Abstract
Background:
Accurate and precise mediolateral foot placement is important for balance during gait, but is impaired post stroke. Mediolateral foot placement may be improved with ankle–foot orthosis use.
Objective:
The purpose of this study was to determine whether an ankle–foot orthosis improves mediolateral foot-placement ability during post-stroke ambulation.
Study design:
Crossover trial with randomized order of conditions tested.
Methods:
The accuracy and precision of mediolateral foot placement was quantified while subjects targeted four different randomized step widths. Subjects were tested with and without their regular non-rigid ankle–foot orthosis in two separate visits (order randomized).
Results:
While ankle–foot orthosis use corrected foot and ankle alignment (i.e. significantly decreased mid-swing plantar flexion, p = 0.000), effects of ankle–foot orthosis use on hip hiking (p = 0.545), circumduction (p = 0.179), coronal plane hip range of motion (p = 0.06), and mediolateral foot-placement ability (p = 0.537) were not significant.
Conclusion:
While ankle–foot orthosis–mediated equinovarus correction of the affected foot and ankle was not associated with improved biomechanics of walking (i.e. proximal ipsilateral hip kinematics or mediolateral foot-placement ability), it may affect other aspects of balance that were not tested in this study (e.g. proprioception, cerebellar, vestibular, and cognitive mechanisms).
Clinical relevance
Studies that investigate the effect of ankle–foot orthosis on gait can help advance stroke rehabilitation by documenting the specific gait benefits of ankle–foot orthosis use. In this study, we investigated the effect of ankle–foot orthosis use on mediolateral foot-placement ability, an aspect of gait important for maintaining balance.
Background
Many stroke survivors consider the ability to independently ambulate integral to successful rehabilitation outcomes. 1 Estimates suggest that 50%–85% of individuals will achieve independent ambulation following a stroke.2–4 Ankle–foot orthoses (AFOs) are often prescribed to provide support when neuromuscular impairments and/or equinovarus deformity of the foot and ankle are present. AFOs typically cover the plantar aspect of the foot (extending just proximal to the metatarsals or to the end of the toes) and the posterior aspect of the lower leg. The device normally extends well above the ankle, with the proximal trim-line usually located 2–4 cm distal to the apex of the fibular head. Anterior straps typically secure the leg into the AFO. While AFOs are prescribed with the intent of improving standing and walking through better foot and ankle alignment and motion control, 5 there are few studies that unequivocally support the use of AFOs in the post-stroke population to improve walking. However, stroke subjects generally perceive that AFOs are comfortable and improve their gait.6–8
One aspect of gait that may be influenced by AFO use is balance, defined in this context as the ability to maintain forward progression without experiencing a fall. Balance impairments are common post stroke as evidenced by the increased incidence of falls9–12 during walking. Falls have been attributed to intrinsic influences (i.e. impaired balance and subject-specific factors) rather than extrinsic influences such as obstacles. 11 Many factors contribute to balance and can be grouped into cognitive, visual, vestibular, proprioceptive, and movement mechanisms. 13 In this study, we investigated one movement mechanism: accurate and precise mediolateral foot placement.
Mediolateral (ML) foot placement is important for maintaining a safe and efficient gait pattern because it defines the base of support and redirects motion of the body center of mass with each step. 14 However, ML foot placement is deficient post stroke, as evidenced by greater ML foot-placement errors during single-step tasks compared to controls, 15 greater ML foot-placement errors during a targeted step width walking task, 16 and difficulty navigating obstacles.17,18
One factor that may contribute to these ML foot-placement deficits is difficulty with preparatory swing leg movement. 15 ML foot placement during walking is planned during the preceding swing phase, which is often characterized by equinovarus posturing of the foot and ankle and insufficient knee flexion in post-stroke individuals. 19 Swing phase impairments contribute to insufficient mid-swing toe clearance, and associated swing phase compensations include hip hiking (i.e. coronal plane pelvic elevation), circumduction (i.e. increased coronal plane thigh angle with respect to vertical), and increased hip abduction of the affected limb during swing phase.20–22 Correction of abnormal swing phase ankle kinematics with an AFO decreases the magnitude of hip hiking, while swing phase hip abduction/adduction angle is unaffected by AFO use. 23 Kinematic coupling between the hip and ankle would suggest that an AFO could also reduce the magnitude of circumduction, but this remains unsubstantiated.
AFO-mediated reduction of abnormal frontal plane motion during affected limb swing phase, including hip hiking and circumduction, may improve an individual’s ability to create a more accurate ML foot placement, which is a key feature of safe ambulation. The purpose of this study was to investigate whether an AFO improves ML foot-placement ability during ambulation in individuals with post-stroke hemiplegia. ML foot-placement ability was investigated by instructing subjects to ambulate at four target step widths, including step widths wider and narrower than preferred. We expected less plantar flexion (i.e. equinus) at mid-swing with AFO use, confirming correction of equinovarus posturing. We hypothesized that circumduction and hip hiking would be reduced with AFO use, but hip abduction/adduction angle would be unaffected. Finally, we hypothesized that affected side ML foot-placement ability (i.e. accuracy and precision) would improve with AFO use, while sound side ability would be unaffected.
Methods
Individuals who met the following recruitment criteria were enrolled in the study: at least 1 year post stroke, at least 18 years of age, able to understand simple instructions, able to walk shod but without any assistive device for at least 12 m, and currently using a non-rigid AFO. Non-rigid AFOs were chosen because it has been suggested that they may be better for dynamic balance conditions than rigid AFOs. 13 Furthermore, these inclusion criteria served to create a more homogeneous study population. Individuals with comorbidities that would affect gait were excluded. Additionally, able-bodied individuals with no gait-related pathologies were recruited for comparative control data. The university’s Institutional Review Board approved this study, and upon arrival at the first study visit, all individuals provided written informed consent.
Gender, age, height, weight, shod foot length (measured using a Ritz stick), and for post-stroke individuals, affected side, time post stroke, and AFO type were recorded. The target step widths for each experimental condition were based upon the subject’s leg length (LL). The distance from the anterior superior iliac spine (ASIS) to the ipsilateral medial malleolus (measured in the supine position) was used for each individual’s LL. When discrepancies were detected between the sound and affected side LL, an average LL was used.
Subjects participated in two separate data collection sessions for the AFO and no-AFO conditions, with the order of testing randomized across participants. Kinematic data were collected at 120 Hz using an eight-camera digital real-time motion capture system. Retro-reflective markers were placed on anatomical landmarks according to a modified Helen Hayes full-body marker set, a standard marker configuration used in clinical gait analysis. 24 Specifically, markers were placed bilaterally over the dorsum of the shod foot immediately proximal to the third metatarsal phalangeal (MTP) joint, on the lateral malleoli (or lateral orthotic ankle joint), anteriorly on the shanks and thighs, on the lateral femoral epicondyles, on the acromion processes, lateral humeral epicondyles, and between the styloid processes of the wrists. Markers were also placed on the ASISs and midway between the posterior superior iliac spines (PSIS). For standing static trials, four additional markers were placed on the medial malleoli and medial femoral epicondyles. The positions of these markers were used in conjunction with all other markers to calculate lower limb joint centers and define locally embedded coordinate axes.
To determine preferred walking speed, six trials of baseline data were recorded as subjects walked back and forth across a 10-m walkway at their preferred walking speed without assistive devices or step-width restrictions (a single pass across the walkway was considered one trial). Subjects whose average preferred walking speed at the no-AFO visit was more than 2 standard deviations (SDs) away from the sample population mean walking speed were excluded from subsequent analyses.
Following collection of baseline walking speed data, subjects were tested at the following randomized step widths: 0%, 15%, 30%, and 45% LL. The range of target step widths was intended to challenge subjects at step widths narrower and wider than their preferred step width during unassisted locomotion (i.e. 20% LL (18.8 cm) 25 ) and is similar to values used in previous studies involving step-width manipulation. 26
Target ML foot placement was indicated using tape lines placed on the laboratory floor at the different step widths. Subjects were instructed to place one foot on each line as close as possible and to walk at a comfortable speed. The task was demonstrated by laboratory personnel to ensure that it was understood by all study participants. Subjects were allowed to rest as needed throughout the experiment.
Whole-body position data were processed using commercial software (Cortex, MAC, Santa Rosa, CA, USA). Marker data were smoothed using a fourth-order bidirectional Butterworth filter with an effective cut-off frequency of 6 Hz. 27 Custom MATLAB (MathWorks, Natick, MA, USA) programs were written to calculate ML foot-placement accuracy and precision, mid-swing ankle angle, hip hiking, circumduction, and hip abduction/adduction range of motion.
ML foot-placement accuracy was defined as the distance between the ankle center and the target line for each step, and an average value was obtained for each subject in each step width condition. ML foot-placement precision was assessed using the standard deviation of all ML foot-placement errors in a target step width condition. For the affected limb, mid-swing ankle angle was calculated at the time point exactly halfway through swing phase for each step. Hip hiking was identified by calculating the peak pelvic angle during swing phase. Circumduction was identified by calculating the maximum lateral distance that the ankle center traveled from just prior to foot off until the end of swing phase. Hip abduction/adduction range of motion was calculated as the peak-to-peak hip abduction/adduction angle during swing phase.
The effect of an AFO on mid-swing plantar flexion was tested with a one-way repeated-measures analysis of variance (ANOVA) (p < 0.05) using commercial software (SPSS version 20, IBM Corporation, Armonk, NY, USA). The hypothesis that ML foot-placement ability would improve with AFO use was tested using a one-way repeated-measures multivariate analysis of variance (MANOVA). The two dependent variables were ML foot-placement accuracy and ML foot-placement precision. AFO-mediated changes to kinematic variables were examined with repeated-measures ANOVAs for each swing phase variable. For the AFO condition, pairwise comparisons among the step width conditions were tested with a Bonferroni correction.
Results
In total, 15 subjects with chronic post-stroke hemiplegia and 6 age-matched, able-bodied individuals completed the study. Two post-stroke subjects, whose preferred walking speeds of 0.09 m/s and 1.27 m/s (without AFOs) were greater than 2 SDs above the group mean, were considered to be outliers and were excluded from subsequent analyses. Subject characteristics recorded at the first study visit are reported in Table 1. All post-stroke subjects were current non-rigid polymer AFO users. Additional AFO characteristics are provided in Table 2 with examples of AFOs shown in Figure 1.
Subject characteristics.
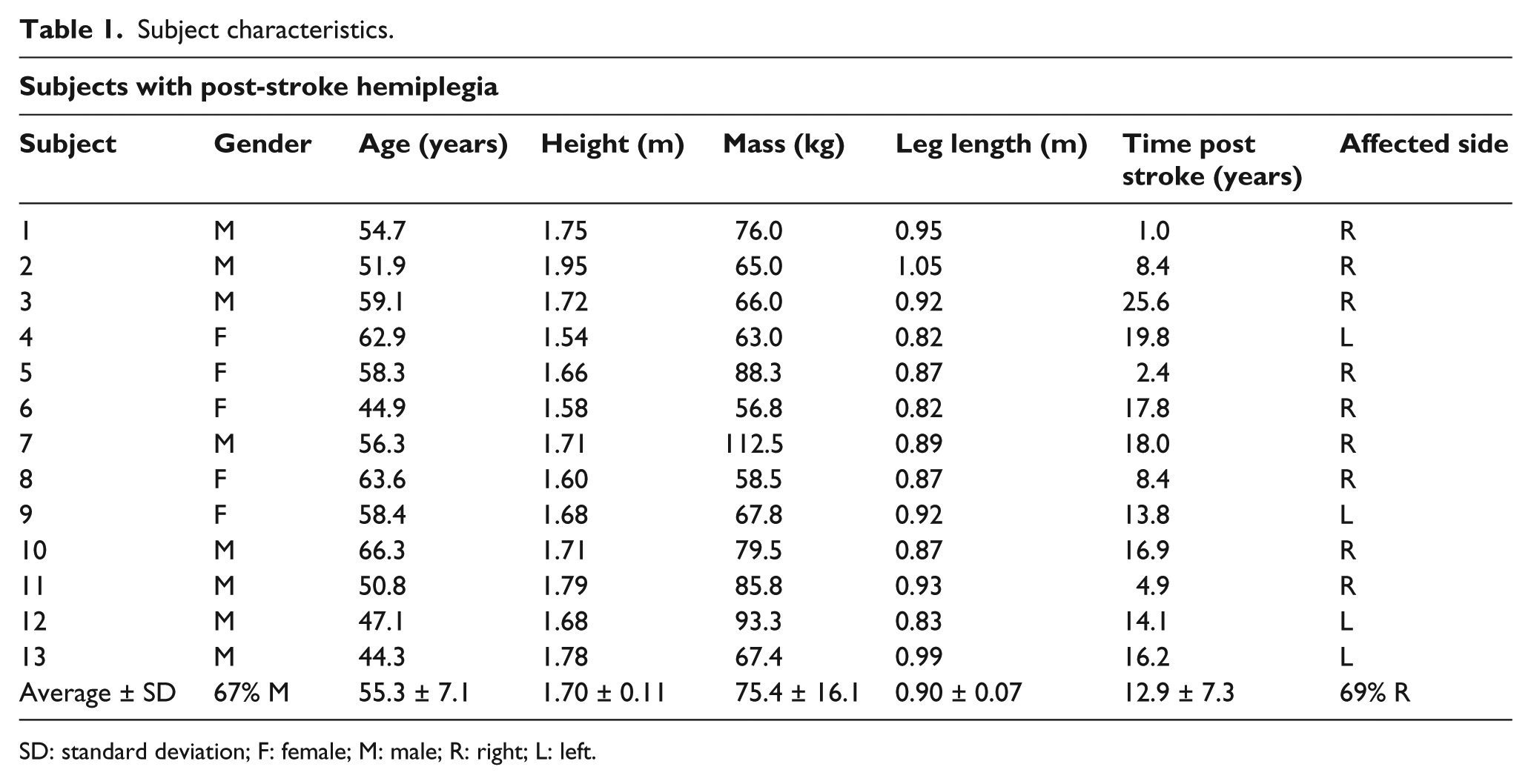
SD: standard deviation; F: female; M: male; R: right; L: left.
AFO characteristics.
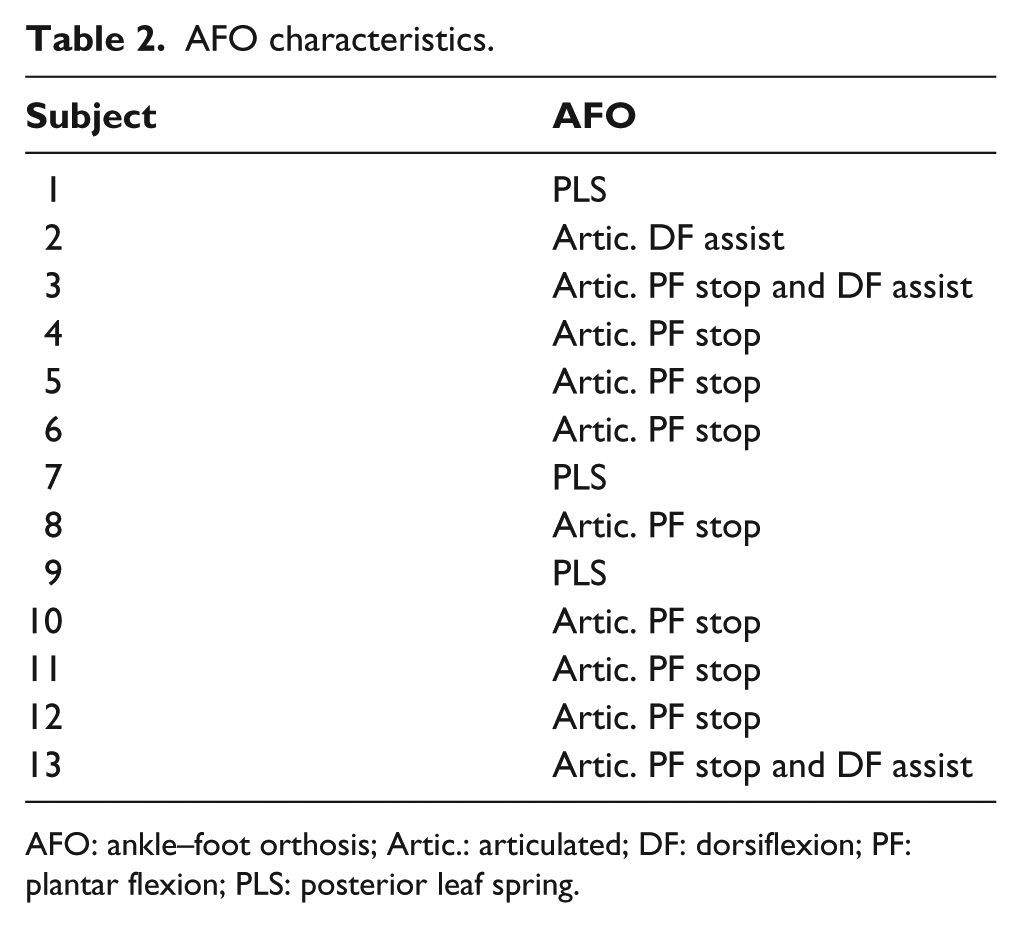
AFO: ankle–foot orthosis; Artic.: articulated; DF: dorsiflexion; PF: plantar flexion; PLS: posterior leaf spring.
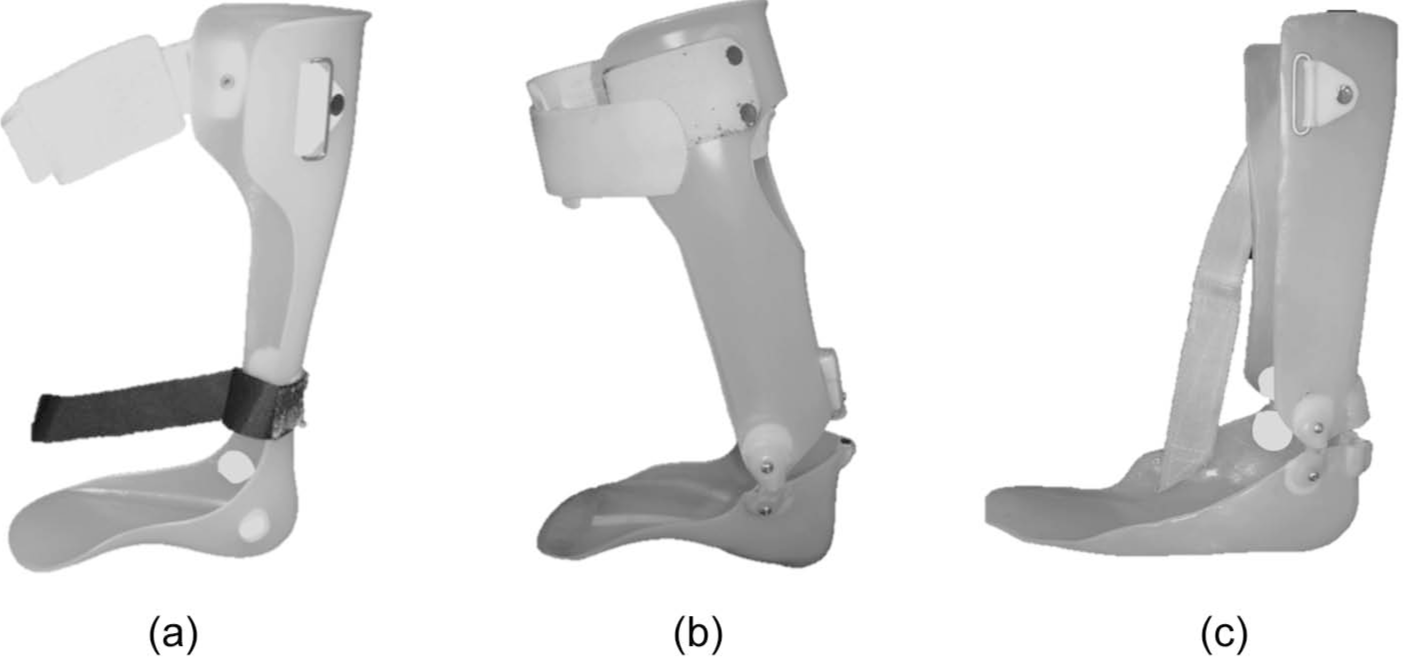
Examples of three polymer ankle–foot orthoses (AFOs) used by subjects in this study. All are considered non-rigid and include (a) posterior leaf spring (PLS), (b) articulated plantar flexion (PF) stop/dorsiflexion (DF) assist, and (c) articulated PF stop. All designs are intended to correct equinus posturing of the foot during swing phase.
There was a statistically significant reduction in ankle plantar flexion at mid-swing with AFO use (F(1, 12) = 25.192, p = 0.000), indicating that, as expected, the orthoses reduced equinovarus posturing of the ankle–foot complex at all step widths. The AFO users’ ankle joint angles at mid-swing for each step width condition are shown in Figure 2.
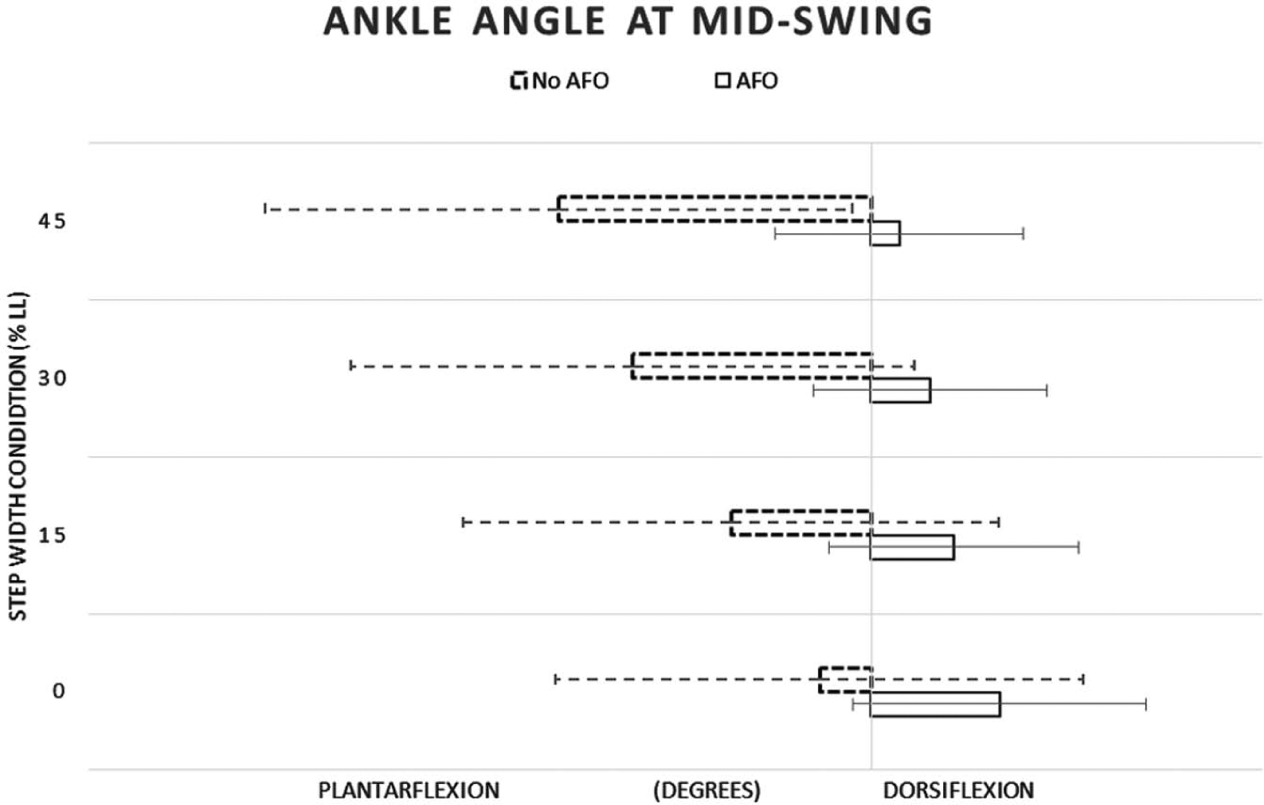
Mid-swing ankle angle with (solid box) and without (dotted box) an AFO for the affected foot.
ML foot-placement error for the affected foot is shown in Figure 3(a) and for the sound foot in Figure 3(b). The precision of foot placement, calculated as the standard deviation of foot-placement accuracy for each condition, is displayed in Figure 4. ML foot-placement ability did not differ with AFO use (F(2, 11) = 0.658, p = 0.537, Wilk’s lambda = 0.893). The step width × orthosis interaction (F(6, 7) = 1.036, p = 0.474, Wilk’s lambda = 0.53) was also not significant, indicating that the effect of the orthosis did not differ with target step width.
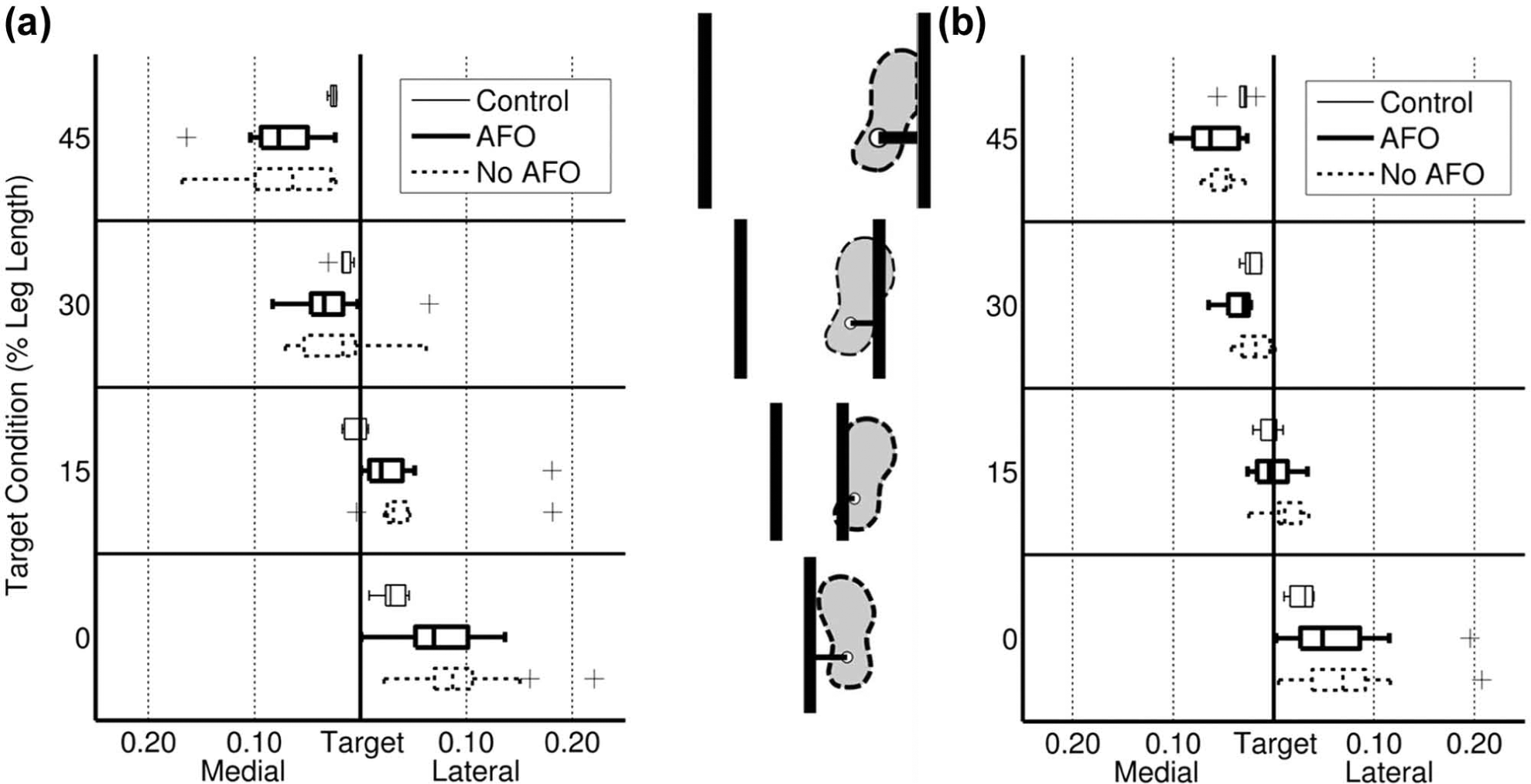
ML foot-placement errors with and without an AFO for the (a) affected and (b) sound foot. Lateral errors are plotted to the right of the target line, while medial errors are plotted to the left of the target line. The data represent ML foot-placement errors when subjects walked without an AFO (dotted lines) and with an AFO (solid lines).
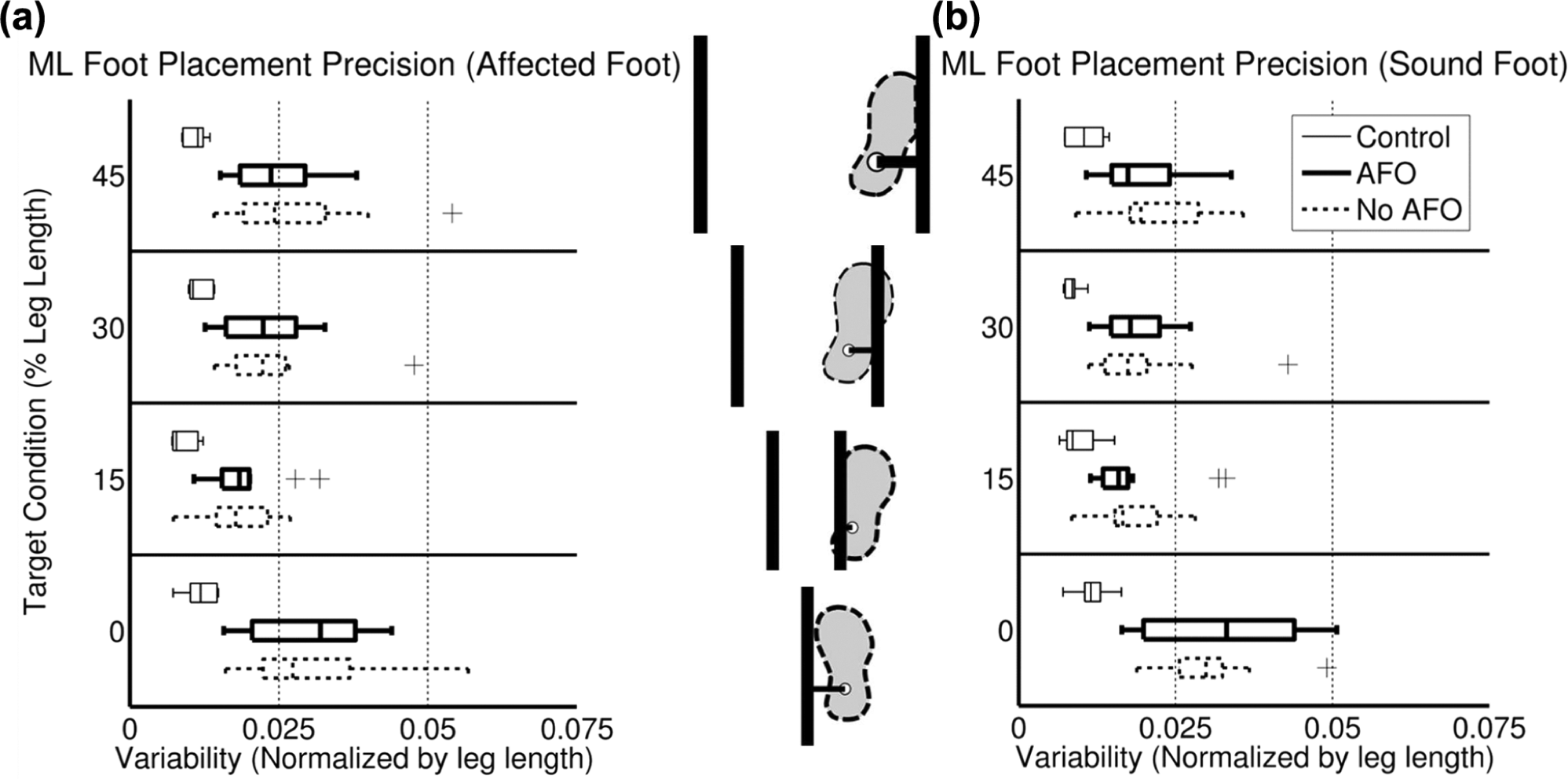
Precision of ML foot placement measured as the standard deviation of error for (a) both the affected foot (a) and the sound foot.
There was no main effect of AFO use on peak pelvic obliquity (F(1, 12) = 0.387, p = 0.545), circumduction (F(1, 12) = 2.037, p = 0.179), or hip abduction/adduction range of motion (F(1, 12) = 4.302, p = 0.06) during affected limb swing phase. These results are summarized in Figure 5.
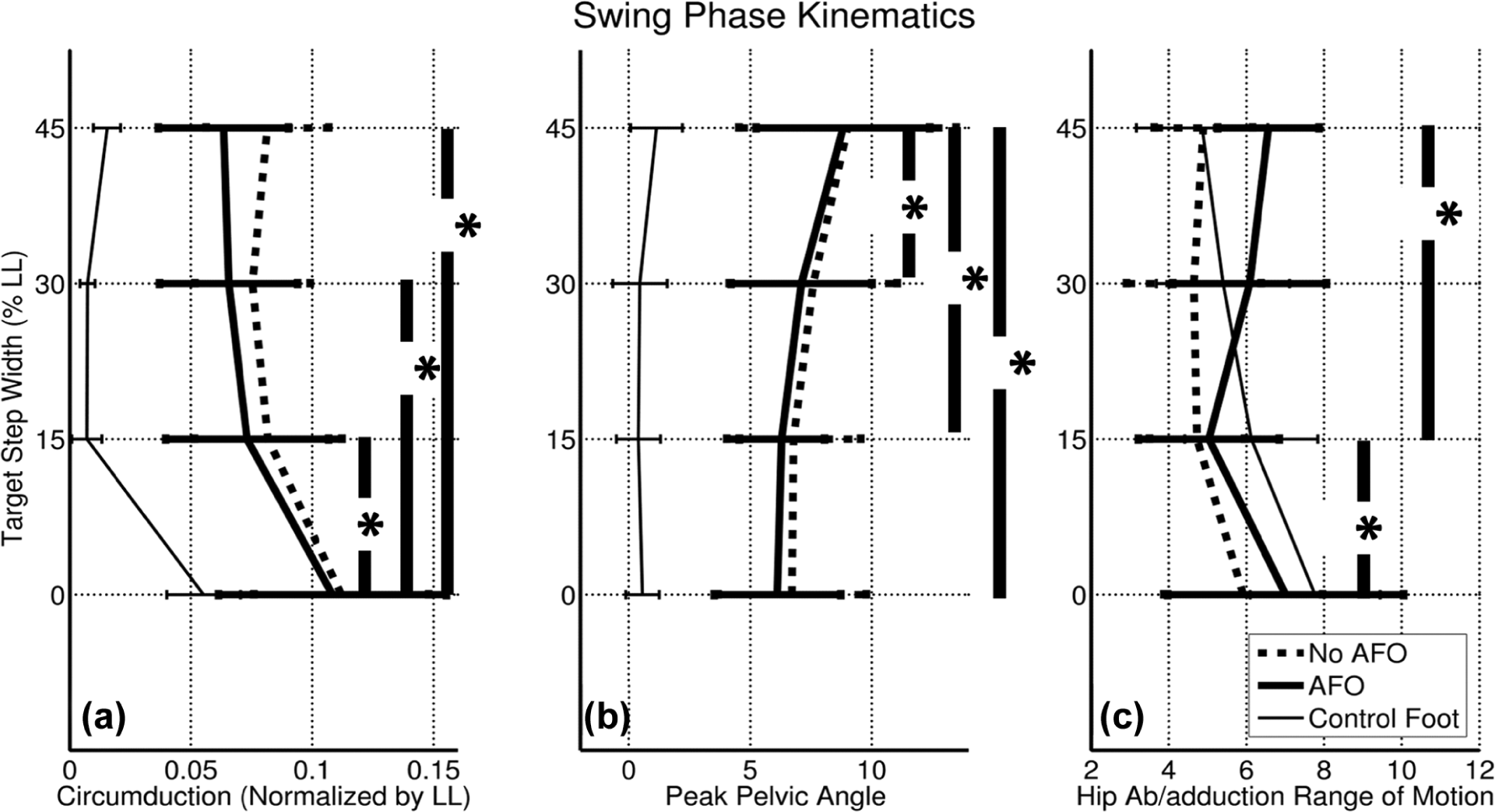
Mean (SD) of kinematic variables: (a) lateral ankle displacement (i.e. circumduction), (b) peak pelvic elevation during swing (i.e. hip hiking), and (c) hip abduction/adduction range of motion at each target step width condition (AFO: thick solid lines; no AFO: dotted lines). Control data are shown (thin solid lines) for comparison purposes.
Discussion
Accurate ML foot placement is important for safe ambulation.14,28 Abnormal frontal plane swing phase movement of the affected lower limb may hinder an individual’s ability to achieve an accurate and precise ML foot placement post-stroke. One common swing phase deficit post stroke is insufficient ankle dorsiflexion at mid-swing, a feature of gait that is important for toe clearance. Individuals often compensate by hip hiking. It has also been suggested that hip abduction is used to create swing phase toe clearance. 22 These actions result in circumduction, defined as “greater than normal coronal thigh angle during midswing of the affected limb.” 21 Swing phase toe clearance has been shown to be sensitive to ankle angle at mid-swing, 29 therefore, we hypothesized that AFO use (designed to maintain a neutral ankle position during swing phase) would reduce adaptive swing phase movements and would improve ML foot-placement ability.
While we verified that the AFO provided a neutral affected side ankle angle during swing phase, AFO use did not result in a significant decrease in hip hiking or circumduction, nor was there an effect of AFO use on hip abduction/adduction range of motion. Furthermore, ML foot-placement ability did not significantly improve with AFO use.
Previous research has suggested that AFO use is associated with a significant decrease in hip hiking. 23 However, in this study, equinovarus correction was not associated with a reduction in the magnitude of swing phase compensations. It is possible that despite achieving a neutral ankle angle, insufficient knee flexion still necessitated the use of compensatory mechanisms. Peak knee flexion was not significantly different between the no-AFO (25.0° (±10.2°), 26.0° (±8.8°), 26.0° (±9.0°), and 22.8° (±9.3°)) and AFO (25.7° (±8.4°), 27.1° (± 7.9°), 26.7° (±8.0°), and 22.4° (±7.6°)) conditions (p = 0.722) for the 0%, 15%, 30%, and 45% LL conditions, respectively. Interestingly, previous efforts to mechanically increase knee flexion in post-stroke individuals yielded more abnormal frontal plane hip kinematics. 30 Consequently, Sulzer et al. 30 suggested that these findings indicate the possibility of an altered motor template in individuals post-stroke. If an altered motor template underlies the abnormal swing phase movement patterns, the effects of AFO use on ML foot-placement ability may be limited. Such a hypothesis requires further research.
Finally, it is important to note that balance during gait is multidimensional in nature. This study focused on biomechanical contributions to dynamic balance, namely, ML foot placement. However, balance requires coordination of proprioceptive, cerebellar, vestibular, and cognitive mechanisms in addition to motor mechanisms. Using the same subjects, we conducted an investigation of the effects of AFO use on balance confidence (a cognitive mechanism) using the Activities-Specific Balance Confidence (ABC) scale, a valid and reliable measure of self-efficacy in the post-stroke population, and a significant improvement in balance confidence was detected with AFO use (p ≤ 0.01). 31 Anecdotal reports of AFO-mediated improvements in confidence have also been reported post-stroke.6,8 These findings suggest that self-perception of one’s ability may be as important as actual ability in determining functioning and mobility. 32 Therefore, an AFO may also influence mobility through improved balance confidence.
Study limitations
It is important to note that the results may not generalize to individuals with greater impairment post-stroke than those who participated in this study since foot-placement impairments will likely be more pronounced in persons with more severe hemiplegia. Additionally, the relatively small sample size of this study may limit generalizability. In this experimental design, post-stroke individuals were always aware of the target step width. Further insight into ML foot-placement ability may be gained from studies in which the ML foot-placement target is altered mid-swing, similar to the single-step study previously mentioned. 15
Conclusion
Balance is one aspect of gait that may be influenced by AFO use. However, balance during gait is multidimensional in nature. In this study, we investigated the effects of AFO use on one movement mechanism: accurate and precise ML foot placement. While AFO-mediated equinovarus correction of the affected foot and ankle was not associated with improved biomechanics of walking (i.e. proximal ipsilateral hip kinematics or ML foot-placement ability), it may affect other aspects of balance that were not tested in this study (e.g. proprioception, cerebellar, vestibular, and cognitive mechanisms).
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Conflict of interest
None declared.
Funding
This research received support from the Orthotic and Prosthetic Education and Research Foundation (Grant # RFA OPERF-2010-FA-1) (Recipient: Angelika Zissimopoulos), the Dr. John N. Nicholson Fellowship (Recipient: Angelika Zissimopoulos), and the National Institute on Disability and Rehabilitation Research (NIDRR) Department of Education (Grant # H133E080009) (Principal Investigators: Steven Gard and Stefania Fatone). The opinions contained in this publication are those of the grantee and do not necessarily reflect those of the Department of Education.
