Abstract
Background: Effects presented on the use of assistive devices such as prosthesis are often based on laboratory findings (i.e. efficacy).
Objectives: To summarise and evaluate findings from studies on effectiveness of lower limb prostheses for adults in real life contexts, primarily in terms of activity, participation, and quality of life (QoL) and secondarily in terms of user satisfaction, use/non-use, and/or cost-effectiveness.
Study Design: Systematic review.
Methods: We included controlled studies and non-controlled follow-up studies including both baseline and follow-up data. Using 14 different databases supplemented with manual searches, we searched for studies published from 1998 until June 2009.
Results: Out of an initial 818 identified publications, eight met the inclusion criteria. Four studies reported on the effectiveness of a microprocessor-controlled knee (MP-knee) compared to a non-microprocessor-controlled knee (NMP-knee). Results were inconsistent except for quality of life and use/non-use, where the authors reported an improvement with the MP-knee compared to the NMP-knee. The remaining four studies included a diversity of prosthetic intervention measures and types of endpoints.
Conclusions: Overall, there was an inconsistency in results and study quality. This review highlights the need for high-quality research studies that reflect the effectiveness of different prosthesis interventions in terms of users’ daily living and QoL.
Clinical guidelines are important to every practitioner. Information on expected effectiveness from assistive devices should be well founded and contain both facts about the device quality and its contribution to users’ daily lives. Thus, studies based on users’ experiences from prosthetic use in everyday life activities are of great importance.
Keywords
Background
Affecting many daily activities, amputation is a life-altering event with an immediate and obvious effect on a person’s mobility, including many daily life activities. During the last decades, new metals and plastics, as well as computer-aided design and new developments in microprocessors, have significantly affected the evolution of lower limb prosthetics, making it possible for amputees to be more independent and physically active.1,2 Nevertheless, earlier studies have reported that a majority of amputees do not consider prosthetic devices useful because of pain, discomfort or misfit.3,4 Karmarkar et al. 5 reported that 31% in a group of veterans with lower-limb prosthesis used wheelchairs as their primary mobility device.
The scientific literature has paid much attention in describing new technical solutions to optimise prosthesis function, e.g. microprocessor-controlled prosthesis, new orthopaedic surgery methods, such as the osseointegrated prosthesis,6,7 new socket solutions, and custom-designed solutions. 1 However, there seems to be a lack of knowledge in how a prosthesis system affects a person’s daily life in the user’s natural context; consequently, there is a need to gather complementary evidence on device effectiveness to enrich best clinical practice. 8 Clinicians and policymakers often distinguish between the efficacy and the effectiveness of an intervention. Efficacy trials determine whether an intervention produces the expected result under ideal circumstances while effectiveness trials measure the degree of beneficial effect under ‘real world’ settings. 9
Thus, the primary purpose of this review is to summarise the current state of knowledge about how lower limb prosthesis for adults can affect activity, participation, and quality of life (QoL) in real life contexts. The secondary purpose of this review is to summarise the state of knowledge concerning user satisfaction, use/non-use, and/or cost-effectiveness when investigated together with primary outcomes.
Methods
This study is a systematic review. Controlled studies and non-controlled follow-up studies, provided that the latter includes both baseline and follow-up data, were included. The focus was on effectiveness studies, thus results collected in patients’ natural contexts. Data from laboratory settings (efficacy studies) or controlled studies, including a population without amputation as controls, were excluded. The study population included lower limb prosthetic users over 18 years of age according to ISO 9999 classification 10 : 062403, partial foot prosthesis; 062406, ankle disarticulation prosthesis; 062409, transtibial (below-knee) prosthesis; 062412, knee disarticulation prostheses; 062415, transfemoral (above-knee) prostheses; or 062418, hip articulation prostheses. The control intervention should include some other prosthesis system or no prosthesis system.
We considered studies focusing primarily on activity and/or participation and/or QoL outcomes. We also considered studies on user satisfaction, use/non-use, and costs outcomes, which we, however, considered to be secondary. That is, we did not include studies that solely reported outcomes based on these dimensions.
Search strategy
The authors selected keywords and synonyms based on the inclusion criteria in collaboration with a research librarian. The search was performed without language restrictions in the following databases: DARE, Cochrane, CIRRIE, Cambell, OVID MEDLINE, ASSIA, PEDro, OTDatabase, Cinahl, OT seeker, Embase, PsychINFO, Agelina, and Social Abstracts. The OVID MEDLINE search strategy was based on the following terms:
Leg prostheses/lower limb prosthetic system/prostheses/partial foot prosthesis/ankle disarticulation prostheses/ transtibial prostheses/knee disarticulation prostheses/transfemoral prostheses/hip articulation prostheses/Amputation/anomalia/Outcome/effect/cost/cost-effectiveness/Use/non-use/ Mobility/activity/occupation/participation/user satisfaction/quality of life/transportation/walking/moving around/running/sports/ADL/PADL/IADL/Assessment/outcome assessment/evaluation/measurement/score/scale/test/Randomized controlled trial, random/cohort studies, follow-up studies, pre-post studies.
The strategy was modified for the other database searches. A manual search of AAATE conference proceedings, RESNA proceedings, and the Assistive Technology and Disability journal completed the searches in different databases. Original studies, as well as reviews, were searched to find possible secondary sources. The search covered January 1998 until June 2009.
Study selection
Two authors (KS and OT) independently went through titles and abstracts of the identified studies and excluded duplicates and studies that obviously were not in accordance with the inclusion criteria. Then, potential studies identified were ordered and read in full text. Next, the same two reviewers independently suggested final inclusion or exclusion for further analysis. In case of disagreement, consensus was sought through discussion or by consulting the other two reviewers. The data extraction followed a defined and formerly used structure,11,12 including a description of the design, methods (Table 1), used instruments (Table 2) and results (Table 3).
Study design, follow-up time, interventions, participants, inclusion/exclusion criteria, and selection in the included studies.

DMERC, Durable Medical Equipment Regional Carrier; Excl, Exclusion criteria; ICEX TBS, Total surface-bearing socket; Incl, Inclusion criteria; MFCL, Medicare Functional Classification Level; MP, Microprocessor control prosthesis; NA, Not applicable; NAIL, Italian Workers’ Compensation Authority; NMP, Mechanical control prosthesis; NR, Not reported; OPRA, Osseointegrated Prostheses for the Rehabilitation of Amputees; PTB, Patellar tendon-bearing socket; TFA, Trans-femoral amputees; TTA, Transtibial amputees; VAPSHCS, Veterans Affairs Puget Sound Health Care System.
Outcome evaluation instruments used.
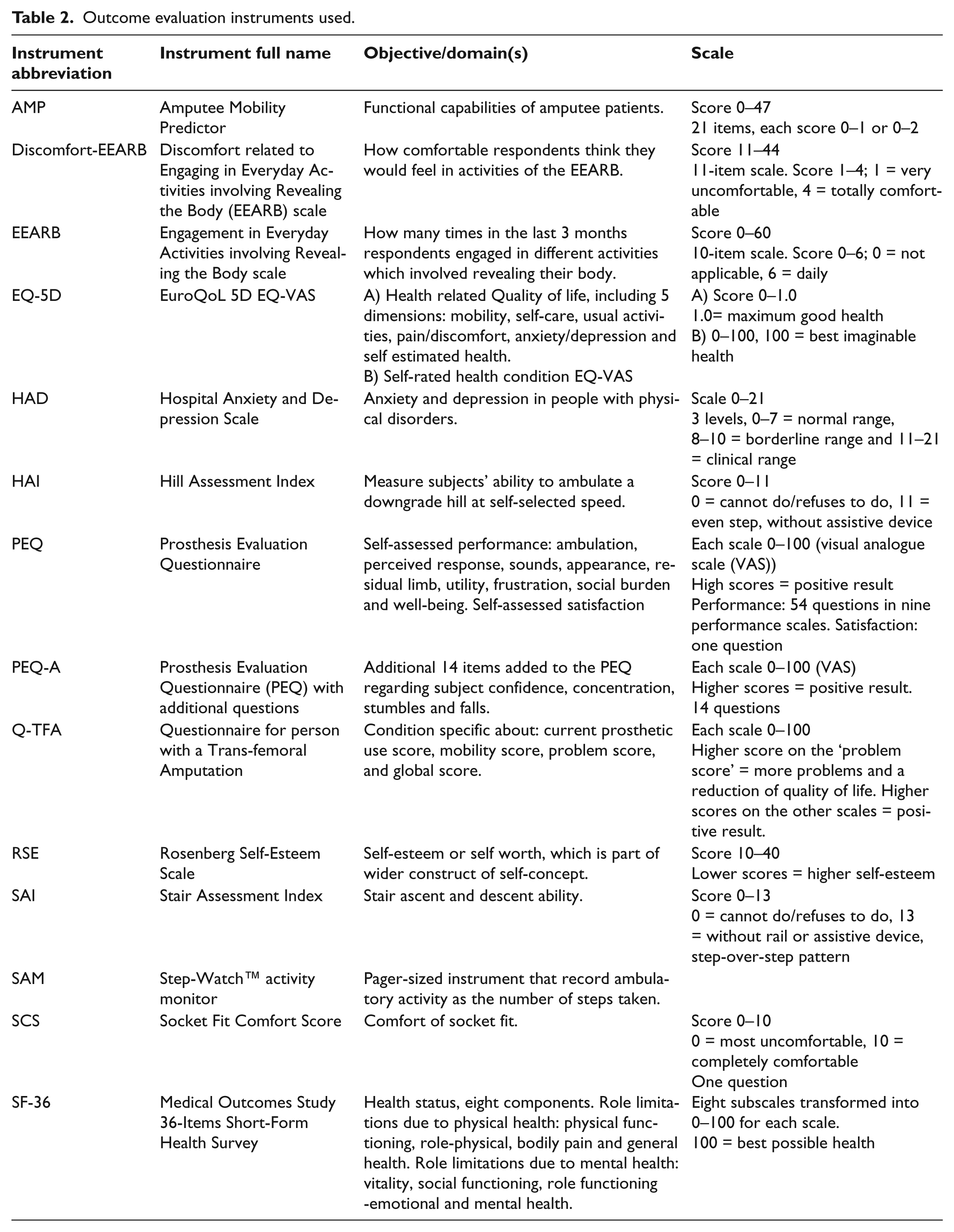
Effectiveness of lower limb prosthesis.

For abbreviations of used instruments and scales, see Table 3. Discomfort-EEARB, Discomfort related to Engaging in Everyday Activities involving Revealing the Body scale; EEARB, Engagement in Everyday Activities involving Revealing the Body scale; EQ-5D, EuroQoL 5D; FPWS, Fastest possible walking speed; HAD; Hospital Anxiety and Depression Scale; HAI, Hill Assessment Index; HRQL, Health Related Quality of Life; MRPP PCS: Montreal Rehabilitation Performance Profile Performance Composite Score; MP, Microprocessor knee; NMP, Mechanical control knee; NR, Not reported; OI-prostheses, Osseointegrated trans-femoral amputation prostheses; PEQ, Prosthesis Evaluation Questionnaire; PEQ-A, Prosthesis Evaluation Questionnaire with additional questions; QALY, Quality adjusted life years; Q-TFA, Condition specific outcome measure that reflects current prosthetic use, mobility, problems and global health; SAI, Stair Assessment Index; SAM, Step-Watch™ activity monitor; SCS, Socket Fit Comfort Score; SF-36, Medical Outcomes Study 36-Items Short-Form Health; Survey SSWS, Self-selected walking speed.
Assessment of study quality
The finally included studies were read, and all four authors assessed study quality. The internal validity of the studies was assessed by a modified criteria list adopted from Borghouts et al. 13 The assessment included a yes/no option to describe the following: a sufficient description of the population selection; inclusion and exclusion criteria described; sufficient study size (> 10 patient years); follow-up > four months; dropouts < 20%; dropouts described; outcome measures and data presentation congruent with the study aims; confounder adjusted in the analysis; and psychometric properties of the instruments reported. The modification of the original assessment schedule meant that the two latter criteria replaced the original criterion: appropriate analysis technique. This modified version has been tested in another recently published review. 11 Criteria for study-size, follow-up, and dropouts were applied (Table 4). The yes/no was transferred into 1/0, making it possible to summarise a score representing internal validity of the included studies (internal validity max score = 10 points).
Summary of internal validity assessment of included studies (modified from Borghouts et al. 1998).*
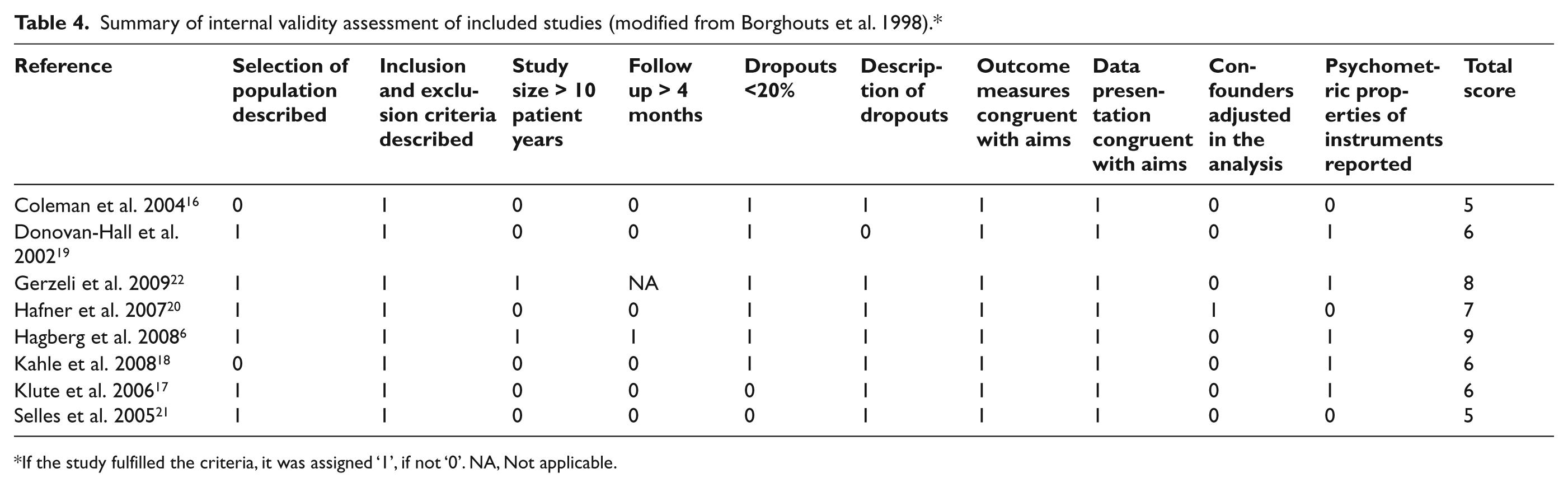
If the study fulfilled the criteria, it was assigned ‘1’, if not ‘0’. NA, Not applicable.
The external validity and applicability was assessed using a modified version of a method described by Shekelle et al. 14 From the original criteria, we replaced one question – treatment benefits in relation to adverse effects – with another question that we considered to be more relevant to the study – effect gain of >10% – adapted from a study on research related to back pain and functionality. 14 Thus, the assessment included a yes/no transferred into 1/0 (maximum score = 4 points) to describe the following: study participants and interventions described in detail; clinically relevant outcomes measured and reported; and size of the effect clinically important (Table 5).
Quality assessment of controlled studies (modified from Van Tulder 2003).

Was the method of randomisation adequate? Yes/No/Don’t know*, 2. Was the treatment allocation concealed?, 3. Were the groups similar at the baseline regarding the most important prognostic indicators?, 4. Were co-interventions avoided or similar?, 5. Was the compliance acceptable in all groups?, 6. Was the timing of the outcome assessment in all groups similar?, 7. Did the analysis include an intention-to-treat analysis?
Furthermore, the quality assessment of the randomised controlled trial (RCT) was based on a modified version of Van Tulder et al. 15 From the original 11 questions, we chose the seven aspects considered most relevant for our purpose (Table 6): the method of randomization was adequate; the treatment allocation was concealed; the groups were similar at baseline; co-interventions were avoided or similar; the compliance was acceptable; the timing of the outcome assessment was similar; and whether the analysis included an intention-to-treat analysis.
Summary of external validity and applicability assessment of included studies (modified from Shekelle et al. 1994).
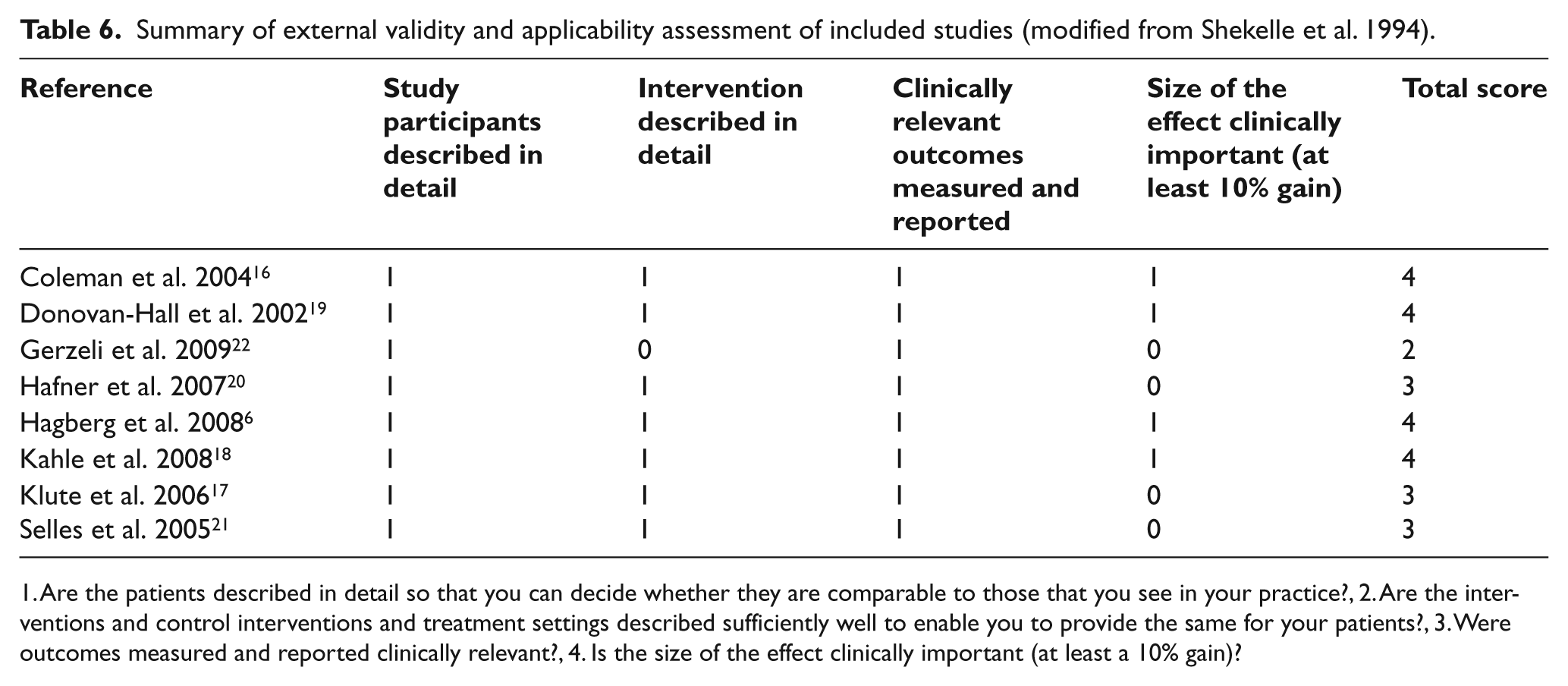
Are the patients described in detail so that you can decide whether they are comparable to those that you see in your practice?, 2. Are the interventions and control interventions and treatment settings described sufficiently well to enable you to provide the same for your patients?, 3. Were outcomes measured and reported clinically relevant?, 4. Is the size of the effect clinically important (at least a 10% gain)?
Results
The initial search resulted in 818 titles (Figure 1). After going through the abstracts, 737 studies were excluded due to not fulfilling the inclusion criteria or being duplicates. The remaining 81 studies were read in full text and another 73 studies were excluded due to not fulfilling the inclusion criteria. The remaining eight studies were controlled studies, all including outcomes of a lower limb prosthetic intervention in terms of activity and/or participation and/or QoL.
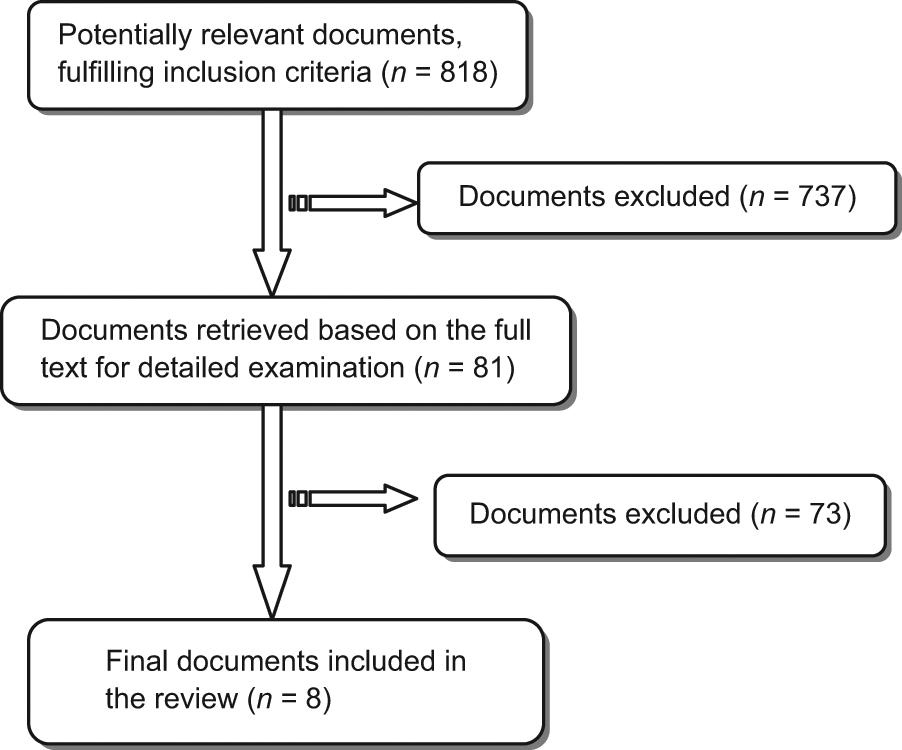
Flowchart illustrating the literature selection process.
Study design and size
All studies were published within the last nine years. Of the eight included studies, seven were experimental6,16-21 and one was an observational study 22 (Table 1). The only RCT-study was the one presented by Selles et al. 21 Two studies had a cross-over design16,17 and one a controlled reversal design. 6 Three studies included a pre/post-design18-20 and one a cross-sectional case-control design. 22 A within-group comparison was used in six studies; in the remaining two, a between-group comparison was used.21,22 The entire study-population included 219 persons (range 10–100 persons), where 158 were transtibial amputees and 61 were transfemoral amputees. Mean age was 48.5 years with a range from 18–83 years.
Type of intervention and control intervention
Seven of the eight studies included a new prosthesis intervention compared to another prosthesis intervention, which, in most cases, was the person’s ‘old’ prosthesis system (Table 1). One study included a comparison of two patient groups already fitted with two different types of prosthesis. 22 The most common type of intervention was the MP-knee, which was compared to a mechanical controlled knee – a NMP-knee.17,18,20,22 Klute et al. 17 also included a comparison between a shock-absorbing pylon prosthesis compared to rigid pylon prosthesis. The remaining four studies concerned different prosthesis interventions: a silicon cover prosthesis compared to a non-silicon prosthesis 19 ; an OI-prosthesis compared to a conventional socket prosthesis 6 ; an elastomeric suspension liner with distal locking pin compared to a liner with neoprene suspension sleeve 16 ; and a total surface-bearing socket compared to a conventional patellar tendon-bearing socket. 21
Outcome evaluation instruments
The included studies reported results from 14 different instruments for outcome evaluation (Table 2). A majority of the instruments focused on activity (n = 8) and/or QoL (n = 8). Some of the instruments targeted user satisfaction and prosthesis use, and one instrument described utility in relation to costs. Another instrument was about risk factors and some instruments included questions from more than one area.
Activity and participation outcomes
Five studies reported significant improvements in activity and/or participation (Table 3).6,16,18,19,20 Neoprene suspension sleeve increased time spent on activities/day. 16 Using prosthesis with silicone cover showed an increase in engagement in predefined everyday activities involving revealing the body. 19 Outdoor mobility was found to improve with the OI-prosthesis 6 and Hafner et al. 20 found that activities that require divided attention such as stair and hill descents went faster with the MP controlled knee-prosthesis compared to the NMP prosthesis. Hafner et al. 20 as well as Kahle et al. 18 found that the frequency of stumbles and falls decreased with the MP-knee. In two studies, the authors reported no effects from intervention on activity and/or participation. Selles et al. 21 compared a total surface-bearing socket with a patellar tendon-bearing socket and found no difference in mobility-related activities. Klute et al. 17 could not demonstrate any difference in level or duration of activity when comparing a shock absorbing versus rigid pylons prosthesis or an MP-knee versus a NMP-knee.
Quality of life outcomes
An increase in aspects related to QoL was reported in four studies (Table 3).6,18,20,22 The intervention in one of these studies was the OI prosthesis 6 and in the other three studies the MP knee. No difference in QoL was reported in the study by Hafner et al. 20 except for one issue – frustration with falling – for the benefit of the MP-knee. Donovan-Hall et al. 19 reported no change in terms of self-esteem, anxiety or depression when using a silicon-covered prosthesis compared to non-silicon prosthesis.
User satisfaction, use/non-use, and costs
Four studies reported results in terms of user satisfaction.16,18,20,21 Two of those studies18,20 reported on satisfaction with the MP-knee versus the NMP-knee. In both studies, a majority of subjects were more satisfied with the MP-knee. Selles et al. 21 reported no difference in user satisfaction with a total surface-bearing socket compared to a conventional. Coleman et al. 16 reported no difference in user satisfaction with the Alpha® versus the Pe-LiteTM liner.
Hagberg et al., 6 Coleman et al. 16 and Hafner et al. 20 all reported on results in terms of use/non-use. An increased use of the OI-prosthesis system compared to the ‘old system’ was reported by Hagberg et al., 6 and Hafner et al. 21 demonstrated an increase use of the MP knee compared to the NMP-knee. Coleman et al. 16 reported more time was spent wearing the Pe-Lite limb compared to the Alpha®.
Gerzeli et al. 22 and Selles et al. 21 included information about costs/cost-effectiveness. Gerzeli et al. 22 analysed cost-utility showing a gain in a five-year healthcare system perspective of 0.09 QALY (Quality Adjusted Life Years) for the MP-knee at a cost of €35.971 per QALY, which should be considered as cost effective. From a societal perspective with respect to costs, there was no difference between interventions in a five-year perspective. Selles et al. 21 presented a comparison of costs for two kinds of sockets; the study reported on significantly higher cost for materials, a shorter manufacturing time, and fewer visits for the total surface bearing socket group compared to the conventional patellar tendon-bearing socket group.
Quality assessment
Internal validity
The evaluation of internal validity resulted in scores ranging from 5–9 (Table 4). The two studies with the highest scores had 8–9/10 ‘yes’.6,22 Inclusion and exclusion criteria were described in all studies. Outcome measures as well as data presentation were considered to be congruent with aims in all studies. Only two studies fulfilled the study size criterion (> 10 patient years),6,22 and one study fulfilled the criterion of follow-up time (> 4 months). 6 The only RCT study 21 fulfilled four of the seven quality assessment criteria (Table 5).
External validity and applicability
The external validity and applicability evaluation process resulted in scores ranging from 2–4 (Table 6). All studies were considered to have ‘study participants described in detail’ and ‘clinically relevant outcomes measured and reported’. Four studies were considered to fulfil the criterion ‘size of the effect clinically important (at least 10% gain)’,6,16,18,19 and only one study did not fulfil the criteria ‘intervention described in detail’. 22
Summary of results
Types of prosthesis interventions as well as assessment tools varied widely in included studies. For example, four of the studies were unique in being the only study reporting on a particular kind of intervention and consequently it was not possible to summarise any outcome from those interventions (Table 1 and 3). Still, four studies compared the MP-knee and the NMP-knee.17,18,20,22 Regarding activity, participation and user satisfaction, the results of these studies were inconsistent; however, in one study the MP-knee was found to decrease stumbles and falls 18 and in another study the MP-knee was found to improve the patient’s ability to descend stairs and hills. 20 Klute et al. 17 reported no difference in terms of daily activity level or duration in transfemoral amputees with MP knee versus NMP knee. As to user satisfaction, the MP-knee was rated higher compared to the NMP-knee in two studies.18,20 All three studies that reported results in terms of QoL presented improvement with the MP-knee.18,20,22 Hafner et al. 20 reported that the MP-knee was used more often than the NMP-knee. Gerzeli et al. 22 included an analysis of cost-utility. From a healthcare perspective, the MP-knee was considered to be cost effective, expressed in terms of QALY. When non-healthcare costs and productivity losses were included, costs did not differ between interventions.
The quality of the included studies varied. The internal validity ranged from 5 to 9 (max 10), and only the study by Hagberg et al. 6 fulfilled the criterion of follow-up time (> 4 months). However, inclusion and exclusion criteria were described in all studies. The external validity and applicability ranged from 2 to 4 (max 4). Four studies were considered to fulfil the criterion ‘size of the effect clinically important, at least 10% gain’.6,16,18,19
Discussion
Geil wrote; ‘Evidence-based practice combines a practitioner’s training and experience with evidence established through scientific research’. 23 This systematic review identifies research presented in scientific literature on effectiveness of lower-limb prosthesis and assesses the quality of the identified research. The target-group for the review was adults and the effects from prosthetic interventions was to be presented as change in activity, participation, and/or QoL as primary outcome variables, and user satisfaction, use/non-use in real life contexts, and cost-effectiveness as secondary outcomes. Results from a well-conducted review should help develop clinical recommendations based on evidence on effectiveness, i.e. outcomes in real life context. 9 Contrary to what we had hoped, we could not, in light of the findings presented, provide any clear recommendations based on available evidence. The small number of studies, varying quality and the variety of prosthetic systems presented, together with a diversity of outcome instruments, make clinical recommendations based on those results unreliable. Although some of the included studies were considered to have high internal and external validity, there were obvious shortcomings in several studies as regards to study size, which only exceeded 26 participants in one study, and follow-up time, which was less than four months in all but one study (Tables 4–5). The RCT study by Selles et al. 21 was found to have shortcomings in one vital aspect – method of randomization (Table 5).
The selection process is a critical phase in a review. To increase the reliability and provide valid results, two researchers independently scanned the search results and selected studies for inclusion and exclusion. The authors of this study are all rehabilitation professionals, but with only little experience in the prosthetic field. This could be considered a weakness, since there was little pre-understanding and prior knowledge in the field of prosthetics and consequently no prior estimations on the scope of findings. However, all authors are experienced in studying outcome from other kinds of assistive technology and in doing reviews in this field.11,12 Internal and external validity as well as applicability and quality assessment were discussed in the group of researchers until consensus was reached. The included methods for assessment have all been used in two former reviews conducted by the same Nordic therapists’ research team.11,12 The overall assessment process was thus well established.
We are fully aware that our review presents significant challenges because it focuses on how a specific intervention such as a lower-limb prosthesis intervention could affect the daily life of its users (effectiveness) and excludes studies that report effects from data collected in a standardised environment or activity (efficacy). However, it is very important to gain more knowledge about how an assistive device, such as prosthesis, affects patients who have left hospital care and are living in their natural contexts. Based on this review, it seems to be a great challenge to perform well-designed studies that can deliver evidence in terms of effectiveness, in the AT field.
By evaluating existing research in a systematic and explicit way, clinical guidelines and recommendations for future interventions could be based on judgements with reduced errors. In addition, evidence-based guidelines could help patients acknowledge realistic expectations with respect to specific interventions. This type of realistic expectation has been found to affect the final outcome positively. 24
Although the included studies had relatively high levels of internal and external validity, the true overview of findings in terms of activity, participation and QoL in real-life contexts was inconclusive and vague and consequently the described interventions could not be recommended based on evidence. Just referring to the design of the included studies (Table 1), one could state that the quality of evidence as it is generally defined (for example, by the Grade Working Group, www.GradeWorking-Group.org) can be considered as low in the referenced studies. The question is then this: is it possible to implement studies with a high level of evidence within the AT-field while still uncovering results related to aspects of daily life? Other authors of systematic literature reviews in this field have made similar conclusions and have also highlighted the need for further research.25-27 Thus, effects from AT, such as the lower limb prosthesis, present a major challenge to standard evidence grading methods. 28
Conclusion
Seven studies reported significant improvements in one or more of the identified outcomes. However, only two of these studies obtained a high score in both internal and external validity. Interventions and outcome measurement methods varied considerably between studies; generally there were also few participants and short follow-up time. Research related to which key factors affecting the effectiveness of prosthetic interventions on user’s everyday life still remains to be done and should be of high interest and priority to clinicians in this field.
Footnotes
Acknowledgements
We would like to thank the librarians at Stakes (now National Institute for Health and Welfare) (Helsinki, Finland) for their substantial help with the electronic searches.
This research was supported in part by Nordic Development Centre for Rehabilitation Technology (now Nordic Welfare Centre).
