Abstract
Histologic grading of canine cutaneous mast cell tumors (cMCTs) has prognostic and therapeutic implications, yet validation for subcutaneous MCTs (scMCTs) is lacking. For scMCTs with or without dermal invasion, determining their biological behavior remains poorly standardized and sometimes sparks controversy. This prospective study aimed to assess the prognostic utility of the 2-tier histologic grading system in MCTs with different growth models (GMs) and explore the prognostic impact of the GM itself. We assessed 6 histologic GM categories: solely cMCT (C-SC0), cMCT with superficial (C-SC1) or deep subcutaneous (C-SC2) involvement, solely scMCT (SC-C0), and scMCT with deep (SC-C1) or superficial (SC-C2) infiltration of the dermis. Ninety-one MCTs from 76 dogs undergoing excision and regional/sentinel lymphadenectomy were examined. GM classification identified 11 (12%) C-SC0 tumors, 12 (13%) C-SC1, 15 (16%) C-SC2, 21 (23%) SC-C0, 15 (16%) SC-C1, and 17 (19%) SC-C2. Mitotic count, 2-tier grade, nodal involvement, surgical margins, and outcome were stratified according to GM. scMCTs lacking dermal invasion, historically associated with a benign clinical course, had a poor prognosis in 10% of cases. cMCTs exhibiting deep subcutaneous involvement included the largest percentage of high-grade tumors (33%), had the highest occurrence of overt nodal metastases (33%), and had the lowest 1-year survival rate (86%). Histologic grade was confirmed as a relevant prognostic factor, surpassing nodal involvement and histologic margin status. The 2-tier histologic grading enabled the identification of all MCTs with aggressive biological behavior, regardless of their cutaneous or subcutaneous location.
Keywords
Mast cell tumors (MCTs) are the most common skin neoplasms in dogs. The prognostication of canine MCTs has historically differed based on their growth model (GM). For MCTs arising primarily in the dermis (cutaneous MCTs [cMCTs]) 2 histologic grading systems have been introduced: the 3-tier (Patnaik) system, which considers both architectural and morphologic criteria, and the 2-tier (Kiupel) system, which exclusively relies on cell morphology.8,16 According to the 3-tier system, cMCTs infiltrating the subcutaneous tissues exhibit a more aggressive behavior and a worse outcome compared with those confined to the dermis. 16
A subset of canine MCTs is confined entirely within the subcutis, with limited or no involvement of the dermal layer (subcutaneous MCTs [scMCTs]). Until recently, these tumors had received limited attention and were either not graded or arbitrarily classified as Patnaik grade II due to their subcutaneous location. Several studies addressing this MCT variant reported extended survival times and low rates of local recurrence and metastasis.4,15,20 However, recent reports have highlighted a small number of cases displaying aggressive biological behavior.3,12,21
Histologically, several negative prognostic factors have been identified for canine scMCT, including mitotic count (MC), infiltrative growth, and multinucleation. 20 However, no specific grading system has been proposed, and it remains uncertain which combination of negative histologic prognostic factors could be considered equivalent to high grade. In a recent study that applied the 2-tier grading system to scMCTs, only one dog was diagnosed with a high-grade tumor, and in no case did the MC reach the cutoff established for high-grade cMCTs. 4 These findings support the hypothesis that scMCTs as an entity are generally lower grade. Alternatively, this may suggest that the histologic prognostic factors identified for cMCTs may not be applicable to the subcutaneous variant.
Additionally, in a significant proportion of canine MCTs, the bulk of the tumor resides within the subcutis, but neoplastic cells extend upward to infiltrate the deep or intermediate dermis, or even reach the superficial dermis. From a clinical perspective, most of these tumors cannot be distinguished from primary cMCTs. This particular GM represents a gap that has not been previously addressed in the literature. As a result, the histologic approach to these tumors is quite confusing; many pathologists grade them as if they were cMCTs with deep subcutaneous invasion, while others recognize their subcutaneous origin and apply the prognostic criteria recommended for scMCTs. If a single grading system could be applied to all canine MCTs, regardless of their primary location and extent of infiltration, this would ensure a reduced level of subjectivity in the histologic interpretation of their biological behavior, ultimately resulting in more informed clinical management.
In this prospective study, the prognostic impact of MCT GM was assessed in a cohort of dogs undergoing primary tumor removal and regional or sentinel lymphadenectomy. For this purpose, tumors were separated into several histologic GM-defined categories, including solely dermal MCTs, solely scMCTs, primarily dermal MCTs extending into the subcutis, and primarily scMCTs extending into the dermis. The primary aim was to compare the biological behavior of cMCTs with deep subcutaneous invasion with that of scMCT displaying dermal invasion, to assess whether these GMs merit distinction in histopathologic reports. Additional aims were to investigate if there were differences in the 2-tier grade distribution based on the tumor GM and if grading correlated with tumor biological behavior across all categories.
Materials and Methods
Study Design and Inclusion Criteria
Client-owned dogs with cMCTs or scMCTs undergoing surgical excision of the primary tumor and regional or sentinel lymphadenectomy at the University Hospital of the Department of Veterinary Medical Sciences (University of Bologna, Italy) between January 2018 and December 2022 were eligible for inclusion. Dogs with more than 2 concurrent MCTs, recurrent MCTs, or with mucosal or muscular MCTs were excluded; comorbidities limiting life expectancy to less than 6 months represented a further exclusion criterion. Dogs with 2 concurrent or asynchronous primary MCTs were included if they underwent surgical excision of both tumors and removal of all regional/sentinel lymph nodes. All dogs had to be negative for distant metastases prior to surgery as assessed by completion of a series of diagnostic procedures, including 3-view thoracic radiographs, abdominal ultrasound, and fine-needle aspiration of the liver and spleen. MCTs were excised according to recent recommendations, with lateral surgical margins proportional to the widest tumor diameter, and deep margins including at least 1 fascial plane.6,17 The obtained surgical samples were subjected to histologic evaluation. In the presence of high-grade tumors 8 and/or overt nodal metastasis, 22 adjuvant vinblastine treatment was recommended, in accordance with prior literature. 11 The remaining dogs were monitored. Dogs were withdrawn from the study if they were lost to follow-up within 120 days of surgery.
Histopathologic Examination
Samples were fixed in 10% neutral-buffered formalin, processed, and embedded in paraffin using a standardized protocol. Four-micrometer-thick histologic sections of the primary tumor, stained with hematoxylin and eosin, were microscopically examined for the assessment of tumor GM, MC, histologic grade according to the 2-tier system, 8 and surgical margins. The histologic GM was assessed as detailed in Table 1. Six categories were defined: solely dermal MCT (C-SC0), dermal MCT with superficial or deep subcutaneous involvement (C-SC1 and C-SC2, respectively), solely scMCT (SC-C0), and scMCT with deep or superficial infiltration of the overlying dermis (SC-C1 and SC-C2, respectively). MC was expressed as the total number of mitotic figures in a 2.37-mm2 area and assessed in contiguous fields within the areas of highest mitotic activity. 13 The 2-tier histologic grading system was applied to all tumors, regardless of the GM. Surgical margins were inked by the pathologist prior to tissue trimming; assessed histologically with combined radial and tangential sections; and defined as complete, clean but close (tumor cells at 1–3 mm from the surgical margins), or incomplete. Formalin-fixed lymph nodes were bisected along their longest axis through the hilum, and both halves were embedded in paraffin. The first section and a step-section at 100 or 200 µm (depending on the lymph node size) and stained with toluidine blue were examined. 19 The histologic node status was evaluated according to criteria established by Weishaar et al 22 as HN0 (non-metastatic), HN1 (pre-metastatic), HN2 (early metastatic), and HN3 (overt metastatic). All histologic evaluations were performed by a board-certified veterinary pathologist (SS).
Growth models of canine mast cell tumor (MCT) identified in the present study.
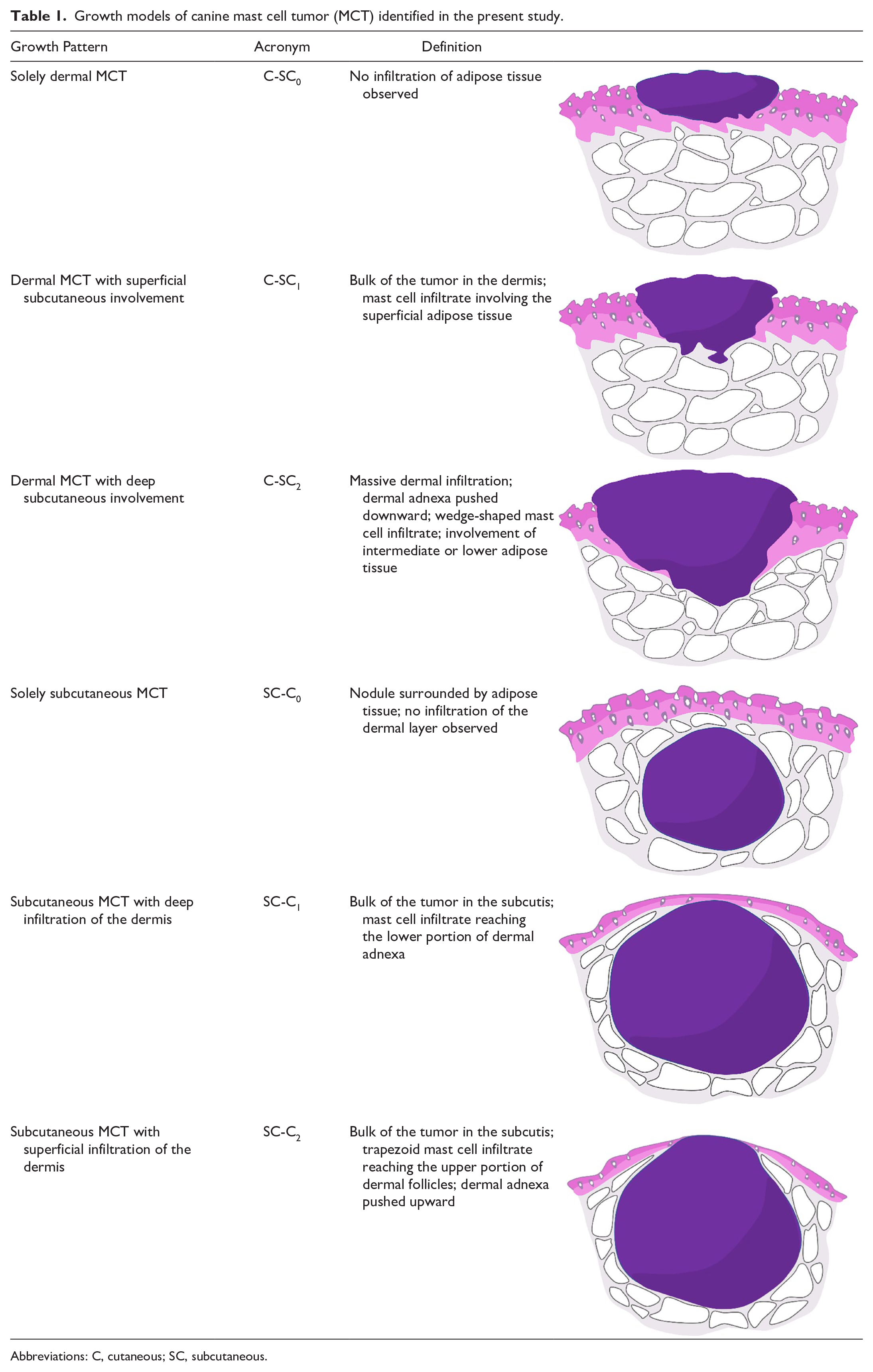
Abbreviations: C, cutaneous; SC, subcutaneous.
Statistical Analysis
Descriptive statistics were used in the analysis of dogs and tumor characteristics. When appropriate, data sets were tested for normality by use of the D’Agostino and Pearson omnibus test. None of the numeric variables had a normal distribution, and therefore, the median and range were used as summary statistics. Information recorded for all dogs included signalment, tumor anatomic location, largest diameter, clinically assessed site (cutaneous or subcutaneous), ulceration, substage, GM, MC, 2-tier grade, surgical margins, extent of nodal involvement, date of tumor progression (if any), and date and cause of death (if any). Time to progression was calculated from the date of surgery to the first occurrence of one or more of the following: local recurrence, nodal metastasis, and distant spread. Dogs with no recurrence or disease progression at the date of the last visit or death were censored. Tumor-specific survival was calculated from the date of surgery to the date of death or to the date of the last visit if death did not occur. Only dogs deceased due to MCT-related causes were considered as events. One-year survival rates were also calculated for each group. Statistical inference was not performed because the assumptions of such methods in terms of sample size or data distribution did not hold. Data were analyzed by use of commercial software programs (SPSS Statistics v. 26, IBM, Somers, NY).
Results
Dogs’ and Tumors’ Characteristics
Ninety-one MCTs obtained from 76 dogs were included. There were 19 (25%) mixed-breed dogs and 57 (75%) purebred dogs; among these, the most represented breeds were Labrador retriever (n = 10, 18%), French bulldog (n = 8; 14%), boxer (n = 7, 12%), and golden retriever (n = 6, 11%). There were 39 females (51%), of which 28 were spayed, and 37 (49%) males, of which 8 were neutered. The median age at presentation was 8 years (range, 3.5–15.0), and the median body weight was 26.5 kg (range, 5.4–58.7). The tumors were located on the trunk and tail (n = 30, 33%), limbs (n = 28; 31%), head and neck (n = 14; 15%), inguinal/perineal area (n = 10, 11%), mammary region (n = 7, 8%), and the digital region (n = 2; 2%). From a clinical perspective, 53 (58%) tumors were cutaneous and 38 (42%) were subcutaneous. The median tumor diameter was 1.5 cm (range, 0.2–16.0 cm). Four (5%) dogs were symptomatic (substage b).
Treatment
Seven (9%) dogs received neoadjuvant chemotherapy, consisting of vinblastine administered intravenously every 2 weeks at the dose of 3 mg/m2 for dogs weighing ≥ 20 kg and 2.5 mg/m2 for those weighing < 20 kg. Dogs also received daily oral prednisolone (1 mg/kg), oral cetirizine (1 mg/kg) twice daily, and oral famotidine (2 mg/kg) twice daily. All dogs underwent surgical excision of their MCTs and lymphadenectomy. A total of 169 regional and 19 sentinel lymph nodes were removed, with a median of 2 lymph nodes for each dog (range, 1–6). Based on the presence of a high-grade tumor and/or HN3 lymph node, 19 (25%) dogs received adjuvant chemotherapy, consisting of vinblastine administered as previously described up to a total of 8 doses.
Histologic Analysis
Eighty-one (89%) tumors were low grade and 10 (11%) were high grade. The median MC was 0 (range, 0–14). Surgical margins were histologically complete in 76 (84%) cases, clean but close in 12 (13%) cases, and incomplete in 3 (3%) cases. Forty-three (47%) dogs had at least 1 HN2 lymph node and 16 (18%) had at least 1 HN3 lymph node.
According to the histologic GM, 11 tumors (12%) were classified as C-SC0, 12 (13%) as C-SC1, 15 (16%) as C-SC2, 21 (23%) as SC-C0, 15 (16%) as SC-C1, and 17 (19%) as SC-C2 (Fig. 1). The main characteristics for each GM-defined group are listed in Table 2. Tumors treated with neoadjuvant chemotherapy included 3 SC-C2, 2 SC-C0, and 2 C-SC2. High-grade MCTs included 5 C-SC2, 2 SC-C0, 2 SC-C2, and 1 SC-C1. Tumors with at least 1 HN3 lymph node included 6 SC-C0, 5 C-SC2, 3 SC-C2, 2 C-SC1, and 1 SC-C1.
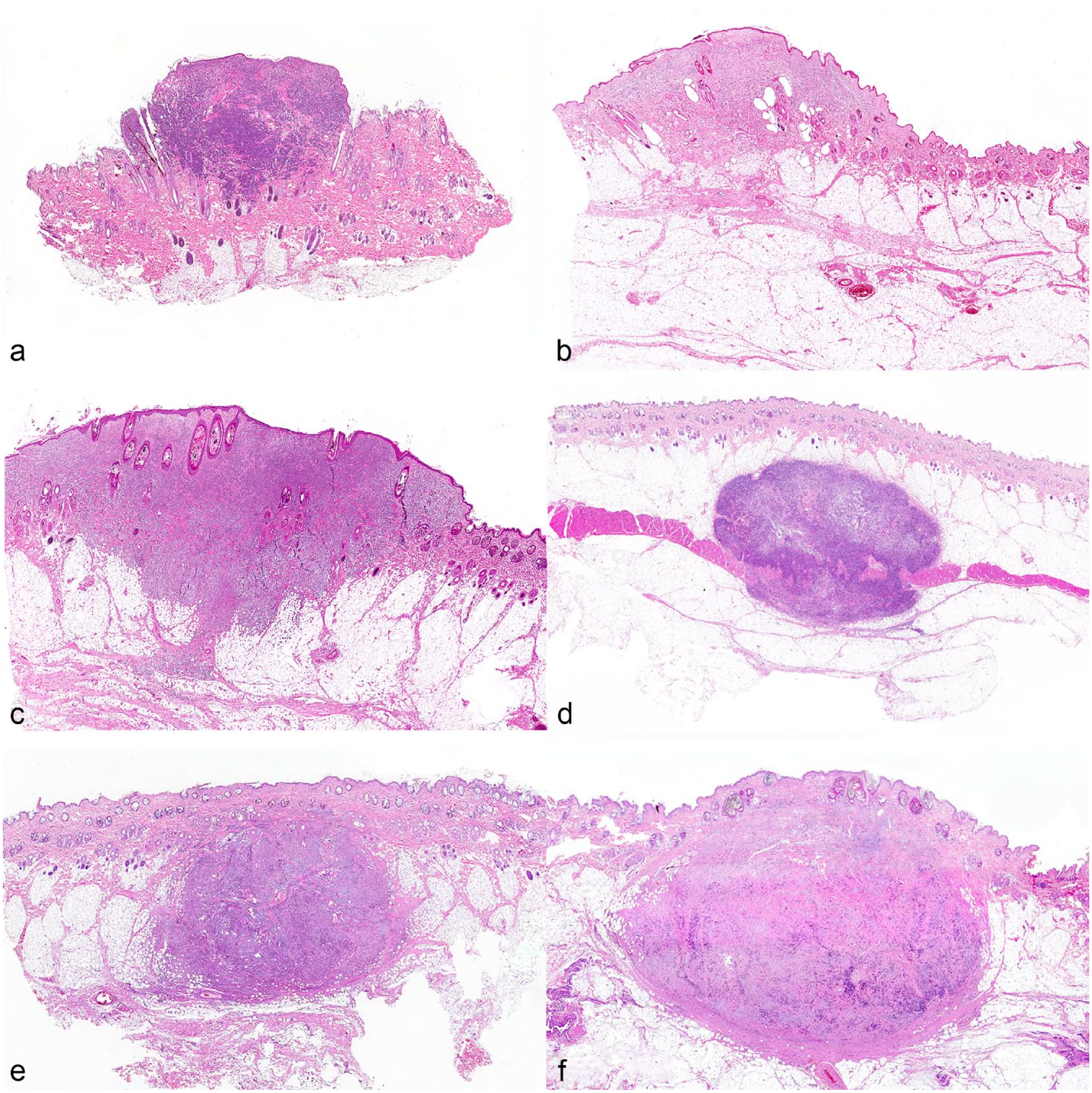
Dog, mast cell tumor (MCT), skin. Hematoxylin and eosin. Representative examples of canine MCT growth models. (a) Solely dermal MCT (C-SC0). (b) Dermal MCT with superficial subcutaneous involvement (C-SC1). (c) Dermal MCT with deep subcutaneous involvement (C-SC2). (d) Solely subcutaneous MCT (SC-C0). (e) Subcutaneous MCT with deep infiltration of the dermis (SC-C1). (f) Subcutaneous MCT with superficial infiltration of the dermis (SC-C2).
Clinicopathologic features and outcome of 91 cases of canine mast cell tumor (MCT) grouped according to their growth model.
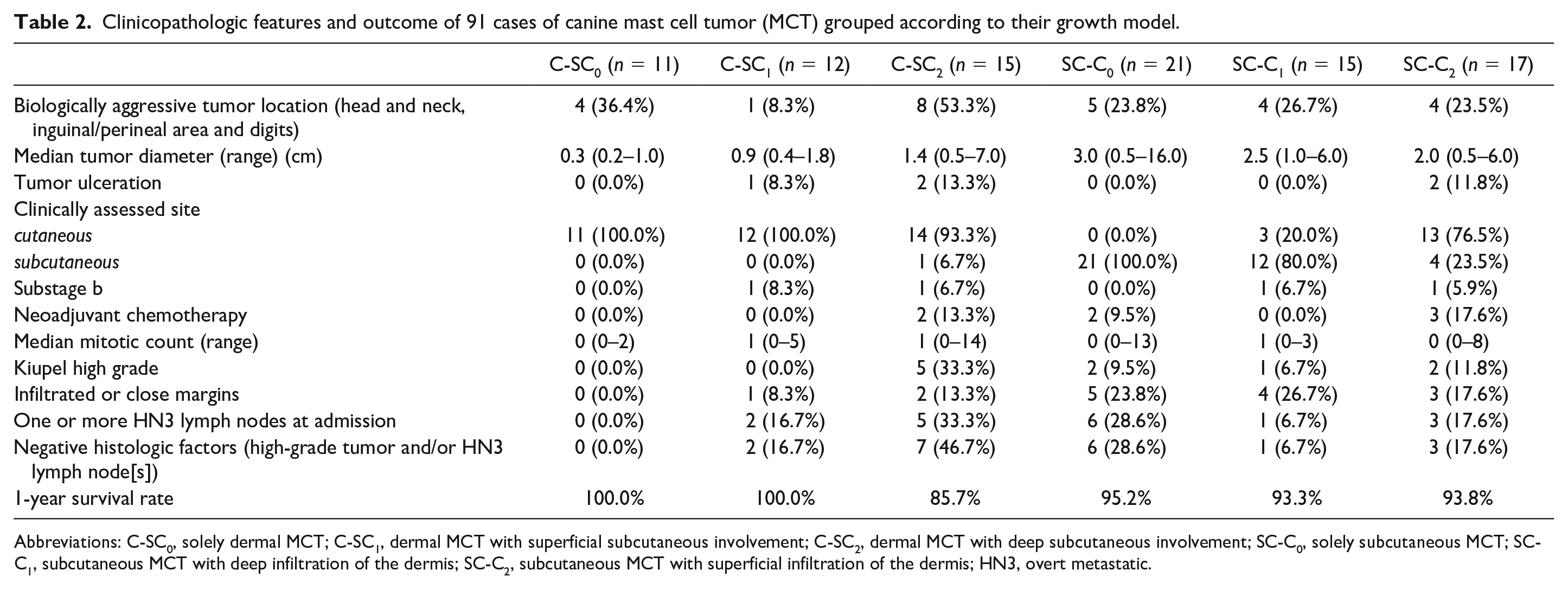
Abbreviations: C-SC0, solely dermal MCT; C-SC1, dermal MCT with superficial subcutaneous involvement; C-SC2, dermal MCT with deep subcutaneous involvement; SC-C0, solely subcutaneous MCT; SC-C1, subcutaneous MCT with deep infiltration of the dermis; SC-C2, subcutaneous MCT with superficial infiltration of the dermis; HN3, overt metastatic.
When specifically considering tumors with extensive involvement of both the cutis and subcutis, C-SC2 MCTs had a median diameter of 1.4 cm (range, 0.5–7.0) and were described clinically as a cutaneous nodule in 14 (93%) cases and as a subcutaneous nodule in 1 (7%) case. Two (13%) tumors were ulcerated. SC-C2 MCTs had a median diameter of 2.0 cm (range, 0.5–6.0) and were described clinically as a cutaneous nodule in 13 (76%) cases and as a subcutaneous nodule in 4 (24%) cases. Two (12%) were ulcerated. The highest percentage of high-grade tumors and HN3 lymph nodes (n = 5, 33%) was found in the C-SC2 category (Table 2).
Outcome and Prognostic Factors
The median follow-up time was 681 days (range, 217–2054). Seven (9%) dogs experienced disease progression after a median of 230 days (range, 49–666), consisting of visceral metastasis (n = 3), nodal metastasis (n = 3), and local recurrence with nodal metastasis (n = 1). All progressions were confirmed by cytologic evaluation. The median time to progression could not be assessed since the estimated survival curve did not fall below 0.5. The GM of tumors associated with disease progression included 3 C-SC2, 2 SC-C0, 1 SC-C1, and 1 SC-C2.
At the end of the study, 63 (83%) dogs were alive and 7 (9%) died because of MCT-unrelated causes, including one each of splenic hemangiosarcoma, heart failure, brachycephalic airway obstructive syndrome, pancreatitis, pancreatic insulinoma, brain neoplasia, and degenerative myelopathy. Six (8%) dogs died because of MCT-related causes after 139, 191, 219, 321, 349, and 422 days due to visceral metastasis. The median tumor-specific survival could not be estimated. The GM of MCTs in the dogs that died of tumor-related causes included 2 C-SC2, 2 SC-C0, 1 SC-C1, and 2 SC-C2. The 1-year survival rate was 100% for C-SC0 and C-SC1 MCTs, 95% for SC-C0 MCTs, 94% for SC-C2 MCTs, 93% for SC-C1 MCTs, and 86% for C-SC2 MCTs (Table 2).
Discussion
Histologic grading is currently validated exclusively for canine cMCTs, and divergent opinions exist regarding the appropriateness of grading MCTs arising in other sites. In solely scMCTs, grading might underestimate a tumor’s biological behavior, while for scMCTs with secondary dermal invasion, there is a lack of established guidelines, and the decision to apply grading is left to the discretion of the pathologist. In the present study, we have tested the prognostic utility of the 2-tier grading system in canine MCTs with different histologic GMs. The prognostic impact of the GM itself was also investigated.
The category of primarily cMCTs with deep subcutaneous infiltration (C-SC2) included the highest proportion of high-grade tumors (33%) and overt metastasis to lymph nodes (33%); moreover, dogs within this group had a higher frequency of tumor progression (20%) and the lowest 1-year survival rate (86%). The C-SC2 GM has been previously acknowledged as a negative prognostic factor, as the replacement of subcutaneous and deep tissues is considered a feature of grade III tumors in the 3-tier grading system. 16 In contrast, the category of scMCTs with superficial dermal involvement (SC-C2) had lower proportions of high-grade tumors, HN3 lymph nodes, tumor progression, and MCT-related deaths. Based on these results, it may be worthwhile to differentiate between the C-SC2 and SC-C2 GMs, rather than generically describing concurrent cutaneous and subcutaneous infiltration. Clinically, most of the tumors with these 2 GMs were identified as cutaneous, appearing macroscopically indistinguishable. Therefore, histology frequently plays a crucial role in discerning between them. scMCTs may infiltrate the adjacent tissues, including a variable degree of dermal invasion. As a result, all MCTs in which the main bulk of the tumor is in the subcutis are likely to originate subcutaneously and SC-C0-2 tumors clearly represent a continuum. By excluding cases with secondary dermal invasion from studies investigating the biological behavior of scMCTs, similar to what other authors have done in the past, we could potentially be omitting cases of advanced disease and/or locally aggressive forms, leading to an underestimation of the true biological behavior of these tumors.
Notably, even considering the category of solely scMCTs (SC-C0), 6 (29%) cases had overt metastasis to lymph nodes, and 2 (10%) tumor-related deaths were recorded. These tumors have been historically associated with a good prognosis and with low metastatic and recurrence rates despite incomplete surgical removal. 15 More recently, several authors hypothesized that they may constitute a more aggressive disease than previously reported.3,12,21 It has been suggested that application of the 2-tier grading system might lead to an underestimation of the true biological behavior of scMCTs, due to lower mitotic rates and different morphologic characteristics. 4 However, no specific grading system is currently available for canine scMCTs, which is a clear limitation in terms of their prognostication and post-surgical treatment decisions. In the present study, both dogs that died due to SC-C0 MCT had high-grade tumors, suggesting that the 2-tier system has prognostic value for scMCT prognostication, and a validation study should be conducted.
Indeed, in the current study, all dogs with disease progression had a high-grade tumor, regardless of their GM. The application of a sole grading system could simplify the work of pathologists, as they would no longer be required to rely on the tumor’s location to predict its behavior. This would eliminate a major source of subjectivity in histopathologic reporting, thereby offering clinicians more reproducible information to manage their patients. Furthermore, this may indirectly validate the application of the Camus cytologic grading system across all MCTs, as it mostly relies on the same parameters included in the 2-tier grading system. 2 This would be of great practical utility, since determining the clinical location of the tumor (cutaneous vs. subcutaneous) might not always be straightforward, as corroborated by the findings of the current study.
Together with grading, the presence of one or more lymph nodes with overt metastasis (HN3) has been confirmed as a negative prognostic factor, although not necessarily associated with tumor-related death in low-grade tumors. This finding further supports the previously demonstrated different significance of nodal metastasis in terms of its impact on low-grade and high-grade tumors. 5 Similarly, we confirmed the limited prognostic relevance of early (HN2) nodal metastases, which did not result in disease progression in any of the cases, even without the administration of adjuvant treatments. 11
Meaningful differences were observed between cMCTs and scMCTs regarding the histologic completeness of surgical excision. The removal of tumors belonging to the subcutaneous categories more frequently resulted in clean but close or incomplete margins (SC-C0, 24%; SC-C1, 27%; SC-C2, 18%). This observation is in line with previous studies that highlighted greater difficulties and less reproducibility in surgical margin planning for scMCTs.14,18 Furthermore, in anatomic sites where maintaining sufficient margins becomes challenging due to limited soft tissue available for deep excision, subcutaneous localization further reduces the chances of obtaining adequate deep margins. These results could also be explained by the larger size of scMTCs compared with cMCTs in this study. However, regardless of surgical margins, tumor progression was only detected in high-grade MCTs. This is consistent with previous studies and provides increasing evidence of the importance of histologic grade in canine MCTs.1,7
This study has several limitations. First, the small number of events (ie, tumor progression and tumor-related death) resulted in limited statistical power, despite a fairly large number of cases. Among the possible explanations, the radicality of the surgical approach likely contributed to this low progression rate, reaffirming its efficacy as a valid strategy in the treatment of these tumors. A further limitation may be the inclusion of dogs receiving neoadjuvant chemotherapy. Although no alterations of the histologic parameters included in the formulation of tumor grade have been reported after neoadjuvant treatments,9,10 these therapies could alter the actual GM of the tumor, potentially affecting the results of this study. However, the exclusion of these cases might have resulted in the preferential selection of mostly low-malignancy tumors, limiting the representation of the entire spectrum of MCTs in the study. Third, tumors with primary muscular locations were not included in the analysis, nor were mucosal MCTs or MCTs located in mucocutaneous junctions. Consequently, it remains to be determined whether the application of the 2-tier histologic grading can be extended to tumors in these locations. Fourth, in advanced tumors with massive dermal and subcutaneous involvement, the identification of the GM might not always be feasible. Therefore, the possibility of grading tumors regardless of their GM becomes even more important in such circumstances. Finally, it is worth considering that further variables related to the growth pattern (eg, circumscribed/expansile, infiltrative, or combined), that have been shown to be prognostic in previous studies, could also influence the tumor’s biological behavior. 20
In conclusion, regardless of the growth pattern, the 2-tier histologic grading system appears to accurately identify canine MCTs with aggressive biological behavior, including scMCTs with or without dermal invasion, for which specific guidelines are currently lacking. Histologic grade was confirmed as the most relevant prognostic factor, surpassing nodal involvement and histologic margin status. cMCTs with deep subcutaneous infiltration may exhibit a more aggressive biological behavior when compared with MCTs with other GMs. Recognizing this distinct pattern could hold prognostic significance.
Footnotes
Author Contributions
SS and AB designed the study and performed the experiments. LM and EF enrolled cases and contributed to the experimental design. SS, AB, RZ, and AR performed histologic evaluations. SS performed the statistical analysis. The manuscript was written by SS and AB with contributions from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
