Abstract
Introduction: Fluoride is known to reduce demineralization and enhance remineralization which is desirable around orthodontic brackets.
Material and methods: This study was carried out to determine the rate of fluoride release, fluoride content of enamel, and surface alteration of enamel from orthodontic bonding agents. Thirty extracted maxillary first premolars were divided into 2 groups of 15 teeth each. Group A—teeth were bonded with a chemically cured orthodontic bonding agent containing fluoride (Rely-a-Bond, Reliance, Itasca) and group B—teeth were bonded with a light-cured orthodontic bonding agent containing fluoride (Light Bond Reliance, Itasca). All individual sample teeth were used to estimate fluoride release and fluoride content of enamel. The same samples were also used to evaluate enamel surface alterations. The rate of fluoride release from the above 2 orthodontic bonding agents were measured in artificial saliva on day 1, 2, 3, 4, 5, 6, 7, 14, 30, 60, and 90. The fluoride content of the enamel was evaluated on day “0” and day “90” of bonding. The samples were also evaluated for the surface changes in enamel using scanning electron microscope (SEM).
Results: The results showed that (a) chemically cured and light-cured bonding agent showed a dramatic decrease in the concentration of fluoride release from day “1” to day “2,” (b) the concentration of fluoride release kept decreasing from day “1” to day “90” of measurement in both the groups, and (c) SEM of the bracket margins bonded with chemically cured bonding agent and light-cured bonding agent revealed particle depositions of irregular shape and size on day “90.”
Conclusion: Light-cured bonding agent (Light Bond) released higher concentration of fluoride as compared to chemically cured bonding agent (Rely-a-Bond). Light-cured bonding agent (Light Bond) showed higher increase in fluoride content of enamel than chemically cured bonding agent (Rely-a-Bond).
Keywords
Introduction
Demineralization of enamel causes an unsightly discoloration of enamel, which can compromise the aesthetics of the final result. The effects of enamel demineralization can vary from microscopic change to the production of white spots, large unsightly areas, and cavitations around brackets during fixed appliance treatment. Poor oral hygiene is clearly a major contributing factor for demineralization beneath and around the orthodontic attachments, orthodontic bands, and brackets.1–6 Thus, one of the most difficult problems in orthodontic treatment with fixed appliances is the control of enamel demineralization around the brackets.4, 7 Measures such as oral hygiene instructions, protection of the tooth surface with a resin sealant, reduction of enamel solubility, and inhibition of bacterial activity via topical fluoride agents have been undertaken extensively, but have proved to be either patient-dependent or of limited clinical significance. 8 An ideal preventive system, therefore, would be one that operates independently of patient co-operation and would release low concentrations of fluoride locally where it is most needed, that is, at the site of plaque stagnation around the brackets.
Fluoride ions interfere with initial bacterial adhesion,9, 10 their colonization,9, 11 and affect bacterial metabolism.12–14 The fluoride ions substitute themselves for the hydroxyl ions of hydroxyapatite crystals in enamel, forming fluorapatite crystal [Ca5(Po4)3F]. This chemical substitution has been known to aid in the remineralization of small decalcified lesions, as well as reducing new lesion formation and reducing tooth decay.15–17 Fluoride has multiple mechanisms of action to reduce decalcification of enamel, such as promoting remineralization, increasing crystallinity, and decreasing the solubility of enamel, reducing the adherence of plaque bacteria on the tooth surface and altering the bacterial metabolism in a way that reduces acid production in plaque.
Fluoride agent has been incorporated to orthodontic adhesives with the aim that it will release fluoride around the bracket and prevent demineralization. If a fluoride-releasing orthodontic bonding agent is to provide protection against decalcification of the enamel adjacent to the orthodontic bracket, then the fluoride released must either exert a local microbiological effect or become incorporated into the adjacent enamel, making the enamel more resistant to acid attack.
The cariostatic potential has been shown to be related to the magnitude of fluoride release and not due to the fluoride content of the bonding agent. Numerous orthodontic bonding agents containing fluoride have been introduced to prevent enamel decalcification around the brackets, and their pattern of fluoride release in de-ionized water,18, 20–24 distilled water,19, 25, 26 demineralizing solution, 27 and artificial saliva 26 has been reported. As it is necessary to know whether the self-cured or light-cured bonding agents release higher concentration of fluoride for longer durations, this study was undertaken to evaluate the fluoride release from different orthodontic bonding agents using artificial saliva as the medium. In addition, it also evaluates the surface characteristics of enamel around the bracket margins bonded with these bonding agents in the same samples, which has not been studied earlier.
Materials and Methods
The study was conducted on 30 human maxillary first pre-molars extracted for orthodontic purposes. The teeth were divided into 2 groups. Group A—15 teeth bonded with a chemically cured orthodontic bonding agent containing fluoride (Rely-a-Bond, Reliance, Itasca) and group B—15 teeth bonded with a light-cured orthodontic bonding agent containing fluoride (Light Bond Reliance, Itasca). Each tooth was mounted up to cemento-enamel junction in one stone block of dimension 1.5 × 1 × 1 cm.
The areas of bracket placement on the buccal surfaces of the teeth were etched with 37% phosphoric acid for 30 seconds, rinsed with adequate amount of distilled water under pressure for 20 seconds. After air drying, the primer provided by the manufacturer was applied by an applicator brush on both the etched enamel surface and the bracket base. Adequate amount of adhesive resin was applied on the bracket base. Standard edgewise premolar brackets of 22″ × 28″ (American Orthodontics) were used in the study. The bracket was pressed firmly against the enamel surface 4.5 mm from the occlusal surface and the excess adhesive was removed with a probe. For group B (Light Bond), after placement of brackets, they were light-cured for 30 seconds. After bonding of brackets, the teeth were removed from the dental stone blocks. The entire surfaces of the teeth were then varnished with the acid-resistant nail polish except the buccal surfaces.
Calibrations 27 were done for the electrode with a series of standard fluoride solutions, ranging in concentration from 0.1, 1, and 10 ppm of fluoride. The above standard fluoride solutions were prepared from a standard solution of fluoride concentration 100 ppm by diluting it with deionized water.
The Carboxymethyl cellulose containing artificial saliva 28 was prepared in the Department of Pharmacology, All India Institute of Medical Sciences, New Delhi and was stored in a plastic container at 4 °C in a refrigerator. A separate plastic vial containing 5 ml of above prepared artificial saliva was included in the experiment as control. 29
2.5 ml of artificial saliva was taken from the above plastic vial and kept in a separate plastic vial. To this, 2 ml of deionized water and 0.5 ml of Total Ionic Strength Adjustment Buffer (TISAB III, Orion Research Inc) was added. The vial was gently agitated to ensure complete mixing. The ionic fluoride concentration in ppm was recorded digitally using an Orion fluoride ion meter (920A) with a fluoride selective electrode (Model 96-09BN) by immersing the electrode in the solution contained in the vial.
Salivary Fluoride Measurement of Samples
Each sample tooth from both the groups was placed in a separate plastic vial containing 5 ml of artificial saliva prepared as described above. Fluoride release from each sample in the artificial saliva was measured on day 1, 2, 3, 4, 5, 6, 7, 14, 30, 60, and 90 from the date of bonding. The artificial saliva was changed 24 hours before the analysis to prevent the reading being cumulative. 25
Enamel Fluoride Measurements
The fluoride content in enamel of each sample tooth from both the groups was measured from the mesial part of the buccal surface occlusal to the bracket on day “0” and from the distal part of the buccal surface occlusal to the bracket on day 90 of saliva fluoride measurements.
For this purpose, 10 mg of powdered enamel was collected from each tooth with the Tungsten carbide bur. The powdered enamel was dissolved in 10 ml of 0.25M HCl in a plastic vial and allowed to stand for 24 hours in order to dissolve the enamel powder. From this, 1 ml of solution was taken and diluted with 9 ml of 15% Sodium citrate in a separate plastic vial. 30 The ionic fluoride concentration in ppm was recorded digitally using an Orion fluoride ion meter (920A) with a fluoride selective electrode (Model 96-09BN).
Scanning Electron Microscopy Evaluation of Samples
The enamel surface around the cervical area of the bracket margin of each sample tooth from both the groups was viewed on day “0” and on day “90” after bonding. The unprocessed samples were visualized under low vacuum mode (LV mode) under Leo, 435 VP SEM using a backscatter electron detector (BSED). Photomicrographs were taken at 1,000× magnification for all the samples. The statistical analysis was done with SPSS software.
Results
Fluoride release by chemically cured (Rely-a-Bond) and light-cured (Light Bond) bonding agents was analyzed at different time intervals. Fluoride content and surface alteration of enamel was also analyzed from the above 2 orthodontic bonding agents. Student t-test was applied to see the significant difference between the 2 groups for each level. Two-way analysis of variance with post-hoc analysis (LSD) was performed to see the trend within the variables for both the groups. Paired Student t-test was applied to see the fluoride content of enamel from day “0” to day “90” of bonding in both the groups. Probability value (p-value) .05 was considered as statistically significant.
The fluoride concentration in the artificial saliva was found to be 0.0912 ppm.
Fluoride Release
The results of fluoride release appear in Table 1 and Figure 1. Both the materials showed maximum release of fluoride on day “1.” The concentration of fluoride release reduced drastically on day “2” in both the groups. However, the decline was steeper in the light-cured group. The rate of fluoride release had decreased from day “1” till the last day of measurement. The fluoride release in both the groups followed a similar trend. The concentration of fluoride release from Rely-a-Bond was less than that of the artificial saliva from day “14” onwards and that of Light Bond from day “30” onwards.
Fluoride Content of Enamel
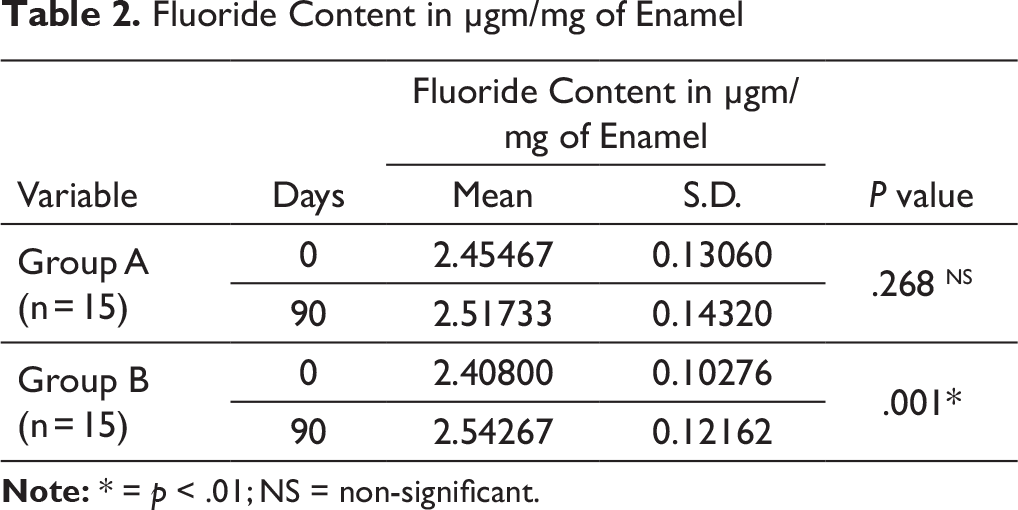
The results of fluoride content of enamel appear in Table 2. Both the groups showed improvements in fluoride content of enamel from day “0” to day “90.” The paired Student t-test from day “0” to day “90” showed statistically significant (p = .001) improvement in fluoride content of enamel bonded with Light Bond while in enamel bonded with Rely-a-Bond it was found to be statistically non-significant (p = .268).
Comparison of Fluoride Release in ppm of Chemically Cured (Group A) With Light-Cured (Group B) Bonding Agent
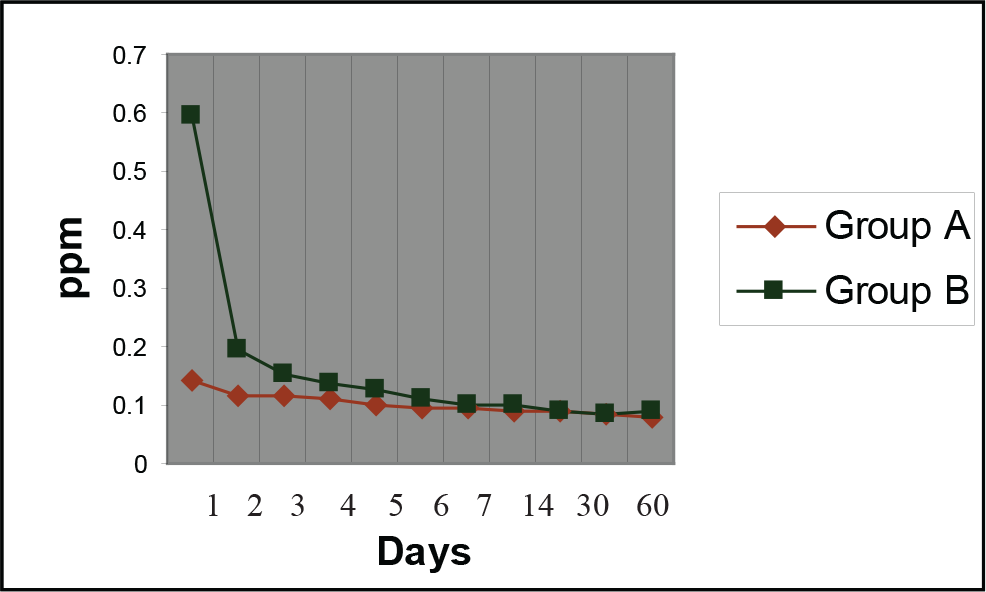
Comparison of Fluoride Release in ppm of Rely-a-Bond (Group A) With Light-Bond Cured (Group B) Bonding Agent
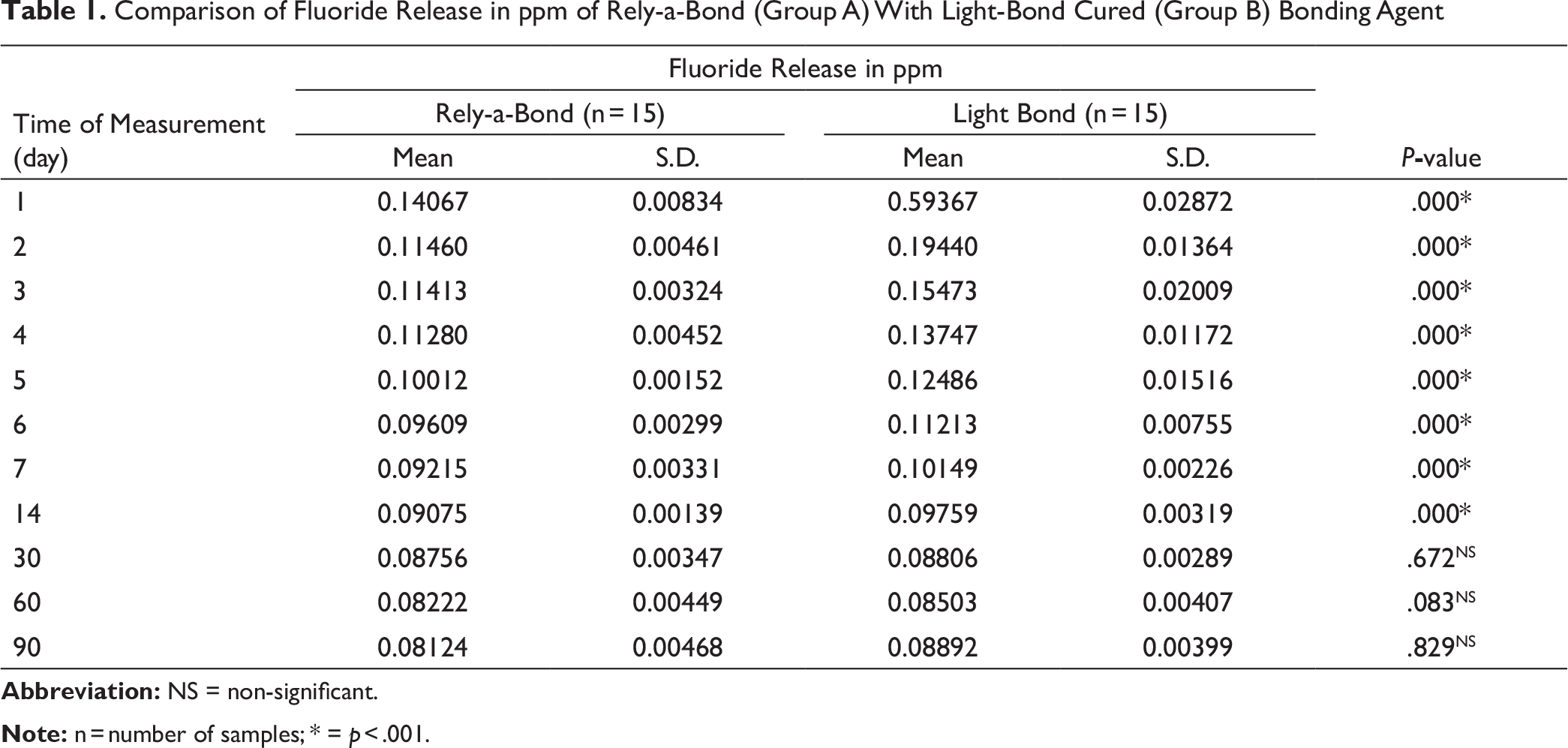
Fluoride Content in µgm/mg of Enamel
Enamel Surface Alterations
Scanning electron microscopy (SEM) at day “0” revealed smooth enamel surface cervical to bracket margin in both the groups (Figures 2 and 3). Some of the samples showed some minute scratches and some etching pattern on the same area. While at day “90,” SEM revealed particle depositions of irregular shape of less than 5 µm in size on the same area (Figures 4 and 5). However, at some places, particle depositions were in the form of spherical globules. These changes were seen in all the samples from both the groups.
SEM of Enamel Surface on Day “0” of Bonding With Rely-a-Bond Showing Some Minute Scratches (Arrow) and Some Etching Pattern

SEM of Enamel Surface on Day “0” of Bonding With Light-Bond Showing Some Minute Scratches and Some Etching Pattern (Arrow)

SEM of Enamel Surface on Day “90” of Bonding With Rely-a-Bond Showing Particle Depositions (Arrow) on Enamel Surface Gingival to Bracket Margin

SEM of Enamel Surface on Day “90” of Bonding With Light-Bond Showing Particle Depositions (Arrow) on Enamel Surface Gingival to Bracket Margin

Discussion
Early enamel demineralization, observed as whitish areas, occurring near orthodontic brackets and bands during treatment is a problem of great concern to the orthodontists. Prevention of such enamel demineralization by different procedures like oral hygiene instructions, protection of the tooth surface with a resin sealant, reduction of enamel solubility, and inhibition of bacterial activity via topical fluoride agents have been carried out extensively. However, all these procedures are either patient-dependent or of limited clinical significance. Thus, an ideal preventive system would be the one that operates independent of patient co-operation and would release low concentrations of fluoride locally where it is most needed, that is, at the site of plaque stagnation around the brackets. To combat decalcification of enamel adjacent to orthodontic attachments, fluoride releasing composite resins has been developed.31, 32
Fluoride Release From Chemically Cured Bonding Agent
The maximum concentration of fluoride release was 0.14067 ppm on day “1.” However, there was a dramatic decrease in the fluoride release from day “1” to day “2.” This could be because of initial release of surface locked fluoride into the sample solution at day “1.” This amount of fluoride released from beneath the brackets was small because only the edges of thin film of adhesive was exposed to the medium. The concentration of fluoride release kept on decreasing until the last day of measurement, but the concentration of the fluoride in the sample solution was less than that of the prepared artificial saliva from day “14” onwards. Absorption of fluoride from artificial saliva on to the enamel surface could be responsible for such type of result. Ghani et al 27 found significant quantity of fluoride release from Mirage Dual Cure® and Reliance® (both are fluoride containing orthodontic bonding agents) in demineralizing solution only for first 3 days and after day “3,” the amount of fluoride release was similar to that of Right-on® and Heliosit® (both were non-fluoride containing orthodontic bonding agents). By the 8th day the amount of fluoride release from all 4 materials was very small but was more than 0.007 ppm. They explained that demineralizing solution contained a fluoride concentration of 0.007 ppm and sound enamel will release small amounts of fluoride. 33 However, in the present study, the fluoride concentration of the artificial saliva was 0.0912 ppm which decreased to values less than 0.0912 ppm after 14 and 30 days in group A and group B, respectively, which might be because fluoride from artificial saliva was absorbed by the enamel. Basdra et al 19 studied the fluoride release from Rely-a-Bond in distilled water and found a dramatic decrease in fluoride release after first 48 hours and continued up to day “60.” They explained that the decrease in fluoride release from the adhesive after first 48 hours could be because of constant dissolution of adhesive fluoride into the sample solution. In the present study, Rely-a-Bond released fluoride up to day “90.” But the concentration of fluoride released by the said bonding agent was less than that of the artificial saliva (storage media) after day “7.” The use of different storage media may be responsible for such variation. Using deionized water as storage media, Young et al 34 observed that composite resin (tetric) released most of the fluoride after 24 hours and the fluoride released appeared to be stabilized after approximately 3 weeks. This longer duration of fluoride release could be because of deionized water used as the storage media.
Fluoride Release From Light-Cured Bonding Agent
There was an initial “burst effect” of fluoride release on day “1,” but the concentration thereafter dropped significantly on day “2.” The surface locked fluoride which was dissolved into the artificial saliva during day “1” could be responsible for such initial “burst effect.” The results of this present study are in conformity to the studies conducted by Chan et al 21 and McNeill et al. 26 Chan et al 21 studied fluoride release from FluorEver OBA (light cure orthodontic bonding agent) in deionized water and found that the fluoride release declined sharply after 24 hours and continued to decline over the rest of the period of experiment. In the present study, the fluoride concentration dramatically decreased on day “2” and continued till day “14” of the measurement. Constant dissolution of adhesive fluoride into the sample solution could be responsible for continued release of fluoride till day “14.” McNeill et al 26 detected fluoride release up to 6 months from discs fabricated from Assure, Fuji Ortho LC, Python, and Transbond XT in distilled water and artificial saliva. They detected similar patterns of fluoride release in both storage media and initial higher rates of fluoride release from all 4 bonding agents which gradually declined to lower but stable levels. However, in the present study, there was an initial higher concentration of fluoride release from Rely-a-Bond and Light Bond in artificial saliva and had gone decreasing till the last day of measurement. Ogaard et al 35 studied fluoride release of the orthodontic cement VP 862 in water and in human saliva. They found that the fluoride release is significantly lower in human saliva than in water and the largest release of fluoride is observed in the first week. However, a significant amount of fluoride was still released after 6 months. But in the present study, Light Bond released fluoride up to day “90” of saliva fluoride measurement. However, the concentration of fluoride release was less than that of artificial saliva from day “14” onwards. This type of result may be because of artificial saliva used as storage media in the present study.
Comparison of Fluoride Release of Chemically Cured Bonding Agent With Light-Cured Bonding Agent
The fluoride release in both the groups followed a similar trend. Light-cured bonding agent (Light Bond) released higher concentration of fluoride as compared to chemically cured bonding agent (Rely-a-Bond) at every time of measurement. This could be because of higher content of fluoride in the light-cured bonding agent (Light Bond) than chemically cured bonding agent (Rely-a-Bond). Earlier studies have shown that fluoride releasing properties of the bonding agents is not affected by their mode of curing, whether chemically or light cure.36, 37
Comparison of Fluoride Content of Enamel Bonded With Chemically Cured Bonding Agent and Light-Cured Bonding Agent
The enamel powder showed improvements in fluoride content of enamel bonded with chemically cured bonding agent (Rely-a-Bond) from day “0” to day “90.” Light-cured bonding agent (Light Bond) showed higher increase in fluoride content of enamel than chemically cured bonding agent (Rely-a-Bond). This could be because of higher concentration of fluoride release by Light Bond than Rely-a-Bond. The individual samples did not show any constant improvement in fluoride content of the enamel. Some samples of Rely-a-Bond showed more fluoride content of enamel on day “90” than Light Bond. Variation in the fluoride content of enamel from surface to the inner layers and difficultly in removing a uniform layer of enamel with the help of tungsten carbide bur could be responsible for such type of results.
Enamel Surface Alterations
The release of fluoride exactly around the bracket margins is important to prevent enamel demineralization. SEM of enamel surface of the 2 fluoride releasing materials revealed particle deposition of irregular shape and size of less than 3 µm on day “90.” Although light-cured bonding agent (Light Bond) released higher concentration of fluoride as compared to chemically cured bonding agent (Rely-a-Bond) at every time of measurement, the nature of surface alteration was similar in both the groups. Thus, varying concentration of fluoride had similar effect on enamel surface. Some of the particle depositions showed spherical globules which resembled to calcium fluoride (spherical globules), a salt with clearly cariostatic properties. The results of the present study are in conformity to the studies undertaken by Nelson et al 38 and Basdra et al. 19 In both the said studies, sample preparation was done by coating the samples with highly conducting layer of gold. Both the studies reported formation of spherical globular agglomerates on the enamel surface as the reaction product of fluoride with enamel.
Thus, the long-term release of fluoride from presently available orthodontic bonding agents seems to be somewhat doubtful. However, an ideal bonding agent should have adequate bond strength, ease of removal, adequate working time, color stability, biocompatibility, and fluoride release. A balance in the fluoride release and bond strength is essential to have a significant reduction in the incidence of decalcification during orthodontic treatment.
Summary and Conclusions
The following conclusions were drawn from this study:
Chemically cured bonding agent and light-cured bonding agent showed a dramatic decrease in the concentration of fluoride release from day “1” to day “2.” The concentration of fluoride release kept decreasing from day “1” to day “90” of measurement in both the groups. Light-cured bonding agent released higher concentration of fluoride as compared to chemically cured bonding agent at every time of measurement. The concentration of fluoride release from chemically cured bonding agent was less than that of the artificial saliva from day “14” onwards, and from light-cured bonding agent from day “30” onwards. Teeth bonded with light-cured bonding agent showed more increase in fluoride content of enamel than teeth bonded with chemically cured bonding agent. SEM of the bracket margins bonded with both chemically cured and light-cured bonding agent revealed particle depositions of irregular shape and size on day “90,” which were probably calcium fluoride, which exert the cariostatic effect around the bracket margins.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
