Abstract
The skeletal anchorage system has emerged with new possibilities in orthodontics, including the correction of maxillary transverse deficiency. The miniscrew-assisted rapid palatal expander (MARPE) is effective for widening the maxilla in adult patients. Among the various designs of MARPE, bone-borne MARPE is exclusively anchored on the palatal bone without any tooth support. Hence, it provides a pure orthopedic force without dentoalveolar side effects. This clinical pearl presents a simple modified bone-borne MARPE design with a clear, flexible template.
Introduction
Transverse maxillary deficiency is a frequently encountered orthodontic malocclusion, with a prevalence of approximately 10% in adults. 1 The quest for a non-surgical treatment of maxillary transverse deficiency in adult patients stimulated the development of miniscrew-assisted rapid palatal expansion (MARPE). MARPE is either a tooth-bone-borne or a solely bone-borne RPE device. 1 For adolescent patients, bone-borne expansion has been shown to produce greater transverse skeletal expansion while minimizing dental side effects compared with tooth-borne RPE appliances. 2
Since the introduction of bone-borne MARPE, various designs have been introduced. We intend to introduce a simple and novel design of bone-borne MARPE supported by a clear, flexible template.
Steps in Fabrication
Take a Hyrax screw of the desired length according to the patient’s maxillary transverse dimension (Figure 1).
Bend the arms of the Hyrax screw using the tip of a spring-forming plier to form helices of about 2–2.5 mm in diameter and adapt them closer to the body of screw (Figure 2).
To ensure that the helix is of the required diameter, superimpose the fabricated helix on a graph with 2 mm helix drawn. Constrict or modify the helix if required.
A 2 mm diameter of the helix is decided, taking into consideration that the miniscrew is around 1.5 mm in diameter. A freedom of 0.5 mm around the miniscrew and the helix helps in the ease of insertion and removal of the appliance.
Position the anterior helices in the T zone3 of the palate (Figure 3).
Adapt the helices closer to the palate on the cast.
Stabilize the well-adapted Hyrax screw on the cast with a thin layer of Plaster of Paris.
Fabricate a template for bone-borne MARPE by using a soft thermoplastic sheet of 1.75 mm thickness (Figure 4).
Adapt the thermoplastic sheet on the cast with the Hyrax screw stabilized in the desired position.
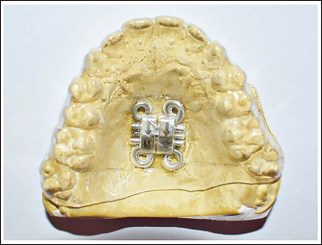
Mark the area of the helix on the thermoplastic template and drill holes to facilitate the placement of the miniscrew (Figure 5).
Hyrax Screw of 11 mm Size.

Bending the Arms of Hyrax Screw to Form Helices.

Positioning of the Hyrax Screw at the Desired Position on the Cast.
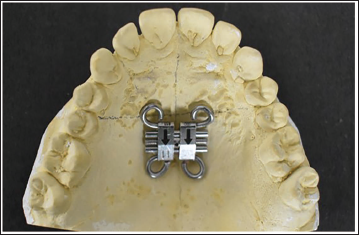
Adaptation of Soft Thermoplastic Sheet on Hyrax Screw Positioned on the Cast.

Drill Holes on the Thermoplastic Template to Facilitate Placement of Miniscrew.
Procedure of Use
Anesthetize the palate with local anesthesia.
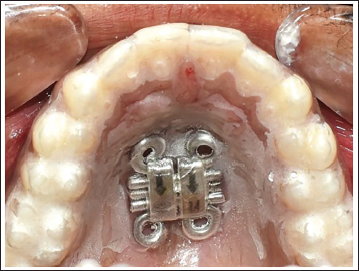
Intraorally, adapt the template with the embedded bone-borne MARPE (Figure 6).
Insert all four miniscrews using a contra-angled driver into the palate through the holes made in the template.
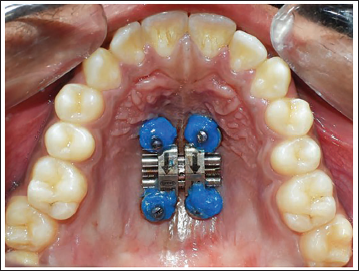
Gently remove the template with the bone-borne MARPE well adapted to the palate (Figure 7).
Intraoral Adaptation of the Template with the Embedded Hyrax Screw in Place.
Bone-borne Miniscrew-assisted Rapid Palatal Expander (MARPE) in Place.
Advantages
By bending the arms of the Hyrax as helices, no additional soldering of rings will be needed.
Can avoid detachment of rings during expansion due to failure of solder joints.
The arms of the Hyrax screw are of sufficient thickness; hence, they can be adapted well without distortion.
Easy to fabricate and economical.
Conclusion
This novel design of bone-borne MARPE with a clear template will be easy to fabricate and simple to use in patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
As it is a routine orthodontic procedure, ethical approval is not needed.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Patient consent for treatment and sharing of photographs was obtained, and they gave their informed consent for participating in the survey.
