Abstract
Canine cutaneous mast cell tumors (MCT) are common, frequently malignant neoplasms that are currently graded histologically for provision of prognostic information. Continuing evidence of subsets of MCT within certain grades (with differing survival times) indicate the need for biomarkers that will facilitate better patient stratification and also provide further information on the biological processes involved in progression. We decided to investigate the expression of p62/sequestosome-1 (p62/SQSTM1), a stress-inducible “hub protein” found in all cell types that shuttles rapidly between the nucleus and cytoplasm and is known to play important roles in protein handling and tumorigenesis. The identity of canine p62/SQSTM1 was confirmed in silico and by validation of a commercial antibody using both Western blotting and functional (pharmaceutical-based) analyses in cell culture. Using immunohistochemistry, 3 patterns of p62 expression were identified based on the predominant intracellular localization, that is, nuclear, mixed (nuclear and cytoplasmic), and cytoplasmic. There was a highly significant association with the 2-tier (Kiupel) grade (P < .0001), with all p62-nuclear immunoreactivity being associated with low grade and most p62-cytoplasmic immunoreactivity (93%) with high grade. Most but not all mixed nuclear-cytoplasmic labeling occurred in low-grade MCT; in other (human) tumor types, this pattern has been interpreted as borderline malignant. These data indicate that there is a shift in protein-handling stress from the nucleus to the cytoplasm in association with increasing malignancy in MCT. Studies to identify the processes and drug-able targets involved in this progression are ongoing.
Keywords
Mast cell tumors (MCTs) are one of the most common cutaneous neoplasms of dogs submitted to veterinary pathology laboratories. 34 In 1 UK study of insured dogs (n = 130 684), the incidence rate of MCT was estimated at 129 per 100 000. 6 It is well known that MCTs vary significantly in their biological behavior, from small well-differentiated tumors frequently amenable to local excision to those that are poorly differentiated and highly likely to metastasize widely. In recent years, a number of large studies have been conducted with the aims of reducing error and interobserver disagreement in prognostic histologic grading systems, most notably including the development and further validation of a 2-tier system. 16 Many diagnostic laboratories have adopted the 2-tier Kiupel system in addition to the widely used 3-tier Patnaik system. 16,26,27 A number of significant problems remain: the first being that in the intermediate Patnaik grade (grade II) and the Kiupel high-grade categories (and in subcutaneous MCT), there are poorly understood subcategories of MCT that differ in terms of behavior and therefore survival time. 27 Patnaik grade II tumors have been documented to result in death in 17% to 56% of dogs due to either recurrent local or metastatic disease. 4 Useful supplementary markers are required to further improve prognostic accuracy for individual cases that fall into these “gray areas.” Estimates of the replicative fraction obtained using Ki67 or MCM7 immunostain have been shown to be of some prognostic value, 1,2,33 although concerns remain over reproducibility and the practicalities of deriving robust replication scores. While grading systems are useful for stratifying patients in terms of the need for adjuvant therapy, they do not indicate exactly what drugs (or other therapies) are most likely to be successful. Thus far, only the identification of overactive c-KIT mutations (CD117; mast/stem cell growth factor receptor) can be used to provide some degree of targeted therapy using tyrosine kinase inhibitors (TKIs); this marker may also predict progression-free survival. 29 However, mutations in that gene arise in only 25% to 50% of canine malignant MCTs (Patnaik grade II or III). 11,19 The development of a personalized anticancer strategy is becoming increasingly important in the medical field and should be so for veterinary patients as well. To be successfully applied, we will need to identify further individually (and biologically) relevant biomarkers that suggest appropriate therapy.
p62/sequestosome-1 (p62/SQSTM1) is a “hub” protein involved in pro-survival nuclear factor (NF)–κB activity, apoptosis, and protein delivery to proteasomes and autophagosomes. 20,22,24 All of these processes and their deregulation are implicated in cellular transformation, and in recent years, there has been an exponential increase in interest in the key role of p62 in the pro-survival strategy of autophagy (occurring only in the cytoplasm and conferring stress tolerance). 22 p62 is known to shuttle extremely rapidly between the cytoplasm and nucleus (using nuclear localization and export signals), with the visualized immunoreactivity in either or both compartments being a “snapshot” of the current dynamic balance (or imbalance). 24 Recently, p62 expression was found to be nuclear-only in normal or hyperplastic prostate tissue, diffusely cytoplasmic in adenocarcinomas, and more frequently cytoplasmic in the subset of (carcinoma precursor) prostatic intraepithelial neoplasia (PIN) lesions that were actually found around cancers. 14 As a biomarker, p62 was therefore used in that scenario to perfectly segregate normal from malignant prostatic tissue and to stratify PIN biopsy material for identification of patients more at risk (ie, needing to be recalled for a second biopsy as opposed to less stringent management).
Compartmentalized and immunohistochemically observable expression of a protein that mitigates the dominant stress pathways active in the cell and that can also inform individual case management is an attractive candidate for use in diagnostic laboratories. p62 immunoreactivity is also an indicator of changes in protein handling (ie, localized protein-handling stress). While proteotoxic stress is commonly associated with highly synthetic tumors such as multiple myeloma, increased ploidy is also now known to impose a protein-handling stress. Nuclear abnormalities involving multinucleate status and ploidy occur in MCTs. 28,30 In murine experimental models, mast cells have been shown to recruit effectors of protein-handling pathways to permit degranulation. 31 While these data have yet to be confirmed in other species, the prospect remains that mast cell protein degradation and secretory pathways may be interlinked. A similar situation may also arise in MCTs, giving rise to the intriguing possibility that each pathway may impose a selective pressure on the other. For example, if granule formation is wasteful energetically and depletes autophagy effectors, then this process could be subject to negative selection in high-grade tumors. Data have also shown that p62 controls the duration of inflammatory signaling (possibly through proteolysis of transcription factors), 12,13 hence the desirability of studying tumors with an immune infiltrate. Taken together, these observations indicate that MCTs express several biologic characteristics that suggest suitability as a test system for the efficacy of p62 as a biomarker.
Our study began with the first in silico and functional analysis of canine p62, followed by development of a classification system for p62 subcellular localization in canine MCTs. We then benchmarked the p62 patterns against the Kiupel grade (ie, related them to the degree of nuclear pleomorphism/mitotic activity and indirectly to prognosis).
Materials and Methods
Cross-Species Analysis of p62/Sequestosome-1
We used an algorithm (basic local alignment search tool [BLAST]) that compares sequence information (DNA and protein) with identify candidate canine p62 genes. tBLASTn searches (that compare translated [t] nucleotide [n] sequences against a protein query) retrieved a predicted canine p62 gene (XM_005626348) and its protein product (XP_005626405.1). The “X” prefix denotes prediction using gene annotation software. 15 XP_005626405.1 expresses greatest similarity to isoform 2 of human p62/sequestosome-1. However, its open reading frame extends upstream of the designed start methionine and encodes further protein sequence with 100% homology to human isoform 1. This suggests that the longer isoform is likely to be expressed in the canine transcriptome and that XP_005626405.1 is (as reported) an incomplete version of p62 isoform 1. Use of the National Center for Biotechnology Information’s domain-detecting tool (conserved domain analysis) revealed N-terminal PB1 and ZZ family domains as well as the LC3 interacting region (LIR) used by p62 to locate to autophagosomes. 24 Two basic monopartite nuclear localization signals (N) previously predicted in human p62 were also identified in canine p62. A nuclear export signal (NE) with the characteristic 3 + 1 clustering of hydrophobic residues on either side of an amphipathic helix (an amino acid organization that typifies nuclear export sequence topology) was also found and experimentally confirmed. A helical wheel representation was used to confirm the appropriate amino acid distribution of the NE signal; this was generated using (http://www-nmr.cabm.rutgers.edu/bioinformatics/Proteomic_tools/Helical_wheel/). The C-terminal ubiquitin-binding domain (UBA) that typifies p62 proteins could not be identified by direct sequence comparisons. However, different amino acid sequences can adopt the same structures by virtue of their shared environmental chemistry, irrespective of primary amino acid sequence order. Therefore, searches for shared structure—in this case, the UBA domain—in the absence of shared primary sequence were attempted for the C terminus of canine p62. This type of search, called threading, was completed using the Fugue algorithm (version 2.s.07); this is freely available and hosted at http://tardis.nibio.go.jp/fugue/. Fugue analyses revealed with certainty (indicated by a z score in excess of 6 [11.51]) that the UBA fold was preserved in canine p62. The relevant human/canine alignments (the canine sequence is capitalized) generated to make this prediction were prlieslsqml/PRLIESLSQML, wltrlLqt/KRSRLTPV, and igaAldtI/SSTEDRGS (Fig. 1).
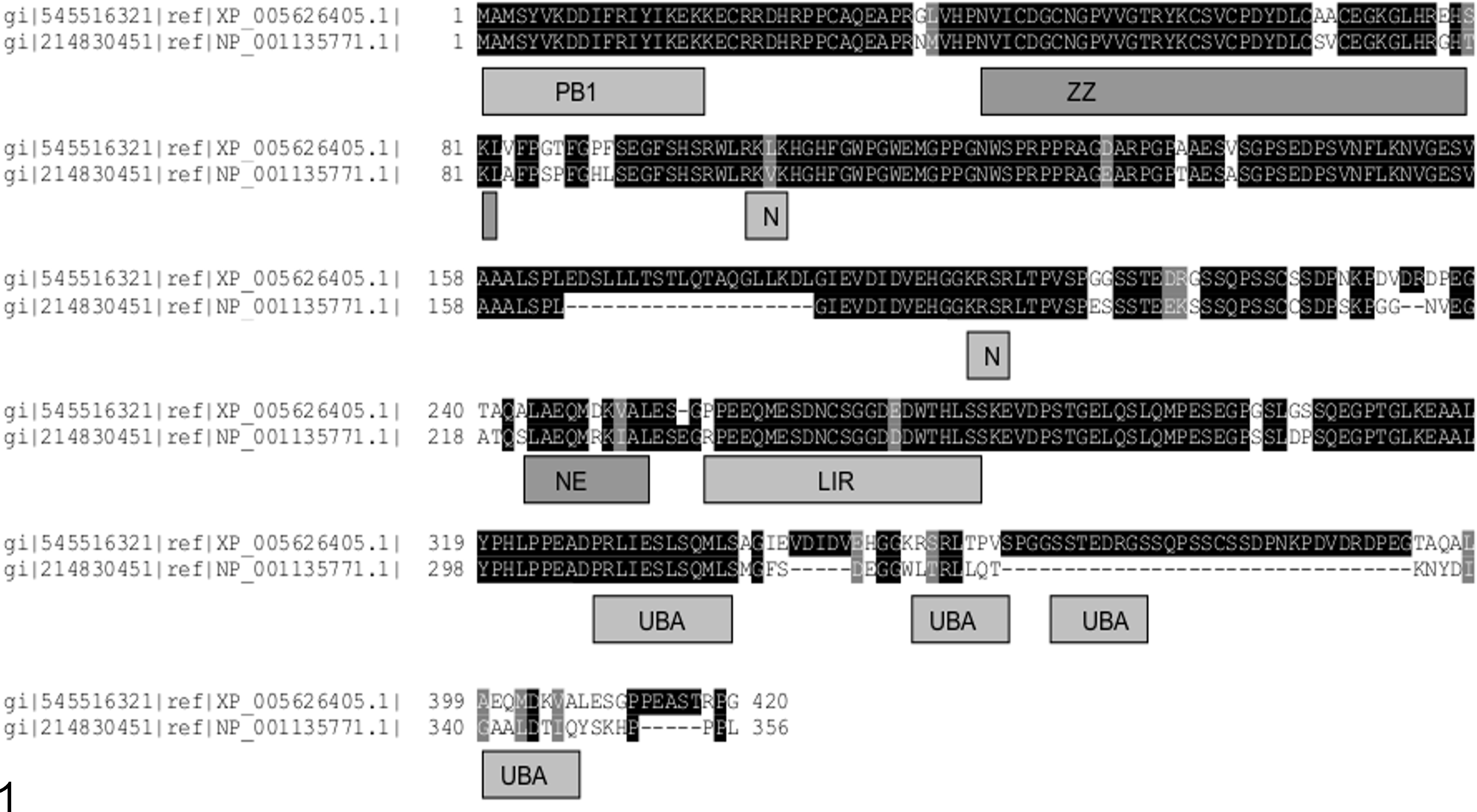
Canine p62 shares functional domains with its human counterpart. A sequence alignment of the canine and human p62 sequences was prepared using ClustalW and displayed using BOXSHADE. Identical residues are indicated by the black background to white text. The gray background indicates conserved sequence; dashes indicate gaps. The “XP/NP” references are the accession numbers for each p62 protein; the canine protein is shown above the human homologue. These sequences represent isoform-2, which is 84 amino acids shorter than isoform 1. Homology to PB1 and UBA domains was identified at the N- and C-terminal portions of canine p62. The PB1 sequence (which is more extensive in isoform 1) permits binding to proteasomes and to other proteins expressing this sequence. The UBA domain binds polyubiquitinated proteins destined for proteasomal or autophagic breakdown. ZZ denotes a zinc finger domain; N and NE indicate nuclear import and export signals, respectively, and LIR indicates the sequence that binds the LC3 Protein, which is critical for autophagosome generation. Sequences contributing to the UBA domain fold were identified at 3 portions of the sequence, the first 2 of which were at identical positions in either protein.
Antibody Validation
Validation of the anti-p62/SQSTM1 antibody (PM045; MBL International, Woburn, MA) was performed first by Western blotting to ensure that it was binding to a protein of the expected molecular weight. This is a polyclonal antibody, the epitopes for which are not mapped. The antibody was then tested in cell culture, using drugs known to target p62 to different cellular compartments; this was to ensure that immunolabeling accurately reflected the switches imposed on p62 targeting (effectively a functional analysis).
Western blot analysis
Madin-Darby canine kidney (MDCK) cells were lyzed with RIPA buffer supplemented with 1% sodium dodecyl sulfate (SDS). Total protein (20 μg) was loaded onto an SDS–polyacrylamide gel electrophoresis (SDS-PAGE) gel (15% Tris-HCL). Proteins were separated at 100 V for approximately 2 hours and then transferred to a nitrocellulose membrane for Western blot analysis. The membrane was blocked for 1 hour using 10% powdered milk w/v in phosphate-buffered saline (PBS)/0.2% Tween 20, followed by a 5-minute wash in PBS/0.2% Tween 20 alone. The membrane was then incubated with anti-p62/SQSTM1 (1:500) for 16 hours at 4°C. This was followed by 3 × 5–minute washes in PBS/0.2% Tween 20, incubation with anti–rabbit immunoglobulin horseradish peroxidase (HRP) conjugate (1:1000, P0448; Glostrup, Denmark) for 3 hours, and then 3 further 5-minute washes. HRP conjugates were detected using the Immobilon Western Chemiluminescent HRP substrate kit (Millipore, Watford, UK) as instructed by the manufacturer.
Drug studies
For drug studies, bortezomib (Velcade) (B1408) and leptomycin B (6100) were purchased from LC Laboratories (Woburn, MA). Bafilomycin A1 (B1793) and etoposide (E1383) were purchased from Sigma-Aldrich (Dorset, UK). Final drug concentrations were 5 nM (bortezomib), 10 nM (bafilomycin A1), 0.5 μg/ml etoposide, and 2 nM leptomycin B; all incubations were for 16 hours at 37°C. For immunofluorescent labeling, MDCK cells were seeded onto glass coverslips in 6-well dishes and cultured to 70% confluence. Fixation was with 4% v/v paraformaldehyde/PBS for 5 minutes, followed by permeabilization with 0.25% Triton X-100/PBS for 5 minutes. Nonspecific binding was blocked by the addition of 10% milk powder/PBS for 1 hour at room temperature (RT). Primary and secondary antibody incubations were carried out overnight at 4°C and 1 hour at RT, respectively. The anti-p62/SQSTM1 antibody (PM045) was used at a concentration of 1:250. Anti–proteasome 20 S antibody (22674; Cambridge, UK) was used at a concentration of 1:200. Alexa Fluor secondary conjugates (1:1000; Life Technologies, Paisley, UK) were used to detect the primary antibodies. Slides were mounted using Prolong-Gold anti-fade with DAPI (Life Technologies). Cells were imaged using the 63× objective lens of an upright Axiophot2 microscope (Zeiss, Cambridge, UK). Fluorescent micrographs were prepared using GIMP digital software (version 2.6.1; www.gimp.org).
Cases, Sample Processing, and Grading
Sixty-six cases of primary canine cutaneous MCTs were selected prospectively by convenience from recent submissions to IDEXX Laboratories Ltd (Sacramento, CA, and Wetherby, UK) (CL, FR-I) in 2013. All specimens had been fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin wax blocks, sectioned at 3 μm, and mounted on glass slides. One slide per case was stained with hematoxylin and eosin (HE), and the diagnosis of MCT was confirmed (J.C.P.-K.).
Immunohistochemistry
Further sections were deparaffinized (Histo-Clear; National Diagnostics, Hull, UK) and then rehydrated using graded alcohols. Antigen retrieval was required for anti-p62/SQSTM1 immunolabeling; this was performed by placing slides in sodium citrate buffer (pH 6.0) for 1 minute and 40 seconds using a pressure cooker (Antigen Access Unit, A. Menarini Diagnostics Ltd, Wokingham, UK). All slides were then loaded into a Dako-Autostainer (Dako UK Ltd, Ely, UK). Endogenous peroxidase and nonspecific staining were blocked using 3% hydrogen peroxide (in PBS) for 5 minutes. All tumors were incubated with a primary antibody against p62/SQSTM1 for 30 minutes at RT. An anti–rabbit secondary antibody conjugated to an HRP-labeled polymer (K4003) (Dako Envision + System-HRP; Dako UK Ltd) was then applied for 30 minutes at RT. Visualization was achieved using 3,3′-diaminobenzidine (DAB+ Substrate Chromagen System; Dako UK Ltd), 20 μl/ml, for 10 minutes. Tissues were counterstained using Gill’s hematoxylin (made in house) for 27 seconds, then dehydrated, cleared, and mounted (DPX mounting medium; Cellpath, Newtown, Powys, UK).
Grading
Following examination of all p62-labeled slides, a 3-tier system was developed. For classification, the entirety of the section was examined in each slide (this being the largest cross section available) and the decision made (consensus by T.R. and J.C.P.-K.) based on the predominant pattern. This consensus allowed the input of a veterinary pathologist alongside that of a cell biologist with expertise in interrogation of cell stress pathways. A “nuclear” pattern was defined as one to multiple (1–8) variably sized but prominent positive puncta in the nucleus with or without a background of diffuse nuclear immunoreactivity; any cytoplasmic immunoreactivity was pale, only noted in small numbers of scattered tumor cells, and/or only seen in a few high-power fields (hpf). In MCTs classified as having the “mixed” pattern, the same nuclear immunoreactivity was noted but was always diffuse in at least some cells; in addition, there was moderately intense to dark, granular (sometimes coarsely clumped) cytoplasmic immunoreactivity in most cells in most hpf. A “cytoplasmic” pattern was defined as diffuse dark, granular cytoplasmic immunoreactivity with either no nuclear reaction or 1 to 4 variably sized nuclear puncta in a variable proportion of tumor cells. Diffuse nuclear immunoreactivity was occasionally noted in this category but did not occur in most fields. Each MCT was scored as low or high grade (without reference to the p62 pattern), with the latter being characterized by 1 or more of the following observations in 10 hpf: at least 7 mitotic figures, at least 3 multinucleated cells (more than 2 nuclei), at least 3 highly atypical nuclei, or at least 10% of the tumor cells varying in nuclear diameter by more than 2-fold. 16 Fields selected in each slide were those with the greatest numbers of mitotic figures and/or most marked anisokaryosis.
Statistical Analysis
A χ 2 test for independence was used to compare the p62 classification with Kiupel grade. The statistical calculations were performed using a commercially available software program (SPSS Statistics version 21.0; SPSS, Inc, and IBM Company, Chicago, IL). P < .05 was considered significant.
Results
In Silico Analyses of Canine p62/Sequestosome-1
A full-length prediction of the canine p62/SQSTM1 protein with 76% sequence identity to the human homologue was obtained as detailed in the Materials and Methods. p62 function is largely predicated by expression of functional domains 9 that tether it to various signaling and protein handling scaffolds; expression of these domains was confirmed in the canine p62 protein (Fig. 1). Several of these domains assist p62 in its delivery of waste proteins (usually ubiquitinated) to proteasomes or autophagosomes. 9 Nuclear import and export signals were also detected, which suggests that canine p62, in common with human p62, shuttles between the nuclear and cytoplasmic compartments. 24
Functional Analyses of Canine p62/Sequestosome-1
Western blots confirmed expression of a protein with the same molecular weight as p62 in the canine MDCK cell line (Fig. 2). Additional lower molecular weight species noted in this blot could indicate the second known p62 isoform, predicted to be 9 kDa smaller than isoform 1, together with posttranslational modifications (eg, phosphorylation and ubiquitination) known to occur on p62. Immunocytochemical and histochemical identification of p62 is most closely linked with its role in protein metabolism. Our in silico analyses suggested preservation of these protein-handling phenotypes for canine p62, together with the appropriate cellular trafficking signals to allow its nuclear entry and export. To test these predictions, we exposed MDCK cells to various agents that cause protein aggregation to assess whether p62 would redistribute to the newly formed aggregates (as would be expected for a protein that metabolizes protein waste). In control MDCK cells, canine p62 was distributed in the cytoplasm as diffuse and punctate structures (Fig. 3). The larger cytoplasmic puncta of p62 are thought to indicate autophagosomes, and the small puncta/speckles indicate proteasomes. Proteasomes were expressed throughout the nuclear and cytoplasmic compartments of control cells, but p62 expression was almost exclusively cytoplasmic (Fig. 4). Exposure of MDCK cells to bortezomib, a drug that inhibits proteasome activity, resulted in larger, more clearly defined (cytoplasmic) puncta of canine p62 (Fig. 5). In cell culture models in which autophagy activity can be absolutely measured (using flux assay) rather than inferred (ie, by detecting autophagosome formation in tissue), this redistribution of p62 has been shown to indicate the increased formation of autophagosomes as increased autophagy substitutes for declining proteasome activity. 3,5 Inhibition of fusion of autophagosomes with lysosomes using bafilomycin A generated even larger irregular cytoplasmic aggregates of p62 at the expense of the micropunctate fraction (Fig. 6). We would suggest that these are autophagosomes that continue to enlarge because they cannot degrade their luminal contents inclusive of p62 (Fig. 6). 17 Proteasomes would be unable to compensate for reduced autophagy function in these cells because the large aggregates of protein waste that form cannot physically access the narrow proteasomal lumens. 18,32 Exposure to DNA-damaging agents can induce pro-survival autophagy signaling in chemoresistant cells, again indicated by increased cytoplasmic p62 expression. 36 The striking increase in large cytoplasmic aggregates of canine p62 following exposure to the genotoxic agent etoposide confirmed the retention of this phenotype in MDCK cells (Fig. 7). Finally, access to all cellular compartments must be ensured if canine p62 is to regulate protein handling as reported for human p62. To test the functionality of the nuclear import and export signals predicted for canine p62, we treated MDCK cells with leptomycin B, which inhibits the process of CRM-dependent nuclear export (the main mechanism of protein egress from the nucleus). We found that leptomycin B treatment provoked the nuclear accumulation of canine p62 in concurrent diffuse and punctate expression patterns (Fig. 8). Punctate nuclear p62 (provoked by leptomycin B exposure) has previously been shown to indicate the accumulation of p62 at sites of dedicated protein breakdown and homeostasis, termed promyelocytic leukemia nuclear bodies (PML-NBs) 24 ; our expression pattern in leptomycin-treated MDCK cells is consistent with that process.
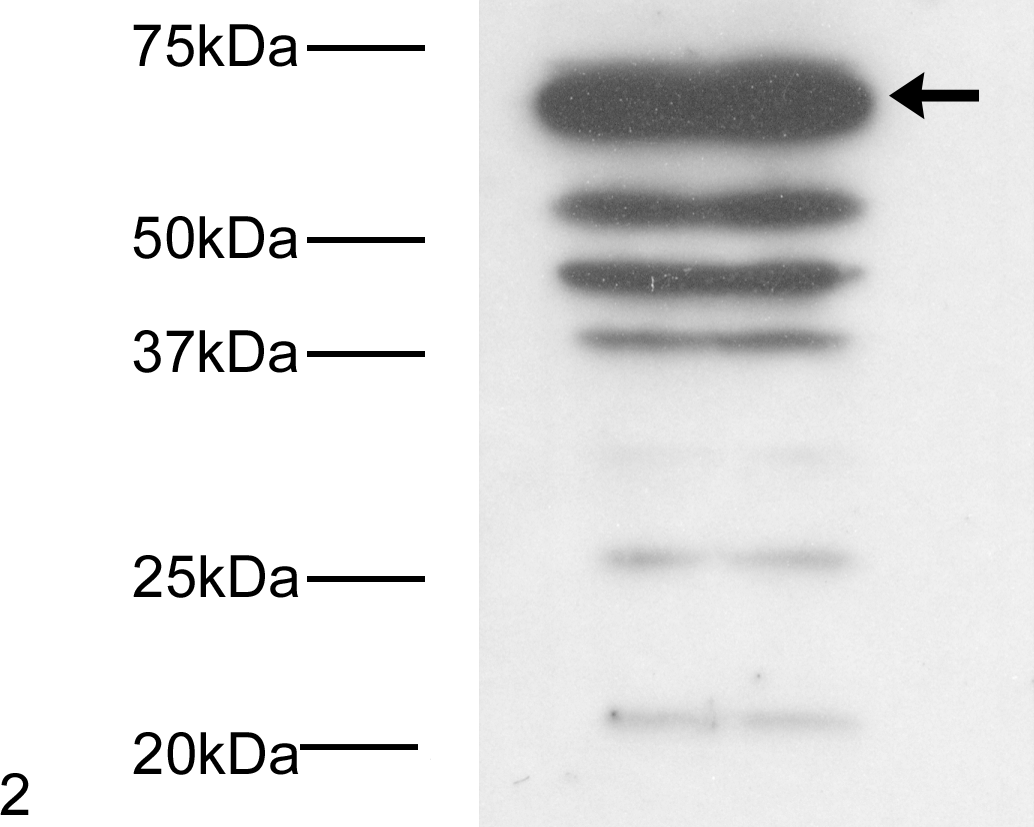
Western blot of canine p62 indicating several species consistent with multiple isoforms and their posttranslational modifications. The arrow shows the position of p62 isoform 1.
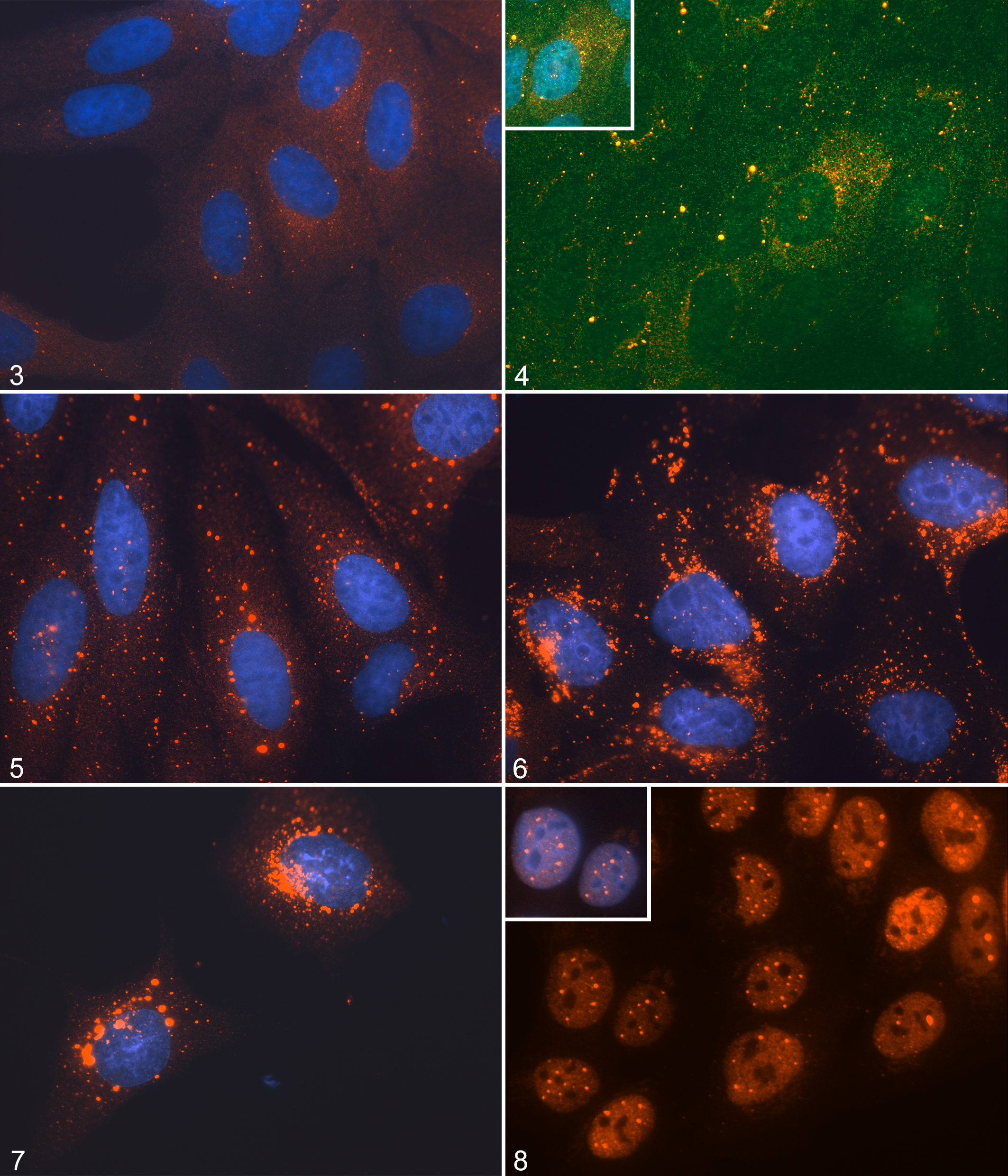
The influence of cellular stress on p62 expression, Madin-Darby canine kidney (MDCK) cells. Functional validation of the anti-p62 antibody for the dog.
Expression of p62/Sequestosome-1 in MCT Biopsy Material
Of the 66 canine cutaneous MCTs, 37 (56%) were low grade and 29 were high grade (44%). 16 In terms of p62 pattern classification, 9 (14%) were nuclear, 29 (44%) mixed, and 28 (42%) cytoplasmic (Figs. 9–12). In 3 cases (2 with a mixed p62 pattern and 1 with nuclear), variable numbers of tumor cells were noted in most fields (these numbers were large in 1 “nuclear” case), in which a juxtanuclear structure of similar size and shape to the nucleus itself was strongly p62 immunoreactive; the nucleus itself was not (Fig. 13). These p62-positive structures showed variable degrees of swelling, vacuolation, and disintegration; in HE sections, they were not clearly identifiable, with minimal to no basophilia and frequent appearance as a large perinuclear vacuole (Fig. 14). There was a highly significant association between Kiupel grade (low vs high) and p62 classification (P < .0001) (Fig. 15). Nuclear p62 immunoreactivity was found only in low-grade MCTs, whereas cytoplasmic p62 was almost always associated with high-grade MCT (93% of cytoplasmic p62 cases were high grade). Most MCTs with the mixed p62 pattern were low grade (Fig. 15). The overlap between cytoplasmic and mixed patterns in terms of 2-tier histologic grade (ie, high-grade MCT with mixed pattern and low-grade MCT with a cytoplasmic pattern) comprised only 5 (8%) total cases.
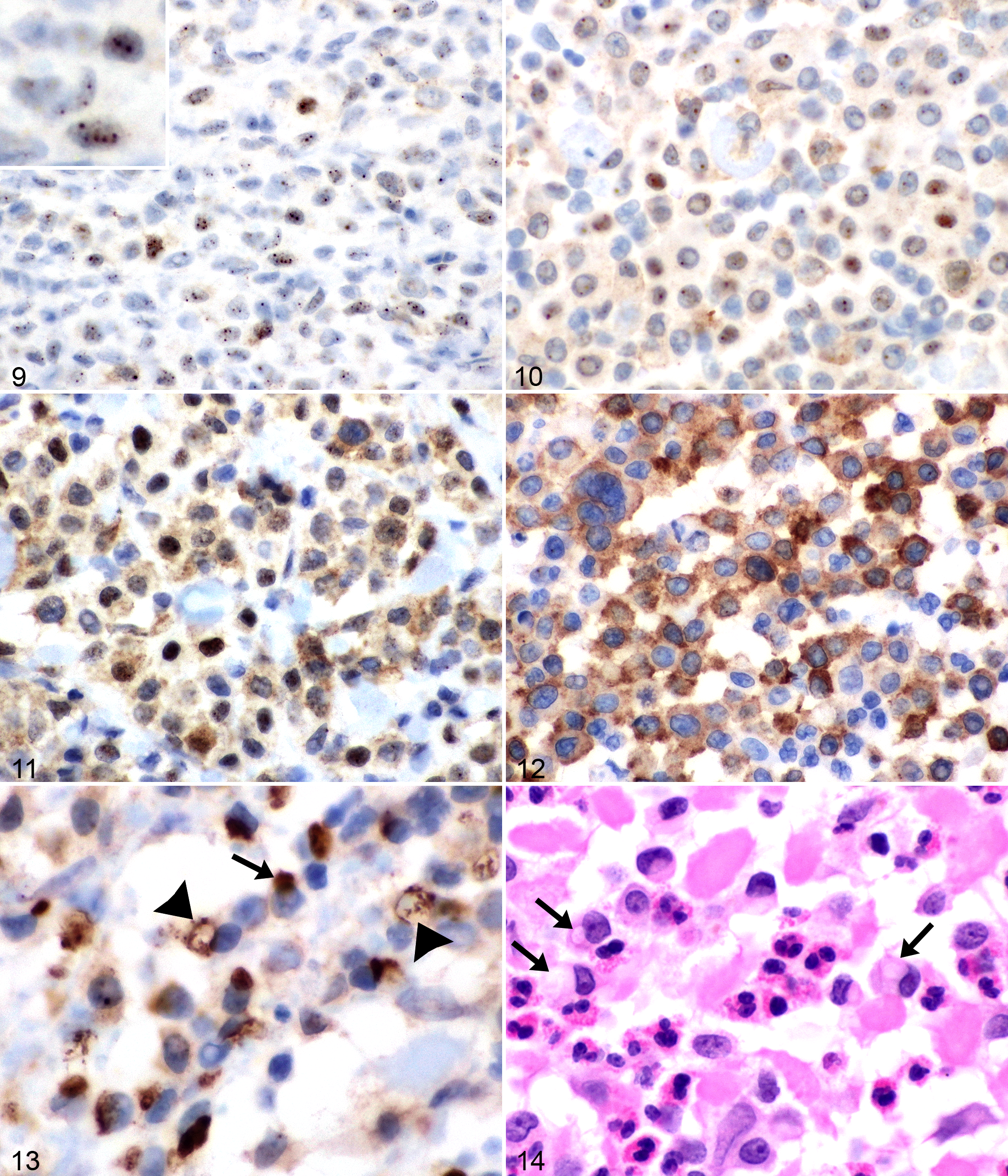
Mast cell tumors (MCTs), dog, haired skin.
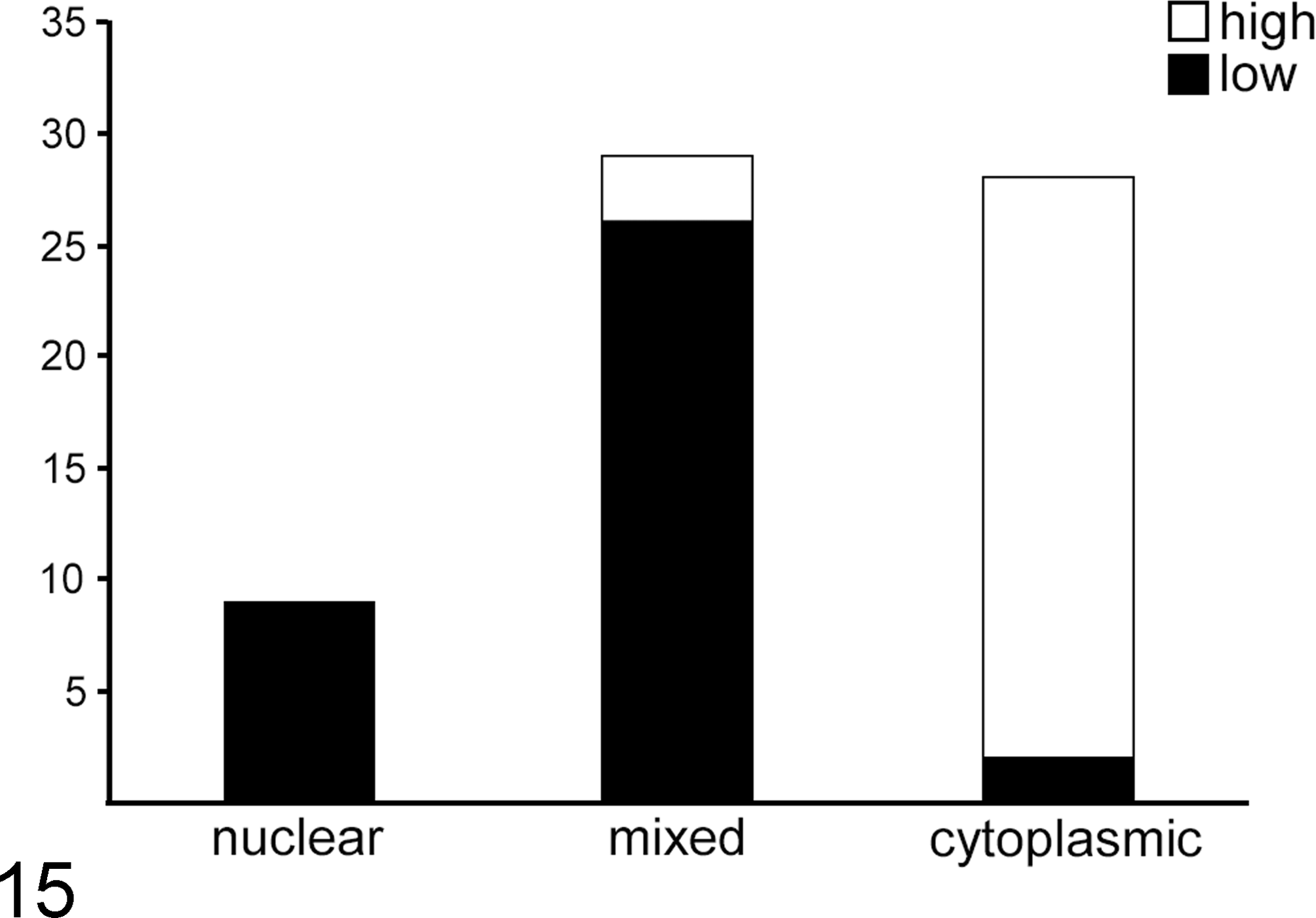
Statistical analysis of p62 pattern (nuclear, mixed or cytoplasmic) in mast cell tumors relative to 2-tier (high or low) histologic grade. 16 χ2 Test for independence. There was a highly significant association (P < .0001).
Discussion
Our data demonstrated a link between predominantly nuclear expression (immunoreactivity) of p62 and low levels of nuclear pleomorphism and mitotic activity (ie, the Kiupel low grade). Conversely, predominantly cytoplasmic p62 localization was linked with malignancy. Although prognostic information was not directly gathered (or the topic of this study), the association of p62 pattern with Kiupel grade was highly statistically significant. The aim was not to replace the current prognostic systems but rather to determine the processes involved in MCT progression that might assist in tumor biology-based therapeutic patient stratification. The two extremes of the p62 pattern (ie, nuclear vs cytoplasmic) provide important clues as to the protein-handling stresses experienced and/or potentially being “ignored” in benign vs malignant tumors. Experiments are currently ongoing to test the efficacy of targeting multiple protein-handling pathways in ex vivo cultures of canine MCTs. Importantly, autophagy (involving lysosomal breakdown) is a process that occurs only in the cytoplasm, while proteasomes (cylindrical structures that break down ubiquitinated proteins) function throughout both the cytoplasm and nucleus. Nuclear accumulation of p62 requires inhibition of its reexport, either by physical anchoring or phosphorylation; under those conditions, it recruits polyubiquitinated proteins to the PML-NBs and assists in their proteasomal degradation in that vicinity. 35 Expression of nuclear p62 in prominent puncta, seen in all patterns but most clearly and exclusively in the “nuclear” category, was consistent with the size and distribution of PML-NBs. We have confirmed nuclear p62/PML co-localization immunocytochemically in other species for which we have the appropriate antibody combinations (data not shown). Furthermore, a drug treatment (leptomycin B) that retains human p62 at PML-NBs 24 generated an identical phenotype in the canine MDCK cells used in this study (Fig. 8). Plausible nuclear stress inducers include inflammation, genomic instability, and DNA damage. Cancers that arise in an inflammatory background (including the eosinophilic infiltrates common in MCT) are susceptible to free radicals that damage proteins and nucleic acids. p62 is known to be involved in regulating the duration of cytokine expression (via transcription factor turnover) in other innate immune cells, including macrophages. 12,13 This raises the question as to whether malignant MCTs in which the p62 is almost completely localized within the cytoplasm are failing to control inflammatory signaling cascades; at this time, we do not have clear information on whether the severity or nature of inflammation within MCTs has a strong relationship with grade and/or prognosis. In situations of DNA damage, DNA damage recognition (DDR) proteins escape translational repression, multiple proteins (including RNA-binding proteins) shift into the nucleus, and the transiently needed multiprotein repair complexes (and damaged proteins) must be cleared. 8 This scenario therefore requires increased nuclear protein turnover. Ubiquitination is known to be involved in negative control of the DDR response, 23 with much of this protein-handling requiring p62-mediated trafficking.
Interestingly, we also found evidence, in a small number of nuclear and mixed tumors, of the ability to control aberrant nuclear phenotypes that are known indicators of high-grade MCT; this might include multinucleation and supernumerary chromosomes. We propose that nuclear pathology of this type is prevented in low-grade MCT cells at least initially, by the expulsion of excess damaged nuclear material. We found evidence to support this hypothesis in the form of juxtanuclear p62-positive structures that were interpreted to be entire nuclei in various stages of degeneration. These cells appear to be resolving a binucleate or multinucleate phenotype (Figs. 13, 14). We are currently further investigating this repair phenomenon (compartmentalization of p62-labeled damage in one nucleus during incomplete mitosis) in cell culture models. The process is consistent with previous reports of a smaller-scale phenomenon in which p62 protein envelops nuclear fragments for autophagic destruction. 25 The p62-positive remnants that we identified showed a variation in staining pattern from strong and diffuse to marked disintegration, consistent with destruction by autophagy; p62 delivers its cargo to autophagosomes and is then itself destroyed. If this mechanism is subsequently confirmed in cell culture, it reveals a prodigious ability to regulate ploidy, as disposal of entire nuclei in this manner has not been observed. This autophagic clearance may also impose a considerable stress on these cells (ie, a selective pressure that could be mitigated by additional “escape” mutations to allow persistence of the second nucleus). Even the presence of a single supernumerary chromosome in a cell has been shown to impose a large protein-handling stress, with increased autophagy and proteasome activity needed to “dial down” the concentration of a subset of the proteins encoded by the additional loci. 28,30
A large percentage of the MCTs (44%) fell into the “mixed” category, with increased p62 nuclear immunoreactivity, including diffuse labeling and varying degrees of retention/redistribution into the cytoplasm. Most but not all of these MCTs were low grade, but this raises important questions: does this represent tumor progression? At what point should a particular tumor be considered potentially malignant, and what is the cause (or causes)? The issue of how or whether canine MCTs progress has largely been ignored in the literature. In these cells, stresses that were previously being handled by p62 at the nucleus are now being partially or largely ignored, cannot be recognized, or are secondary to (or less important than) cytoplasmic stress. There are a number of possible pathways by which p62 might be prevented or diverted from accumulating in the nucleus—for example, inhibition of nuclear import by regulatory phosphorylation, a switch to preferential trafficking of proteins into the more potent (and solely cytoplasmic) autophagy pathway (when nuclear pathways are overwhelmed), or cytoplasmic anchorage due to significant polymerization and self-aggregation. 24 Typically, immunohistochemical detection of increased amounts of cytoplasmic p62 has been suggested to indicate upregulation in autophagy activity and therefore a poorer prognosis because of the selective advantage provided by this pro-survival signaling to tumor cells, including resistance to ischemia-hypoxia. 7 Autophagy-mediated delivery of nutrients and biosynthetic intermediates for cell growth is also a significant hindrance to the success of chemotherapy but could be itself targeted. 10,21,22 Autophagy can also be a tumor suppressor, but the p62 patterns in high-grade MCTs indicate that is not the case for this tumor type.
To summarize, in canine cutaneous MCTs, we have identified a spectrum of nuclear vs cytoplasmic cellular localization of the proteotoxic stress protein, p62. If nuclear or cytoplasmic-predominant p62 labeling is identified in an individual MCT (cytologically or histologically), these results indicate that classification of the tumor as benign or malignant, respectively, would be appropriate. The cytosolic overexpression of p62 has been identified in human prostate cancer as a marker of malignancy and, as a general marker of protein-handling stress, might have similar associations in other canine tumor types. 14,24 The large percentage of mixed-p62 pattern MCT (most of which were low grade) suggests that mechanisms of tumor progression are associated with a gradual shift from nuclear to cytoplasmic protein-handling stress, but the “tipping point” at which this can be considered to represent true malignancy is not known. These results do, however, give us some indication of where to look for markers of progression that may assist in further patient stratification and decision making in the well-known “gray area” encountered in the prognostic grading systems. Further studies are currently ongoing to assess how p62 immunoreactivity correlates with outcome. The likelihood that cytosolic p62 is linked with increased autophagy also suggests that this process might be targeted therapeutically in high-grade or incompletely excised MCTs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
