Abstract
Background:
Research on adaptation to advanced upper limb prostheses is needed.
Objectives:
To (1) examine change in function, quality of life and community integration after prosthetic training, (2) determine whether change in outcomes varied by prosthesis complexity, and (3) compare patterns of change at 1 month for those who withdrew from the study and those who did not.
Study design:
Quasi-experimental time series.
Methods:
Data were analyzed for 22 participants (18 completers). Performance and self-report outcome measures were collected after in-laboratory training (Part A) and every 4 weeks of home use (Part B). Outcomes from End of A to End of B were compared statistically. Outcomes across assessments and by configuration level were compared graphically. Changes in scores were compared graphically for completers and non-completers.
Results:
Quality of life scores did not change between End of A and End of B, whereas scores improved for one activity measure, two measures of self-reported function, and three dexterity measures (p < 0.05). Outcomes of community integration, self-reported function, four dexterity measures, and one activity measure varied by prosthesis level. For participants who withdrew early, dexterity and activity scores worsened, perceived disability increased, and prosthesis satisfaction decreased after 4 weeks of home use.
Conclusion:
Study completers adapted to the DEKA Arm.
Clinical relevance
Findings suggest that for the majority of upper limb amputees discharged from prosthetic rehabilitation, function continues to improve with home use. However, a minority experience a decline in function, greater perceived disability, and greater dissatisfaction after 4 weeks, suggesting a need for continued therapy after intensive prosthetic training ends.
Background
Prosthetic training is recommended to maximize functional use of an upper limb prosthetic device. 1 Prosthetic training protocols for upper limb amputees have been largely published in textbooks, 2 a handful of peer-reviewed papers,3–5 or through websites of manufacturers. 6 Typical training programs last approximately 5 h for people with transradial (TR) amputation, 10 h for persons with transhumeral (TH) amputations, and 12–20 h for those with bilateral amputations. 2 Training programs are commonly delivered over the course of 3–5 weeks for persons with TR amputation 7 but may last longer. Evidence-based clinical practice guidelines suggest that upper limb amputees should have annual visits to their amputation care team and that prosthetic training should be provided whenever a new type of device is received. 1 However, these guidelines do not specifically recommend follow-up visits in the months after initial prosthetic training is complete. 1
After initial prosthetic training ends, we expect that many amputees continue to adapt to their devices, gaining proficiency and confidence in their use. However, we suspect that for amputees lacking a therapist’s oversight for coaching and troubleshooting, the proficiency of prosthesis use might decline. If this happens, it may result in decreased satisfaction with the device and contribute to eventual abandonment. Therefore, it is important to understand the process of adaptation to a prosthesis and to determine whether and how prosthetic outcomes change in the first few months following prosthetic training. This information may be useful in determining when and whether ongoing or follow-up prosthetic training is needed.
The purposes of this article are to (1) examine change in measures of function, quality of life, and community integration in the first few months after prosthetic training; (2) determine whether change in outcomes varied by complexity of the prosthesis; and (3) compare change in the first month of home use for participants who withdrew without completing all weeks of the home study and those who completed.
Methods
Study design
This multi-site study used a quasi-experimental time series design with repeated measures. Data were included from subjects who completed the VA Home Study of an Advanced Upper Limb Prosthesis (Home Study). Part A of the study involved in-laboratory training with the DEKA Arm. Part B of the study was trial of up to 3 months of home use of the DEKA Arm. The study was approved by the Institutional Review Boards of the Providence VA Medical Center, the VA NY HHS, the James A. Haley VA, and the Center for the Intrepid at Brooke Army Medical Center. All participants provided voluntary informed consent.
DEKA Arm
The Gen 3 DEKA Arm comes in three configurations: the shoulder configuration (SC) for persons with forequarter amputation, shoulder disarticulation, or very short TH amputation; the humeral configuration (HC) for persons with TH amputation; and the radial configuration (RC) for those with TR amputation. 8 All levels have six powered grip patterns and have powered wrist flexion/extension and pronation/supination. The HC and SC configuration levels also have powered elbow flexion/extension and humeral internal/external rotation (with an axis located just above the elbow joint). The SC has two degrees of powered shoulder movement degrees of freedom (flexion/extension and abduction/adduction). The SC device uses endpoint control to control most of the joints, rather than control of individual shoulder and elbow movements. 9 With endpoint control, the user controls movement of the endpoint of the prosthesis in space by moving it up/down, right/left, or forward/back. However, control of shoulder abduction/adduction movement, referred to as voluntary elbow positioning is controlled directly and is not included in endpoint control.
The HC and SC DEKA Arms have two operation modes: hand mode and arm mode. In hand mode, the controls are used to operate hand and wrist movements, while in arm mode, the controls operate elbow or shoulder movements. The DEKA Arm is controlled primarily by inertial measurement units (IMUs), placed on the top of the feet. 10 This control method can be supplemented by other control types including pneumatic bladders, linear transducers, and surface electromyography (EMG) controls.
Subjects
Subjects were eligible for enrollment in Part A, if they were at least 18 years old; had an upper limb amputation at the TR, TH, shoulder disarticulation/scapulothoracic level (shoulder); and had sufficient control sites available to operate a DEKA Arm. Subjects were excluded if their residual limb length or skin conditions prohibited socket fitting or if they had health conditions that the study staff believed might limit their future participation. At the completion of Part A, the Principal Investigator and study staff used clinical judgment to determine whether subjects had at least fair functional skills with the DEKA Arm, good safety awareness, and sound judgment in troubleshooting common operational problems. If so, they were invited to progress to Part B and a subset of those considered eligible agreed to participate in Part B. All videotaped testing sessions and study notes were reviewed by the analytic team. Each case was discussed by the entire team before a decision to progress to Part B was made.
Data collection
Self-report and performance measures (described in Table 1) were administered at the End of Part A, and after 4, 8, and 12 weeks of home use (Part B). One measure, the T-MAP, 16 was only administered at the End of Part A and at week 12.
Brief description of outcome measures used in the study.
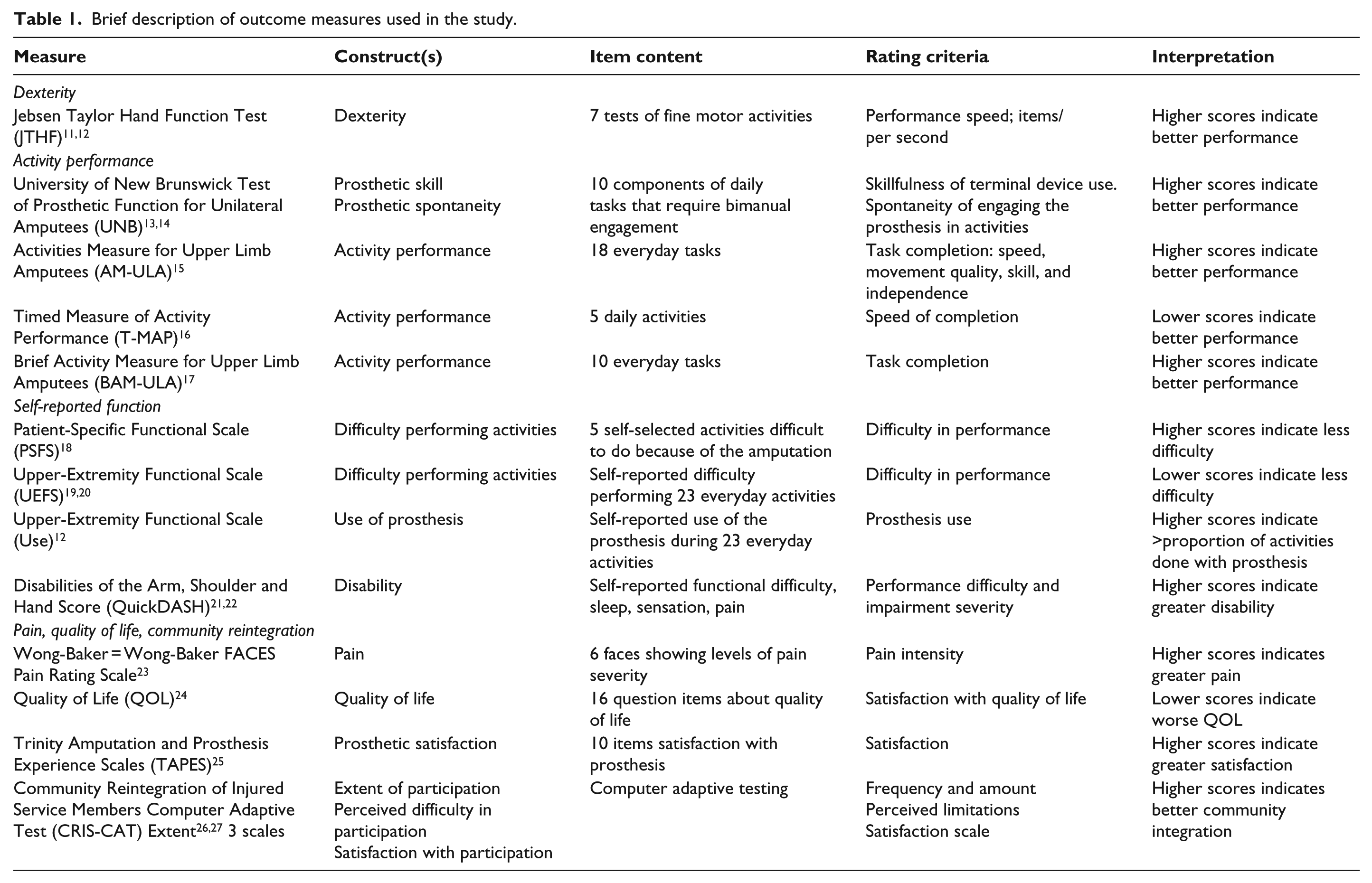
Data analyses
All measures were scored using published algorithms. Descriptive statistics were used to examine means and distribution of scores at each assessment point. Using data from the 18 study completers, outcomes at End of A and End of B were compared using two-tailed t tests. Multiple categories were identified to adjust for false discovery rates in “families” or categories of tests. The following categories were used: dexterity, activity performance, self-reported function, pain, quality of life, community integration, and satisfaction with prosthesis. The Benjamini–Hochberg method was used to maintain a false discovery rate of 0.10 within each category of tests. 28
Effect size (ES) and standardized response mean (SRM) response means were calculated to determine the magnitude of change between End of A and End of B. ES was calculated as the difference in mean scores divided by the pooled standard deviation (SD). SRM was calculated as the mean difference in scores divided by the SD of the difference. Trends in outcomes over time were examined by creating line graphs that show measures at End of A, 4, 8, and 12 weeks of home use. Variation by DEKA Arm configuration level was compared by graphically displaying outcomes at End of A and End of B by configuration level. Similar comparisons were made between outcomes at End of Part A and after 4 weeks of home use, for those who did not complete Part B.
Results
Characteristics of the sample are shown in Table 2. A total of 22 subjects began Part B of the study, and 18 completed End of Part B testing, though 1 of the completers had missing data for week 4 and 8 testing. All subjects who withdrew had acquired amputation and 50% were prosthesis users at baseline. Amputation level in the full sample of completers was 56% TR, 39% TH, and 5.6% shoulder level. The sample was predominantly male (89%) and White (89%), with mean age of 45 years. About 89% of completers were prosthesis users at baseline, and 66% used a myoelectric device at baseline testing. One participant, who identified as a prosthesis user (but only used his device some of the time), did not bring his prosthesis to baseline testing, thus no prosthesis description was provided. On average, completers had 8.6 (SD: 3.6) 2-h training visits.
Characteristics of subjects who withdrew from the study during Part B but completed some week 4 testing, completed testing at the End of Parts A and B, and completed testing at weeks 4 and 8.
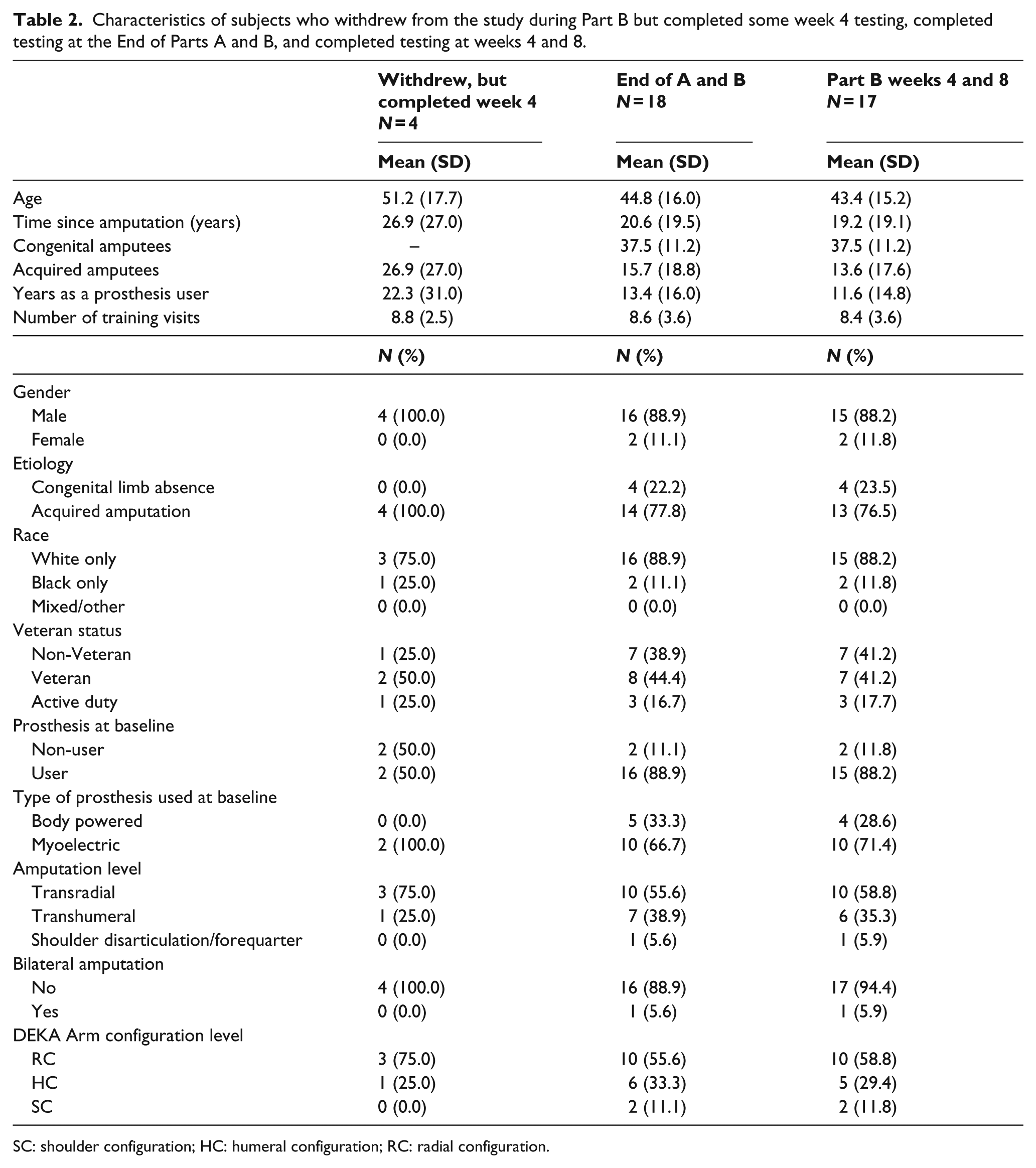
SC: shoulder configuration; HC: humeral configuration; RC: radial configuration.
Table 3 shows the results of t tests comparing End of A and End of B dexterity and activity measures and ES and SRM values. In dexterity, scores significantly improved in the JTHF Writing, Page Turning, and Heavy Cans tasks. Among measures of activity, only the AM-ULA scores improved significantly at End of B. After correcting for multiple comparisons with the Benjamini–Hochberg procedure, all findings remained significant. No statistically significant differences were observed in any other measure.
Comparison of scores of performance at End of Part A and End of Part B.
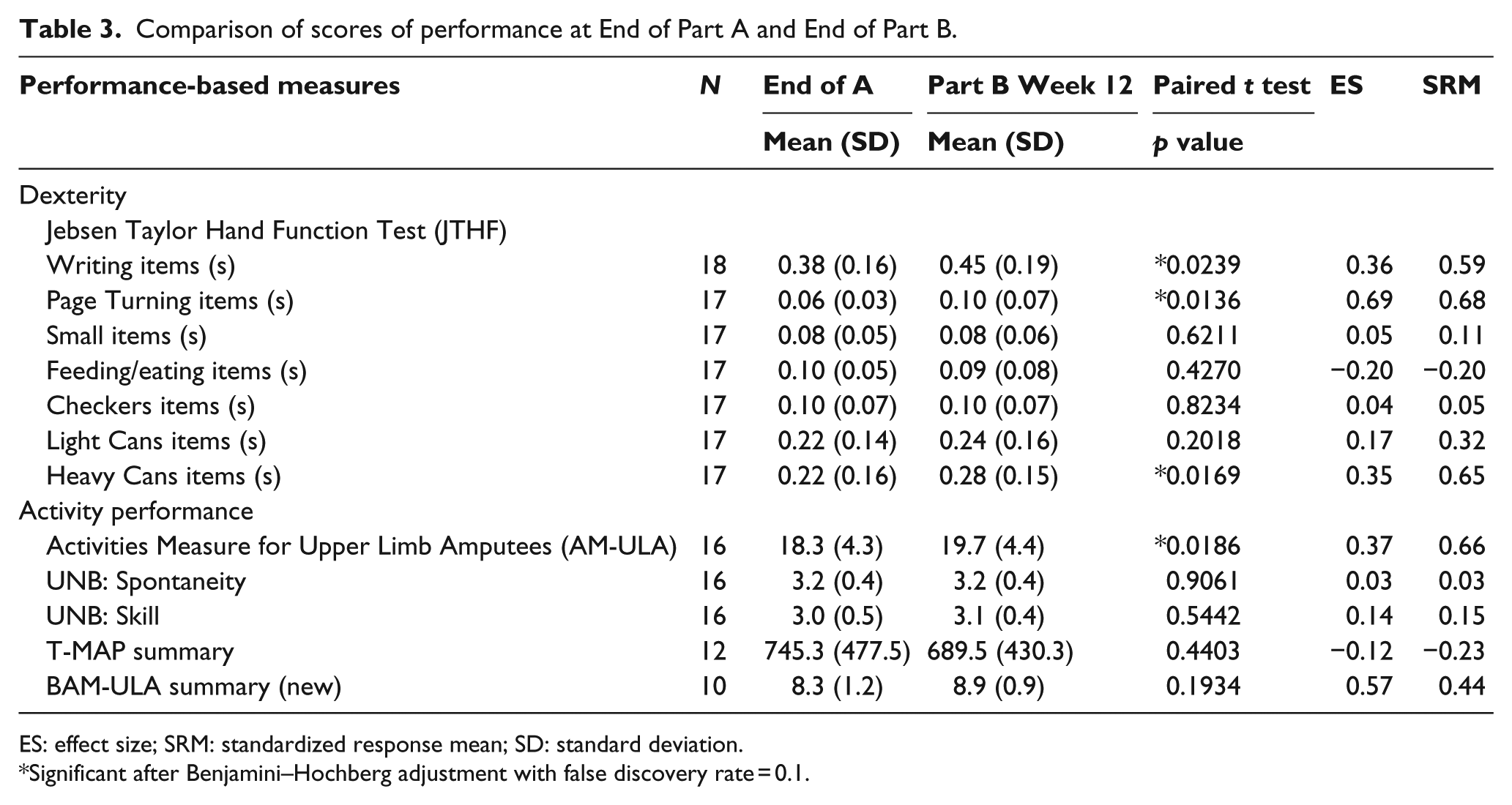
ES: effect size; SRM: standardized response mean; SD: standard deviation.
Significant after Benjamini–Hochberg adjustment with false discovery rate = 0.1.
Table 4 shows results comparing End of A and End of B for self-report, pain, quality of life, and community integration measures. Scores of the Upper-Extremity Functional Scale (UEFS) and Patient-Specific Functional Scale (PSFS) were significantly better at the End of Part B, while scores of UEFS use were worse. No statistically significant differences were observed in any other measure. After correcting for multiple comparisons with the Benjamini–Hochberg procedure, all three self-reported function findings remained significant. Outcomes over each month of time are shown in Table 5. Overall, there was a trend toward improvement in dexterity and activity performance with each month of home use experience for those measures that did improve.
Comparison of self-report measures at End of Part A and End of Part B.
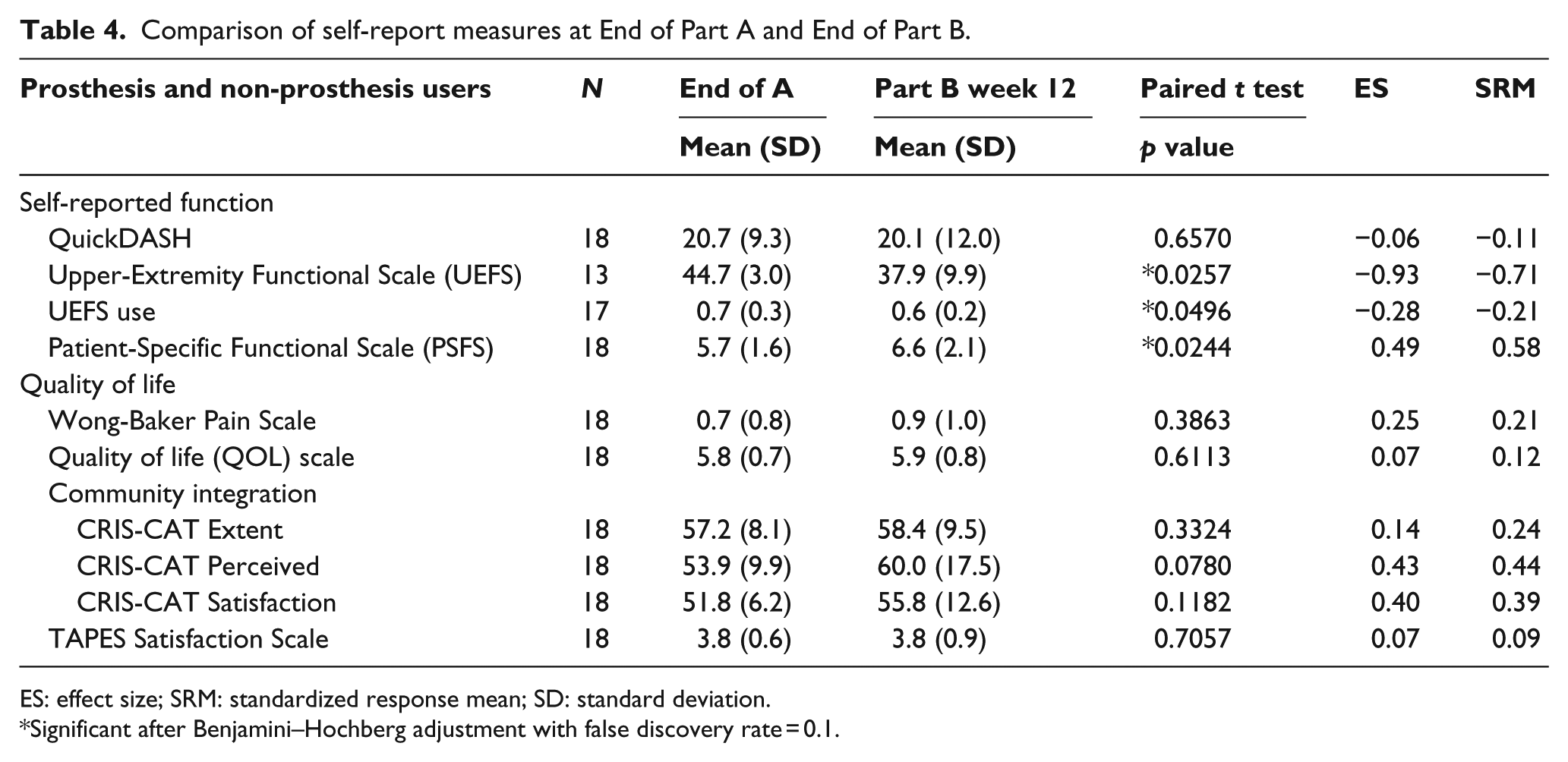
ES: effect size; SRM: standardized response mean; SD: standard deviation.
Significant after Benjamini–Hochberg adjustment with false discovery rate = 0.1.
Outcome measures across assessment time points.
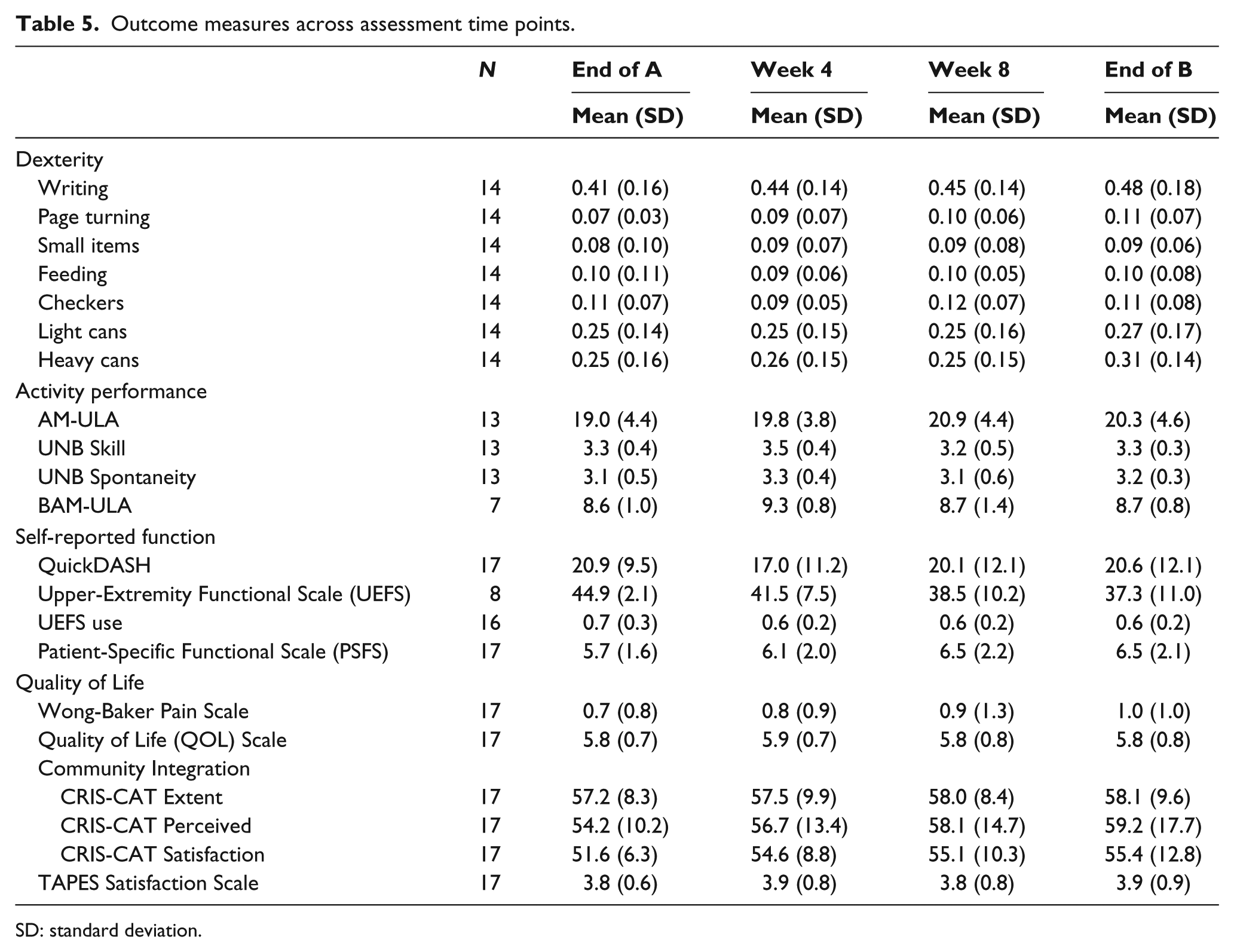
SD: standard deviation.
Descriptive comparisons of Part A and Part B outcomes by configuration level are shown in Appendix 1—Figure 1(a)–(d). Patterns of change appeared to differ for SC users as compared to RC and HC users, in that SC users had higher scores of CRIS-CAT Perceived limitations and CRIS-CAT Satisfaction with participation, and lower QuickDASH scores. In addition, SC users had similar improvements in the three JTHF tests as compared to RC and HC users but did show improvements in two additional tests (small items and light cans) as well. Scores of the UEFS use scale indicated that HC device users were using their prosthesis for fewer activities at the End of B as compared to End of A, while RC and SC users’ scores were equivalent. However, HC users’ BAM-ULA scores improved, whereas RC users’ scores remained constant.
Those who withdrew showed a decrease in JTHF scores from End of Part A to 4 weeks of Part B for 6/7 tests, while those who completed Part B improved in 1 test, stayed the same in 4, and decreased in 2 tests (Appendix 1—Figure 2(a)–(d)). Measures of activity performance were similar for completers and non-completers, except for the BAM-ULA, where scores of those who withdrew decreased and scores of those who completed increased. In self-reported function, subjects who withdrew reported an increase in disability (QuickDASH) and lower scores on the PSFS, while those who completed reported a decreased disability and greater function on the PSFS. Finally, those who withdrew reported decreased satisfaction with the prosthesis (TAPES (Trinity Amputation and Prosthesis Experience Scales)), while the satisfaction ratings of those who completed did not change between End of A and week 4.
Discussion
This study examined change in outcomes after initial prosthetic training as measured in the first few months of home use of a new prosthesis, the DEKA Arm. Overall, the findings demonstrate that the majority of new prosthesis users experienced an adaptation effect, in that their physical performance improved after formal training ended and they had the opportunity to use the device at home. We found that for persons who continued to use the device for approximately 3 months, there was evidence of medium to large improvements in some measures of dexterity and activity performance and small improvements in measures of self-reported disability. At the same time, there was also evidence that use of the prosthesis during everyday activities declined, although this was a small effect. However, a greater decline in prosthesis engagement in daily tasks was found among users of the HC arm as compared to users of the RC arm. For the full group of subjects who completed the home use study, we did not observe any impact of the DEKA Arm on quality of life or community integration. The findings demonstrate that use of the DEKA Arm at home was associated with improvements in community integration for the small number of persons in our sample who used an SC device.
About 25% of subjects who began Part B of the study withdrew without completing all home use weeks. Two-thirds of these subjects did complete some testing 4 weeks after beginning the home trial. To test the hypothesis that experiencing a decrease in function would be associated with device abandonment (as measured by study withdrawal during Part B) and decreased prosthetic satisfaction, we compared the outcomes from End of Part A to 4 weeks for the subset of participants who withdrew from Part B early but had completed some week 4 testing. Overall, in the first month of home use, those who withdrew had worse dexterity and activity, greater perceived disability, and lower prosthetic satisfaction. We did not observe the same pattern among those who remained in the study. These findings suggest that worsening function and decreased satisfaction are associated with device abandonment. Of those who withdrew during Part B, two participants relocated and were unable to meet the demands of the study. Two others withdrew due to musculoskeletal pain which may have been related to operation of the foot controls and/or the weight of the device. Although testing sessions took more than 30 min to complete, we do not believe that the length of sessions was a factor in participants’ desire to continue in the study. In a separate paper, we reported major reasons for attrition which included scheduling and factors related to the DEKA arm. 29
This study has several limitations. First, our findings are limited to upper limb amputees who were new users of the DEKA Arm and cannot be generalized to other types of prostheses. The study results not only provide empirical data to support an adaptation effect but also demonstrate that for one-fifth of upper limb amputees trained to use the DEKA Arm, their function declines after discharge from training. Although it is likely that similar patterns of adaptation and decline occur among new users of other devices, we cannot be certain that this is the case for other types of devices or initial training protocols. Further research is needed to understand the optimal progression of care. Another study limitation is that we were unable to compare change in outcomes over time for all participants who dropped from the study because two of six did not return for testing visits. One did not return due to family and personal reasons, while the other said he was too busy to travel back to the study site for testing.
Conclusion
These findings provide empirical evidence to support the widely held clinical opinion that the function of upper limb amputees continues to improve after formal prosthetic training ends, as patients gain greater experience and home use. However, in 20% of subjects, who later withdrew from the study, there was a decline in function, greater perceived disability, and greater dissatisfaction after 4 weeks of home use, suggesting that these subjects might have benefited from continued occupational therapy follow-up after the intensive training period had concluded.
Footnotes
Appendix 1
Acknowledgements
The view(s) expressed herein are those of the author(s) and do not reflect the official policy or position of the US Government.
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by VA RR&D A9226-R. The information in this manuscript does not necessarily reflect the position or policy of the government; no official endorsement should be inferred.
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Boards of each participating data collection site (Center for the Intrepid at Brooke Army Medical Center (CFI), VA New York Harbor Healthcare System, the James A. Haley VAMC), and the Coordinating site, the Providence VA Medical Center.
Informed consent
Written informed consent was obtained from all subjects before the study.
