Abstract
Background:
Evaluating the effectiveness of a novel prosthetic device during walking is an important step in product development.
Objective:
To investigate the efficacy of a novel quasi-passive ankle prosthetic device, Ankle Mimicking Prosthetic Foot 4.0, during walking at different speeds, using physiological determinants in transtibial and transfemoral amputees.
Study design:
Nonrandomized crossover design for amputees.
Methods:
Six able-bodied subjects, six unilateral transtibial amputees, and six unilateral transfemoral amputees underwent a 6-min walk test at normal speed, followed by series of 2-min walking at slow, normal, and fast speeds. The intensity of effort and subjective measures were determined. Amputees performed all walking tests on a treadmill with current and novel prostheses. Shapiro–Wilk normality tests and parametric and nonparametric tests were conducted (p < 0.05).
Results:
Compared to able-bodied individuals, the rating of perceived exertion levels were significantly elevated in transtibial and transfemoral amputees for both prostheses (p ≤ 0.016). Compared to able-bodied individuals transfemoral amputees also showed significantly elevated heart rate for both prostheses at normal speed (p ≤ 0.043). Within-group comparisons demonstrated that walking with Ankle Mimicking Prosthetic Foot significantly increased the heart rate in transfemoral amputees and transtibial compared to current prosthesis (p = 0.002). Furthermore, transfemoral amputees reached a significantly higher rating of perceived exertion levels.
Conclusion:
Intensity of effort during walking with Ankle Mimicking Prosthetic Foot is higher compared to current prostheses.
Clinical relevance
Ankle Mimicking Prosthetic Foot 4.0 is a novel quasi-passive ankle prosthesis with state-of-the-art technological parts. Subjective measures show the importance of this technology, but the intensity of effort during walking still remains higher compared to current passive prostheses, especially in transfemoral amputees.
Keywords
Background
Lower-limb amputees depend on assistive devices to cope with daily activities. One of these assistive devices is prostheses which provide the possibility of bipedal locomotion, which is of utmost importance for an independent lifestyle, directly influencing the quality of life. Current ankle-foot prostheses are fixed at a 90° angle between foot-tibia and mimic normal ankle motion by deformation of the device during gait. The lack of mobility and flexibility of these passive prostheses still create discomforts at the other joints (e.g. knee, hip, and back) of the body. Next to basic prostheses, articulated and dynamic-response prosthetic feet are available on the market. The incapability to restore a normal gait and posture affects the prevalence of back pain and knee osteoarthritis, especially in more proximal amputations. 1
The challenge for robotics engineers is to design and construct prostheses that simulate walking of able-bodied individuals and thus optimize comfort during daily activities. Recently, a novel quasi-passive lower-limb prosthesis, named Ankle Mimicking Prosthetic foot or AMP-Foot 4.0, was developed. The prosthesis contains an actuator, which is responsible for the coupling–decoupling mechanism in which a spring set stores and releases energy at the correct moment during walking. The resultant lift of the prosthetic forefoot should simulate walking of able-bodied individuals.
AMP-Foot 4.0 is a new type of energy-efficient below knee prosthesis providing improved passive adaptive and propulsion characteristics to its wearer. The device mainly distinguishes itself from commercial prostheses thanks to its new type of actuation providing a compact and energy-efficient solution to the challenge of ankle-foot actuation. This new actuation method consists of using springs, a servo motor, and a locking mechanism, coupled with a sensory network providing intelligence to the robotic device. The AMP-Foot 4.0 design is also based on previous research conducted on AMP-Foot 22 and 3.3 However, it is important to note that in contrast to its preceding designs, AMP-Foot 4.0 does not provide active propulsion at push-off.
During walking, the AMP-Foot 4.0 working principle is divided into two main logic sequences, that is, the stance and the swing phase. These two phases are detected by analyzing gyroscope and acceleration measurements from an inertial measurement unit chip. During the stance phase, the ankle performs a dorsi-flexing movement while a plantar-flexing torque is applied at the ankle. The person’s gravitational potential energy is stored into elastic potential energy by means of the used spring. It is this spring that provides the plantar-flexing torque required at the ankle as reaction to the movement of the user. Thanks to the use of a locking system, the prosthesis can adapt its so-called zero torque rest point depending on the slope or stride length of the user. This provides adaptability and therefore greater comfort compared to prostheses with a fixed zero torque rest point. During the swing phase, the locking mechanism unlocks to free the ankle movement. Parallel springs external to the stance system are then activated to reset the foot to its initial position. From that moment, the device is ready for a new step.
Evaluation of the effectiveness of a novel prosthesis is an important step in the iterative process of product development. In the literature, functional ambulation is commonly evaluated using the 6-min walk test (6MWT). The 6MWT is easy to administer, better tolerated, and more reflective of daily life activities compared to other walking tasks. 4 The 6MWT can be performed in the hallway or on the treadmill. 5 , 6 However, the latter is easier to perform, allows the monitoring of subjects more easily, and demonstrated high test–retest reliability in many subject groups. Another walking task is the 2-min walk test and has previously been used in lower-limb amputees. 7 This task strongly correlates with the 6MWT. 8
The goal of this experiment is to evaluate the novel ankle prosthesis AMP-Foot 4.0 during walking at different speeds. Comparisons were made with the current prosthesis in transtibial (TTA) and transfemoral amputees (TFA) using physiological measures. Able-bodied individuals were included to investigate walking performance differences between amputees and able-bodied individuals.
Methods
Subjects
Three subject groups were included in this study, that is, six able-bodied individuals (five men and one woman aged 26 ± 5 years, height 1.75 ± 0.10 m, and weight 72 ± 12 kg), six unilateral TTA (six men aged 54 ± 14 years, height 1.76 ± 0.08 m, and weight 80 ± 13 kg), and six unilateral TFA (five men and one woman aged 53 ± 14 years, height 1.79 ± 0.09 m and weight 89 ± 16 kg). TTA and TFA had experience with a prosthetic leg for 18 ± 18 and 15 ± 13 years, respectively. The amount of physical activity was 5.0 ± 3.9 and 4.3 ± 3.8 h per week for TTA and TFA, respectively. The current prosthetic knees of TFA were mechanical knees from Ottobock® (types 3R15 or 3R80). The prosthetic foot of all amputees was SACH-foot (Ortho Europe). Preceding the experiment, nine TTA and nine TFA were recruited, but a total of six dropouts (TTA: n = 3; TFA: n = 3) were registered throughout the whole experiment due to injury (mainly caused by friction of the residuum leg and socket), surgery, and sickness.
The experiment was approved by the institutional medical ethics commission of UZ Brussel and Vrije Universiteit Brussel (Belgium, B.U.N.143201526629) and the Federal Agency for Medicines and Health Products (FAMHP reference: AFMPS/80M0641). Subjects were provided written and oral information about the experimental procedures and potential risks before giving informed consent to participate in this study.
Protocol
TTA and TFA visited the laboratory three times, whereas able-bodied individuals visited the laboratory once. The laboratory was situated in an orthopedic center (VIGO, Wetteren, Belgium). The environmental temperature and humidity of the laboratory was set at 26.5°C ± 0.4°C and 43% ± 2%, respectively. During the first visit, subjects were instructed about the experimental protocol, the 6-min treadmill walking test (6MWT), the 2-min slow, and the 2-min fast walking protocol. During 6MWT, subjects were instructed to walk at a self-selected comfortable pace for 6 min on a treadmill (inclination treadmill: 0.1%). No motivational feedback was provided. In addition, biometrical measurements were obtained.
After the familiarization trial, TTA and TFA conducted two experimental trials in a nonrandomized order; the first experimental trial was performed with the current prosthetic foot and the second experimental trial with the AMP-Foot 4.0. The AMPFoot was fitted by a professional prosthetist. Besides the ankle foot component, the tibial extension tube and adaptors were individually adjusted. The average acclimation period of the novel device was about 30 min.
The walking task was 6-min treadmill walking at the same speed as the 6MWT conducted during the familiarization trial. Able-bodied individuals walked at a constant speed of 5.2 km/h, slow and fast speeds were set at 3.9 (−25% of the normal speed) and 6.5 km/h (+25% of the normal speed), respectively. The walking speed was limited to 5.2 km/h in order to avoid the transition from walking to running during fast speed walking trials. During the familiarization trial, TTA and TFA performed the 6MWT at self-selected speed. This walking speed was kept constant for all experimental trials in order to compare physiological measures. After 6-min treadmill walking, subjects walked 2 min at slow and 2 min at fast speed. TTA and TFA walked at a normal speed of 4.1 ± 1.2 km/h (slow speed: 3.1 ± 0.9 km/h and fast speed: 5.1 ± 1.5 km/h) and 2.8 ± 0.9 km/h (slow speed: 2.2 ± 0.7 km/h and fast speed: 3.5 ± 1.2 km/h), respectively. Each amputee walked at the same speed during the two experimental trials.
To investigate the intensity of the effort, heart rate (HR) data were gathered with an HR monitor (Polar RS400®). During 6-min treadmill walking, HR was monitored and registered at baseline and each minute. During the 2-min walking sessions, HR was monitored at the end of the session. Furthermore, the rating of perceived exertion (RPE) was rated on a scale of 6 (“no exertion”) to 20 (“maximal exertion”) 9 at the beginning (first min) and at the end of the 6-min walking trial, and at the end of the 2-min walking sessions. Furthermore, ergo-spirometrical data (VE, VO2, VCO2, respiratory quotient (RQ) and metabolic equivalents (METS)) were continuously gathered during the 6-min treadmill walking using a wearable ergo-spirometrical system (Cosmed K5®, Cosmed, Rome, Italy). Preceding each experiment and after a warm-up period of 30 min, flowmeter, scrubber, reference gas, and room air calibrations were performed to ensure accurate and reliable measurements. Mixing chamber data were stored in the portable unit and after the experiment data were transferred to the program Omnia of Cosmed. The K5 device was mounted on the back of the subject using a harness.
To investigate the subjective value of the assistive technology for end-user purposes, the Quebec User Evaluation of Satisfaction with assistive Technology (QUEST) Version 2.0 was used. The questionnaire consists of 12 items, but only the first 8 items regarding the satisfaction of the assistive device (e.g. dimensions, weight, ease in adjusting, and safety) were relevant. Subjects had to respond to the questions using a scale that ranges from 1 (“not satisfied at all”) to 5 (“very satisfied”). Further questions were asked from the prospect of marketing the novel device: Question 1: Would you consider buying the novel technology if AMPFoot is available on the market?; Question 2: Is it useful to further develop and test prototypes of this specific type of technology with the aim to bring the product on the market and compete against current existing ankle prostheses?; Question 3: Would you like to have the AMPFoot if the product is available on the market?
Statistical analysis
SPSS Version 24.0 was used for statistical analyses. Shapiro–Wilk normality tests of the different datasets were performed to determine the distribution of the data. Nonparametric tests were conducted for RPE and QUEST. For RPE, Kruskal–Wallis H tests were performed between able-bodied individuals, TTA, and TFA as well as post hoc Mann–Whitney U tests. Within TTA and TFA, Friedman analyses of variance (ANOVAs) were performed as well as Wilcoxon-signed rank tests. For QUEST, Wilcoxon-signed rank tests were conducted.
To determine the effect of AMPFoot on HR, mixed ANOVAs (between groups TTA and TFA; within: prosthetic device and time) at all walking speeds were conducted. Repeated-measures ANOVAs were performed for ergo-spirometrical data (VE, VO2, VCO2, RQ, and METS: between groups TTA and TFA; within: prosthetic device). To compare data between the groups TTA and TFA with able-bodied individuals, repeated-measure ANOVAs were performed for HR (between able-bodied individuals, TTA, and TFA; within: time). If significant differences were found, post hoc comparisons with Bonferroni correction were performed. In addition, for HR, one-way ANOVAs (between able-bodied individuals, TTA, and TFA) were conducted at slow and fast walking speeds. Also, for ergo-spirometrical data one-way ANOVAs were performed between able-bodied individuals, TTA, and TFA at normal speed. Since ergo-spirometrical datasets were missing for two TFA subjects at slow and fast speeds, comparisons were made between TTA and able-bodied individuals using independent samples T-tests and within TTA using paired sample T-tests. p was set at 0.05.
Results
Subjective measures
All amputees responded confirmative on additional question 2 regarding future technological prospects compared to current existing ankle prosthetic devices. In total, 10 (out of 12) participants also responded positive regarding buying the device if it was available on the market. Only in the TFA, significant lower values for questions 1 and 2 (of the QUEST questionnaire) related to the satisfaction of the dimensions and the weight of AMPFoot compared to the current prosthetic device were observed (p = 0.038 and 0.042, respectively).
Intensity of the effort (HR and RPE)
Figure 1 shows HR during walking at different speeds for each group. The main effects of the used prosthetic device at normal (F(1,10) = 16.322, p = 0.002) and other speeds (F(1,10) = 17.547, p = 0.002) were found. At fast speed, no significant differences in HR were observed, whereas at slow speed higher HR data were observed in TFA and TTA with AMPFoot compared to current prosthetic devices (p = 0.026 and 0.018, respectively, F(1,10) = 20.883; p = 0.001). Furthermore, compared to able-bodied individuals both the current and the novel prosthesis showed an interaction effect time ×groups (p = 0.003 and 0.001, respectively) and a between-subject effect (p = 0.045 and 0.009, respectively) at normal speed. Pairwise comparisons showed significant higher HR measures for TFA compared to able-bodied individuals for the current and novel prosthetic device (p = 0.043 and 0.008, respectively; Figure 1). At other speeds, no significant differences were revealed.
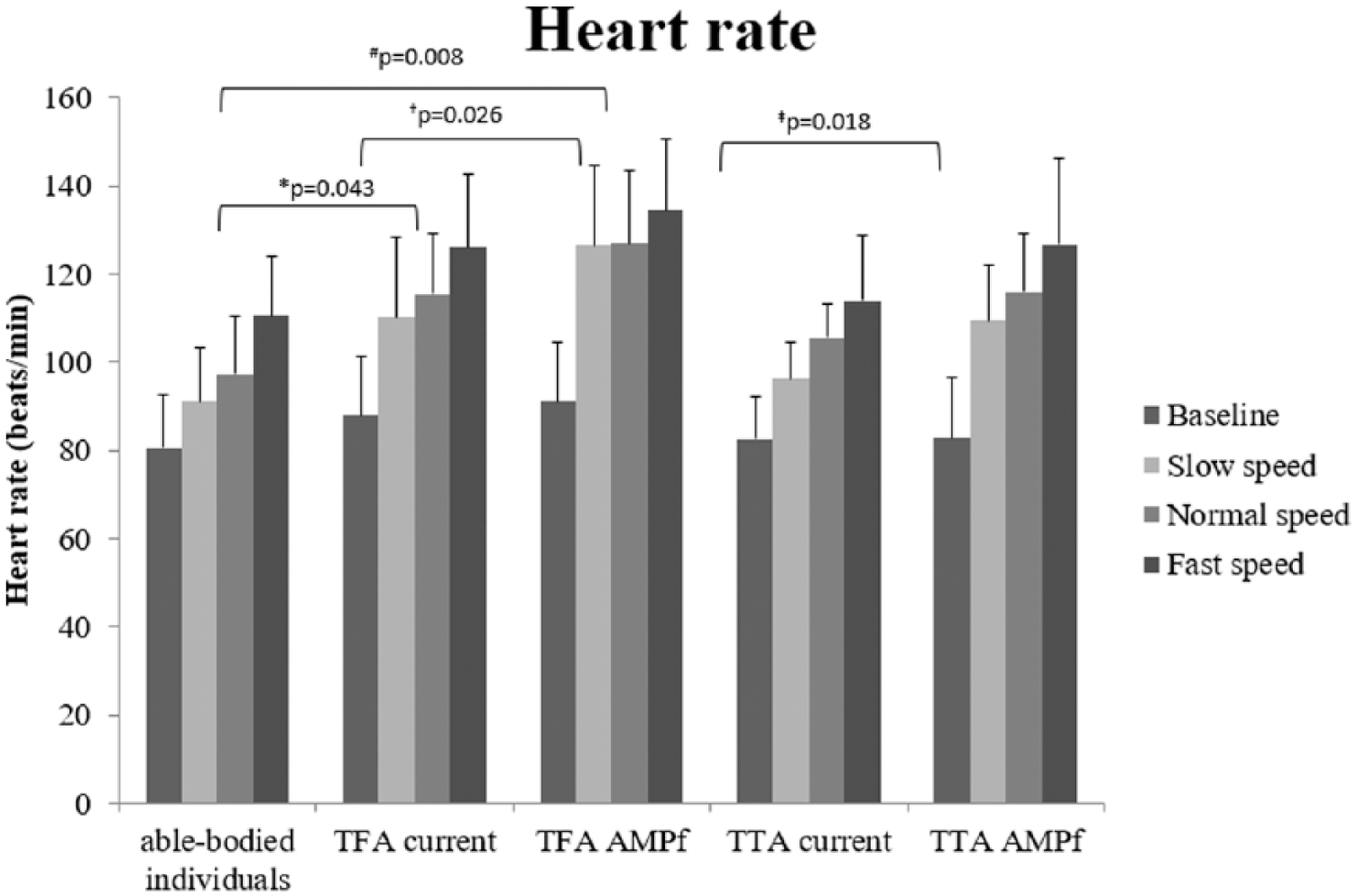
Heart rate (mean and SD) of able-bodied individuals, TFA, and TTA with the current prosthetic device and AMPFoot (AMPf). Significant pairwise comparisons with Bonferroni correction are displayed at normal speed (* and #) and slow speed († and ‡).
At normal speed, RPE levels were significantly higher in the TFA and TTA wearing the current prosthesis and AMPFoot compared to able-bodied individuals at the first (p ≤ 0.016 and p ≤ 0.004, respectively) and sixth minute (p ≤ 0.003 and p ≤ 0.004, respectively; Table 1). No significant differences were observed between TFA and TTA when wearing the current prosthesis. Significant higher RPEs were observed for TFA compared to TTA when wearing AMPFoot (p = 0.027).
RPE values of the different subject groups (mean ± SD) at different time points of the experiment.

TFA: transfemoral amputees; AMPFoot: Ankle Mimicking Prosthetic Foot; TTA: transtibial amputees.
Compared to able-bodied individuals: p ≤ 0.016.
Compared to able-bodied individuals: p ≤ 0.004.
Compared to TTA AMPFoot: p = 0.027.
Compared to TFA current: p ≤ 0.042.
Table 1 shows RPE values of the different subject groups (mean ± SD) at different time points of the experiment.
At slow and fast walking speeds, significantly higher values for TFA and TTA compared to able-bodied individuals for the current and novel prostheses were revealed (slow speed: p ≤ 0.004 and p ≤ 0.003, respectively; fast speed: p ≤ 0.005 and p ≤ 0.009, respectively). No significant differences in RPE were observed between TFA and TTA. In addition, at fast speed significantly lower RPE values were observed for TTA compared to TFA when subjects wore AMPFoot (p = 0.042). Furthermore, in TFA significantly higher RPE levels were observed with AMPFoot compared to the current prosthesis at 1 and 6 min (p = 0.027 and 0.042, respectively).
Ergo-spirometry
At normal speed, no significant differences between groups were observed for VE, VO2 (Figure 2), VCO2, RQ, and METS. At normal speed, RQ showed a significant within-subject effect for prosthetic use (F(1,10) = 8.003; p = 0.018). In TTA, significantly higher RQ values were found with AMPFoot compared to the current prosthesis (p = 0.017). At other walking speeds, no significant differences were found.
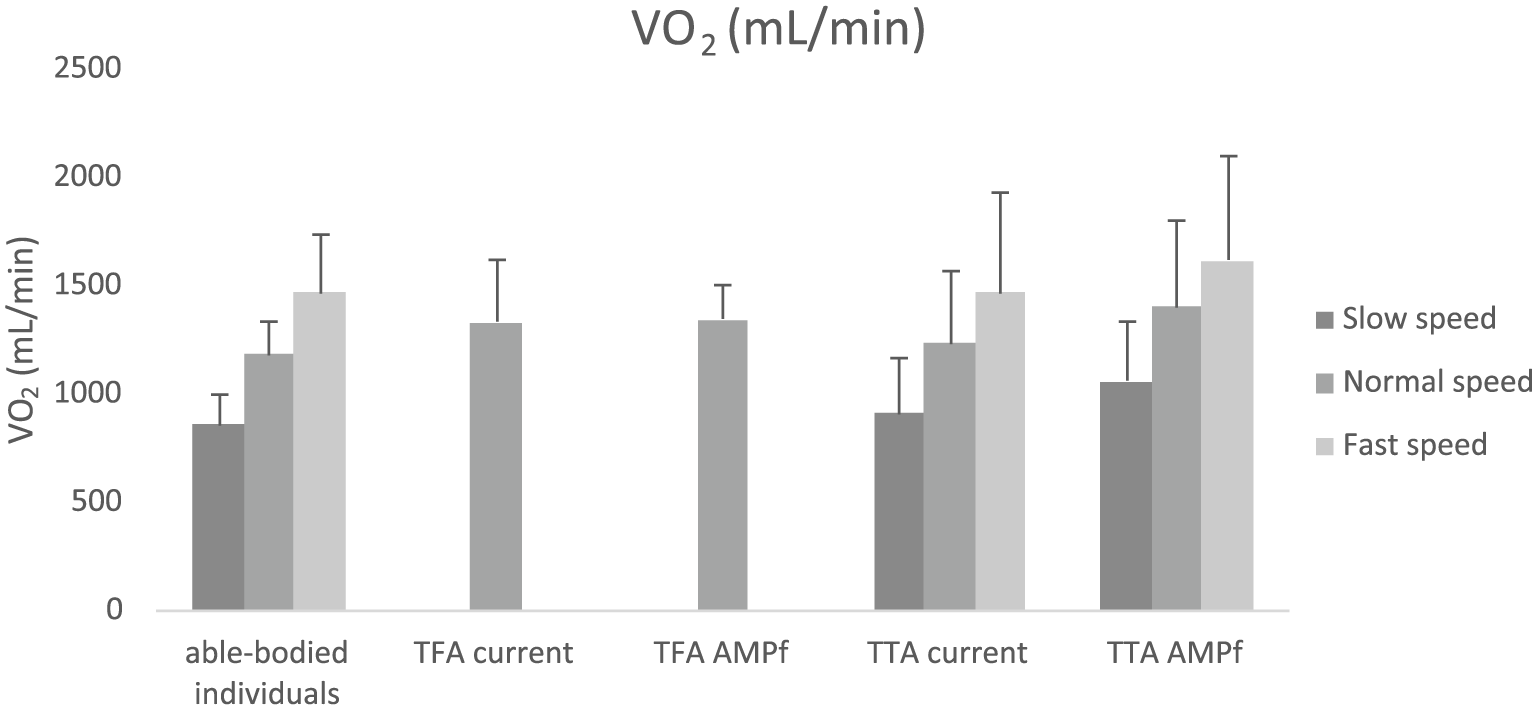
Oxygen consumption for able-bodied individuals, TFA, and TTA during different conditions. Note that missing data are present for TFA at slow and fast speeds.
Discussion
Robotic innovations of AMPFoot and a case study with AMPFoot prototypes 2.0 and 3.0 were published in Cherelle et al.2,3 Research on human biomechanics in order to mimic its behavior in intelligent robotic systems2,3,10 has eventually led to AMP-Foot 4.0. This study included TTA and TFA subject groups, and aimed at investigating the efficacy of AMP-Foot 4.0 during walking at different speeds compared to current passive prostheses. Able-bodied individuals were included to investigate the level of resemblance of walking performance measures between able-bodied subjects and amputees. The results show that AMPFoot increases HR and RPE during walking in TTA and TFA, regardless of the walking speed. Subjective measures demonstrate that TFA are less satisfied regarding the dimensions and weight of the AMPFoot. On the other hand, all TTA and TFA recognize the added value of the locking–unlocking spring set mechanism used in the design and support the further development of this technology.
TTA and TFA require a higher metabolic cost during walking at the same speed as able-bodied individuals. 11 This study confirms the increased intensity of effort during walking in TTA and TFA (TFA: higher HR; TTA and TFA: higher RPE) compared to able-bodied subjects, even though the average normal walking speeds were lower in TTA and TFA compared to able-bodied individuals. In recent years, lower-limb prosthetic devices, such as BiOM® (Ottobock), Meridium® (Ottobock), and Proprio Foot® (Össur) were designed and constructed to counteract the additional metabolic cost of transport in TTA and TFA. BiOM is a commercialized bionic foot that provides propulsive forces, whereas the novel AMP-Foot 4.0, Meridium, and Proprio Foot are microcontroller feet. This study showed that walking with AMPFoot increased HR and RPE in TTA and TFA at normal walking speeds.
A possible explanation for the increased intensity of walking effort with AMPFoot is the limited acclimation time. This is due to the restricted financial support and thus the available prototypes. Another explanation for the increased intensity of walking effort with the AMPFoot could be the weight. Previous reports mention a higher energy consumption with more load at the distal end of the prosthesis. 12 A general limitation of this study is that treadmill walking was used, which is more challenging for patients to pace themselves compared to ordinary walking. However, all subjects were familiar with walking on a treadmill and during the familiarization trial, the self-selected speed was determined. Regarding AMPFoot, some current limitations need to be put forward: (1) the coupling–decoupling system of coil spring set functions for over 95% of the time, but when the system fails to unlock the stored energy in the coil spring set, amputees, especially TFA, might trip and fall; (2) The current device requires customization regarding the coil springs (spring rate: between 90 and 180 N/mm) depending on the body weight of the subject; (3) the current weight of the AMPFoot is comparable to the weight of a normal foot. The weight can be reduced using carbon fiber–reinforced polymers; however, it should be mentioned that some TTA outlined that the “heavy” weight might be beneficial to counteract atrophy of the calf musculature, which plays a key role in human walking and affects optimal prosthetic fitting; (4) AMPFoot when turned on makes quite some noise, which gives participants the feeling that the prosthetic device will disconnect from the shank. A major concern of AMPFoot for TFA is that this group of amputees does not lift the heel of the prosthetic device high enough to trigger the release of energy of the spring set (mechanical part of the prosthetic device).
Subjective measures were determined using QUEST, by which the reliability, validity, and applicability have been investigated in different population subgroups.13–15 Most amputees would consider buying the novel device if it is available on the market, since they feel that the novel device increases perceived comfort and better mimics normal gait. Therefore, the technology used in AMP-Foot 4.0 should be further developed. The current findings are important for engineers to continue fine-tuning the mechanical system of the prosthesis. In the near future, experiments will evaluate AMPFoot prototypes during additional daily activities such as stair climbing, sit-to-stance, and uphill and downhill walking. Furthermore, brain dynamics will be explored to optimize human–robot interaction.
Conclusion
In conclusion, walking with AMPFoot increases the intensity of effort compared to current existing passive prostheses. Especially for TFA, a redesign of AMPFoot is required. All TTA and TFA encourage to further explore the prosthetic technology used in AMPFoot.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
