Abstract
Although waterfowl are less susceptible to Newcastle disease (ND) virus (NDV) infection compared with chickens and turkeys, lethal ND in waterfowl has been sporadically reported. Factors underlying the high pathogenicity of certain NDV strains in waterfowl remain unclear. In ducks, the NDV 9a5b isolate shows low pathogenicity while the d5a20b isolate shows high pathogenicity. This study aimed to identify the definitive lesions that led to the lethal virulence of d5a20b by comparing the histopathology of 9a5b- or d5a20b-inoculated ducks in order to elucidate lesions related to the enhanced pathogenicity of certain NDV strains in ducks. Herein, 7-day-old ducks were intranasally inoculated with either 9a5b or d5a20b NDV strains. The neurological signs were more severe in the d5a20b-inoculated group than in the 9a5b-inoculated group. Ducks in the d5a20b-inoculated group exhibited more severe lymphoid depletion in immune organs than those in the 9a5b-inoculated group, which may have caused an immunosuppressive state in the d5a20b-inoculated ducks. Ducks in the d5a20b-inoculated group had more severe nonsuppurative encephalitis with increased NDV nucleoprotein than those in the 9a5b-inoculated group. Additionally, pancreatic necrosis, with intralesional NDV nucleoprotein, was more severe in the d5a20b-inoculated group than in the 9a5b-inoculated group. Our results showed that the immune organs, brain, and pancreas were significant targets of the NDV d5a20b infection in ducks.
Newcastle disease (ND) is one of the most prevalent poultry diseases that significantly impedes the poultry industry worldwide. 36 ND is caused by virulent strains of avian paramyxovirus 1 (species Orthoavulavirus javaense), also known as the Newcastle disease virus (NDV), which belongs to the Orthoavulavirus genus, Avulavirinae subfamily, and Paramyxoviridae family.17,29 NDV is an enveloped virus with a single-stranded, negative-sense, and non-segmented RNA genome containing 6 genes (3′-NP-P-M-F-HN-L-5′) that encode nucleoprotein (NP), phosphoprotein (P), matrix protein (M), fusion protein (F), hemagglutinin-neuraminidase (HN), and large RNA-dependent RNA polymerase protein (L). 7 Additionally, the phosphoprotein encoding gene can express non-structural proteins V and W in virus-infected cells through RNA editing. 35
As per the World Organization for Animal Health, virulent NDV strains are defined as viruses with an intracerebral pathogenicity index exceeding 0.7 in 1-day-old chicks or containing phenylalanine at position 117 along with multiple basic amino acids at the cleavage site of the fusion protein. 29 Based on the clinical signs observed in infected chickens, NDV strains have been categorized into 5 pathotypes, from the least to the most virulent: asymptomatic, lentogenic, mesogenic, and velogenic with neurotropism or viscerotropism. 29 However, pathotype categories are often ambiguous, and infections of specific-pathogen-free birds can show considerable overlap. 29
NDV can naturally or experimentally infect over 200 avian species; however, the disease severity varies with both host species and virus strains. 29 Among the host species, chickens and turkeys are more susceptible to NDV infection than waterfowl, such as ducks and geese. 32 Generally, waterfowl are resistant to velogenic NDV and show few or no signs post-infection, whereas chickens often develop a more severe disease with a high mortality rate.1,36 This difference in susceptibility to NDV infection has been explained by differences in the innate immune system between waterfowl and chickens; waterfowl have stronger innate immune responses than chickens and are considered a natural reservoir for NDV.11,32
The number of ND outbreaks in waterfowl has been increasing in China since the late 1990s.8,18,23,39,41,42,44,46 In 2007, a velogenic NDV strain was isolated from the carcass of a mallard duck found in a field in Serbia. 37 These findings indicate that waterfowl are not only natural reservoir of the virus but can also exhibit clinical signs of ND. Although virologic, clinical, and histologic studies on experimental NDV infections in waterfowl have been reported,4,9,10,19,24,28,39,40,43,45 few detailed and temporal histologic studies have been conducted on the systemic organs of NDV-infected waterfowl, and the factors underlying the high pathogenicity of certain NDV strains in waterfowl remain unclear.
Previously, Shengqing et al 34 generated a velogenic NDV mutant isolate (9a5b) by passaging a lentogenic Goose/Alaska/415/91 isolate in chickens. Our previous histologic studies have demonstrated that although it is more virulent in chickens, 9a5b induces mild lesions in ducks,2,3 indicating its marginal pathogenicity in ducks, similar to other virulent NDVs. Hidaka et al 16 reported that 9a5b acquired high pathogenicity in ducks via serial passaging in domestic ducks. The intracerebral pathogenicity index value in 2-day-old domestic ducks increased from 1.10 to 1.88, and the passaged NDV mutant isolate (d5a20b) efficiently replicated in tissues. 16 To date, 11 amino acid substitutions have occurred between 9a5b and d5a20b, 16 and these affect the function and activity of viral proteins, which might underlie the change of the pathogenicity of the virus. However, histologic analyses of d5a20b-inoculated ducks have not yet been performed, and the definitive lesions underlying the high pathogenicity of d5a20b in ducks remain unclear.
We aimed to elucidate the lesions associated with the enhanced pathogenicity of certain NDV strains in ducks by comparing the clinical signs, gross and histologic lesions, and distribution of viral antigens in 9a5b- or d5a20b-inoculated ducks by immunohistochemistry.
Materials and Methods
Virus and Animals
The 9a5b 34 and d5a20b 16 NDV mutant isolates were obtained from the Department of Veterinary Public Health at Tottori University, Japan. One-day-old domestic ducks (n = 40) (Takahashi Artificial Hatchery, Osaka, Japan) were randomly divided into groups 1 (n = 18, ducks 1–18), 2 (n = 18, ducks 19–36), and 3 (n = 4, ducks 37–40), which consisted of 9a5b-inoculated, d5a20b-inoculated, and control ducks, respectively. All groups were kept separately in bird-breeding isolators in biohazard rooms and were provided water and food ad libitum. After an acclimation period, 7-day-old ducks in the infected groups (groups 1 and 2) were intranasally inoculated with 107 egg infective doses (EID50)/0.2 ml of the virus. From group 1, 3 ducks each were euthanized at 2, 4, 5, 6, 7, and 8 days post-infection (dpi). Ducks in group 2 were euthanized at 1 dpi (n = 4), 2 (n = 4), 3 (n = 3), and 4 (n = 1), respectively. Also, dead ducks at 3 dpi (n = 3) and 4 dpi (n = 3) were sampled immediately. Ducks in the control group (group 3) were intranasally inoculated with saline solution and euthanized at 4 days after inoculation. The ducks were euthanized using isoflurane (FUJIFILM Wako Pure Chemical Corporation, Tokyo, Japan). The Ethics Committee on Animal Experiments of Tottori University approved the experimental protocol (approval number 18-T-17), and the experiments were performed in accordance with the guidelines for animal experiments in the same facility.
Necropsy and Sampling
Necropsy and tissue sampling were performed immediately after the ducks were euthanized. During the period of severe clinical signs in ducks, observations were made every 2 h, and dead ducks were collected as soon as possible to minimize postmortem autolysis. The brain, spinal cord, nasal mucosa, trachea, lung, heart, proventriculus, gizzard, duodenum, jejunum, ileum, cecum, colorectum, liver, pancreas, spleen, bursa of Fabricius, thymus, and kidney were collected and fixed in 10% neutral-buffered formalin or preserved at −80°C for virus titration.
Histology and Immunohistochemistry
After approximately 72 h of fixation, samples from the aforementioned organs were dehydrated, paraffin embedded, and sectioned for hematoxylin and eosin staining (3-μm thick sections) or immunohistochemistry (IHC; 5-μm-thick sections on positively charged slides).
The primary antibody for NDV-NP detection was prepared in rabbits as described by Kommers et al. 21 Briefly, a polyclonal rabbit anti-NDV-NP was raised against the synthetic peptide TAYETADESETRRIC. Sections were dewaxed, rehydrated, and treated with 3% H2O2 in distilled water for 15 min at room temperature to block endogenous peroxidase activity, followed by antigen retrieval in citrate buffer (pH 5.4) and microwaving at full power for 10 min. Sections were blocked with 10% normal goat serum for 30 min at room temperature and then incubated with the primary antibody (1:8000 dilution) for 12 hours at 4°C.2,3 After stringent washing with phosphate-buffered saline, the sections were incubated with a labeled polymer (ChemMate Dako EnVision/HRP [DAP]; Dako, Carpinteria, California, CA) for 30 min at room temperature and subjected to stringent washing. Labeling was visualized using 3,3′-diaminobenzidine tetrahydrochloride (Dako), and the sections were counterstained with hematoxylin and covered with DPX mounting media (Sigma Life Science, Steinheim, Germany). Cleaved caspase-3 was detected with an anti-ACTIVE caspase-3 polyclonal antibody (Promega, Madison, WI, USA) (1:350 dilution), using a protocol similar to that for NDV-NP, except blocking with normal goat serum was not performed, the detection was performed using a polyclonal biotinylated goat anti-rabbit secondary antibody (Dako), and the enzyme-substrate reaction involved a streptavidin-biotin complex (Dako). 3 As positive control, the sample from previous study 13 was used. As negative controls, the tissues from uninfected ducks were used, and slides incubated with phosphate-buffered saline (omitted antibodies) were prepared. The cytoplasmic signals were considered as positive immunoreactions in both NDV-NP and cleaved caspase-3.
Grading of Histologic Lesions and IHC
The scoring systems used for histologic lesions and IHC labeling of NDV-NP are provided below and in Supplemental Table S1. Histologic and NDV-NP IHC scores were compared between ducks in groups 1 and 2 at 2 and 4 dpi. Data were tested using the Mann-Whitney U tests. All the slides were graded simultaneously by 3 veterinary pathologists.
Scoring of the immune organs
Histologic lesions manifesting as lymphoid depletion were scored. The severity of lymphoid depletion was evaluated across the entire section of each immune organ per animal and scored in 5 grades (Supplemental Table S1). For IHC scoring of NDV-NPs in immune organs, 5 different high-power fields (0.982 mm2) were randomly selected. In brief, the area containing the cortex and medulla was randomly selected in the thymus, and the bursa of Fabricius, and for the spleen, the area containing the white pulp and red pulp was randomly selected. Subsequently, the number of positive cells was counted, and the mean was calculated and scored in 5 grades (Supplemental Table S1). Statistical analyses (Mann-Whitney U tests) were performed to compare the difference in the NDV-NP IHC scores of the thymic medulla, bursal medulla, and splenic peri-arteriolar lymphoid sheath in ducks between 2 groups at 2 and 4 dpi. The thymic medulla, bursal medulla, and splenic peri-arteriolar lymphoid sheath were selected because the NDV-NP IHC scores were similar within the thymic areas (the cortex and medulla), bursal areas (the cortex, medulla, and follicle-associated epithelium), and splenic areas (the peri-ellipsoidal lymphoid sheath, peri-arteriolar lymphoid sheath, germinal center, and red pulp) of the ducks in each group by preliminary statistical analyses (Wilcoxon signed rank test of the thymus and Friedman test for the bursa of Fabricius and spleen; p-value >0.05). Active caspase-3-positive cells were counted in 5 randomly chosen high-power fields within each immune organ of the ducks at 4 dpi in each group (3 9a5b-inoculated ducks, 4 d5a20b-inoculated ducks, and 4 control ducks). The data were analyzed using a nonparametric Kruskal-Wallis test, followed by the Steel-Dwass multiple comparisons test.
Scoring of the central nervous system
Grading of histologic lesions and IHC labeling of NDV-NP were performed on 6 distinct areas of the brain—namely the cerebrum (CBR), diencephalon, tectum, reticular formation of the midbrain (RF), medulla oblongata, and cerebellum (CER)—and sections from the cervical spinal cord, thoracic cord, and lumber cord. Histologic lesions and IHC labeling of NDV-NPs were evaluated according to the scoring system as shown in Supplemental Table S1. Statistical analyses (Mann-Whitney U tests) were performed to compare the difference of the CBR, RF, CER, and cervical spinal cord in ducks between 2 groups at 2 and 4 dpi. RF (brain stem) and cervical spinal cords were selected because both the histologic and NDV-NP IHC scores were similar within the brain stem (diencephalon, tectum, RF, and medulla oblongata) and the cervical, thoracic, and lumber spinal cord of the ducks in each group by preliminary statistical analyses (Friedman test; P-value >.05).
Scoring of other organs
The histologic lesions and IHC labeling of NDV-NPs were evaluated across the entire section of each organ per animal and scored in 5 grades (Supplemental Table S1).
Virus Titration of the Brain of d5a20b-Inoculated Ducks
Virus titers were measured in 10-day-old embryonated specific pathogen-free eggs. Briefly, the brain tissue from each duck was homogenized in phosphate-buffered saline (1: 10 [wt: vol]), and clear homogenate (100 µl) obtained following a 10-fold serial dilution was inoculated into the allantoic cavity of the eggs. The eggs were incubated at 37ºC for 2 days and then at 4ºC for 1 day, and hemagglutination assays were performed. Viral titers are expressed as EID50 per gram.29,31
Statistical Analyses
Statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). Data were tested for significant differences using the Mann-Whitney U tests, Wilcoxon signed-rank test, Friedman test, Kruskal-Wallis tests, or Steel-Dwass multiple comparison test. The correlation between the data was evaluated using Spearman’s rho correlation. Statistical significance was set at a p-value <0.05. The exact method of Mann-Whitney U test was adopted to scrutinize the difference between 2 independent samples (2 groups) across 2 variables (histologic and IHC scores). The exact method of this test is more suited for analyzing small sample sizes (n = 7) following nonparametric distribution. All statistical tests were validated using IBM SPSS (v27.0.1, IBM, IL, USA) as well as Python scripts in Google Colab environment.
Results
Clinical Signs
Ducks in the 9a5b-inoculated (group 1) showed an unsteady gait at 2 dpi and additional dysstasia at 6 dpi; however, no obvious respiratory or gastrointestinal signs or deaths were observed. Clinical signs in ducks in the d5a20b-inoculated (group 2) appeared at 2 dpi and included unsteady gait, which worsened until the end of the experiment, with depression, anorexia, gasping, eyelid edema, dysstasia with unilateral or bilateral weakness of the legs, twisting of the head and neck, shivering, and muscular incoordination. Ducks in group 2 died at 3 and 4 dpi. Gastrointestinal signs including diarrhea were not observed. The control ducks remained healthy throughout the observation period. The clinical signs are summarized in Supplemental Table S2.
Gross Lesions
Gross lesions are summarized in Supplemental Table S3. Ducks in group 1 showed mild thymic atrophy and pancreatic petechiae at 2–6 dpi and 4 dpi, respectively. Thymic atrophy of varying severity was observed in ducks in group 2 at 1–4 dpi. In the pancreas from ducks in group 2, petechiae were observed at 1 dpi, and pale white foci were evident at 2–4 dpi. In addition, splenic atrophy with white or dark red spots at 2–4 dpi, bursal atrophy at 3 and 4 dpi, brain edema and hyperemia at 3 and 4 dpi were observed in ducks in group 2. No gross lesions were observed in control ducks.
Histology and Immunohistochemistry
Histologic and immunohistochemical findings are summarized in Supplemental Tables S4–S7 and the results of the semi-quantitative analysis are shown in Supplemental Table S8.
Thymus
Compared with control ducks (Fig. 1a), the number of apoptotic and necrotic lymphocytes was mildly increased in ducks in group 1 at 4–6 dpi, and mildly-to-severely increased in ducks in group 2 at 2–4 dpi (Fig. 1d, g). The cortex and medulla of the thymus from ducks in group 1 were infiltrated with cells that were morphologically compatible with macrophage-like cells (macrophages and dendritic cells), whereas thymus from ducks in group 2 were infiltrated with heterophils in addition to these cells. The lymphoid depletion score of ducks in group 2 was nominally higher than that of ducks in group 1 at 2 dpi, as revealed by the near-significant-level P-value (P = .057). There was no significant difference in scores between the groups at 4 dpi (P > .05); however, ducks in group 2 exhibited severe lymphoid depletion at 3 and 4 dpi. The number of cleaved caspase-3-positive cells was significantly higher in the thymus from ducks in group 1 than in those from control ducks (P < .01) and was significantly higher in the thymus from ducks in group 2 than in those from control ducks (Fig. 1b) and ducks in group 1 (P < .01) (Fig. 1e, h, Supplemental Figure S1). NDV-NP immunolabeling was detected mainly in macrophage-like cells in the cortex and medulla of the thymus from ducks in group 1 at 2–8 dpi and ducks in group 2 at 2–4 dpi (Fig. 1f, i). There was no significant difference in the NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05), whereas only ducks in group 2 showed numerous NDV-NP immunolabeling at 3 and 4 dpi.
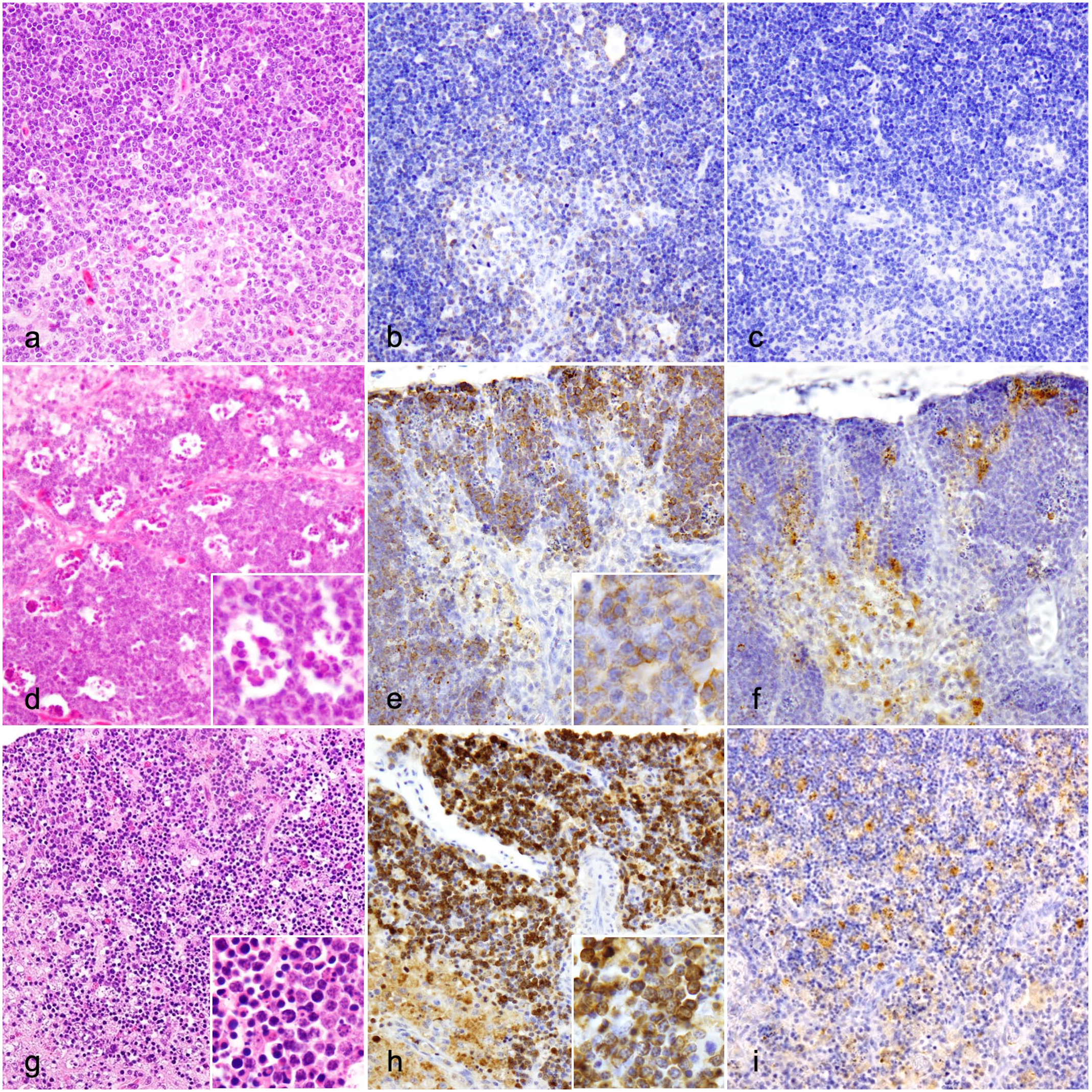
Histologic and immunohistochemical findings of the thymus in control, 9a5b-inoculated (group 1), and d5a20b-inoculated (group 2) ducks. (a-c) Control duck. (a) Few apoptotic lymphocytes are in the cortex and medulla. Hematoxylin and eosin (HE). (b) Cleaved caspase-3 immunolabeling is present in a few lymphocytes. Cleaved caspase-3 immunohistochemistry (IHC). (c) No Newcastle disease virus-nucleoprotein (NDV-NP) immunolabeling is detected. NDV-NP IHC. (d-f) Group 1 duck, 4 days post-infection (dpi). (d) Compared with control ducks, an increase in the number of apoptotic and necrotic lymphocytes (inset) is observed with mild lymphoid depletion in the cortex and medulla. HE. (e) Compared with control ducks, an increase in the number of cleaved caspase-3-positive cells is observed in the thymus (inset). Cleaved caspase-3 IHC. (f) NDV-NP immunolabeling is detected mainly in macrophage-like cells. NDV-NP IHC. (g-i) Group 2 duck, 3 dpi. (g) Compared with ducks in group 1, a marked increase in the number of apoptotic and necrotic lymphocytes (inset) is observed with severe lymphoid depletion and infiltration of macrophage-like cells and heterophils in cortex and medulla. HE. (h) Compared with ducks in group 1, a marked increase in the number of cleaved caspase-3-positive cells is observed in the cortex and medulla (inset). Cleaved caspase-3 IHC. (i) NDV-NP immunolabeling in macrophage-like cells. NDV-NP IHC.
Bursa of Fabricius
Compared with control ducks (Supplemental Figure S2a), a mild increase in the number of apoptotic lymphocytes was observed in some lymph follicles from ducks in group 1 (Supplemental Figure S2d) at 4 and 6 dpi. Mild-to-severe increase in the number of apoptotic and necrotic lymphocytes, as well as the infiltration of macrophage-like cells and heterophils, was observed in ducks in group 2 at 2–4 dpi (Supplemental Figure S2g). The lymphoid depletion score of ducks in group 2 was nominally higher than that of ducks in group 1 at 4 dpi, as revealed by the near-significant-level P-value (P = .057). The number of cleaved caspase-3-positive cells was significantly higher in the bursa from ducks in group 2 than in those from ducks in group 1 (P < .01) (Supplemental Figures S1 and S2b, e, and h). NDV-NP immunolabeling was observed mainly in macrophage-like cells in the lymph follicles and follicle-associated epithelium of the bursa from ducks in group 1 at 4–6 dpi and ducks in group 2 at 2–4 dpi (Supplemental Figures S2f, i). There were no significant differences in NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05).
Spleen
Compared with control ducks (Supplemental Figure S3a), a mild increase in the number of apoptotic lymphocytes was observed in the white pulp and red pulp of spleens from ducks in group 1 at 6 and 8 dpi (Supplemental Figure S3d). Mild-to-severe increase in the number of apoptotic and necrotic lymphocytes, as well as the infiltration of macrophage-like cells and heterophils, was observed in ducks in group 2 at 1–4 dpi (Supplemental Figure S2g). The lymphoid depletion score of ducks in group 2 was nominally higher than that of ducks in group 1 at 2 and 4 dpi, as revealed by the near-significant-level P-value (P = .057). The number of cleaved caspase-3-positive cells was significantly higher in the spleens from ducks in group 2 than in those from ducks in group 1 (P < .01) (Supplemental Figures S1 and S3b, e, and h). NDV-NP immunolabeling was observed mainly in macrophage-like cells in the white pulp and red pulp of spleens from ducks in group 1 at 5–8 dpi and ducks in group 2 at 2–4 dpi (Supplemental Figures S3f, i). At 4 dpi, the NDV-NP IHC scores of the spleen in ducks in group 2 were relatively higher than that of ducks in group 1, as revealed by the near-significant-level P-value (P = .057).
Pancreas
Mild-to-moderate pancreatic necrosis was observed in ducks in group 1 at 4–6 dpi, which was characterized by multifocal vacuolar degeneration, necrosis, and apoptosis of exocrine cells, with occasional infiltration of lymphocytes and macrophages in the interstitial tissue of the exocrine tissues (Supplemental Figure S4a). In ducks in group 2, similar lesions were first observed at 2 dpi, which worsened at 3 and 4 dpi. In addition, infiltration of heterophils and degeneration and necrosis of islet cells were detected in ducks in group 2 (Supplemental Figure S4c). There were no significant differences in histologic scores between the groups at 2 and 4 dpi (P > .05); however, compared with group 1, more ducks in group 2 had diffuse and severe lesions. NDV-NP immunolabeling was observed in the cytoplasm of degenerated and necrotic exocrine cells, macrophages, pancreatic duct epithelium, and islet cells in group 1 ducks at 4–8 dpi and group 2 ducks at 2–4 dpi (Supplemental Figures S4b, d). There were no significant differences in the NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05); however, compared with group 1, more ducks in group 2 had NDV-NP immunolabeling.
CNS
Mild-to-moderate nonsuppurative lymphocytic encephalitis and myelitis were observed throughout the CNS from ducks in group 1 at 2–8 dpi, which was characterized by lymphocytic perivascular cuffing, microgliosis (glial nodules), neuronophagia, and swelling of vascular endothelial cells (Fig. 2a). In ducks in group 2, similar lesions, along with macrophage-dominated perivascular cuffing and extensive neuronal tissue necrosis in all CNS areas, were observed at 2–4 dpi, which were more severe than those in ducks in group 1 (Fig. 2c). Regarding the RF and CER, the histologic score of ducks in group 2 were nominally higher than that of ducks in group 1 at 4 dpi, as revealed by the near-significant-level P-value (P = .057). Other CNS regions showed no significant differences in histologic scores between the groups (P > .05); however, extensive necrotic lesions were detected only in ducks in group 2. NDV-NP immunolabeling was observed in cells that were morphologically compatible with neurons, macrophages, lymphocytes, and glial cells in both groups. NDV-NP immunolabeling was detected in the RF, medulla oblongata, CER, and spinal cord from ducks in group 1 at 2–8 dpi and throughout the CNS of ducks in group 2 at 3 and 4 dpi (Fig. 2b, d). Regarding the CBR, RF, and CER, the NDV-NP IHC scores of group 2 ducks were nominally higher than those of group 1 ducks at 4 dpi, as revealed by the near-significant-level P-value (P = .057).
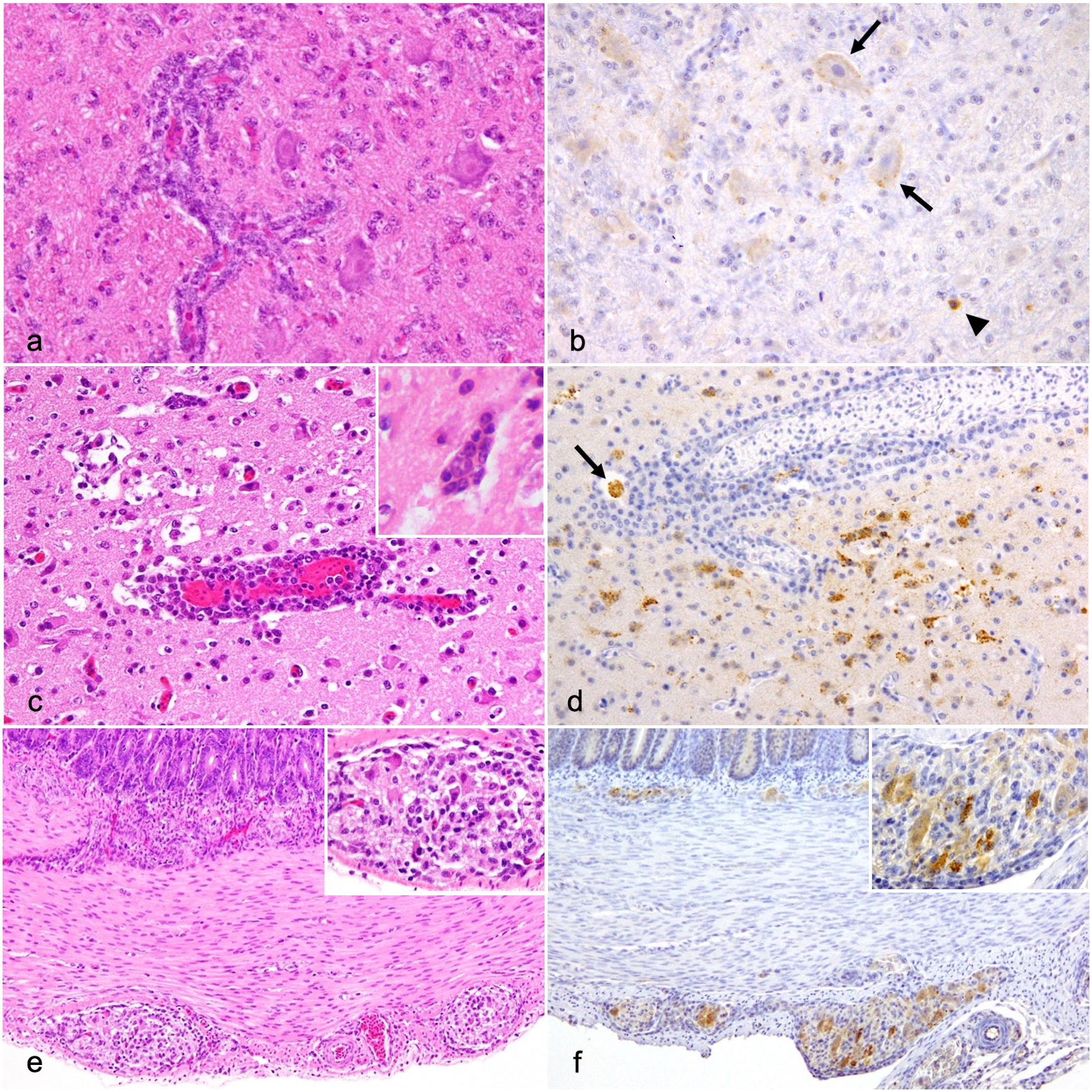
Histologic and immunohistochemical findings of the neural tissue in 9a5b-inoculated (group 1) and d5a20b-inoculated (group 2) ducks. (a-b) Medulla oblongata, group 1 duck, 7 days post-infection (dpi). (a) Mild nonsuppurative encephalitis with perivascular cuffing. Hematoxylin and eosin (HE). (b) Weak Newcastle disease virus-nucleoprotein (NDV-NP) immunolabeling in cells morphologically compatible with neurons (arrows) and glial cells (arrowhead). NDV-NP immunohistochemistry (IHC). (c-d) Cerebrum, group 2 duck, 4 dpi. (c) Severe nonsuppurative encephalitis with neuronophagia (inset) and perivascular cuffing. HE. (d) NDV-NP immunolabeling in the cells morphologically compatible with neurons, glial cells, and macrophages (arrow). NDV-NP IHC. (e-f) Duodenum, group 2 duck, 3 dpi. (e) Degeneration of enteric neurons and the infiltration of inflammatory cells in Meissner’s plexus and Auerbach’s plexus (inset). HE. (f) NDV-NP immunolabeling in enteric neurons (inset). NDV-NP IHC.
Spearman’s correlation analysis showed a significant but weak correlation between the histologic score of the CBR and the lymphoid depletion score of the thymus in ducks in group 1 (r = 0.584, P = .01) (Supplemental Table S9). In contrast, a significant and strong correlation between the histologic score of the CBR and the lymphoid depletion score of each immune organ was identified in group 2 ducks (thymus: r = 0.776, P < .01; bursa: r = 0.771, P < .01; spleen: r = 0.712, P < .01).
Respiratory system
Lymphocytic rhinitis and tracheitis with mild epithelial cell degeneration were observed in ducks in group 1 at 6 and 7 dpi, respectively. In ducks in group 2, lymphocytic rhinitis and tracheitis with mild degeneration and necrosis of epithelial cells were observed at 2–4 dpi. NDV-NP immunolabeling was occasionally observed in epithelial cells, glandular epithelial cells, and macrophages of the nasal mucosa and/or trachea in ducks in group 1 at 2–8 dpi and ducks in group 2 at 1–4 dpi. Mild pneumonia was observed in ducks in group 1 at 4 and 5 dpi, which was characterized by infiltration of macrophages, lymphocytes, and heterophils and degeneration and shedding of alveolar epithelial cells. In group 2 ducks, mild-to-moderate pneumonia was observed at 1–4 dpi. NDV-NP immunolabeling was occasionally detected in macrophages and epithelial cells within lesions in both groups. The histologic scores of the lungs from group 2 ducks were nominally higher than that of ducks in group 1 at 4 dpi, as revealed by the near-significant-level P-value (P = .057). In addition, ganglionitis was observed in the lungs from ducks in group 2 at 4 dpi, characterized by mild degeneration of neurons and infiltration of macrophages and lymphocytes. NDV-NP immunolabeling was occasionally detected in neurons and macrophages. These pathological lesions were not observed in the ganglion of group 1 ducks.
Heart
Focal to multifocal degeneration of myofibers was observed in ducks in group 1 at 4, 6, and 7 dpi, which was partly associated with NDV-NP immunolabeling. Multifocal mild-to-moderate degeneration and necrosis of myofibers with occasional mineralization was observed in ducks in group 2 at 2–4 dpi, which were partly associated with NDV-NP immunolabeling. There were no significant differences in the histologic and NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05). In both groups, ganglionitis was observed, which was characterized by neuronal degeneration and infiltration of macrophages and lymphocytes. NDV-NP immunolabeling was occasionally detected in neurons and macrophages.
Digestive tract
Gizzards from ducks in group 1 at 4-7 dpi showed mild degeneration and necrosis of epithelial cells and infiltration of macrophages and heterophils into the mucosa, which were associated with NDV-NP immunolabeling. Compared with control ducks, no obvious lesions were detected in other areas of the digestive tract from ducks in group 1. Mild enteritis was also observed from the duodenum to colorectum of ducks in group 2 at 3 and 4 dpi, which was characterized by degeneration of the mucosal epithelium and infiltration of lymphocytes, macrophages, and heterophils. NDV-NP immunolabeling was occasionally observed in epithelial cells and macrophages. No mucosal lesions were observed in the proventricular region. There were no significant differences in histologic and NDV-NP IHC scores between the groups at 2 and 4 dpi (p>0.05). Only ducks in group 2 exhibited plexitis from the proventriculus to colorectum at 3 and 4 dpi, which was characterized by the degeneration of enteric neurons and the infiltration of lymphocytes, macrophages, and heterophils (Fig. 2e). NDV-NP immunolabeling was detected in enteric neurons and macrophages (Fig. 2f).
Liver
In ducks in group 1 at 4 and 5 dpi and in group 2 ducks at 3 and 4 dpi, mild infiltration of macrophages, lymphocytes, and heterophils was observed in the sinusoids, and NDV-NP immunolabeling was occasionally observed in macrophages in liver. There were no significant differences in the histologic and NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05).
Kidney
Mild degeneration of the renal tubules and infiltration of macrophages and lymphocytes were observed in ducks in group 1 at 4–7 dpi and in group 2 ducks at 3 and 4 dpi, which were associated with NDV-NP immunolabeling. There were no significant differences in the histologic and NDV-NP IHC scores between the groups at 2 and 4 dpi (P > .05).
Control group
The histologic features of all organ tissues were within normal limits. No NDV-NP immunolabeling was detected in any organ by IHC.
Virus Titration of the Brain of d5a20b-Inoculated Ducks
The virus was detectable in 2/3 ducks at 3 dpi and in all (3/3) ducks at 4 dpi (Supplemental Figure S5). The results of virus titration evaluation were approximately correlated with the degree of NDV-NP immunolabeling (r = 0.919, P < .01) (Supplemental Table S7).
Discussion
In this study, we conducted comparative histologic analyses of ducks that were experimentally inoculated with low-virulence (9a5b) and virulent (d5a20b) NDV-mutant strains. Herein, neurological signs and gross lesions in the immune organs, pancreas, and brain in d5a20b-inoculated ducks were more severe than those in 9a5b-inoculated ducks. Various clinical signs and gross lesions have been previously reported in waterfowl that were naturally or experimentally infected with NDV, including gastrointestinal and respiratory lesions,9,24,32,39,40 which were not observed in this study. In chickens, the pathotypes of NDV strains are diverse. 29 Therefore, in waterfowl, as in chickens, the pathotypes exhibited by different virus strains may be diverse, and histologic lesions and virus antigen distribution in organs of waterfowl are important for understanding the virulence of NDV.
In the immune organs, the lesions in d5a20b-inoculated ducks were similar to those previously reported in NDV-infected geese24,39 and ducks, 45 suggesting that severe lesions in the immune organs might be a common sign of infection with virulent NDVs in waterfowl. Cleaved caspase-3-positive cells in immune organs from d5a20b-inoculated ducks indicates that apoptosis, in addition to necrosis, played a role in lymphocyte depletion, as reported in geese 24 and chickens.3,14,20 In an experiment using geese, it has been suggested that NDV infection may decrease immune function. 24 The severe lesions in the immune organs observed in d5a20b-inoculated ducks might be associated with an immunosuppressive state and contribute to the lethal virulence in ducks.
In the pancreas, pancreatic necrosis was observed mainly in the exocrine portion in ducks inoculated with either isolate, and these lesions were similar to those observed in the pancreas of chickens13,27,33 and geese.39,40 It has been suggested that damage mainly to the exocrine portion after NDV infection may be due to the protease content in exocrine cells, which may enhance viral replication via the activation of the viral fusion protein precursor.13,26 Our findings suggested that the pancreas might be one of the most important target organs for either isolate in ducks, which is consistent with previous reports.
Nonsuppurative encephalomyelitis was more severe in d5a20b-inoculated ducks than in 9a5b-inoculated ducks, which might be responsible for the lethal pathogenicity of d5a20b. NDV-NP immunolabeling was morphologically identified in the neurons, glial cells, and macrophages of ducks inoculated with either isolate, as previously reported in chickens.6,12,25 The strong correlation between the severity of histologic lesions and the number of viral antigens in d5a20b-inoculated ducks suggests that replication of d5a20b isolate directly induce severe lesions in the CNS of ducks. Although encephalitis caused by NDV infection has been reported in waterfowl,4,28,39 herein, CNS lesions in d5a20b-inoculated ducks were more severe than those previously reported in waterfowl. In chickens, it has been observed that nearly all NDV strains, except for the least virulent ones, can invade the CNS and cause pathological damage; however, the distribution and intensity of histologic lesions and viral antigens in the CNS vary among NDV strains. 12 The differences between the velogenic and mesogenic NDV strains suggest differences in the rate of NDV replication in nervous tissue. 25 As in chickens, the high replication rate of d5a20b in the nervous tissue might have contributed to its high virulence. Furthermore, the degree of lymphoid depletion in the immune organs correlated with the severity of encephalitis in d5a20b-inoculated ducks, suggesting that immunosuppressive states might have contributed to the development of neural lesions in d5a20b-inoculated ducks.
Plexitis associated with NDV-NP immunolabeling was observed in d5a20b-inoculated ducks despite the absence of other gastrointestinal lesions. Moreover, ganglionitis with NDV-NP immunolabeling was observed in the lungs and heart of d5a20b-inoculated ducks. NDV-NP immunolabeling was detected in the peripheral nervous system as well as in the CNS, which might suggest enhanced tropism for neural tissue of d5a20b. Previous reports have shown that virulent NDVs in waterfowl tend to replicate in the brain, whereas most velogenic NDVs without virulence in ducks do not.10,19 Accordingly, increased infectivity to neural tissues might be involved the enhanced pathogenicity of the d5a20b in ducks.
In ducks inoculated with either isolate, NDV-NP-positive macrophages were detected early in the respiratory organs, which served as a portal of entry for both isolates. Additionally, NDV-NP immunolabeling was detected in the systemic organs of the ducks inoculated with either isolate. NDV-NP-positive macrophages were observed in nearly all systemic organs, whereas few NDV-NP immunolabeling was observed in vascular endothelial cells. NDVs replicate in avian macrophages, leading to functional alterations and subsequent viral dissemination.3,5,22,24,28,30,38 Results of this study, as well as those of previous studies, suggest that macrophages play a major role in the systemic dissemination of NDV in ducks.
Genetic comparisons between the 9a5b and d5a20b strains have demonstrated that d5a20b has 11 amino acid substitutions in its phosphoprotein/V, matrix, fusion, HN, and large RNA-dependent RNA polymerase proteins compared with 9a5b. 16 Furthermore, experiments using recombinant NDVs with these amino acid substitutions have suggested that substitutions I142M in the fusion protein and Q44R in the matrix protein contribute to the enhanced virulence in ducks due to an increase in fusion activity and replication efficiency. 15 These substitutions in the fusion or matrix proteins might be responsible for the efficient infectivity and replicability of d5a20b in the nervous tissue, pancreas, and macrophage-like cells, especially in the thymus and spleen, of ducks. The d5a20b strain used in this study was mutant virus, and further studies using naturally circulating virulent NDVs in waterfowl are needed to elucidate the mechanism of enhanced pathogenicity of NDV in waterfowl.
In conclusion, the present study showed that the immune organs, pancreas, and neural tissues were significant targets of NDV d5a20b isolate in ducks. In particular, the damage of immune organs and increased infection in neural tissues may contribute in enhanced pathogenicity of NDV.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241286817 – Supplemental material for Comparative and experimental pathology of passaged Newcastle disease virus isolates in ducks
Supplemental material, sj-pdf-1-vet-10.1177_03009858241286817 for Comparative and experimental pathology of passaged Newcastle disease virus isolates in ducks by Sohta Hishikawa, Yuji Sunden, Aoi Imamura, Chiharu Hidaka, Hiroshi Ito, Toshihiro Ito and Takehito Morita in Veterinary Pathology
Footnotes
Authors’ Note
The manuscript has been prepared in accordance with the Uniform Requirements format.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially, in part, by a grant from the MEXT for the Joint Research Program of the Research Center for Zoonosis Control, Hokkaido University, Japan.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
