Abstract
Background:
Obstacle crossing is an important activity of daily living, necessary to avoid tripping or falling, although it is not fully understood how transtibial amputees adapt to performing this activity of daily living following discharge from rehabilitation.
Objectives:
The objective of this study was to investigate the longitudinal adaptations in obstacle crossing in transtibial amputees post-discharge from rehabilitation.
Study design:
Longitudinal repeated measures.
Methods:
Seven unilateral transtibial amputees crossed an obstacle 0.1m high positioned along a walkway while kinematic and kinetic data were recorded at 1, 3 and 6 months post-discharge.
Results:
At 6 months post-discharge, walking velocity had increased (0.17 m.s−1) with most participants self-selecting an intact lead limb preference. During swing phase, peak knee flexion (p = 0.03) and peak knee power absorption (K4; p = 0.01) were greater with an intact versus affected lead limb preference. Having crossed the obstacle, intact limb peak ankle power generation in pre-swing (A2; p = 0.01) and knee power absorption (K3; p = 0.05) during stance phase were greater when compared to the affected limb.
Conclusions:
Obstacle crossing improved, although a greater reliance on intact limb function was highlighted. Results suggested that further improvements to locomotor performance may be obtained by increasing affected limb knee range of motion and concentric and eccentric strength of the knee extensors and flexors.
Clinical relevance
The novel objective data from this study establish an understanding of how recent transtibial amputees adapt to performing obstacle crossing following discharge from rehabilitation. This allows for evidence-based clinical interventions to be developed, aimed at optimising biomechanical function, thus improving overall locomotor performance and perhaps subsequent quality of life.
Background
Transtibial amputees must perform numerous activities of daily living (ADLs) of varying difficulty, including obstacle crossing. Obstacle crossing is an important ADL and is necessary to avoid a potential trip or fall and any subsequent falls-related injury. Therefore, the successful crossing of obstacles influences an individual’s ability to maintain independence and subsequent quality of life.
Previous studies have shown that lower limb amputees are able to negotiate obstacles successfully, albeit with an inherent degree of altered mechanical functioning.1–6 However, when compared to able-bodied individuals, transtibial amputees negotiated obstacles more slowly 4 and made contact with obstacles more often under increasing time pressure.3,5 Encouragingly, these deficits have been shown to diminish in individuals with greater time since amputation in cross-sectional studies. 3 Therefore, there is a need to monitor adaptations longitudinally.
A lead limb preference (LLP) (Figure 1) reflects an amputee’s preferred obstacle crossing strategy, and the lack of a clear LLP may be indicative of increased adaptability when performing this motor task. Equivocal findings with regards to LLP, and the potential mechanisms responsible for LLP selection, highlight the lack of a clear consensus within the literature as to the best strategy of crossing an obstacle in lower limb amputees.1,4 The propulsive mechanism achieved via ankle plantarflexion in pre-swing, prior to the limb crossing the obstacle, has been cited as a reason for choosing an intact LLP. 2 Conversely, reduced knee joint range of motion (ROM) owing to the physical constraints of the posterior shell of the prosthesis and socket fit have been proposed to reduce the suitability of an affected trail limb.1,2 When leading with the affected limb, compensatory mechanisms such as increased intact limb ankle plantarflexion and affected limb knee and hip flexion have been reported to facilitate obstacle clearance. 1 Moreover, reduced affected limb knee ROM upon landing purportedly indicates an inability to effectively control musculature about the knee in preparation for the subsequent stance phase.2,3
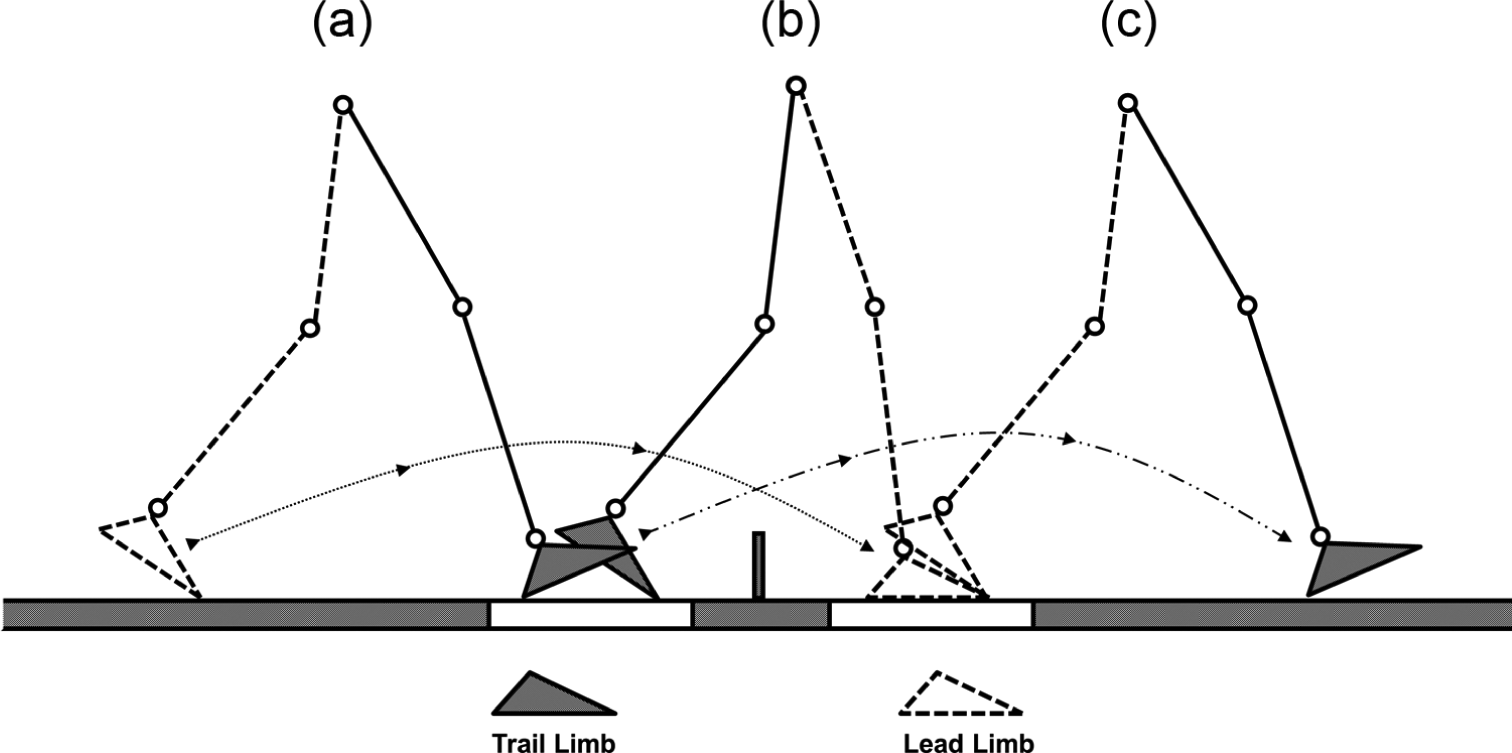
Schematic diagram of obstacle crossing with lead limb gait cycle defined from toe-off (a) to toe-off (c) and trail limb gait cycle defined from foot contact (a) to foot contact (c). Transition step represented by schematic (b) with force plate locations indicated.
Results from previous studies have been largely obtained from amputees with a number of years of prosthetic experience1–5 with few investigations assessing the longitudinal changes that occur in more recent amputees. 6 However, given that recent amputees are likely to be more receptive to adaptations to their movements, these investigations have important implications with regards to improving locomotor function, avoiding trips and falls, falls-related injuries and subsequent loss of mobility and independence in this population. The aim of this study therefore was to investigate the longitudinal adaptations in recent transtibial amputees when crossing an obstacle positioned along a level walkway, during the 6-month period following discharge from rehabilitation. It was predicted that walking velocity, an indicator of overall performance, would increase over time following discharge. 7 It was also predicted that improvements in overall performance would be due to the increased joint mobility and power bursts associated with the intact limb. Finally, it was predicted that LLP would change over time as participants adapted their movement strategies when crossing an obstacle.
Methods
Participants
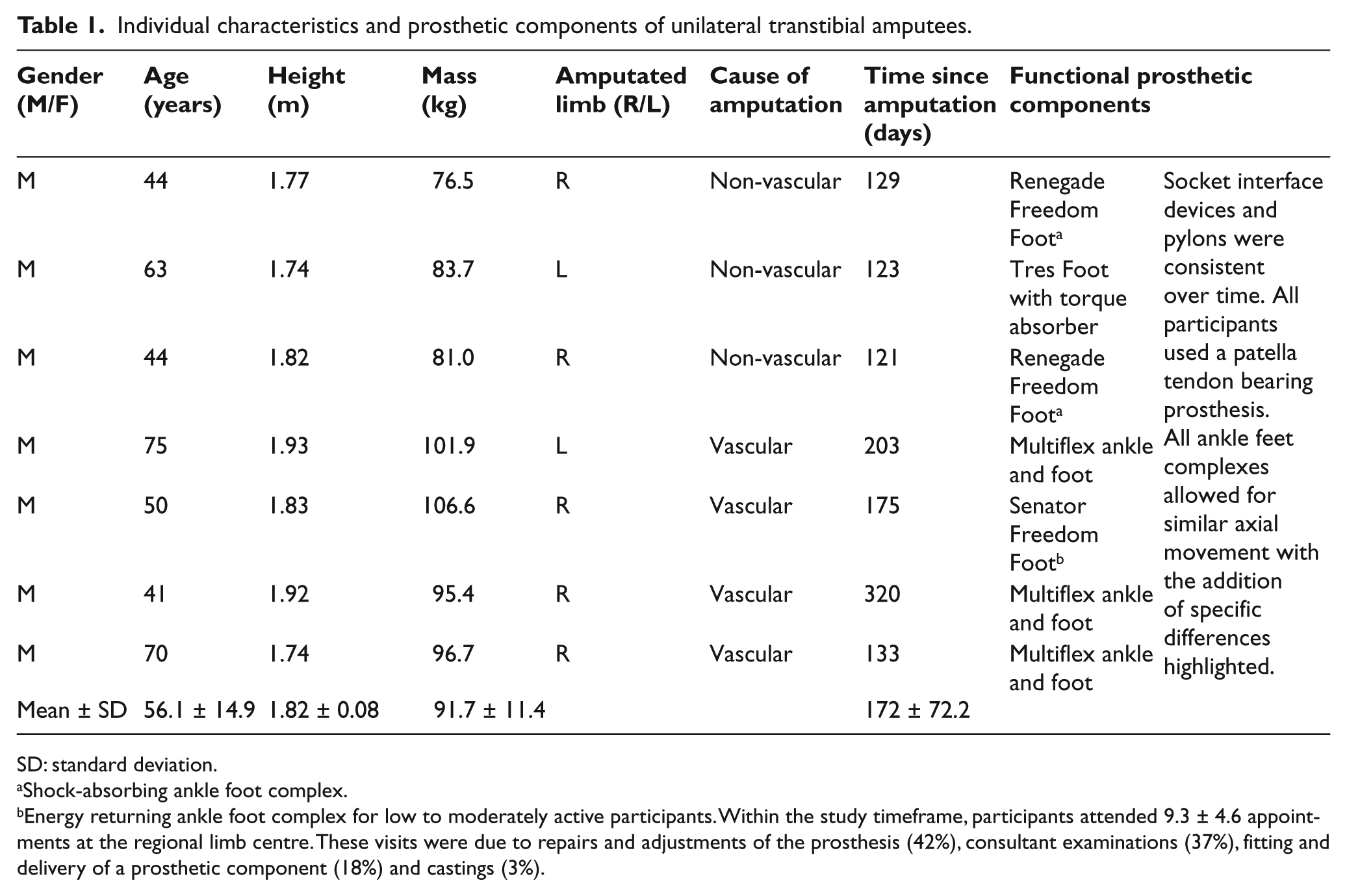
Seven unilateral transtibial amputees gave informed consent to participate in this study having completed a course of rehabilitation within a National Health Service (NHS) physiotherapy department (Table 1). Participants’ rehabilitation was conducted by the same clinicians in the same department and followed similar pathways including the initial use of early walking aids, followed by the practice of ADLs with an initial prescribed prosthetic limb. Participants were excluded if they had any current musculoskeletal injuries, cognitive deficits or experienced pain or discomfort while using their prostheses. Participants were included if they could complete a number of functional tasks without the use of a walking aid, including walking a distance of 5m and stepping over an obstacle. The study was approved by a local NHS research ethics committee (08/H1304/10).
Individual characteristics and prosthetic components of unilateral transtibial amputees.
SD: standard deviation.
Shock-absorbing ankle foot complex.
Energy returning ankle foot complex for low to moderately active participants. Within the study timeframe, participants attended 9.3 ± 4.6 appointments at the regional limb centre. These visits were due to repairs and adjustments of the prosthesis (42%), consultant examinations (37%), fitting and delivery of a prosthetic component (18%) and castings (3%).
Experimental set-up
A polystyrene obstacle of 0.1m (height) and 1.0m (width) with supporting legs was positioned between two force platforms along a 10-m walkway. Obstacle dimensions were wide enough to prevent negotiation of the obstacle by walking around it and high enough to represent items encountered on the floor during everyday living and corresponded to those previously reported.1,4,6 A 10-camera motion capture system (Qualisys, Gothenburg, Sweden) and two force platforms (Model 9281B, Kistler, Winterthur, Switzerland) sampled kinematic (100Hz) and ground reaction force (GRF) (1000Hz) data synchronously via Qualisys Track Manager software (Qualisys, Gothenburg, Sweden).
Experimental design and protocol
This study utilised a repeated measures design with participants attending standardised data collection sessions at 1, 3 and 6 months following discharge from rehabilitation. Participants wore their own comfortable, flat footwear and were able to fit and readjust their own prostheses prior to data collection. In accordance with the six degrees-of-freedom marker set,8–10 14-mm reflective markers were attached bilaterally to the calcaneus; first, second and fifth metatarsals; medial and lateral aspects of the malleoli and femoral epicondyles; greater trochanter; iliac crest and anterior and posterior superior iliac spines. Four-marker rigid clusters were attached to the thigh and shank segments. This marker set allowed for six degrees-of-freedom segmental kinematics to be recorded. 8 Marker placement on the affected limb was estimated from intact limb anatomical landmarks.7,11 A static calibration was performed by collecting kinematic data of each participant standing in the anatomical neutral position. Following completion of several practice trials, participants self-selected a starting position which was typically around 4 m from the obstacle, before walking along the walkway and stepping over the obstacle at a self-selected velocity. A minimum of five trials were recorded.
Data analysis
A large number of gait variables were computed from this analysis and key variables are presented in this study. Raw kinematic and GRF data were interpolated using a cubic-spline algorithm and filtered using a fourth-order low pass Butterworth filter in Visual 3D (C-Motion, Inc., Germantown, MD, USA) with cut-off frequencies of 6 and 30Hz, respectively. Anatomical frames were defined using medial and lateral landmarks from which segment co-ordinate systems were defined following the right-hand rule. 8 As participants crossed the obstacle, the transition steps were analysed (Figure 1). The lead limb was defined as the first limb to cross the obstacle, with the contralateral limb designated as the trail limb. Lead limb selection was not controlled for and was noted during each trial in order to assess LLP (Figure 1). Walking velocity (m.s−1) and stance duration (% gait cycle) were calculated along with joint angles (°) from the ankle, knee and hip. Peak vertical GRF was normalised to body weight (BW) with corresponding braking (Fz1) and propulsive (Fz2) peaks labelled. Normalised joint power (W/kg) data were calculated for the ankle, knee and hip with peak power burst values being presented. 12 Kinetic data were measured following obstacle crossing for the lead limb and prior to obstacle crossing for the trail limb (Figure 1). The gait cycle was normalised from toe-off to the subsequent toe-off for the lead limb and from foot contact to subsequent foot contact for the trail limb (Figure 1).
Statistical analysis
Group mean data were analysed using a linear mixed model, Limb (Affected, Intact) × Time (1, 3 and 6 months) with repeated measures on the last factor. This design allowed for the analysis of changes in multiple gait variables. 13 Each feature of the design (Time and Limb) was modelled as a fixed effect with the appropriate model being selected according to the lowest value for Hurvich and Tsai’s Criterion (Akaike information criterion–corrected (AICC)). Underlying assumptions were checked using conventional graphical methods and were deemed plausible unless stated otherwise. In the instance of a significant result, post hoc comparisons were conducted using a Sidak adjustment in SPSS v.17.0 (SPSS Inc., Chicago, IL, USA). The alpha level of statistical significance was set at p ≤ 0.05.
Results
No participants made contact with the obstacle during any visit to the laboratory. The majority of participants favoured an intact LLP, although this preference reduced by 10.6% from 68.8% to 58.2% between 1 and 6 months post-discharge (Table 2). As predicted, participants’ walking velocity when crossing the obstacle increased by 0.17 m.s−1 between 1 and 6 months post-discharge, regardless of LLP (Table 2). Intact limb stance duration was significantly greater whether it acted as the lead (p < 0.01) or trail limb (p < 0.01), with differences of 6% (lead limb) and 8% (trail limb) at 6 months post-discharge from rehabilitation.
Group lead limb preferences (A) and
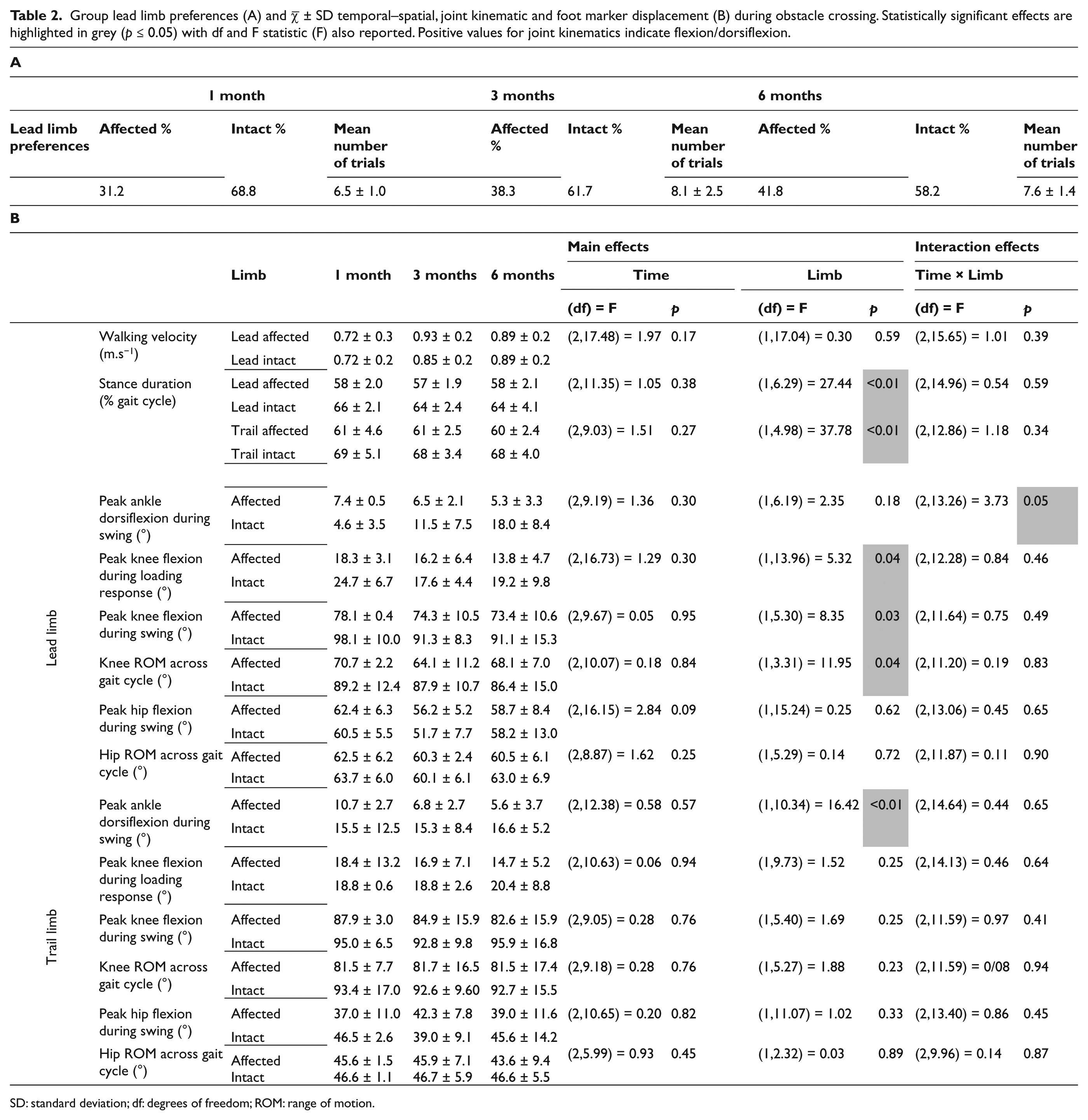
SD: standard deviation; df: degrees of freedom; ROM: range of motion.
Lead limb comparisons
A significant interaction effect was reported for peak ankle dorsiflexion during swing (p = 0.05) due to the increased ROM associated with the intact ankle joint when compared to the prosthetic ankle joint. With an intact LLP, peak knee flexion during swing (p = 0.03) and peak knee flexion during loading after touch-down (p = 0.04) were significantly greater when compared to an affected LLP (Table 2). No statistically significant effects were observed for variables pertaining to the hip in the lead limb.
Having crossed the obstacle, load rate (p = 0.05) and second peak vertical GRF (Fz2) (p = 0.03) were significantly higher when leading with the intact versus affected limb (Figure 2, Table 3). Statistically significant time main effects were reported for Fz2 (p = 0.05) and decay rate (p = 0.05) (Figure 2, Table 3). There were no statistically significant effects associated with lead limb first peak vertical GRF (Fz1).
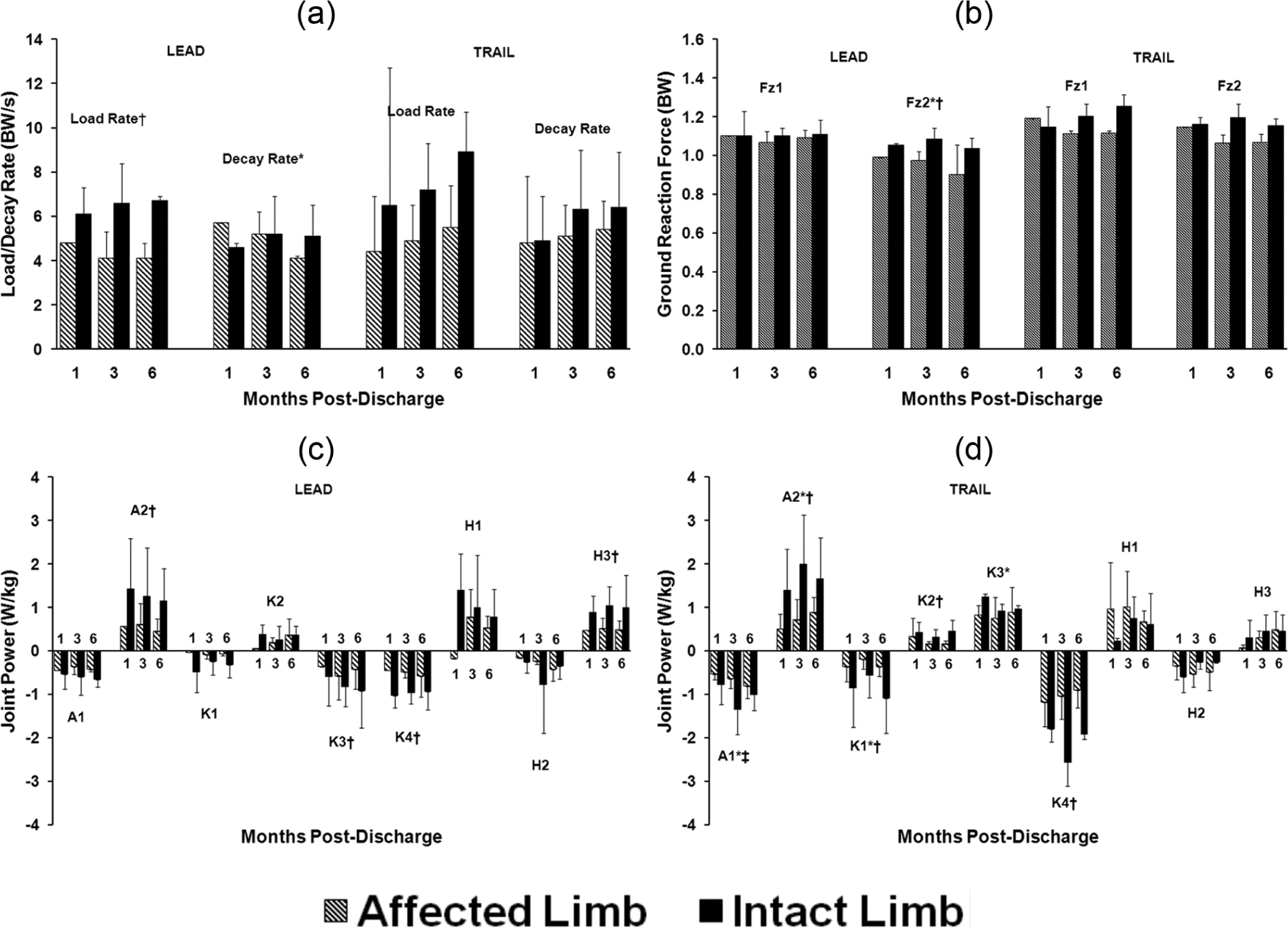
Group
Statistical analysis of group
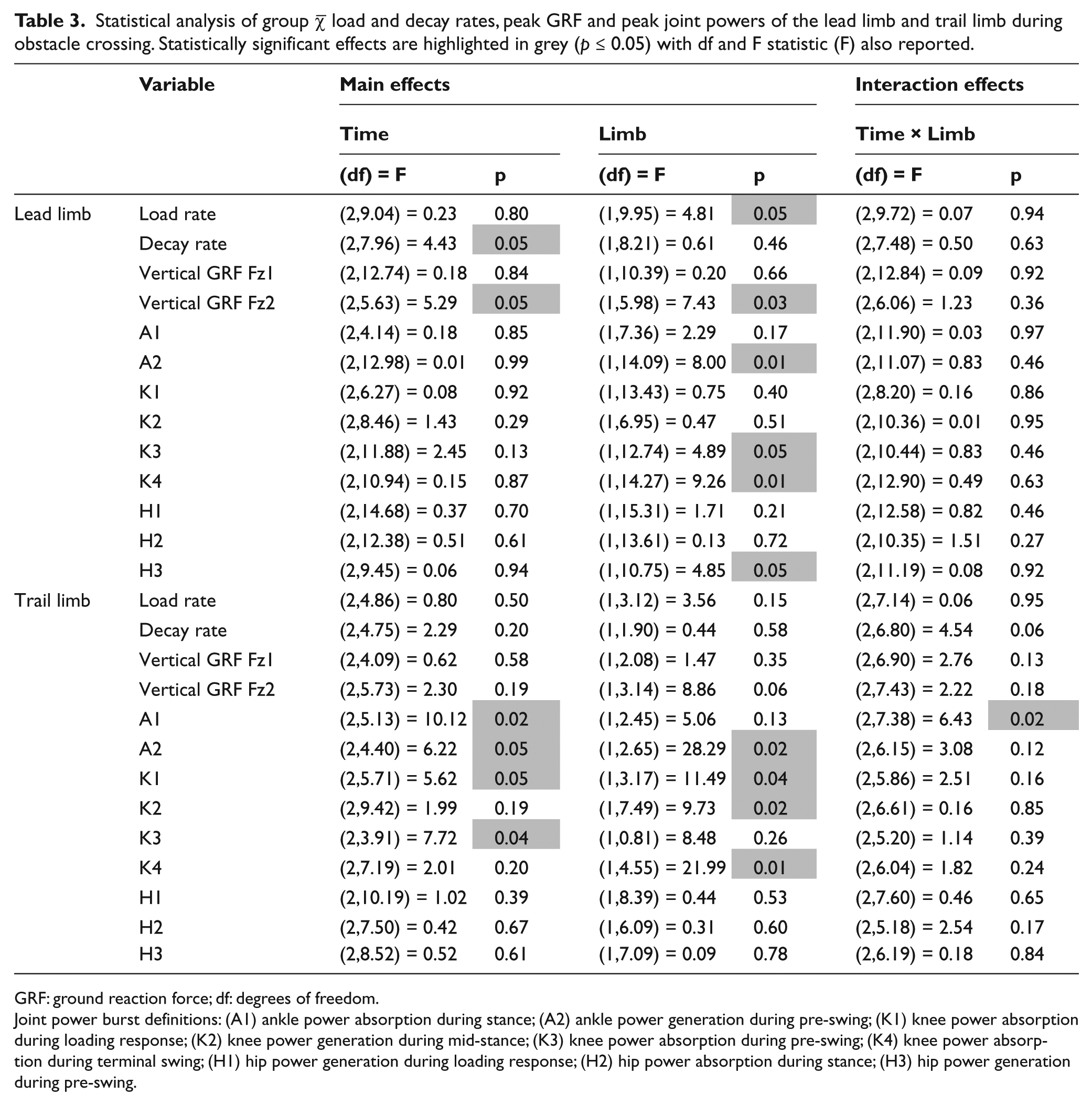
GRF: ground reaction force; df: degrees of freedom.
Joint power burst definitions: (A1) ankle power absorption during stance; (A2) ankle power generation during pre-swing; (K1) knee power absorption during loading response; (K2) knee power generation during mid-stance; (K3) knee power absorption during pre-swing; (K4) knee power absorption during terminal swing; (H1) hip power generation during loading response; (H2) hip power absorption during stance; (H3) hip power generation during pre-swing.
Peak ankle power generation (A2) (p = 0.01), knee power absorption (K3) (p = 0.05) and hip power generation (H3) (p = 0.05) during pre-swing following obstacle crossing as well as peak knee power absorption during swing (K4) (p = 0.01) prior to obstacle crossing were all higher when leading with the intact limb compared to leading with the affected limb (Figure 2, Table 3). There were no further statistically significant effects associated with lead limb peak joint power bursts.
Trail limb comparisons
During swing, as the trail limb crossed the obstacle, peak ankle dorsiflexion was greater with the intact versus affected limb (p < 0.01). No other statistically significant effects were noted for trail limb joint kinematics or variables associated with GRF.
A significant interaction effect (p = 0.02) was found for peak ankle power absorption during stance (A1) which increased steadily between 1 and 6 months when trailing with the affected limb but was reduced in magnitude when compared to the intact limb (Figure 2, Table 3). Similarly, increases observed in peak ankle power generation (A2) between 1 and 6 months were statistically significant (p = 0.05), although the magnitude of power burst A2 was consistently greater throughout with an intact versus affected trail limb strategy (p = 0.02) (Figure 2, Table 3). Peak knee power absorption (K1) (p = 0.04) and generation (K2) (p = 0.02) during early stance were greater when trailing with the intact limb versus affected limb (Figure 2, Table 3). In addition, the increase and subsequent decrease in K1 between 1 and 6 months resulted in a significant time main effect (p = 0.05) (Table 3). Changes in peak knee power generation during pre-swing (K3) were statistically significant between 1 and 3 months (p = 0.05) (Table 3). However, these changes were not uniform, with a decrease associated with an intact limb trail strategy and an increase associated with an affected limb trail strategy. There was a large, statistically significant inter-limb difference in peak knee power absorption during terminal swing (K4), due to the increased magnitude of power absorption when trailing with the intact limb (p = 0.01) (Figure 2, Table 3). There were no statistically significant effects associated with trail limb peak hip joint power bursts.
Discussion
The aim of this study was to investigate the biomechanical adaptations in recent transtibial amputees when crossing an obstacle, during the 6-month period following discharge from rehabilitation. As predicted and independent of LLP, walking velocity increased by 0.17 m.s−1 between 1 and 6 months post-discharge. While not statistically significant, this 24% increase was considered a clinically meaningful improvement in performance within the 6-month timeframe as walking velocity reflects an individual’s overall locomotor ability. 7 In addition, no trips or falls occurred during the performance of the task, which was important given the safety concerns of performing such ADLs for this group. These results corroborated findings from previous studies and the assertion that transtibial amputees are able to negotiate obstacles successfully.1–6
Results from this study suggest that the increased capacity of the intact limb to perform the role of the lead limb may explain the LLP observed. As predicted, knee flexion and power absorption during swing were greater when leading with the intact limb compared to the affected limb. This increased intact limb knee ROM and control during the approach and initial stage of obstacle crossing may reflect participants’ increased confidence of avoiding contact of the intact limb with the obstacle. Unintentional lead limb contact with the obstacle would necessitate corrective movements in order to avoid tripping or falling which may be more effective with the intact limb. Concurrently, the affected limb, which is supporting bodyweight during the critical single limb support phase, may also be required to provide corrective movements in the case of obstacle contact. Previous research has suggested that postural adjustment originating from the affected limb during stance phase may not be as complex as kinematic adjustments during swing. 3
As predicted, a number of variables indicative of stance phase function, such as stance duration, knee joint ROM, load rate and peak power generation (A2) and absorption (K3), were increased upon landing after the obstacle when leading with the intact limb. Previous literature has suggested a number of mechanisms responsible for the selection of an intact LLP such as the enhanced ability to push off with the intact limb at the end of the preceding stance phase 2 and the reduced affected limb control during swing, resulting in instability in preparation for the subsequent stance phase.2,3 This highlights the importance of the role of the lead limb having crossed the obstacle during a potentially vulnerable stage of obstacle crossing when the contralateral (affected) limb is in swing. This is an important consideration for those involved in the rehabilitation of lower limb amputee obstacle crossing as lead limb stance phase function will help prevent tripping or falling. Results from this study suggest that in the early stages following rehabilitation, the intact limb was not more accomplished or preferred in performing this role. However, circumstances may require the use of an affected lead limb strategy. Therefore, additional gains in affected lead limb function are important for further overall improvement in function and adaptability of amputee obstacle crossing performance.
Previous literature has reported equivocal findings with regards to LLP,1,4 although individual and study sample differences may partially account for these discrepancies. In this study, participants generally self-selected an intact LLP although, as predicted, there was an increase in the use of an affected limb LLP over time providing an insight into the obstacle crossing strategies of transtibial amputees. Significant time main effects were observed in ankle and knee kinetic variables during the stance phase of the trail limb, including increased power generation and absorption at the ankle. This suggests that participants improved their ability to utilise the passive function of the prosthetic ankle and active function of the biological ankle during stance which may help explain the changes in LLP over time.
The influence of rehabilitation practices must also be considered. Initially, amputees are often advised to cross obstacles leading with their ‘strongest’ limb which, during and shortly following rehabilitation, is likely to encourage an intact LLP. However, with time following rehabilitation, improved prosthetic confidence and practice of locomotor tasks, the LLP is liable to change3,7 as observed in this study, which is possibly reflective of increased and more adaptable obstacle crossing ability. These results also suggest that immediately following discharge from rehabilitation and for at least 6 months, amputees’ locomotor function is malleable and particularly sensitive to intervention, whether through formal clinical treatment or home-based activity. These novel findings advocate the importance of continuing strength and flexibility training following discharge from rehabilitation, with recommendations for follow-up visits at regular intervals to monitor progress.
Previous studies have suggested that an affected LLP allows amputees to control the limb during swing via visual feedback1,4 and provides increased time to prepare the limb for stance. 4 However, the intact LLP observed and inter-limb differences outlined in this study are in contrast to these suggestions. Despite these equivocal findings, one implication of these results is that the flexibility to adopt an affected LLP may be necessary when encountering an unexpected obstacle. Practising obstacle crossing during rehabilitation in addition to improving joint ROM, muscle strength and enhanced prosthetic design may increase amputees’ ability to perform these tasks safely and confidently.3–6 The findings of this study advocate these suggestions, which have implications for those involved in the care and rehabilitation of transtibial amputees. Given the plasticity of the locomotor system, the identification of a timeframe during which the system is more responsive to further change could be very important for improving an amputee’s confidence and performance of more complex ADLs. This may in turn help reduce the intact LLP bias established during rehabilitation and thereby improve the ability to cross unexpected obstacles safely and reduce the potential for subsequent falls and falls-related injury. Future investigations should focus on examining the effects of interventions, such as advanced rehabilitation or home-based therapy, aimed at improving affected limb strength, on amputees’ performance of complex ADLs following discharge from rehabilitation when amputees’ motor patterns are more receptive to change.
The results from this study have highlighted a number of possible mechanisms that lead to the establishment of an intact LLP and have outlined the key role played by the intact limb in the 6-month period following rehabilitation. However, limitations of this study must be acknowledged. Several variables were adapted favourably and often improved in the 6-month period following discharge, with some of these effects being statistically significant. This was encouraging in that performance of obstacle crossing improved without the specific clinical interventions or guidance advocated in this study. However, the magnitude of time main effects was not as great as the limb main effects. It is likely that the relatively small sample size and subsequently reduced statistical power may have resulted in the more subtle changes over time not reaching statistical significance. In addition, it could be suggested that the variation in the cause of amputation may have introduced some additional variance in the measures reported. However, more recent amputees are likely to still be adapting to the novelty of the mechanical constraints of the lower limb in the 6 months following discharge from rehabilitation. Participants in this study had an amputation related to either traumatic or vascular reasons, and irrespective of cause, lower limb amputees are likely to be responsive to further treatment in the 6 months following discharge from rehabilitation. Future research should attempt to investigate the long-term adaptations in function of lower limb amputees secondary to a range of causes, as this information would be valuable to those involved in the care and rehabilitation of lower limb amputees by highlighting cause-specific patient requirements. Finally, participants were discharged from rehabilitation once they had achieved the individual goals established with their care team and had a comfortable level of function. This process varies in length of time and number of treatments depending on the individual. However, as this is more reflective of the population’s experience, the results are more generalisable to the wider amputee population.
Conclusion
Despite the greater reliance on intact limb function, changes in walking velocity, LLP and lower limb kinetics suggested that obstacle crossing in the current participant group improved over 6 months with inter-limb biomechanical mechanisms being highlighted. In the 6-month period following discharge from rehabilitation, amputees may be positively susceptible to further improvements in performance and prosthetic confidence. The findings from this study suggest that the introduction of obstacle crossing during rehabilitation, improvements to prosthetic design and therapeutic interventions addressing the joint ROM and limb strengthening issues may help improve amputees’ capacity when performing obstacle crossing.
Footnotes
Acknowledgements
Authors would like to thank Mrs Amanda Hancock, Mrs Barbara Brown and Mrs Lynne Smith for their help during patient recruitment.
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
This study was financially supported by the Owen Shaw Award from the Circulation Foundation, UK.
