Abstract
The lymphatic system plays an essential role in the drainage of fluids, proteins, and cells from tissues. However, it facilitates the dissemination of cancer cells, favoring progression and the development of metastases. The present study aimed to characterize the intratumoral and peritumoral lymphatic vessel density and to evaluate their prognostic value in canine cutaneous mast cell tumors (MCTs). The lymphatic vessels from intratumoral and peritumoral areas were identified and quantified in 57 tumor samples from 51 dogs (28 low-grade and 29 high-grade MCTs), using immunohistochemistry with an anti-LYVE1 antibody. The results were compared with histological grades, mortality due to the disease, and post-surgical survival of the patients. The number of peritumoral lymphatic vessels was higher in high-grade MCTs (P = .0370) and in dogs that died due to the disease (P = .0215), while no statistically significant differences were found for intratumoral lymphatic vessel counts. Dogs that had MCTs with peritumoral lymphatic vessel counts higher than 113 had shorter post-surgical survival times, with a median survival of 122 days (P = .0080; hazard ratio = 4.886). No association was detected between lymphatic vessel counts and the presence of lymph node metastases. Our results suggest that peritumoral lymphatic vasculature is a prognostic indicator for post-surgical survival in cases of canine cutaneous MCTs.
Keywords
Canine cutaneous mast cell tumor (MCT) is one of the most common tumors routinely seen in veterinary oncology.4,21,33,37,42,47,52 The clinical presentation of this neoplasm is heterogeneous and the biological behavior is extremely variable.14,31,36,40,43 Histological classification remains the most consistent prognostic tool for this neoplasm.7,14,31,43 However, other factors also contribute to predicting tumor behavior and guiding the choice of the most appropriate treatment, such as the Ki67 index, KIT expression, 32 detection of cKIT mutations, and clinical staging.14,31,43,55
Among the characteristics acquired during cancer development, the dissemination of tumor cells that colonize other tissues and organs, resulting in metastases, stands out as particularly significant. 25 The lymphatic system represents an important route for the dissemination of tumor cells, as well as a potential target for antitumor therapy. 18 Lymphatic flow is subject to lower shear stress compared to blood vessels, 58 which facilitates the survival of cancer cells and contributes to more efficient metastasis. In addition, lymphatic capillaries are composed of a single layer of lymphatic endothelial cells (LECs) and a minimal basement membrane and are not covered by pericytes or smooth muscle cells, which makes them extremely permeable. In this way, they exert their physiological action of absorbing interstitial fluids, macromolecules, and cells while also facilitating the intravasation and extravasation of tumor cells. 2
Neoplastic cells can enter the lymphatic vessels either passively or actively. During transport, neoplastic cells can traverse the lymph node and exit via the efferent lymphatics or veins. Alternatively, they can enter the lymph node and either interact with immune cells to generate an immune response or can colonize the lymph node with an insufficient immune response, resulting in the establishment of a metastasis. 20 In MCTs, metastases occur more frequently in the locoregional lymph nodes and may also affect other organs, particularly the spleen and liver.14,36,44,54 The presence of lymph node metastasis may be an indicator of poor prognosis and has a direct influence on the treatment to be implemented.51,57
Despite the significant importance of the lymphatic system in both physiological and pathological processes, research on this system has progressed slowly due to the histological similarity between lymphatic vessels and blood vessels. 3 However, the identification of specific markers for lymphatic endothelium has enabled more precise and in-depth investigations. In 1999, Banerji et al 5 described the first receptor involved in the binding of hyaluronic acid and its entry into lymphatic vessels, lymphatic vessel endothelial receptor-1 (LYVE1), a homolog of CD44. This membrane-bound protein is absent in blood vessel endothelial cells, lymphocytes, macrophages, fibroblasts, and epithelial cells, making it a valuable marker for lymphatic vessels and useful in distinguishing them from blood vessels.
Anti-LYVE1 antibodies have also been used in immunohistochemistry and immunofluorescence to assess the prognostic significance of the lymphatic vascularization in several human cancers, including breast, lung, ovarian, uterine, hepatocellular, and head and neck cancers, as well as melanoma.6,8,12,15,17,18,28–30,34,39,45,48,53
In veterinary medicine, LYVE1 has been used to determine the histogenesis of neoplasms in cats and to generate an immunohistochemical panel for vessel differentiation.16,49 Halsey et al 24 used an anti-LYVE1 antibody to differentiate cutaneous angiosarcomas in dogs and, more recently, this lymphatic vessel marker was used to evaluate the intestinal lymphatic vasculature of biopsy samples from dogs with chronic inflammatory enteropathy. 26
The present study aimed to characterize the intratumoral (IT) and peritumoral (PT) lymphatic vasculature of canine cutaneous MCTs and to assess their prognostic value, as well as to evaluate the correlation between lymphatic vessel density and lymph node status.
Materials and Methods
Clinical Samples and Data
Canine cutaneous MCT samples were obtained from the Tumor Bank of the Laboratory of Comparative and Translational Oncology of the School of Animal Science and Food Engineering of the University of São Paulo, Pirassununga/SP; the Veterinary Pathology Department of the São Paulo State University “Júlio Mesquita Filho,” Jaboticabal/SP; the Veterinary Hospital of the School of Veterinary Medicine and Animal Science of the University of São Paulo, São Paulo/SP; specialized oncology clinics (E+ Especialidades Veterinárias and Veros Hospital Veterinário, São Paulo/SP, and Oncospes Oncologia Veterinária, Ribeirão Preto/SP); and Diagnostic Laboratories (Centro Veterinário de Anatomia Patológica—CVAP, São Paulo/SP and TECSA Laboratory, Campinas/SP). The project was approved by the Ethics Committee of Animal Use of the School of Veterinary Medicine and Animal Science of the University of São Paulo (protocol #2821020222). The cases were randomly selected to form 2 groups of cutaneous MCTs with a similar number of lesions per grade, according to the 2-tier grading system. 31
All dogs underwent surgical treatment with wide margins (2–3 cm), and most had 1 or more lymph nodes excised with the aim of achieving disease cure. The decision to perform lymphadenectomy was made by the surgeon responsible for each case. Sentinel lymph nodes were located by lymphography, performed with patent blue. Only cases with available lymph node tissue for histology were used in statistical comparisons regarding lymph node metastasis. Clinical data were obtained during consultations and analysis of medical records and/or interviews with owners and veterinarians in charge. Cases of death due to the MCT, development of severe paraneoplastic syndromes (such as severe hemorrhage and anaphylactic reactions 7 ), and/or indication of euthanasia due to MCT by the veterinarian were considered deaths due to the disease for statistical analysis purposes. Dogs that were alive at the end of the study, deaths unrelated to the disease, and dogs lost to follow up before a minimum of 180 days were censored.
Samples were routinely processed for histology and reviewed and graded according to the proposals of Patnaik et al 43 and Kiupel et al 31 by an experienced pathologist (RFS). In cases where dogs had more than 1 MCT, the tumor with the highest grade was considered for survival analysis. Lymph nodes were stained with toluidine blue to facilitate visualization of mast cells. In cases where there was more than 1 lymph node available for examination, the case was classified as HN3 (overt metastasis defined as disruption or effacement of the normal nodal architecture by discrete foci, nodules, sheets, or overt masses composed of mast cells) 57 if overt metastases were identified in any of the lymph nodes.
Immunohistochemistry
Four micrometer-thick sections of MCTs were adhered to silanized glass slides (Starfrost), deparaffinized and rehydrated. Antigen retrieval was performed by heating the slides in Tris EDTA buffer solution (pH 9), in a steamer at 95°C for 25 minutes. Endogenous peroxidase and protein block were performed using a commercial kit (Leica, Novolink Max Polymer Detection System). The samples were incubated with the primary antibody anti-LYVE1 (ab33682, Abcam, 1:5000) for 16 hours. After washing, they were incubated with the post-primary (Novocastra Post Primary, Leica), followed by the polymer (Novolink Polymer, Leica), following the manufacturer’s instructions. The reactions were visualized with 3,3’-diaminobenzidine chromogen (Leica) and were counterstained with Harris hematoxylin. Normal skin or canine intestine samples were used as positive controls. For negative controls, the primary antibodies were replaced with Tris-buffered saline with 1% bovine serum albumin buffer, and the slides were processed under the same conditions as the test slides.
The lymphatic vessels were evaluated in the IT and PT areas by 1 observer (KCB). The histological sections were scanned using a 10× objective to subjectively identify the hot spot areas and 5 photomicrographs were captured with a 40× objective (total area = 0.40 mm2). The PT area images were acquired immediately adjacent to the tumor margins. Lymphatic vessels were counted according to the criteria described by Weidner et al 56 and Kato et al, 29 ie, any immunolabeled endothelial cell or cluster of endothelial cells, whether or not they contained a visible lumen, was considered a single countable microvessel. When lymphatic vessels presented well-defined lumen, each visible luminal opening was counted as 1 vessel.
Statistical Analysis
Data normality was assessed using the Anderson-Darling test. The Mann-Whitney test was used to verify the association between IT and PT lymphatic vasculature and histological grades or mortality due to the tumor. Receiver operating characteristic curve analysis was performed to determine the cutoff value for lymphatic vessels in dogs with MCTs, comparing censored cases vs dogs that died due to the disease. Survival analysis was performed using the Kaplan-Meier method, followed by the Mantel-Cox test (log-rank). Lymphatic vessel counts and lymph node status were compared using the Fisher exact test. Only cases with adequate lymph node tissue were included in the statistical analyses of lymph node status. Statistical analysis was conducted using GraphPad Prism software (GraphPad Inc., version 9.5.1 for MacOS), and the significance level was set at 5%.
Results
Fifty-seven cutaneous MCT samples from 51 dogs were immunolabeled for the identification and quantification of lymphatic vessels. Four dogs had 2 MCTs, and 1 dog had 3 MCTs. The mean age at diagnosis was 9.6±3 years (range: 4–16 years), and 29/51 were female (57%). Mixed breed dogs were the most frequent breed (17/51, 33%), followed by Labrador retrievers (6/51, 12%), pitbulls (6/51, 12%), boxers (5/51, 10%), golden retrievers (3/51, 6%), and Yorkshire terriers (3/51, 6%). The remaining dogs included a blue heeler, bull mastiff, Doberman pinscher, English cocker spaniel, French bulldog, Italian greyhound, Lhasa Apso, pug, Shiba, and Shih Tzu. The breed was not registered for 1 dog. Most MCTs were found on the extremities/limbs (28/57, 51%), followed by the thorax (11/57, 19%), head/neck and inguinal regions (7/57, 12% each), and the abdomen (2/57, 4%). In 1 case, the location of the lesion was not available. Fifteen dogs received adjuvant chemotherapy with vinblastine and prednisone, and of these, 8 cases were also treated with a tyrosine kinase inhibitor (Palladia). The main clinical data of the patients are summarized in Supplemental Table S1.
According to the grading system of Patnaik et al, 43 1 MCT was grade I (1/57, 2%), 46 were grade II (46/57, 81%), and 10 were grade III (10/57, 18%). According to the grading system of Kiupel et al, 31 28 MCTs were low grade (28/57, 49%) and 29 were high grade (29/57, 51%). In 32 cases, a lymph node was available for evaluation, and some had more than 1 lymph node removed and analyzed for the presence or absence of metastasis. The majority of lymph nodes were axillary (10/40, 25%) and inguinal (9/40, 23%) lymph nodes. Other lymph nodes included the cervical (6/40, 15%), popliteal (6/40, 15%), prescapular (5/40, 13%), mandibular (3/40, 8%), and iliac (1/40, 3%) lymph nodes. Of the 40 lymph nodes analyzed, 16 had overt metastases (HN3), 13 were classified as HN2, 3 as HN1, and 8 as HN0.
LYVE1 immunolabeling was cytoplasmic in LECs and absent in blood vessels. The LYVE1-positive cells were arranged in a single layer, forming lymphatic vessels with or without clear lumens, or in groups and, in some cases, as isolated LECs (Fig. 1). In 8 samples (4 low-grade and 4 high-grade), the PT region was not available for evaluation in the histologic sections.
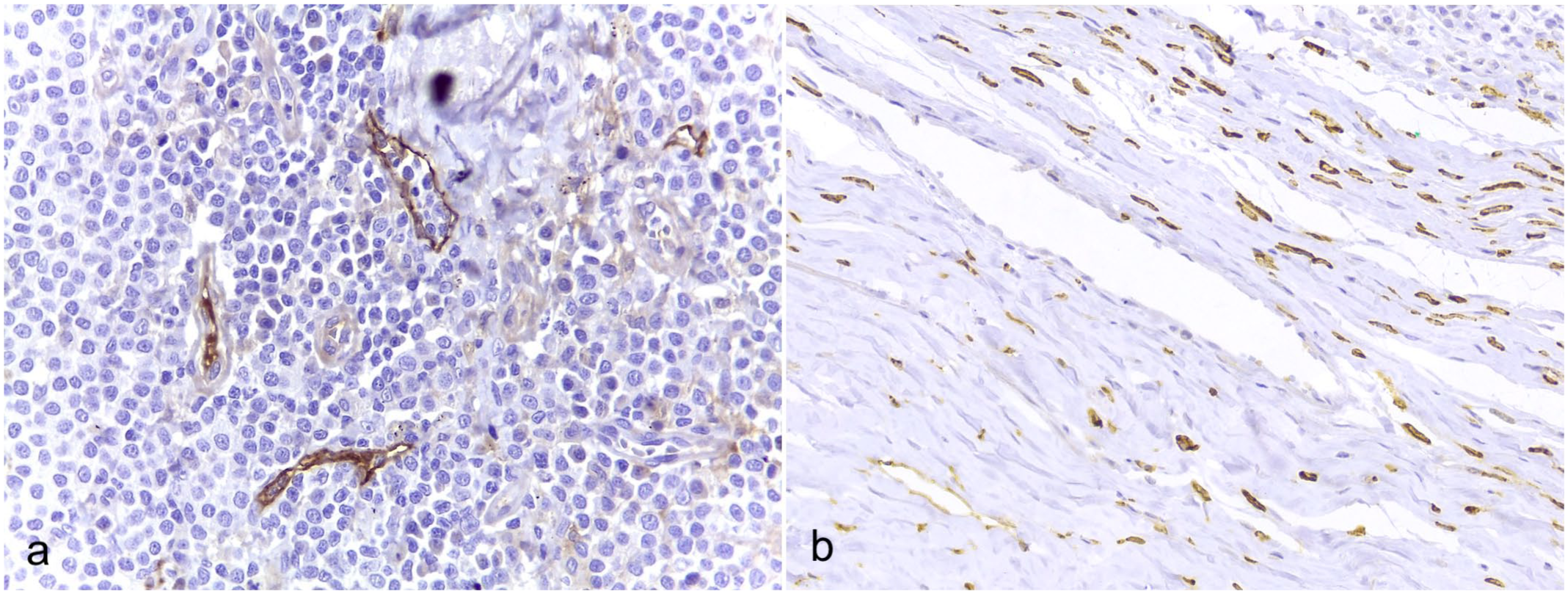
LYVE1 immunohistochemistry in canine cutaneous mast cell tumors showing (a) labeled lymphatic vessels containing tumor cells in the intratumoral region and (b) high lymphatic vessel density, with or without visible lumens, in the peritumoral region. LYVE1 immunohistochemistry.
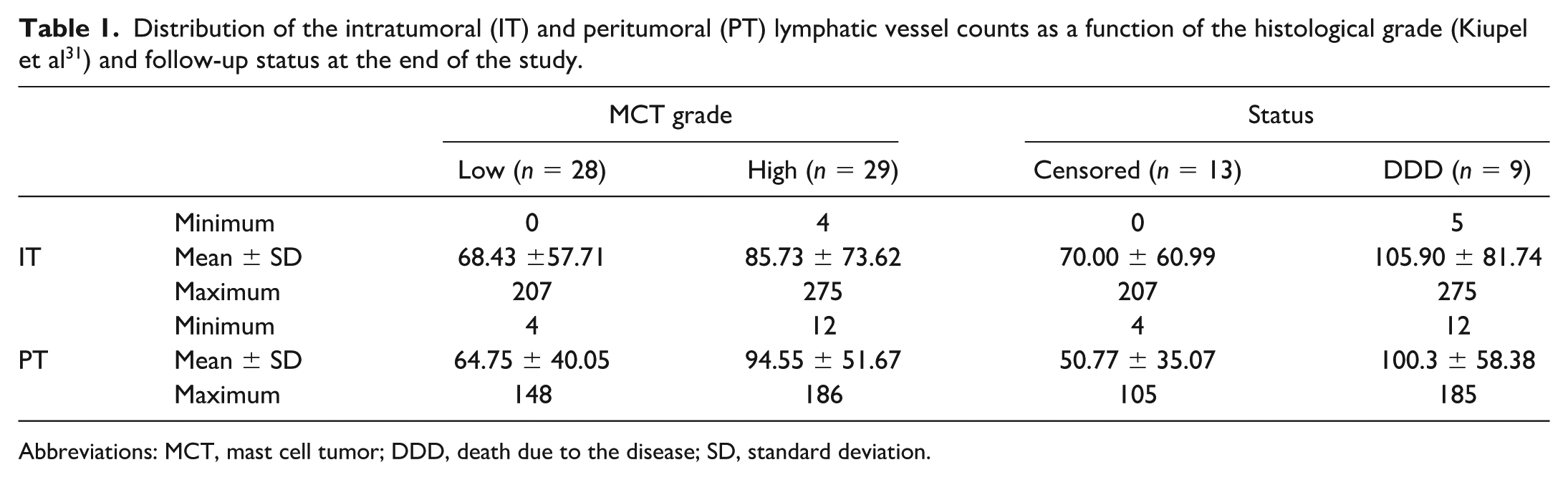
The number of PT lymphatic vessels (PTLV) was higher in high-grade MCTs than in low-grade MCTs, 94.55±51.67 vs 64.75±40.05, respectively; P = .0370 (Fig. 2(a) and Table 1). The IT lymphatic vessel (ITLV) counts were also higher in high-grade MCTs (85.73±73.62 vs 63.43±57.71, respectively; P = .3391) but were not statistically significant.
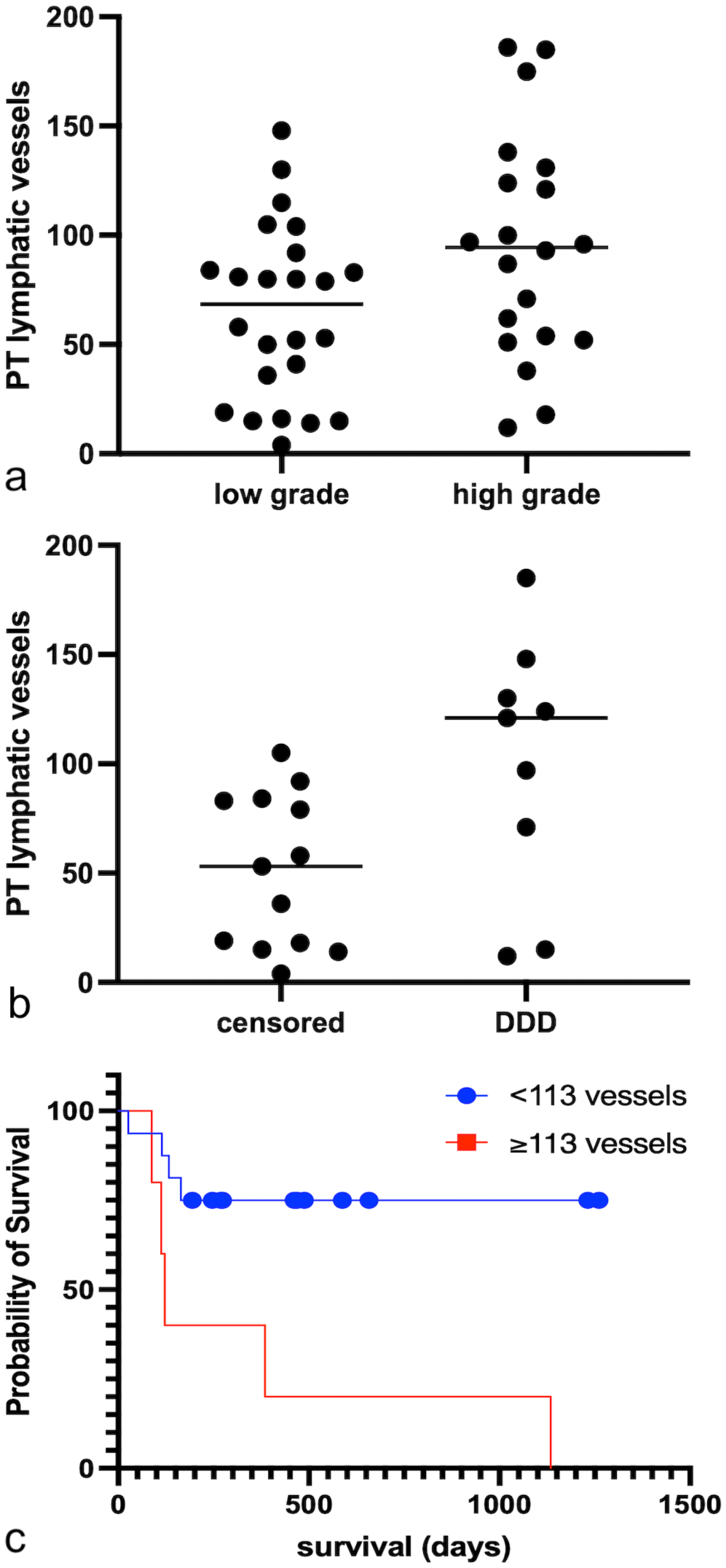
Distribution of the peritumoral (PT) lymphatic vessel counts showing higher PT lymphatic vessel counts in (a) high-grade than in low-grade mast cell tumors (MCTs) (P = .0370, Mann-Whitney test; horizontal lines represent the average number of lymphatic vessels) and in (b) dogs that died due to the disease (DDD) than those that were censored in the statistical analysis (P = .0215, Mann-Whitney test; horizontal lines represent the average number of lymphatic vessels). (c) Kaplan-Meier survival curves demonstrating that dogs with high PT lymphatic vessel count MCTs (≥113 vessels, n = 5) have shorter survival times compared with those with low PT lymphatic vessel counts (<113 vessels, n = 13). Kaplan-Meier followed by Mantel-Cox post-test, P = .0080; χ2= 7.029; hazard ratio = 4.886; median survival for ≥113 vessels = 122 days. Median survival time was not reached for dogs with <113 vessels.
Distribution of the intratumoral (IT) and peritumoral (PT) lymphatic vessel counts as a function of the histological grade (Kiupel et al 31 ) and follow-up status at the end of the study.
Abbreviations: MCT, mast cell tumor; DDD, death due to the disease; SD, standard deviation.
Dogs that died due to disease had higher PTLV counts than censored cases, 100.3±58.38 vs 50.77±35.07, respectively; P = .0215 (Table 1; Fig. 2(b)), while no statistically significant difference was found for ITLV counts (105.90±81.74 vs 70.00±60.99, respectively). Using receiver operating characteristic curve analysis, we established a cutoff point of 113 PTLV (area under the curve = 0.7564; P = .0451; sensitivity = 55.56%; specificity = 100%). Five dogs had tumors with ≥113 PTLV counts, all of which died due to the disease. Sixteen dogs had tumors with <113 PTLV counts; 4/16 (30%) died due to the disease. Survival analysis revealed that dogs with higher PTLV counts (≥113 vessels) had shorter survival times, P = .0080; χ2= 7.029; hazard ratio = 4.886; median survival time for cases with ≥113 LVs = 122 days; median survival time was not reached for dogs with <113 LVs; Fig. 2(c). No association was detected between the PTLV (P = .1393) or ITLV (P = .1160) counts and the presence of lymph node metastasis.
Discussion
Our results show that a higher PTLV count is associated with increased tumor-related mortality and shorter post-surgical survival times for dogs with cutaneous MCTs.
Lymphatic vessels can directly contribute to the dissemination of cancer cells and, consequently, to the formation of metastases, which are among the main causes of death in human cancer patients.2,58 Solid tumors exhibit a significant increase in interstitial fluid pressure. Lymphatic vessels play a fundamental role in helping to reduce this pressure, which can mechanically influence and direct cell migration through interstitial flow, thereby contributing to metastasis and positively modulating tumor proliferation.11,19,41 This process is also facilitated because the intravasation of tumor cells into lymphatic vessels can be influenced by the reduced physical barriers imposed on invasive tumors. Unlike blood vessels, lymphatic capillaries consist of a single layer of LECs and possess a minimal basement membrane. They also lack coverage by pericytes or smooth muscle cells, making them extremely permeable and reducing barriers to the entry of tumor cells.1,2 Furthermore, the lymphatic system has lower intravascular pressure compared to blood vessels, which favors the dissemination of cancer cells by facilitating their survival and enhancing metastatic efficiency. 58
However, it is important to emphasize that, despite all these contributing factors, the dissemination of cancer cells is not driven solely by mechanical forces. For the metastatic cascade to initiate, cancer cells must acquire an invasive phenotype, enabling them to detach from their tissue niche and invade the surrounding tissue. Subsequently, they enter blood and lymphatic vessels through a process known as intravasation. 10 In addition, the ability of neoplastic cells to migrate to and infiltrate lymph nodes is often associated with the expression of specific receptors and hyaluronic acid-binding proteins, which facilitate immune evasion and/or modulation, thereby promoting the establishment of metastases within the lymph nodes. 59
The anti-LYVE1 antibody has been used in immunohistochemistry and immunofluorescence to identify lymphatic vessels in several human cancers.6,12,28–30,39,48 In veterinary medicine, angiogenesis in canine tumors has been a subject of interest, especially in mammary tumors.22,23,50 Sleeckx et al 49 found that anti-podoplanin (D2-40) and anti-CD34 antibodies did not show immunoreactivity with canine tissue, while PROX1 and CD31 were considered the most suitable markers for lymphatic and blood vessels, respectively, since they were associated with lower background labeling compared to anti-von Willebrand factor and anti-LYVE1 immunolabeling. Halsey et al 24 used the anti-LYVE1 antibody to differentiate lymphangiosarcomas from hemangiosarcomas in dogs, and, more recently, this antibody was used to evaluate the intestinal lymphatic vasculature in biopsy samples from dogs with chronic inflammatory enteropathy. 26 The present study confirmed the efficiency of the anti-LYVE1 antibody and indicates that it can be applied in future research to correlate the presence and distribution of lymphatic vessels with other pathological processes.
The number of ITLVs in MCTs did not show prognostic value, consistent with findings from other studies in which only the PT vasculature was associated with unfavorable outcomes.8,13,18,60 Some authors report that lymphatic vessels are frequently identified in the tumor periphery, while their presence within the neoplastic parenchyma is rarely observed or absent.35,41 The hypothesis regarding the reason for this selective location of lymphatic vessels is that, due to the high interstitial pressure present in expanding tumors, the vessels collapse, making their identification difficult. 41 In support of this hypothesis, we observed that some of the lymphatic vessels did not have visible lumens, with LECs being found in isolation or in small groups. The absence of vascular lumens caused by the high interstitial pressure generated by the tumor makes the lymphatic vessels unsuitable for the dissemination of neoplastic cells.18,41 The absence of functional lymphatic vessels can further contribute to the increase in interstitial pressure within the tumor, hindering the diffusion of therapeutic agents.9,27
In humans, increased lymphatic vasculature densities are associated with higher rates of lymph node metastasis in breast, lung, gastric, prostate, cervical, pancreatic, and uterine cancers, as well as in melanomas.8,12,13,15,17,18,34,45,53,60 It is also known that MCTs, especially high-grade lesions, have metastatic potential, and that lymph node metastasis often precedes distant metastasis and may be a negative prognostic factor.14,36,44,54 However, no associations between the lymphatic vasculature and lymph node metastasis were observed in the present study. Although present in higher quantities in more aggressive tumors, the lack of association with the presence of lymph node metastasis may be related to the persistent activation of lymphangiogenic signaling pathways, which culminates in the formation of dysfunctional lymphatic vessels. 46 As mentioned before, metastasis is a complex process that does not depend exclusively on physical factors such as the presence and distribution of lymphatic vessels but also involves an important interaction of molecular mechanisms. 10
One limitation of the present research is that some of the dogs underwent adjuvant chemotherapy, which may have influenced the outcome of the patients. However, cases with neoadjuvant chemotherapy were excluded, and adjuvant chemotherapy was performed in accordance with recommendations in the veterinary literature.7,14 In addition, lymphadenectomy may have therapeutic potential, reportedly reducing local recurrence and progression to distant metastasis in 1 study of dogs with stage II cutaneous MCTs. 38 Another limitation of the present study was the small number of cases, particularly grade I MCTs, which hindered statistical comparisons based on the 3-tier grading system. The number of events did not allow for a robust multivariate model without risking overfitting and compromising the reliability of the results. For this reason, we restricted our analyses to univariate approaches, which we believe are still informative within the context of our data set. Future studies with larger case numbers are necessary to validate our findings.
Our study is the first to investigate lymphatic vasculature and its prognostic value in canine MCTs using an anti-LYVE1 antibody, demonstrating that this is an efficient marker, as in human tumors. The MCTs have ITLVs and PTLVs, but only PTLVs have prognostic value for this cancer in dogs, and these vessels may play an important role in lymphatic metastasis.
The results presented in this study reinforce the importance of lymphatic vessels in the tumor microenvironment. Future investigations into the mechanisms of intravasation in lymphatic vessels and related receptors and chemokines may provide important information and may generate new therapeutic strategies to prevent lymphatic dissemination of neoplastic cells.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251400715 – Supplemental material for Prognostic significance of lymphatic vessel density in canine cutaneous mast cell tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858251400715 for Prognostic significance of lymphatic vessel density in canine cutaneous mast cell tumors by Karina C. Bittencourt, Adriana T. Nishiya, Juliana V. Cirillo, Karine G. Cadrobbi, Rodrigo Ubukata, Alessandra R. N. Voges, Paulo C. Jark, Carolina G. Pires, Daniel S. Sanches, Felipe A. R. Sueiro and Ricardo F. Strefezzi in Veterinary Pathology
Footnotes
Acknowledgements
We thank Lindsay Baltel Paskoski for technical support.
Supplemental Material for this article is available online.
Author Contributions
KCB collected samples and clinical data, performed and quantified the immunohistochemistry reactions, analyzed the slides, contributed to the statistical analysis, and wrote the manuscript. KCB and RFS performed histological analysis and grading of tumor samples. ATN, JVC, KGC, RU, ARNV, PCJ, CGP, DSS, and FARS provided samples and clinical data for the study. RFS guided the study and performed the statistical analysis. All authors reviewed the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, grants #2020/10582-0 and #2022/09378-5), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Código de Financiamento 001), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—grant #303748/2021-4).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
