Abstract
Background and aim:
Thermal discomfort often affects prosthesis wearers and could be addressed by increasing liner thermal conductivity. This note explores a liner made from thermally conductive silicone and two additional alternative liner designs.
Technique:
Thermally conductive silicone was used to create a conductive liner and a hybrid liner. Additionally, one with open elements was made. These were compared with a plain silicone liner and a no liner scenario. Scaled down liner prototypes were used due to the high-cost of the thermally conductive silicone. Temperature decay profiles were collected by attaching thermistors to a heated liner phantom and used to evaluate scenarios.
Discussion:
No scenario performed much better than the plain silicone liner. Implementation of passive solutions may be easier, but alternative liner materials are unlikely to affect dissipation enough to address thermal discomfort. Based on this work, future research efforts may be better spent developing active thermal discomfort solutions.
Clinical relevance
Thermal discomfort can increase the probability of skin damage, reduce prosthesis satisfaction and, ultimately, the quality of life. The prosthesis-wearing experience could be improved if thermal discomfort can be addressed by technological improvements.
Keywords
Background and aim
Lower limb prosthesis wearers often report a moderate level of satisfaction with their artificial limbs; listing excess sweating, foul odors, and sounds as their top 3 annoyances. 1 Thermal discomfort is highly prevalent among prosthesis wearers, affecting more than 53% of all amputees. 2 In lower limb prosthesis wearers, even light exercise causes an increase in temperature of the skin–prosthesis interface3–7 that dissipates away slowly.3,5,7 Prosthesis impermeability means sweat cannot evaporate, and when this hot and sweaty interface is subjected to ambulation forces, skin damage can rapidly occur.6,8–11
Components that aim to minimize or delay heat and sweat discomfort are emerging,12,13 but more studies are required to comprehensively determine their efficacy.14,15 Klute et al. 16 suggested that increasing the thermal conductivity of interface components could improve heat transport and suggested that the liner material has a greater effect on skin temperature, in comparison to the socket component as it is thinner.
This note explores potential liner design solutions in response to lower limb thermal discomfort. To evaluate potential designs, an easy-to-implement experimental method is described. Most importantly, by investigating a liner material with a higher thermal conductivity, this article progresses prosthetic thermal discomfort research.
Technique
Thermally conductive and plain silicone were used to create liner scenarios called the mini thermal liner (MTL) which was made using thermally conductive silicone, the mini open liner (MOL) which was made using plain silicone, and a mini hybrid liner (MHL) which was made using both plain and thermal silicone (Figure 1(a)). These were compared to a mini ‘plain’ silicone liner (MPL) and a no-liner scenario. Commercially available elastomeric materials with thermal conductivities above 0.266 W/m°C, and Shore 00 hardness between 0 and 60 were sourced (Table 1). Existing liners have conductivities lower than this 16 and similar hardness. Silcotherm materials (ACC Silicones, UK) were the only candidates near these criteria, with SE2010 meeting them exactly. Only liquid samples of SE2010 could be sourced at a cost of nearly £70/50 mL. The high cost meant that mini liner designs were preferred over full-sized prototypes, as this was not believed to significantly alter the underlying thermodynamics. The prototypes were designed to be a simplified anatomical shape, to enable a realistic liner donning procedure. Prototypes were digitally designed, three-dimensional (3D) printed, and cast in the appropriate material (Table 1). SE2010 was difficult to mold, which meant that the MTL required minor surface repairs that added up to 1 mm to the thickness in those locations (Figure 1(a)).
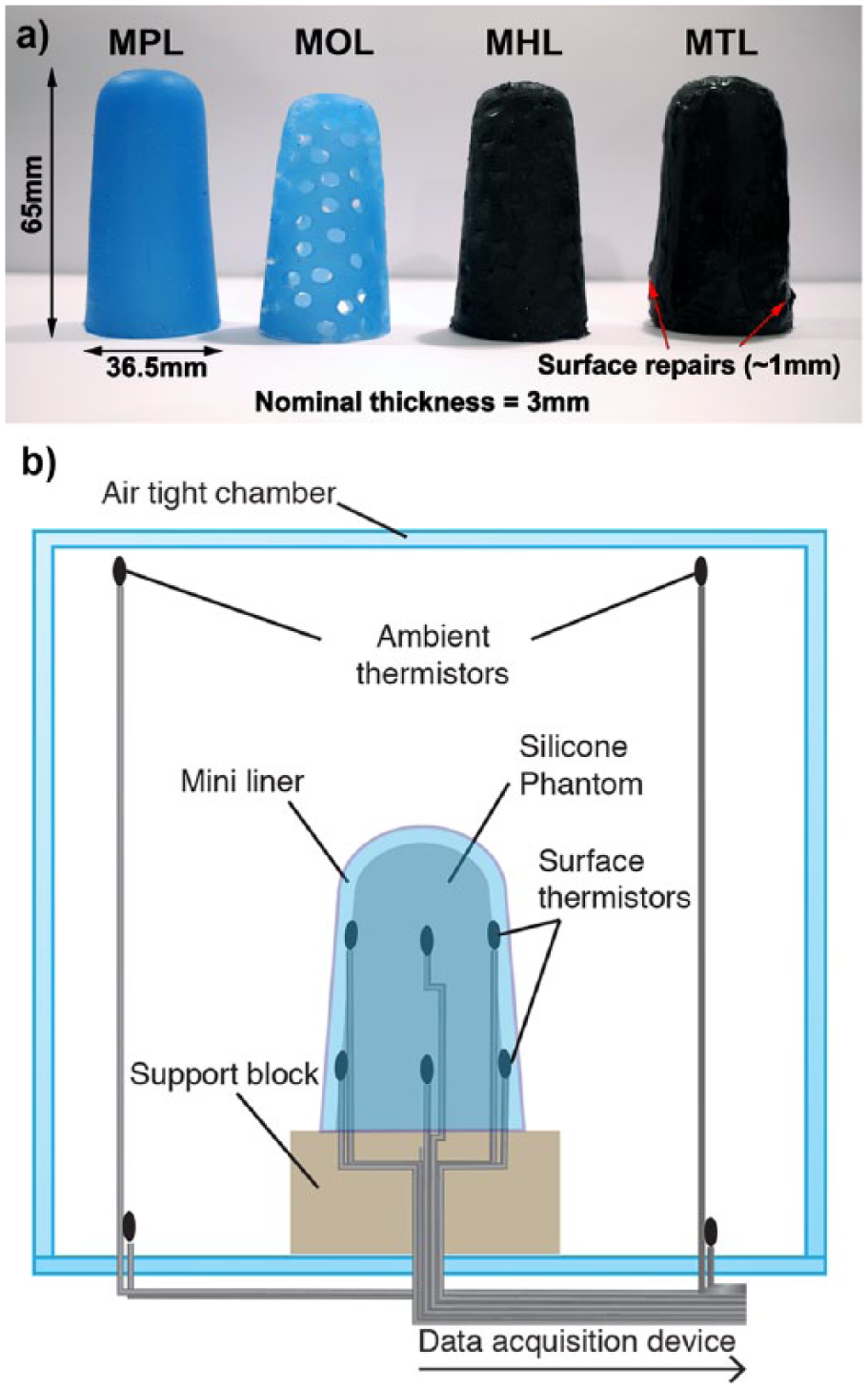
(a) Scaled-down liners were used to test the mini plain liner (MPL) made from standard silicone, mini open liner (MOL) featuring 30% surface perforations, mini hybrid liner (MHL) featuring 30% conductive silicone and 70% plain silicone, and mini thermal liner (MTL) which was 100% thermally conductive silicone and (b) a silicone tissue phantom was instrumented with thermistors and sealed in an acrylic box during experiments for each liner scenario.
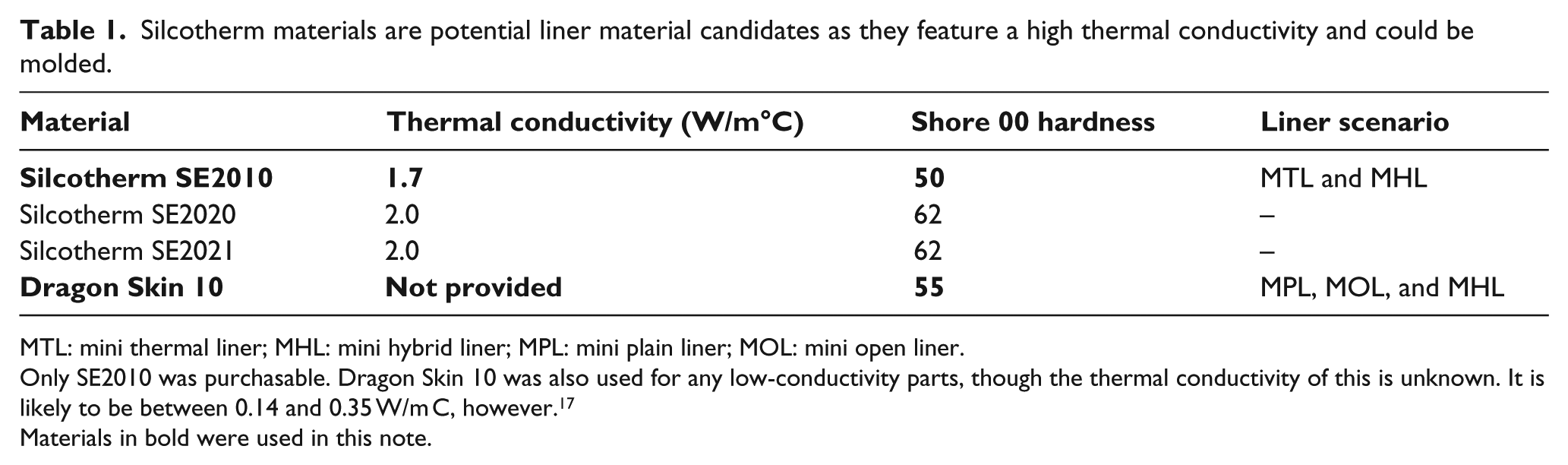
Silcotherm materials are potential liner material candidates as they feature a high thermal conductivity and could be molded.
MTL: mini thermal liner; MHL: mini hybrid liner; MPL: mini plain liner; MOL: mini open liner.
Only SE2010 was purchasable. Dragon Skin 10 was also used for any low-conductivity parts, though the thermal conductivity of this is unknown. It is likely to be between 0.14 and 0.35 W/m C, however. 17
Materials in bold were used in this note.
Due to the early stage of this project, a controlled silicone limb phantom was used to test the effect of each liner scenario on heat decay, rather than an amputee’s residual limb. This removed physiologically related temperature variations 16 which would have been difficult to control. This ensured a consistent base thermal decay profile for each experiment. Microwave heating was used as it quickly heated the phantom volume, not just the surface, and has been used previously to heat limb phantoms. 18 The homogeneity of the silicone phantom meant surface temperature was approximately the same over the entire surface. This differs from residual limbs, which have locational temperature differences due to anatomical features. 4 This extra layer of control meant that the average of surface temperature data could be used in the analysis. Data were collected with an Arduino Mega 2560 (Arduino, Italy), as it supported 16 analog inputs, which were connected to sixteen 10-kΩ B57863S103F40 negative temperature coefficient (NTC) thermistors (Epcos, Germany). The Arduino was interfaced and programmed using LabVIEW 2015 (National Instruments, USA) and the LabVIEW LINX interface. Data were acquired at 2 Hz and stored on a connected laptop. This acquisition rate is above the thermoregulatory response time, which is an order of multiple seconds 19 and lies within the range of other prosthesis temperature studies (0.125–4 Hz3,5,6). Thermistors were each connected to a standard bridge circuit, supplied by a 5-V direct current (DC) laboratory power supply and calibrated using the Steinhart–Hart equation, resulting in an accuracy of ± 0.2°C between 0°C and 70°C. Once collected, data were analyzed using MATLAB 2016a (Mathworks, USA).
The phantom was irradiated for 45 s in a category D 700-W microwave and eight thermistors were attached using Kapton tape (DuPont USA) and evenly spaced around the perimeter of the phantom, with four on the upper and lower halves respectively (Figure 1(b)). Room temperature liners were rolled over the thermistors and phantom, and then placed into an acrylic box (Figure 1(b)), and data were collected for 35 min. The time from removing the phantom, post-heating, to donning the liners was under 30 s. Thermal grease was not applied to maintain similarity with the natural prosthesis interface. Eight thermistors recorded ambient conditions inside the box, and each scenario was repeated seven times to enable easy recognition of anomalous data, though the number of repetitions is arbitrary. After microwaving, the phantom was much hotter (>50°C) than skin. Thus, when the phantom registered 33.0°C ± 0.1°C, the time was recoded as t = 0, to represent the highest temperature found post-exercise for transtibial amputees, in three studies3,5,6 to the nearest integer. The average of phantom surface and ambient data was calculated (one ambient thermistor broke and was excluded). Despite the airtight chamber, some coupling existed between ambient and phantom surface data.
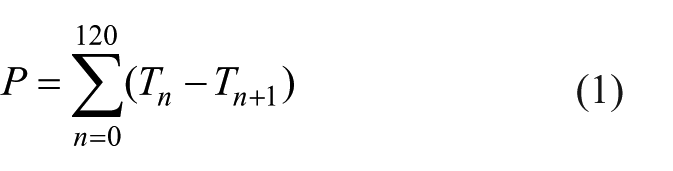
To remove this coupling, the difference between surface and ambient temperature data was calculated. Equation (1) was used to find the surface temperature decay, P, after a noise reducing 60-s moving average filter was applied (filter window = 120 samples at a collection rate of 2 Hz). This metric shows changes in phantom surface temperature in 1 min during the experiment.
Results and discussion
First-degree polynomial correlations were extracted from the data after applying equation (1). The combined data sets make it possible to see the effect of the liners on phantom surface temperature decay (Figure 2). The no-liner scenario demonstrated the greatest decay per minute, per °C of ambient-phantom temperature difference.
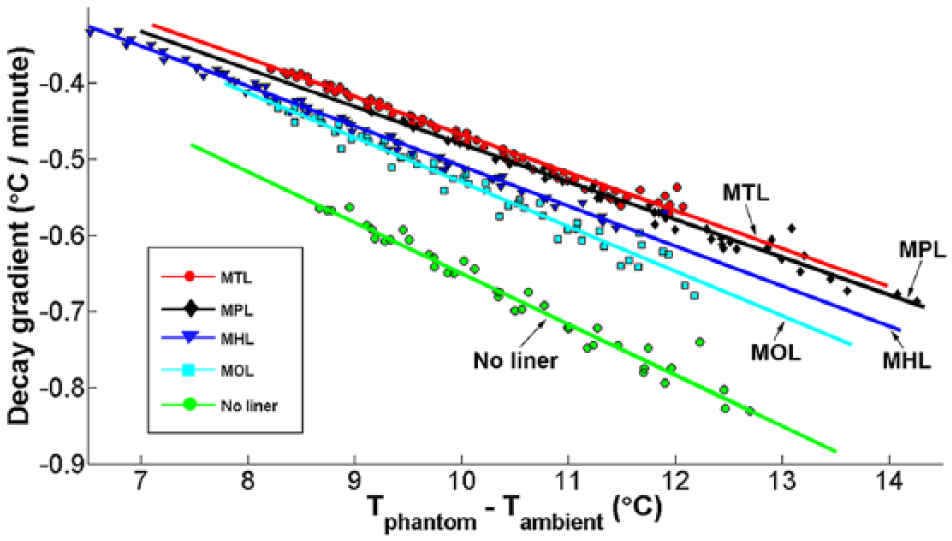
Data from all of the scenarios were plotted, and the gradient of each was subsequently extracted.
With ambient coupling removed, the MHL had superior dissipation performance over the MPL; however, only the MOL had a gradient close to the no-liner scenario. To contextualize the data in Table 2, if the MOL was used in an ambient temperature of 20°C, with a phantom surface temperature of 30°C, the phantom surface could decrease by 0.6°C/min. Open elements were the most effective at increasing heat decay; however, an open liner design with large open elements, such as those on the MOL, may reduce suspension and durability.20,21 Additionally, although there are liners on the market with small perforations, 13 large open elements may also introduce high shear stresses that could harm skin.
The dissipation gradient for each scenario’s correlation.
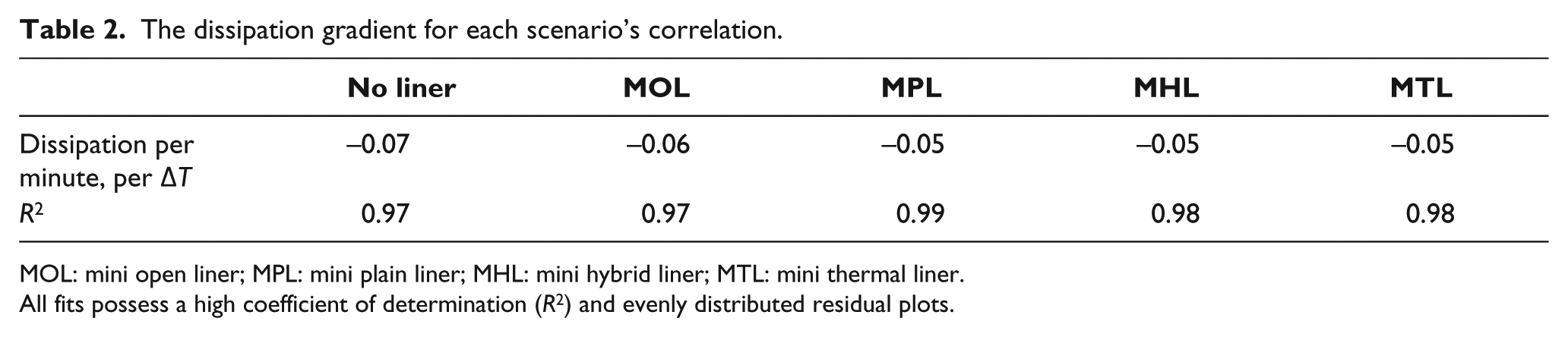
MOL: mini open liner; MPL: mini plain liner; MHL: mini hybrid liner; MTL: mini thermal liner.
All fits possess a high coefficient of determination (R2) and evenly distributed residual plots.
Increased thermal conductivity has been suggested as a way to improve heat dissipation in liners; 16 this study indicates that it may not effectively minimize or prevent thermal discomfort for lower limb prosthesis wearers. There are caveats to this conclusion: surface repairs of the MTL may have affected heat dissipation, but as these were minor and isolated, this is unlikely to significantly affect this finding. The applied technique also did not include a simulated socket layer which is an interface layer with a low thermal conductivity. However, they are thinner than liner components and will, therefore, have a smaller impact on heat dissipation in comparison to liners. 16 The thermodynamic properties of the phantom may also differ from human tissue, but it provided a consistent heat decay profile necessary for evaluation. Finally, as this experiment was informed by studies that only recruited transtibial amputees,3,5,6 an appropriate level of caution must be applied when extrapolating findings to other lower limb amputee populations. This highlights an important reminder that future thermal discomfort studies should actively seek to recruit participants with varying levels of amputations, including both unilateral and bilateral amputees to broaden understanding of the phenomenon. To conclude, this note suggests that in scaled-down liner scenarios, passive heat transport solutions are unlikely to improve heat decay at a simulated prosthesis interface. Unless the thermal conductivity of elastomers can be increased beyond the elastomers used here, or suspension liners are radically rethought, active solutions used in the study of Han et al. 22 and Ghoseiri et al. 23 may be a more promising avenue to mitigate and prevent thermal discomfort for prosthesis wearers.
Key points
Increasing thermal conductivity of liners was suggested by Klute et al. 16 as a potential solution to prosthesis heat and sweat discomfort.
The only viable commercially available material found had a thermal conductivity of 1.7 W/m°C and a Shore 00 hardness of 50.
Experiments revealed that of the four scaled-down liner scenarios proposed, only the open liner notably improved heat decay but may be impractical due to mechanical and durability issues.
Active cooling solutions may present a more promising research direction in the future, in favor of increasing the thermal conductivity of prosthetic components.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Rhys Williams is funded by the UCL Doctoral Training Programme in Medical Device Innovation. This programme is funded by UCL, EPSRC Doctoral Training Grants and the National Institute for Health Research, University College London Hospitals, Great Ormond Street Hospital and Moorfields Eye Hospital Biomedical Research Centres.
