to 3.1°C in controlled laboratory settings;6,7,20 however here, in similar conditions, the largest temperature change was 3.7°C (P1).
The box plot diagram shown in Figure 3 shows that all participant’s residual limbs were, on average, hotter in the out-of-lab phase than in the in-lab phase. This is confirmed by a significant increase of 1.9°C (95% CI = 1.2°C–2.6°C; p = 0.043) between participants’ mean limb temperature in phase 1 and mean limb temperature in phase 2. Differences in ambient temperatures between phases make this a relatively unsurprising finding; however, it subtly signifies the value of out-of-lab studies which have natural variety to prosthetic thermal comfort research. In addition, during phase 2, P5’s residual limb changed temperature the most, at 5.1°C. This appears higher than increases found by Segal and Klute, 21 who found a mean residual limb temperature change of 3.9°C. However, as only the mean residual limb temperature changes between subjects were reported and not the range, it is difficult to compare.
The data from our study will help inform the development of comfortable prostheses. However, the small participant size (n = 5) means that generalizations about the wider amputee population cannot be made. It is important to highlight that our participants were also relatively active and therefore, our findings may not translate to lower activity prosthesis wearers. One additional limitation inherent to most prosthetics research is that prosthesis prescription was not consistent between participants, as recruitment would have been prohibitively difficult. However, prescription differences do not reduce the validity of the presented findings.
Finally, we contribute a number of practical considerations. Although our study suggests differences in mean residual limb temperature when comparing in-lab and out-of-lab environments, in-lab testing still has a place in future research. However, to progress prosthesis thermal comfort research, it may be beneficial to also consider simulating indoor activities such as task-based ambulation or leisure activities, rather than exclusively implementing treadmill ambulation. This will situate our understanding firmly in the real-world lived experience of prosthesis wearers. When designing out-of-laboratory protocols, researchers must acknowledge reductions in control. For example, participant autonomy means that researchers should not expect to control participants’ activities, nor the environments that they encounter. Out-of-lab protocols, therefore, lend themselves to exploratory research, such as the study presented here. To account for reduced control, environmental sensors and activity monitors can provide useful additional data for analysis. Although the latter sensors were not used in this study, they would have added greater depth to the analysis. The impact of variety of activities and environmental contexts may also be mitigated by collecting data from participants at scale. For example, if experiments lasted multiple days, similar participants will likely experience comparable situations.
Long-term experiments still pose significant practical challenges. Although our study aimed to be minimally invasive and provided participants with more freedom than previous studies, sensor application is laborious and requires assistance. Thermistors are also prone to breakages, as demonstrated here and in other studies.6,21 It is unclear how previous studies have handled thermistor breakages, however, we excluded broken thermistor data from mean limb temperature calculations. As residual limb temperature varies over the residuum surface, 6 this could introduce positive or negative bias to mean limb temperature calculations. However, in the majority of our experiments, no more than two thermistors broke, therefore, the consequences to mean limb temperature are likely minimal. In the case of PT3’s phase 2 data, one anterior and three posterior thermistors broke. Therefore, the presented mean limb temperature may be lower than a mean limb value calculated if no thermistors had broken, as TTA posterior limb surfaces have been found to be warmer than anterior surfaces. 6 Multi-day or multi-week experiments, therefore, need robust temperature sensors that can be independently applied and removed by participants. Data collection systems also need to have high battery and data storage capacities for sustained data collection and recording. Small participant sizes also appear to be a common limitation in prosthetic thermal comfort research and thus the community should explore ways to safely lower participation barriers. One possible solution would be to devise ways for participants to independently apply sensors and to explore data redundancy 28 between sensing sites to make application simpler. If these challenges can be overcome, long-term remote studies may become a feasible and safe reality.
Conclusion
Our study shows that it is possible to conduct autonomous out-of-lab temperature investigations among lower-limb prosthesis wearers for multiple hours. In both experimental phases, residual limb temperature did not return to pre-prosthesis donning temperatures and residual limb temperature changes across both phases varied from 1.6°C to 5.1°C. This appears higher than previous studies and raises the specification for future technologies that hope to prevent lower-limb prosthetic thermal discomfort. Thermal comfort research in real-world contexts may also provide valuable novel insights, as indicated by significant increases in mean limb temperature in real-world environments, compared to lab environments.
Footnotes
Acknowledgements
The authors extend sincere thanks to the Japan Society for the Promotion of Science, who generously funded this research project as part of a 10-week summer fellowship.
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Engineering and Physical Sciences Research Council (grant/award no.: ‘Doctoral Training Grants’), Japan Society for the Promotion of Science (grant/award no.: ‘Summer Fellowship’) and UCL Engineering, University College London (grant/award no.: ‘Doctoral Training Programme in Medical Device Innovation’).
