Abstract
Immunohistochemistry for E-cadherin (ECAD) has been used to distinguish canine cutaneous histiocytoma from other leukocytic neoplasms (“round cell tumors”). To determine the specificity of this test, 5 types of canine cutaneous round cell tumors were evaluated for immunohistochemical expression of ECAD. Tumors of all 5 types had variable cytoplasmic, plasma membrane, and/or paranuclear ECAD expression: All 13 cutaneous histiocytomas were ECAD+; all but 1 of 14 mast cell tumors expressed ECAD; 10 of 12 epitheliotropic lymphomas reacted with E-cadherin antibody; of 72 plasmacytomas, 54 were ECAD+; and 5 of 5 histiocytic sarcomas were positive. Conclusions based on these results include the following: First, immunoreactivity for ECAD is not limited to leukocytes of cutaneous histiocytoma; second, antibody to ECAD also labels neoplastic cells in most mast cell tumors, plasmacytomas, cutaneous histiocytic sarcomas, and epitheliotropic lymphomas; third, although most histiocytomas have membranous ECAD expression, the immunoreactivity varies among round cell tumors and is frequently concurrent in different cellular compartments; fourth, the distinctively paranuclear ECAD expression pattern in epitheliotropic lymphomas might distinguish them from other round cell tumors; and, fifth, ECAD should be used with other markers (eg, MUM1 for plasmacytomas, KIT for mast cell tumors, CD3 and CD79a for lymphomas) to distinguish among canine round cell tumors.
Keywords
Cadherins are calcium-dependent, transmembrane homophilic adhesion molecules with roles in morphogenesis and maintenance of tissue integrity. 31 In the epidermis, E-cadherin (ECAD) is present in basilar and other keratinocytes; its intracytoplasmic domain anchors to other cytoskeletal proteins, such as catenin and actin. 4,33 In addition to its expression in the epidermis, ECAD is expressed in organs derived from epithelia, Langerhans cells, immature thymocytes, some lymphocyte subtypes, mast cells, and nucleated erythroid cells. 1,2,4,15,18,22,27,29,30,34 Epidermal keratinocytes and Langerhans cells interact via adherens junctions, which contain ECAD. 12 Intracellular ECAD distribution depends in part on the cell type and its functional status. 5,6,11 ECAD has been considered a marker for diagnosis and prognosis in human cancer because of its frequent loss in many invasive cancers, including carcinomas of the head and neck and urogenital and digestive systems. 6 In addition to labeling canine epithelial cells and their tumors, ECAD has been used as a marker for canine cutaneous histiocytomas. 4,18 However, an observation of strong immunoreactivity for ECAD in a routine diagnostic workup of a canine cutaneous plasmacytoma prompted an evaluation of the cross-reactivity of ECAD among nonhistiocytic canine round cell tumors (plasmacytomas, mast cell tumors [MCTs], and epitheliotropic lymphomas) that commonly occur in the skin.
Materials and Methods
For this study, 116 canine round cell tumors were examined, including 13 cutaneous histiocytomas, 72 plasmacytomas (60 cutaneous, 8 oral, and 4 intestinal), 12 epitheliotropic (T-cell) lymphomas (n = 10 cutaneous, 1 nodal, 1 intestinal), 14 cutaneous MCTs (n = 5, grade 2; n = 9, grade 3), and 5 cutaneous histiocytic sarcomas. In addition, 2 normal lymph nodes were evaluated for ECAD expression. Tumor diagnosis was based on histologic features in hematoxylin and eosin–stained slides. 32 Grading of MCTs was done according to Patnaik’s system. 16 In addition, immunohistochemistry (IHC) was done in many of these tumors to confirm the diagnosis: MUM1 protein for all plasmacytomas, 23 KIT for some MCTs, 35 and CD3 for all T-cell lymphomas. 21 Histochemistry for metachromatic granules was used in poorly granulated MCTs (toluidine blue and/or Giemsa stain).
For IHC for ECAD, formalin-fixed, paraffin-embedded tissue sections were deparaffinized and hydrated before antigen retrieval in a decloaker (Biocare Medical, Concord, CA) with citrate buffer (pH 6.0). Slides were incubated at room temperature with a mouse monoclonal antibody to ECAD (clone 36, BD Transduction, San Jose, CA) for 60 minutes. According to the manufacturer, this antibody recognizes the ECAD cytoplasmic domain, regardless of its phosphorylation status. The antibody recognizes a 120-kDa protein upon Western blot. A streptavidin–biotin peroxidase detection system (LSAB+, Dako, Carpinteria, CA), with diaminobenzidine as chromogen, was used to detect the immune reaction. 20
The intensity of the immunohistochemical reaction for ECAD was assessed semiquantitatively: 0, no reaction; 1, weak (10–20% positive cells); 2, moderate (< 50% positive cells); 3, strong (≥ 50% positive cells). The cellular location of ECAD was recorded as plasma membrane, cytoplasmic, or paranuclear. Nuclear immunolabeling was not observed. Expression of ECAD was not unusual in multiple cellular compartments within the same tumor or even within a single cell. To confirm the specificity of the reaction, the primary antibody was replaced with mouse immunoglobulin-negative reagent control (Dako).
Results
Most canine round cell tumors (95 of 116) expressed ECAD immunohistochemically. Of the positive tumors, ECAD expression was moderate or strong in all but 19 (1 histiocytoma, 1 MCT, 4 T-cell lymphomas, and 13 plasmacytomas). Tumors of all 4 types had variable cytoplasmic, plasma membrane, and/or paranuclear ECAD expression. Multiple patterns were commonly present within the same tumor or cell.
All 13 cutaneous histiocytomas were ECAD+ (7 plasma membrane, 4 cytoplasmic/plasma membrane, 2 cytoplasmic/plasma membrane/paranuclear). The expression of ECAD did not vary with tumor depth except in cutaneous histiocytomas, in many of which the ECAD labeling faded with increasing distance from the epidermis (Figs. 1, 2 ). Of the 72 plasmacytomas, 54 (75%) were ECAD+ (30 cytoplasmic/plasma membrane, 12 cytoplasmic, 5 cytoplasmic/plasma membrane/paranuclear, 4 cytoplasmic/paranuclear, 2 plasma membrane, 1 paranuclear) (Figs. 3–7). All but 1 of 14 MCTs expressed ECAD (7 plasma membrane/cytoplasmic, 5 plasma membrane/cytoplasmic/paranuclear, 1 plasma membrane). Cytoplasmic labeling for ECAD was also present in the negative reagent slide of 7 MCTs, with an intensity similar to that in the positive reagent slide. Given the possibility that endogenous biotin-like activity was involved in the cytoplasmic labeling of MCTs, immunostaining with a polymer-based (non–avidin–biotin) detection system was used for this group of tumors with results similar to those obtained using the avidin–biotin system (Figs. 8, 9 ). This diffuse cytoplasmic labeling of the negative reagent slide was not observed with other tumor types. Of 12 epitheliotropic lymphomas, 10 expressed ECAD (9 paranuclear, 1 paranuclear/cytoplasmic) (Fig. 10 ). All 5 cutaneous histiocytic sarcomas had reactivity for ECAD (3 cytoplasmic/plasma membrane, 1 cytoplasmic/paranuclear, 1 cytoplasmic/plasma membrane/paranuclear). The ECAD-positive cells in the lymph node were follicular dendric cells, paracortical dendritic cells, and numerous histiocytic cells populating the sinusoidal system. Normal lymphocytes did not express ECAD.
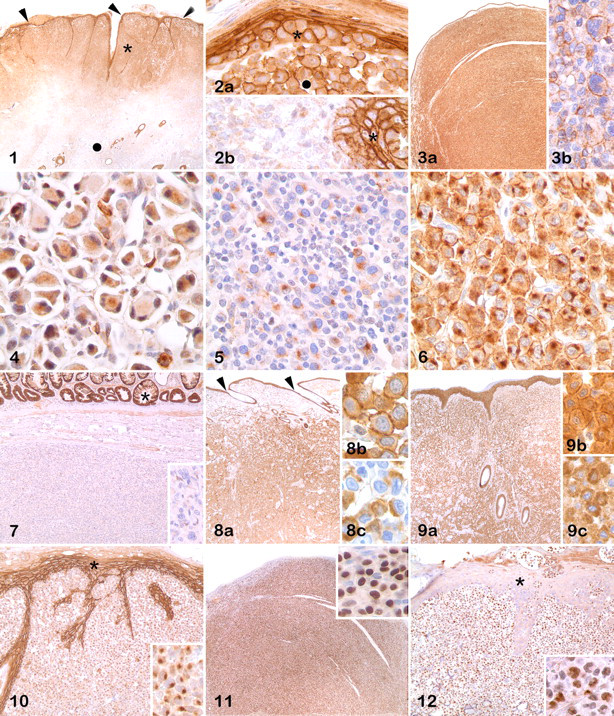
MUM1 protein was detected in all 72 plasmacytomas and in 1 of 12 epitheliotropic T-cell lymphomas but in none of the MCTs examined (Figs. 11, 12 ). All T-cell lymphomas were CD3 positive. All MCTs evaluated for KIT expression or presence of metachromatic granules were positive.
There was strong cytoplasmic membrane and less intense cytoplasmic in follicular dendritic cells and in dendritic cells of the paracortex and medullary cords of reactive lymph nodes. Histiocytic sinusoidal cells had also a similar and strong labeling.
Discussion
ECAD, a glycoprotein involved in intercellular adhesion, has been used as a marker of epithelial differentiation in neoplasms. ECAD has been detected in human lymphocytes (by flow cytometry), 1 lymphomas and leukemias (by IHC, gene expression by polymerase chain reaction) 3,14 and myelomas (IHC, polymerase chain reaction, and circulating ECAD by ELISA), 8,13,25,28,36 human and canine meningiomas (IHC), 22 and canine Merkel cell tumors (IHC), in addition to carcinomas of various types. 10,26 Human and murine mast cells also express ECAD, as detected by flow cytometry, polymerase chain reaction, immunoblotting, and IHC. 9,15,29,30
ECAD is consistently expressed in canine cutaneous histiocytomas. 4,18 In the study reported here, however, antibody to ECAD labeled neoplastic cells in 5 types of canine leukocytic neoplasms, so it was not diagnostic for cutaneous histiocytoma. The intensity of ECAD immunohistochemical labeling in canine cutaneous histiocytoma characteristically decreases with increasing distance from the epidermis. 18 This reduction in ECAD expression is probably the result of Langerhans cell maturation 12,18 or the immune response during tumor regression. 17 The same reduction in intensity of ECAD immunoreactivity was observed in many but not all cutaneous histiocytomas in this series. However, this inverse correlation of immunoreactivity with tumor depth was not obvious in the other types of round cell tumors, including cutaneous histiocytic sarcomas.
Although ECAD is typically expressed in the plasma membrane, other cellular locations have been reported (cytoplasmic and nuclear), particularly in neoplastic processes. 6,7,24 Interestingly, ECAD nuclear distribution correlates with lymph node and liver metastases in human pancreatic endocrine tumors. 6 Nuclear labeling was not observed in any of the canine round cell tumors in this study.
Expression of ECAD has been reported in human mast cells by some researchers but not others; see references 9 and 15. Interestingly, the same mast cell line was used in those 2 studies, so differences in the antibody to ECAD might have accounted for the contradictory results. In the present study, most cutaneous MCTs had strong expression of ECAD, with plasma membrane and cytoplasm labeled in most cases and with only a rare paranuclear pattern. Initially, a streptavidin–peroxidase IHC method was used; however, because of the strong ECAD reactivity in most MCTs and even in some negative-reagent control slides, the possibility of nonspecific binding of avidin to mast cells was considered. 19 When IHC was repeated using a non–avidin–biotin system, the negative-reagent control sections had cytoplasmic labeling in 7 cases, but no plasma membrane labeling. Based on the difference in labeling patterns between the positive and negative slides, only those MCTs with plasma membrane labeling (13 of 14) were counted as ECAD positive.
As in MCTs, ECAD immunoreactivity was detected in different cellular compartments in most canine plasmacytomas. Of 54 positive plasmacytomas, 12 had only cytoplasmic reactivity, which was usually weak; however, because the negative-reagent control slide lacked reactivity, this was interpreted as specific labeling. Immunohistochemical ECAD expression has been observed in 46% of human myelomas; increased ECAD expression in tumor cells or increased circulating soluble ECAD correlated with poor prognosis. 13,28 Moreover, hypermethylation of the ECAD gene has been linked to progression in human multiple myeloma in some studies. 8,25,36 The significance of ECAD expression in canine plasmacytomas is unknown, but based on the clinical history in this canine series and the well-known benign behavior of most canine cutaneous plasmacytomas, ECAD expression or lack thereof is not predictive of tumor outcome.
In one study, 3 pan-cadherin was expressed in 13 of 14 human anaplastic lymphomas, 50% of T-cell lymphomas, and less commonly in other lymphomas; interestingly, lymphocytes in reactive lymph nodes did not express pan-cadherin. In the same study, when based on an antibody specific for ECAD, neither lymphomas nor normal lymphocytes expressed this marker. In our series of canine epitheliotropic lymphomas, 10 of 12 expressed ECAD in a distinct paranuclear pattern, with only 1 having both paranuclear and cytoplasmic labeling. Although paranuclear ECAD expression was observed in all 4 types of canine round cell tumors, this was the only expression pattern in 9 of 10 epitheliotropic lymphomas. Normal canine lymph node lymphocytes did not express ECAD, but expression was strong in nodal histiocytes and dendritic cells.
Among canine round cell tumors, MUM1 protein nuclear expression is typically limited to plasmacytomas and a few lymphomas. 23 In the present study, no tumor except all the plasmacytomas and 1 epitheliotropic lymphoma expressed nuclear MUM1 protein. This result confirms the specificity of MUM1 for canine plasmacytomas. The strong cytoplasmic labeling with antibody to MUM1 in some MCTs was considered nonspecific because (1) MUM1 cytoplasmic labeling in plasmacytomas is not the criterion to determine the positive/negative status (only nuclear labeling is considered diagnostic) and (2) the negative-reagent control slides had similar cytoplasmic reactivity to that in the positive-reagent control in 7 MCTs.
In conclusion, immunoreactivity for ECAD is not exclusive to leukocytes of cutaneous histiocytoma. Antibody to ECAD also labels neoplastic cells in most MCTs, plasmacytomas, cutaneous histiocytic sarcomas, epitheliotropic lymphomas, and normal histiocytic and dendritic cells of lymph nodes. Although histiocytomas usually have a membranous component to ECAD expression, the pattern of immunoreactivity varies among round cell tumors and is frequently concurrent in different cellular compartments. The distinct paranuclear ECAD expression pattern in epitheliotropic lymphomas might distinguish them from other round cell tumors. ECAD IHC should be used only in conjunction with other markers (eg, MUM1 for plasmacytomas, KIT for MCTs, CD3 and CD79a for lymphomas) to distinguish canine cutaneous histiocytoma from its cutaneous mimics.
Footnotes
Acknowledgements
We appreciate Dee DuSold’s expertise in immunohistochemical procedures. Preliminary results of this study were presented at the 2009 American College of Veterinary Pathologists annual meeting (Vet Pathol
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
