Abstract
Cutaneous mastocytosis (CM) is a rare condition in young dogs characterized by multicentric cutaneous proliferation of neoplastic mast cells. Clinical data from 8 dogs that met inclusion criteria (age of onset less than 1.5 years, greater than 3 lesions) were obtained via a standardized survey. Biopsy samples were classified by the Kiupel/Patnaik grading systems and analyzed for c-KIT mutations. The median age of onset was 6 months (range: 2–17 months). Dogs had 5 to more than 50 lesions characterized as nodules, plaques, and papules. Seven dogs were pruritic. Clinical staging in 2 dogs did not reveal visceral involvement. No dogs had systemic illnesses at diagnosis. Histologically, CM was similar to cutaneous mast cell tumor (cMCT). Two dogs had neoplasms classified as high-grade/grade II while 6 dogs had low-grade/grade II neoplasms. No dogs had mutations in c-KIT exons 8 and 11. Treatment included antihistamines (8/8), corticosteroids (7/8), lokivetmab (3/8), and toceranib (1/8). Six dogs were alive with lesions at the end of the study with a median follow-up time of 898 days, while 2 dogs were euthanized. In dogs with high-grade/grade II neoplasms, one continued to develop lesions at 1922 days post-diagnosis, while the other dog was euthanized at 56 days post-diagnosis. One dog was euthanized 621 days post-diagnosis due to rupture of a neoplasm. CM occurs in young dogs and is histologically indistinguishable from cMCT. Current histologic grading systems did not apply uniformly to the dogs of the study and further studies are needed.
Cutaneous mast cell tumor (cMCT) is common in dogs, representing up to 21% of cutaneous neoplasms.8,24 Canine cMCT develops most commonly as a solitary nodule, 9 which is in contrast to humans where multiple mast cell neoplasms are more common and the disease is referred to as “mastocytosis.” 4 Solitary cMCT is mostly seen in aged dogs, but multiple mast cell neoplasms have been reported in few young dogs and were referred to as “mastocytosis” or “urticaria pigmentosa-like disease” in the veterinary literature.3,5,14,16,18,19 Canine cutaneous mastocytosis (CM) has been reported to spontaneously regress in one dog 3 and shares some similarities with a subtype of mastocytosis in humans, namely maculopapular CM (previously termed urticaria pigmentosa). Little is known about the prognosis of this condition due to the small number of cases reported. This study aimed to characterize the clinical and histopathologic features and outcomes of canine CM in 8 young dogs.
Materials and Methods
Signalment, Clinical History, and Clinical Follow-up
The surgical biopsy database of the School of Veterinary Medicine of the University of Pennsylvania was searched between 1 January 2015 and 19 March 2022 with the following keywords: “canine” and “mastocytosis” or “mast cell tumor.” Inclusion criteria include (1) multiple (greater than 3) cutaneous lesions, (2) age of onset of less than 1.5 years, and (3) histologic evidence of dermal and/or subcutaneous mast cell neoplasm. Hematoxylin and eosin (HE)-stained sections from all cases were reviewed. Histologic grade was determined based on both Kiupel and Patnaik grading schemes.10,17 A histochemical stain of toluidine blue was performed in 1 case (Dog 3).
A written survey (Supplemental Material S1) and a request for medical records were sent to the submitting clinician for each case fulfilling the inclusion criteria. Historical information was gathered including signalment (age, sex, breed), weight, age at onset, lesion location, dermatologic signs, presence of Darier’s signs (ie, whealing and reddening of lesions upon mechanical stroking or rubbing), size and number of the lesions, mucosal involvement, pruritus (scored using a previously published numerical rating scale [0–10]), 15 and other systemic clinical signs (eg, pain, lymphadenomegaly, vomiting, hematemesis, hematochezia, melena, diarrhea, lethargy, anorexia, and fever). Prior and current medications and diagnostic tests/laboratory data, including complete blood count, biochemical analysis, diagnostic imaging, prior cytology, or biopsies, were recorded. Treatment type, effect on lesion size and pruritus (complete, partial, or no response), and outcome (lesion status and patient status) were recorded as available or gathered in a follow-up conversation with the veterinarian. Complete response was defined as the disappearance of all lesions or pruritus. Partial response was defined as the reduction of the number of lesions or pruritus level. No response was defined as no reduction in the number of lesions or pruritus level. When more than 1 treatment regimen with different combinations of medications was given to the same dog at different times, the response to each treatment will be documented separately.
Immunohistochemistry
The mast cell neoplasm with the highest mitotic count from each case was selected for evaluation of KIT localization and c-KIT mutation analysis. Immunohistochemistry (IHC) was performed in all cases. Five-micrometer-thick paraffin sections were mounted on ProbeOn™ slides (Thermo Fisher Scientific, Waltham, MA, USA). The IHC procedure was performed using a Leica BOND RXm automated platform combined with the Bond Polymer Refine Detection kit (Leica, Allendale, NJ, USA). After dewaxing and rehydration, sections were pretreated with the BOND epitope retrieval solution 2 (Leica) for 20 minutes at 98°C. Endogenous peroxidase was inactivated with 3% hydrogen peroxide for 10 minutes at room temperature. Nonspecific tissue-antibody interactions were blocked with PowerVision IHC/ISH Super Blocking solution (Leica) for 30 minutes at room temperature. The same blocking solution also served as the diluent for the primary antibody. A monoclonal rabbit anti-human c-KIT (CD117) antibody (Cell Signaling Technology, Danvers, MA, USA) was used at a concentration of 1/500 and incubated on the sections for 45 minutes at room temperature. A biotin-free polymeric IHC detection system consisting of horseradish peroxidase (HRP)-conjugated anti-rabbit IgG was then applied for 25 minutes at room temperature. Immunoreactivity was revealed with the diaminobenzidine (DAB) chromogen reaction. Slides were counterstained in hematoxylin, dehydrated in an ethanol series, cleared in xylene, and permanently mounted with a resinous mounting medium (Thermo Scientific). Sections of canine cMCT were included as positive controls. Negative controls were obtained either by omission of the primary antibody or replacement with an irrelevant isotype-matched rabbit monoclonal antibody. The predominant KIT protein staining pattern was assigned as described previously. 11
c-KIT Mutation Analysis
Polymerase chain reaction (PCR) of DNA extracted from formalin-fixed, paraffin-embedded skin was performed by Michigan State University Veterinary Diagnostic Laboratory for mutational analysis of c- KIT exons 8 and 11. 2
Results
Clinical Characteristics
Eight cases from the histopathology database met the inclusion criteria (Supplemental Table S2). Follow-up information was available in all cases. There were 5 mixed-breed dogs, 1 German shorthaired pointer, 1 Labrador retriever, and 1 Newfoundland dog. Five were female (2 intact and 3 spayed) and 3 were male (2 intact and 1 castrated). The median weight at initial diagnosis was 26.9 kg (range: 16.9–43.7 kg). The median age of onset was 6 months (range: 2–17 months) and the median age of diagnosis by histopathology was 12 months (range: 2–30 months). The number of lesions ranged from 5 to more than 50 and the size of lesions ranged from 0.4 to 6 cm. The lesions (Fig. 1a–d) observed at the initial diagnosis were nodules (4/8), plaques (3/8), papules (2/8), erythema (2/8), and alopecia (2/8). Other lesions included hemorrhagic bullae (1/8), crusts (1/8), pustules (1/8), and ulcers (1/8). The lesions were multifocal (3/8) to generalized/disseminated (5/8). Darier’s signs were evaluated in 3 dogs and were positive in 2 dogs. Lesions within individual dogs were similar in shape, color, and size in 3 of 8 dogs. Waxing and waning of lesions were reported in 2 of 8 dogs. Pruritus was noted in 7 of 8 dogs. Pruritus score at the time of presentation was available in 3 dogs and was 4/10, 5/10, and 6/10. No mucosal involvement was reported in any case. At the time of initial diagnosis, concurrent clinical signs, including pain, vomiting, hematemesis, hematochezia, melena, diarrhea, lethargy, anorexia, and fever, were not reported in any dogs.
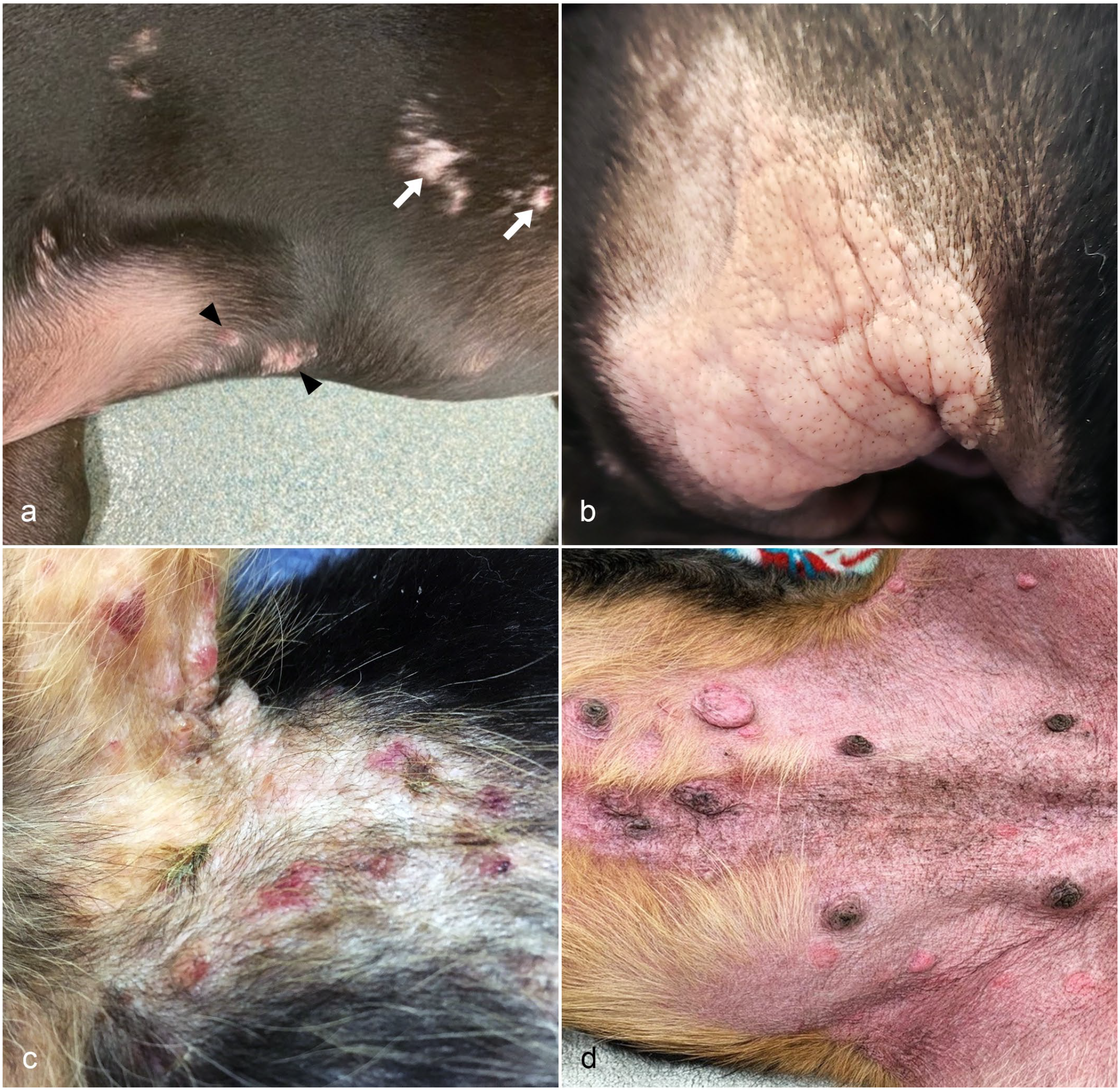
Clinical lesions of cutaneous mastocytosis in young dogs. (a) Multifocal plaques (black arrowheads) with alopecia (white arrows) on the ventral thorax and flank (Dog 6). (b) Coalescing plaques on the right concave pinna (Dog 2). (c) Multifocal papules, vesicles, hemorrhagic bullae, and crusts on the axilla and ventral thorax (Dog 1). (d) Multifocal plaques and nodules with diffuse erythema on the caudal ventrum and groin (Dog 1).
Laboratory Results
Complete blood counts were performed in 6 of 8 dogs at initial diagnosis with significant changes in 1 dog (Dog 1). Dog 1 had leukocytosis (17700/µL; reference interval: 6000–17000/µL), lymphocytosis (4903/µL; reference interval: 1000–4800/µL), and monocytosis (1717/µL; reference interval: 150–1350/µL) 2 weeks after biopsy. Serum biochemistry profiles were performed in 5 dogs at initial diagnosis with significant changes in 2 dogs (Dog 1 and Dog 3). Dog 1 had increased serum calcium (12.8 mg/dL; reference interval: 8–12 mg/dL), phosphorus (9 mg/dL; reference interval: 3–7 mg/dL), and alkaline phosphatase (219 U/L; reference interval: 10–84 U/L), and Dog 3 had decreased phosphorus (2.1 dL/L; reference interval: 2.5–6.8 dL/L). Mast cells were identified on cytologic examination of the cutaneous lesions in 5 of 5 dogs examined. Clinical staging was performed in 2 dogs via fine-needle aspiration of the lymph node and abdominal ultrasound (Dogs 1 and 3) at the time of diagnosis. Fine-needle aspiration of the regional lymph node in Dog 1 revealed rare mast cells. Abdominal ultrasound of Dog 3 revealed an enlarged right medial iliac lymph node. The node was aspirated and rare mast cells were identified by cytologic examination. The rare mast cells in both dogs were interpreted as drainage reactions from the cutaneous lesions.
Histopathologic Features
A total of 33 biopsy samples, including 21 punch biopsy samples and 12 excisional biopsy samples, from 8 patients were evaluated (Fig. 2a–f). The number of biopsy samples reviewed was 3 (5/8), 4 (2/8), and 10 (1/8) per patient. All samples contain cellular infiltrates expanding the dermis with variable extension to the subcutis and the underlying skeletal muscle (Fig. 2c). The infiltrates were unencapsulated, poorly to moderately demarcated, and composed of well-granulated mast cells amid the pre-existing collagen fibers. The mast cell infiltrates either had poor demarcation (22/33 samples; Fig. 2a) dissecting between dermal collagen fibers or formed dense sheets with moderate demarcation (11/31 samples; Fig. 2b). Mitotic count was determined in 32 of 33 samples, as the remaining sample was a small biopsy and did not have 10 high-power fields (2.37 mm2) for evaluation. The median mitotic count was 1 and ranged from 0 to 8 per 2.37 mm2. Mitotic count was 0 (9/32 samples), 1 (9/32), 2 (7/32), 4 (1/32), 5 (1/32), 6 (2/32), 7 (2/32), and 8 (1/32) per 2.37 mm2. No multinucleated cells or cells with bizarre morphology were identified. Three of 32 (9.4%) samples were graded as Kiupel high-grade/Patnaik grade II, including 2 neoplasms from Dog 1 (Fig. 2d) and 1 neoplasm from Dog 4 (Supplemental Table S2). The neoplasms in the remaining 6 patients, including 29 of 32 (90.6%) samples, were graded as Kiupel low-grade/Patnaik grade II. Other histologic features included eosinophilic infiltrate (33/33), edema (21/33), discrete intracytoplasmic vacuoles within mast cells containing aggregates of granules, erythrocytes, or pyknotic debris (11/33; Fig. 2e), hemorrhage (3/33), hyalinized collagen (3/33), ulcers (3/33), and desmoplasia (1/33). Additional findings in the skin included acanthosis (15/33), hyperpigmentation (2/33), vesicle formation (2/33), folliculitis/furunculosis (2/33), and serocellular crusts (1/33). Most lesions (23/33) had compromised surgical margins, including 19 punch biopsy samples and 4 excisional biopsy samples. The margins were near (<1 mm) in 6 of 33, all of which were excisional biopsy samples. The margins were clean (>3 mm) in 4 of 33 samples, including 2 punch biopsy samples and 2 excisional biopsy samples. The KIT pattern was determined in 1 sample from each dog. There were 2 samples with KIT pattern I (Dogs 4 and 8) and the remaining 6 samples had KIT pattern II (Fig. 2f). Mutational analysis for c-KIT exons 8 and 11 was performed in all dogs and no mutations were detected.
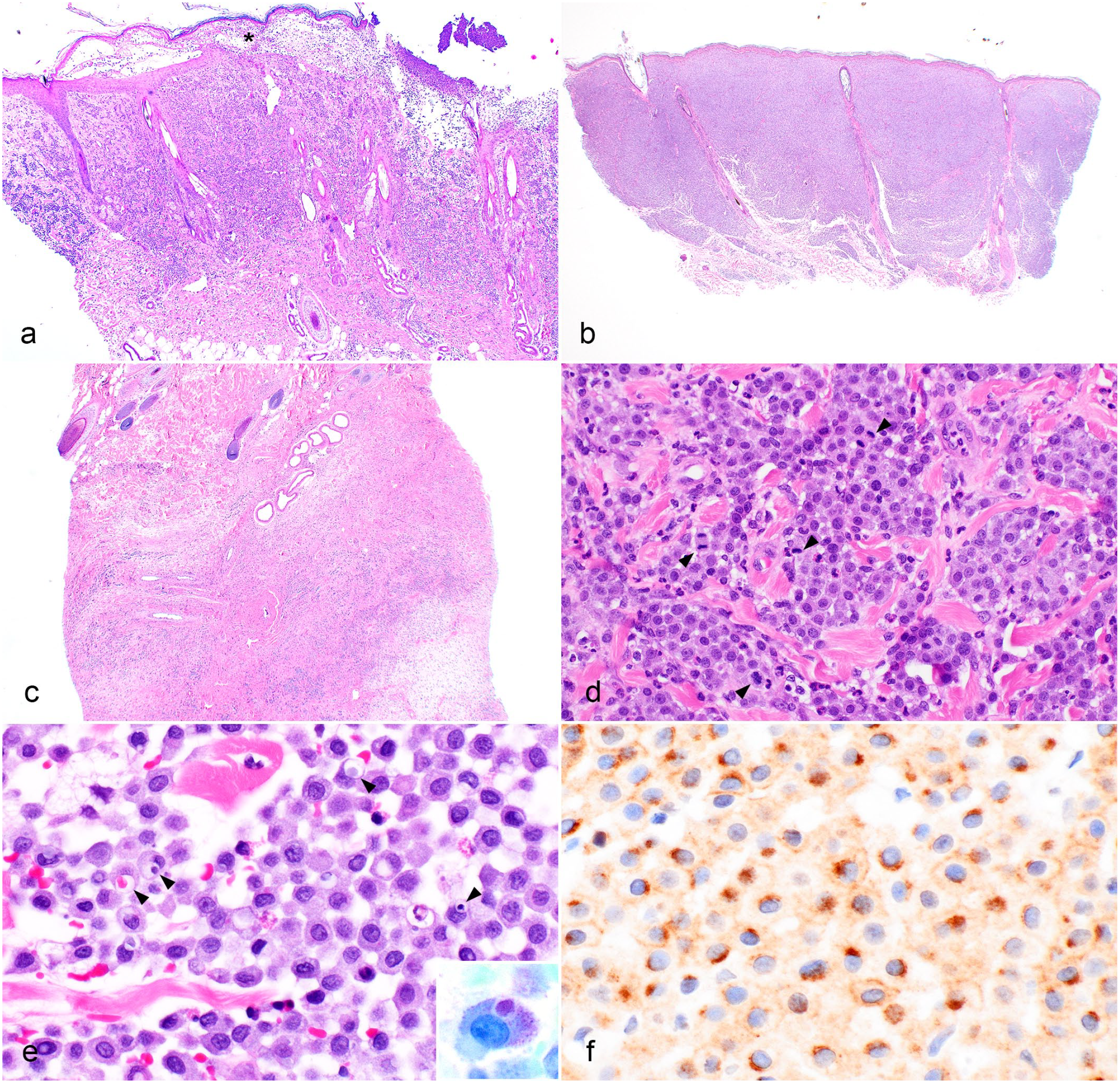
Histopathology of cutaneous mastocytosis in young dogs. (a) A punch biopsy sample from a vesicle of haired skin. There is a poorly demarcated dermal proliferation of neoplastic mast cells with vesicle formation (asterisk) and serocellular crusts (Dog 1, Hematoxylin and eosin [HE]). (b) A punch biopsy sample from a plaque on the pinna. The neoplastic mast cells form a moderately demarcated dense sheet expanding the dermis and extending to the lateral margins of the sample (Dog 2, HE). (c) A punch biopsy sample from a nodule. Expanding the deep dermis and subcutis are neoplastic mast cells with regions of edema (Dog 7, HE). (d) High magnification from a Kiupel high-grade/Patnaik grade II mast cell neoplasm from a dog with cutaneous mastocytosis. There are several mitotic figures (arrowheads) (Dog 1, HE). (e) High magnification from a Kiupel low-grade/Patnaik grade II mast cell neoplasm from a dog with cutaneous mastocytosis. Some mast cells contain aggregates of granules, erythrocytes, or pyknotic nuclei, within a discrete intracytoplasmic vacuole (arrowheads). Admixed with mast cells are eosinophils (Dog 4, H&E). Inset: The cytoplasmic granules and aggregates of granules within a discrete intracytoplasmic vacuole are metachromatic in the toluidine stain (Dog 3). (f) Intense focal cytoplasmic immunolabeling and weak membranous labeling of mast cells in a Kiupel low-grade/Patnaik grade II mast cell neoplasm in a dog with cutaneous mastocytosis, consistent with KIT staining pattern II (Dog 6). Immunohistochemistry for KIT.
Treatments
Dogs were treated with antihistamine (8/8), corticosteroids (7/8), lokivetmab (3/8; anti-canine IL-31 monoclonal antibody, Cytopoint, Zoetis; Parsippany, NJ, USA), and toceranib phosphate (1/8; Palladia, Zoetis) (Supplemental Table S2). Additional treatments included antacids (3/8) and antibiotics (5/8) for concurrent dermatitis. Surgical excision of the mast cell neoplasms was performed 5 times on 1 dog (Dog 8), but this patient continued to develop additional lesions. The dosage for prednisone or prednisolone was 0.2–1.3 mg/kg/d and the dosage for methylprednisolone was 0.3 mg/kg/d. When prednisone was used alone with a dosage between 0.9 and 1.1 mg/kg/d in 3 dogs (Dogs 1, 2, and 4), skin lesions were reported as a partial resolution in 1 dog (Dog 1), no response in 1 dog (Dog 4), and unknown response in 1 dog (Dog 2). When prednisolone was used alone with a dosage between 0.2 and 0.3 mg/kg/d in 2 dogs (Dogs 7 and 8), skin lesions recurred in 1 dog (Dog 7) and had no response in 1 dog (Dog 8). When corticosteroid was used in combination with antihistamines (5 dogs) and with antihistamines and lokivetmab (1 dog), skin lesions were reported as a complete resolution in 1 dog (dosage range, 0.8–1.6 mg/kg/d; Dog 7), partial resolution in 2 dogs (dosage range: 1.0–2.1 mg/kg/d; Dogs 3 and 5), and minimal to no response in 2 dogs (dosage range: 0.4–0.9 mg/kg/d; Dogs 1 and 2), and unknown response in 1 dog (Dog 4). Skin lesions recurred when corticosteroid was discontinued or tapered in 2 dogs (Dogs 5 and 7). When corticosteroid was used alone or in combination with other drugs, response to pruritus was reported as a complete response (Dog 5), partial response in 1 dog (Dog 1), and no response in 1 dog (Dog 4). Only one dog (Dog 3) received chemotherapy, which included 3 doses of toceranib. However, this treatment was discontinued due to the severe side effects of the development of cutaneous ulcers and open wounds secondary to tissue necrosis. Dog 3 was then given prednisone (2.1 mg/kg/d) and cetirizine (1.8 mg/kg/d) and the neoplasms decreased in size. The same dosage of prednisone was continuously given for 2 months inadvertently. After the initial response, the neoplasm continued to increase in size until Dog 3 was euthanized due to the rupture of 1 large neoplasm. When antihistamine was used alone (Dogs 3, 4, 6, 7, and 8), skin lesions were reported as no response in 4 dogs and partial response in 1 dog (Dog 6); response to pruritus was partial in 1 dog (Dog 3) and no response in 1 dog (Dog 4). Response to pruritus after lokivetmab injection alone was reported as partial in 1 dog (Dog 1) and unknown in 1 dog (Dog 4). Response to pruritus was complete in 1 dog (Dog 5) after lokivetmab injection in combination with prednisone and antihistamines.
Follow-up
Follow-up information was available for 8 of 8 dogs. At the time of manuscript preparation, 6 dogs were alive, and 2 dogs were euthanized (Dogs 3 and 4). The median follow-up time was 898 days (range: 56–1922 days). Six dogs that were alive continued to have additional lesions after removal by surgical excision, including 1 dog (Dog 1) that initially had 2 Kiupel high-grade/Patnaik grade II neoplasms and continued to have additional neoplasms at 1922 days post-diagnosis. During follow-up evaluation, Dog 1 had neutrophilia (27191/µL; reference interval: 2940–12670/µL) and monocytosis (1793/µL; reference interval: 130–1150/µL) 4 months later, and continued to have neutrophilia (from 27370 to 35120/µL) and monocytosis (from 2254 to 1760/µL) in the third and fourth years post-diagnosis. Dog 1 was further investigated for underlying infections, but the cause of neutrophilia and monocytosis was not identified. Dog 1 also developed bilateral lymphadenomegaly of the inguinal lymph nodes 3 years after the initial diagnosis. Aspiration of the enlarged inguinal lymph nodes demonstrated few mast cells, which were interpreted as drainage reactions. Dog 1 also had transient lethargy, inappetence, and episodes of vomiting 5 years after the initial diagnosis. One of the dogs that was euthanized (Dog 4) had 1 Kiupel high-grade/Patnaik grade II mast cell neoplasm at initial diagnosis. This dog was presented to an emergency service for lethargy and inappetence approximately 1 year after the onset of the disease and 56 days post-diagnosis. On presentation, the dog had elevated temperature (103.7°F), panting, extreme pain, yellow mucous membrane, and a slightly delayed capillary refill time of 2–4 seconds. The owner elected euthanasia and an autopsy was not performed. Five hundred days after the initial diagnosis, Dog 3 had mild neutrophilia (10160/µL; reference interval: 2700–9400/µL) and mild hyperalbuminemia (3.8 g/dL; reference interval: 2.5–3.7 g/dL) before the treatment of toceranib. One hundred and twenty-one days later, Dog 3 was presented to the emergency service due to the rupture of a large cutaneous neoplasm after running. Dog 3 was euthanized 621 days post-diagnosis and an autopsy was not performed.
Discussion
This study reports the clinical and histopathologic features with follow-up information in 8 young dogs with CM. CM in young dogs is characterized by multiple to generalized nodules, plaques, and papules composed of neoplastic mast cells that are histologically indistinguishable from those in solitary cMCT. The lesions responded to corticosteroids in combination with antihistamines in some cases, but persisted for years with a median follow-up time of 898 days.
CM in dogs has several features distinct from CM in humans. It is worthwhile noting that the terminology and diagnostic criteria used for mast cell neoplasms differ between humans and dogs. In humans, mastocytosis is characterized by the clonal proliferation of mast cells in different organs and is subclassified into cutaneous form affecting the skin or systemic form affecting bone marrow and/or other organs. 4 CM is further classified into 3 types: (1) maculopapular cutaneous mastocytosis, which was previously termed urticaria pigmentosa and is defined as patients with 4 or more lesions, (2) diffuse cutaneous mastocytosis, and (3) mastocytoma defined as solitary neoplasms with a maximum of 3 lesions. 4 This is distinct from the conventional terminology used in veterinary medicine where a solitary neoplasm composed of mast cells is more commonly called a mast cell tumor instead of mastocytoma. Despite the name “maculopapular” CM, the lesions in humans can also present as nodules. Maculopapular CM is further subdivided into 2 variants, including a monomorphic variant with small maculopapular lesions that are typically seen in adults and a polymorphic variant with larger asymmetric macular, plaque-type, or nodular lesions that are seen in pediatric patients. In pediatric patients, the monomorphic variant tends to persist into adulthood, whereas most cases of the polymorphic variant spontaneously regress. 4 While the cases in our study are most compatible with a polymorphic variant of maculopapular CM, based on the definition from human literature, none of our cases had spontaneous regression and only one case of CM in a dog spontaneously regressed in the literature. 3 A recent study reported a case of the bullous form of diffuse cutaneous mastocytosis in a 7-month-old puppy. 18 The lesions were controlled with systemic corticosteroids, antihistamines, and masitinib, and the dog was alive at 10 months post-diagnosis. Due to the small number of cases reported, whether the subclassification of CM is meaningful in dogs is yet to be investigated.
Diagnostic criteria used for mast cell neoplasms also differ between humans and dogs. Diagnosis of CM in human patients includes one major criterion, which is typical clinical cutaneous lesions of mastocytosis associated with Darier’s sign. 4 In addition, systemic involvement of the disease should be excluded. 21 Two minor criteria are usually present, including increased numbers of mast cells in biopsy sections of lesional skin and activating c-KIT mutation in lesional skin. In contrast, canine cMCT is typically diagnosed via fine-needle aspiration with or without histopathologic examination and the occasional use of IHC. While Darier’s sign was reported, this is not always a feature of cMCT in dogs. This is consistent with our findings and the literature, where only 2 of 3 dogs in this study and 4 of 6 dogs in the literature had a positive Darier’s sign. In addition, c-KIT mutations are not routinely present in canine patients with cMCT and they are more common in high-grade cMCT. 22 Studies reported that mutation in c-KIT exon 11 in dogs with cMCT ranged from 9.55% to 17.6%.22,23 All cases of CM in dogs in the literature and our case series did not carry mutations in c-KIT exons 8 and 11, which contrasted the childhood-onset CM in pediatric patients where c-KIT mutations are relatively common. In one study, 86% of children were reported to have c-KIT mutations, including 42% of cases with mutations in exon 17 codon D816V and 44% of cases with mutations outside exon 17. 1 Future studies investigating additional c-KIT mutations, other than exons 8 and 11, in dogs with CM may be warranted.
Eight prior publications on canine CM were identified in the literature.3,5,7,12,14,16,18,19 With the exclusion of 2 publications due to the focal nature of the lesion reported and the later onset of the disease,7,12 these studies included 7 dogs and are summarized in Supplemental Table S3. In prior reports, the median age of onset and diagnosis was 12 months (range: 3 weeks–27 months) and 24 months (range: 2.5–84 months), respectively. While the age of onset and diagnosis was later than what we reported in this study, the skin lesions described in the literature were similar to our study. The number of lesions varies from 3 to approximately 100 and ranges in size from 0.4 to 5 cm. The lesions observed were papules (3/7), nodules (3/7), macules (2/7), and hemorrhagic bullae (1/7). The lesions were reported as multiple (6/7) or generalized (1/7) with pruritus (4/5). No mucosal involvement was reported in any cases. Unlike our study, concurrent systemic signs were reported in 3 of 7 dogs at the time of diagnosis, including vomiting (2/7), hematochezia (1/7), lethargy (1/7), diarrhea (1/7), and tenesmus (1/7).3,16,19 One dog was diagnosed with concurrent pancreatitis. 16 Four dogs in the literature were clinically staged with various methods, including a combination of fine-needle aspiration of the regional lymph node, imaging of the visceral organs, cytologic examination of the blood smear, and/or bone marrow aspirate, and systemic involvement was not reported at the time of initial diagnosis.3,5,14,19 Systemic involvement cannot be completely ruled out in our study because most dogs were not staged at the initial diagnosis, and 1 dog (Dog 1) eventually developed transient clinical signs of lethargy, inappetence, and vomiting 5 years post-diagnosis.
The proliferation of mast cells and associated histologic changes in CM and cMCT in dogs are histologically indistinguishable based on our study. The associated histologic changes such as eosinophilic infiltrates, edema, and occasional hyalinized collagen reported in typical canine cMCT were present in our cases of CM. This finding is similar to humans where subtypes of CM cannot be distinguished histologically. Intracytoplasmic vacuoles within neoplastic mast cells containing aggregates of metachromatic granules, erythrocytes, or pyknotic debris were observed at some frequency in this study (Fig. 2e). This finding has been observed in typical solitary canine cMCT in the authors’ experience. Intracytoplasmic vacuoles containing erythrocytes or degenerate neutrophils have been observed previously in feline cMCT and these findings were interpreted as endocytosis of erythrocytes or neutrophils.6,13 Therefore, a diagnosis of CM in dogs cannot be made by histopathologic findings alone and requires a supporting clinical history of a young age of onset.
Unlike the literature where 1 case had spontaneous regression, none of the dogs in our study had spontaneous regression or resolution of lesions despite various treatments. In the literature, CM was treated with antihistamine alone or in combination with corticosteroids (Supplemental Table S3). Antihistamines did not appear to be an effective treatment in our study when used alone with a partial response in only 1 of 5 dogs. This is consistent with the literature; 3 dogs were treated with antihistamines alone, and only one dog had resolution of skin lesions 5 while the other 2 had minimal to no response.16,19 When antihistamines were used in combination with corticosteroids (including 1 dog with an additional combination of lokivetmab in our study), there was a partial to complete response in 3 of 4 dogs in the literature and 3 of 5 dogs in our study. The lesions recurred when corticosteroids were tapered in 2 dogs in our study. The use of chemotherapy for CM in dogs has not been reported. One dog in our study was given toceranib, but this was discontinued due to the severe side effects of tissue necrosis. Whether toceranib can be used as a treatment for CM in dogs requires further investigation. Lokivetmab was reported to resolve pruritus and flare in 1 dog in the literature. 14 In our study, lokivetmab partially and completely resolved pruritus in 1 dog each, whereas the response was unknown in the third dog. Overall, skin lesions do not appear to respond to antihistamines alone and likely require a combination with corticosteroids. Skin lesions recurred when corticosteroids were tapered. Lokivetmab resolved pruritus in CM to various degrees.
The Kiupel 2-tier system and Patnaik grading systems were originally developed for solitary lesions but not multicentric lesions. This alone suggests that these grading systems are inappropriate for CM. Also, while the mean and median survival times for Kiupel high-grade tumor are 6.29 and 3.65 months, respectively, 10 1 dog in our study that initially was given a grade of Kiupel high-grade/Patnaik grade II was still alive at 1922 days post-diagnosis. It is important to emphasize that the Kiupel 2-tier system and Patnaik grading systems were established based on dogs that were 1–14 years old and 2–19 years old, respectively.10,17 It also seems to be inappropriate to use these grading systems in dogs with mast cell neoplasm(s) with an early age onset of less than 1 or 2 years. In support of this, a favorable prognosis was identified in dogs that were less than 1 year of age with solitary cMCT in 1 study. 20 The authors in that study identified 4 Kiupel high-grade cMCT among 16 dogs aged less than 1 year of age with solitary cMCT, and the dogs were all alive and disease-free after various treatments at follow-up times of 972, 1017, 1769, and 2628 days post-diagnosis, respectively. 20 Therefore, future studies investigating prognostic indicators for young dogs with CM or solitary cMCT are warranted.
This study has several limitations. Systemic involvement cannot be completely ruled out because clinical staging was only performed in 2 dogs at the time of diagnosis. Also, clinical staging and autopsy were not performed in 1 dog that initially had Kiupel high-grade/Patnaik grade II neoplasms. While systemic involvement at the initial diagnosis appeared to be less likely because all dogs did not have systemic signs, one of the dogs eventually developed lethargy, inappetence, and episodes of vomiting 5 years post-diagnosis. Whether these clinical signs were related to CM was not investigated.
Conclusion
Diagnosticians should be aware of CM, which is characterized by multifocal to generalized nodules, plaques, and papules composed of dermal neoplastic mast cells with variable extension to the subcutis in young dogs. This condition is histologically indistinguishable from typical cMCT and the diagnosis requires correlation with signalment and clinical signs. None of the dogs had spontaneous regression and the lesions persisted for years. The lesions responded to corticosteroids in combination with antihistamines in some cases. While metastatic spread was not identified in our study, monitoring for systemic involvement over the years may be required. Future studies on histologic grading systems for young dogs with CM are needed.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231174452 – Supplemental material for Cutaneous mastocytosis in 8 young dogs and review of literature
Supplemental material, sj-pdf-1-vet-10.1177_03009858231174452 for Cutaneous mastocytosis in 8 young dogs and review of literature by Ching Yang, Charles W. Bradley, Diane Preziosi and Elizabeth A. Mauldin in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858231174452 – Supplemental material for Cutaneous mastocytosis in 8 young dogs and review of literature
Supplemental material, sj-xlsx-2-vet-10.1177_03009858231174452 for Cutaneous mastocytosis in 8 young dogs and review of literature by Ching Yang, Charles W. Bradley, Diane Preziosi and Elizabeth A. Mauldin in Veterinary Pathology
Supplemental Material
sj-xlsx-3-vet-10.1177_03009858231174452 – Supplemental material for Cutaneous mastocytosis in 8 young dogs and review of literature
Supplemental material, sj-xlsx-3-vet-10.1177_03009858231174452 for Cutaneous mastocytosis in 8 young dogs and review of literature by Ching Yang, Charles W. Bradley, Diane Preziosi and Elizabeth A. Mauldin in Veterinary Pathology
Footnotes
Acknowledgements
We thank Drs Allison Inga, Drew Fleischman, Naomi Rich, Brian L. Dvorsak, Greg Bogle, Noel Radwanski, and Lois Rich for providing the clinical history, clinical photographs, and follow-up information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Immunohistochemical staining has been provided by the Penn Vet Comparative Pathology Core, which is partially supported by the Abramson Cancer Center Support Grant (P30 CA016520). The Aperio Versa 200 scanner used for whole slide imaging and the image analysis software was supported by a NIH Shared Instrumentation Grant (S10 OD023465-01A1).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
