Abstract
In the past few years, Newcastle disease virus (NDV) strains with epizootic characteristics belonging to subgenotypes VIIi and XIIIb emerged in the Middle East and Asia. In this study, 2 NDV strains—1 representative of subgenotype VIIi isolated in Israel (Kvuzat/13) and 1 representative of subgenotype XIIIb isolated in Pakistan (Karachi/07)—were characterized by intracerebral pathogenicity index and detailed clinicopathologic assessment. The intracerebral pathogenicity index values for Kvuzat/13 and Karachi/07 were 1.89 and 1.85, respectively, classifying these strains as virulent by international standards. In 4-week-old White Leghorn chickens, both strains caused 100% mortality within 4 (Kvuzat/13) and 5 (Karachi/07) days postinfection. Histopathology and immunohistochemistry for NDV nucleoprotein showed that both strains had wide systemic distribution, especially targeting lymphoid organs and mucosa-associated lymphoid tissues in the respiratory and intestinal tracts. Results of the animal experiment confirm that both Kvuzat/13 and Karachi/07 are highly virulent and behaved as velogenic viscerotropic NDV strains.
Keywords
Newcastle disease (ND) is an important disease of poultry caused by virulent strains of ND virus (NDV). 1,8 ND is a global concern for the poultry sector because of the high mortality rates and trade restrictions imposed on countries that declare outbreaks. 1,8 NDV is synonymous with avian paramyxovirus serotype 1, a member of the Paramyxoviridae family and Avulavirus genus. 6 Based on their virulence, NDV isolates are divided, from least to most virulent, into asymptomatic enteric, lentogenic, mesogenic, and velogenic. 3 The intracerebral pathogenicity index (ICPI) in day-old chicks is the internationally recognized method to categorize the virulence of NDV strains: those with ICPI values ≥0.7 are considered virulent and notifiable to the international community. 10
Genetically, NDV strains are divided into 2 distinct classes: classes I and II. 7 Strains of genotype VII within class II, especially subgenotypes VIIb and VIId, have been most commonly associated with ND outbreaks in the Middle East and Asia. 2,7 A recent study showed that NDV strains of novel subgenotype VIIi have been spreading rapidly through these regions, possibly becoming more prevalent than previously circulating strains. 9 Because of high infectivity and mortality rates and the ability for rapid spread, strains of subgenotype VIIi have been regarded as the possible cause of the fifth and latest ND panzootic. 9 Similarly, in Pakistan, strains of novel subgenotype XIIIb have become more prevalent than strains of genotype XIIIa, commonly present in that region. 4,9
In this study, we describe the clinicopathologic characteristics of 1 representative strain of subgenotype VIIi (chicken/Israel/Kvuzat-Yavne/50-826/2013, referred here as Kvuzat/13 strain) and 1 representative of subgenotype XIIIb (chicken/Pakistan/Karachi/SPVC/33/2007, referred here as to Karachi/07). 9 This is the first report of detailed clinicopathologic assessment of these 2 newly described NDV subgenotypes in chickens.
NDV strains Kvuzat/13 and Karachi/07 were obtained from the repository of the Southeast Poultry Research Laboratory (SEPRL; Athens, GA, USA). The experiment was conducted in rigorous accordance with the recommendations of the “Guide for the Care and Use of Laboratory Animals” of the National Institutes of Health, as implemented by the Institutional Animal Care and Use Committee of the SEPRL (Animal Welfare Act, US Department of Agriculture, SEPRL, A4298-01; expiration date, May 31, 2018). For all experiments, eggs and chickens were obtained from the SEPRL White Leghorn specific–pathogen–free (SPF) flock, and experiments were conducted in negative-pressure isolators under biosafety level 3–enhanced (E) agriculture conditions.
The ICPI test was performed for both strains as prescribed by the World Health Organization for Animal Health. 10 Clinicopathologic assessment was carried out by inoculating groups of 4-week-old White Leghorn SPF chickens with Kvuzat/13 strain, Karachi/07 strain, or brain-heart infusion broth (negative control). Each group consisted of 10 birds, and the target virus dose was 105.5 embryo infectious dose 50% diluted in 0.1 ml of brain-heart infusion broth, half (0.05 ml) delivered in the right conjunctival sac and half (0.05 ml) in the choanal slit. After infection, birds were monitored daily. At days 2, 5, 10, and 14 postinfection (pi), 2 birds per each group were scheduled to be swabbed (oral and cloacal) and euthanized (sodium pentobarbital, 100 mg/kg, intravenous) for necropsy. Terminally ill birds were sampled regardless of the sampling schedule. After euthanasia, tissues (n = 26; eyelid, spleen, thymus, bursa of Fabricius, Harderian gland, crop, esophagus, proventriculus, duodenum and pancreas, jejunum, ileum, Meckel diverticulum, large intestine, cecal tonsils, larynx, trachea, air sac, lung, heart, kidney, liver, brain, bone marrow, nasal turbinates, comb) were fixed in 10% neutral buffered formalin and processed into paraffin for standard histopathology. Immunohistochemistry for NDV nucleoprotein (NP) was performed with a polyclonal rabbit antibody 5 (dilution, 1:8000) coupled with a polymer detection system, applied according to the manufacturer’s instruction (Novolink; Leica Biosystems, Newcastle, UK). Grading for histopathology and immunohistochemical signal was carried out as specified in Table 1. Virus titers in oral and cloacal swabs were assayed in 9- to 10-day-old embryonated SPF chicken eggs. 1
Severity of Microscopic Lesions (HE) and Distribution of Newcastle Disease Virus Nucleoprotein (IHC) in Chickens Infected with Kvuzat/13 and Karachi/07 Strains.
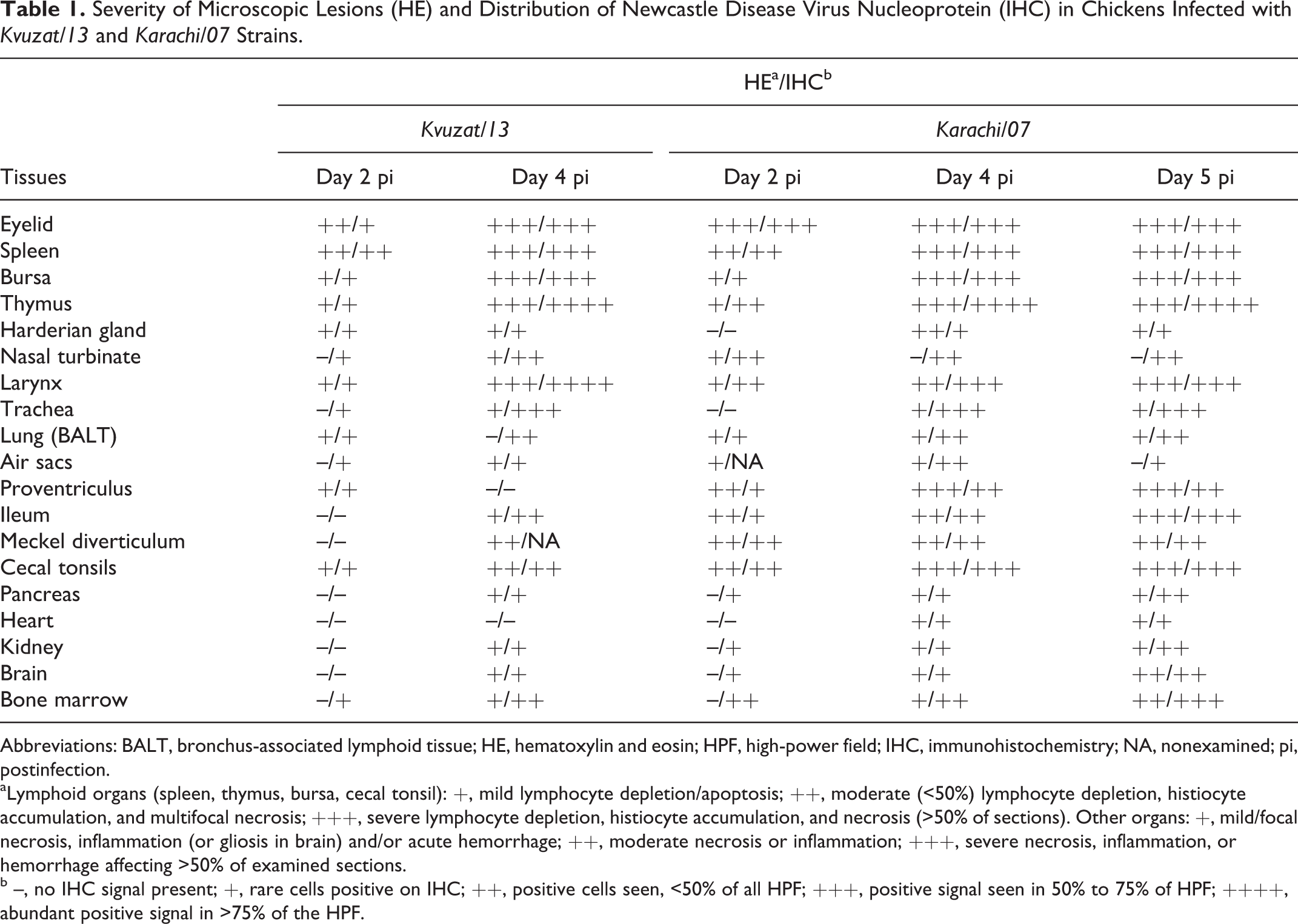
Abbreviations: BALT, bronchus-associated lymphoid tissue; HE, hematoxylin and eosin; HPF, high-power field; IHC, immunohistochemistry; NA, nonexamined; pi, postinfection.
aLymphoid organs (spleen, thymus, bursa, cecal tonsil): +, mild lymphocyte depletion/apoptosis; ++, moderate (<50%) lymphocyte depletion, histiocyte accumulation, and multifocal necrosis; +++, severe lymphocyte depletion, histiocyte accumulation, and necrosis (>50% of sections). Other organs: +, mild/focal necrosis, inflammation (or gliosis in brain) and/or acute hemorrhage; ++, moderate necrosis or inflammation; +++, severe necrosis, inflammation, or hemorrhage affecting >50% of examined sections.
b –, no IHC signal present; +, rare cells positive on IHC; ++, positive cells seen, <50% of all HPF; +++, positive signal seen in 50% to 75% of HPF; ++++, abundant positive signal in >75% of the HPF.
Pathologic Observation
The ICPI values for Kvuzat/13 and Karachi/07 strains were 1.89 and 1.85, respectively. Detailed description of clinical signs and macroscopic lesions is reported in Supplemental Table 1. All Kvuzat/13- and Karachi/07-inoculated birds developed clinical signs and were euthanized by days 4 and 5 pi, respectively. For both strains, clinical signs consisted of conjunctivitis, ruffled feathers, and diarrhea starting at day 2 pi, progressing to severe depression at days 4 and 5 pi. No abnormal clinical signs were observed in the control group. Macroscopic lesions consisted of conjunctivitis, splenomegaly associated with multifocal to coalescing foci of necrosis throughout the parenchyma (Fig. 1), severe diffuse thymic atrophy (Fig. 2), diffuse bursal atrophy, and hemorrhages scattered throughout the proventriculus and intestine (Fig. 3).
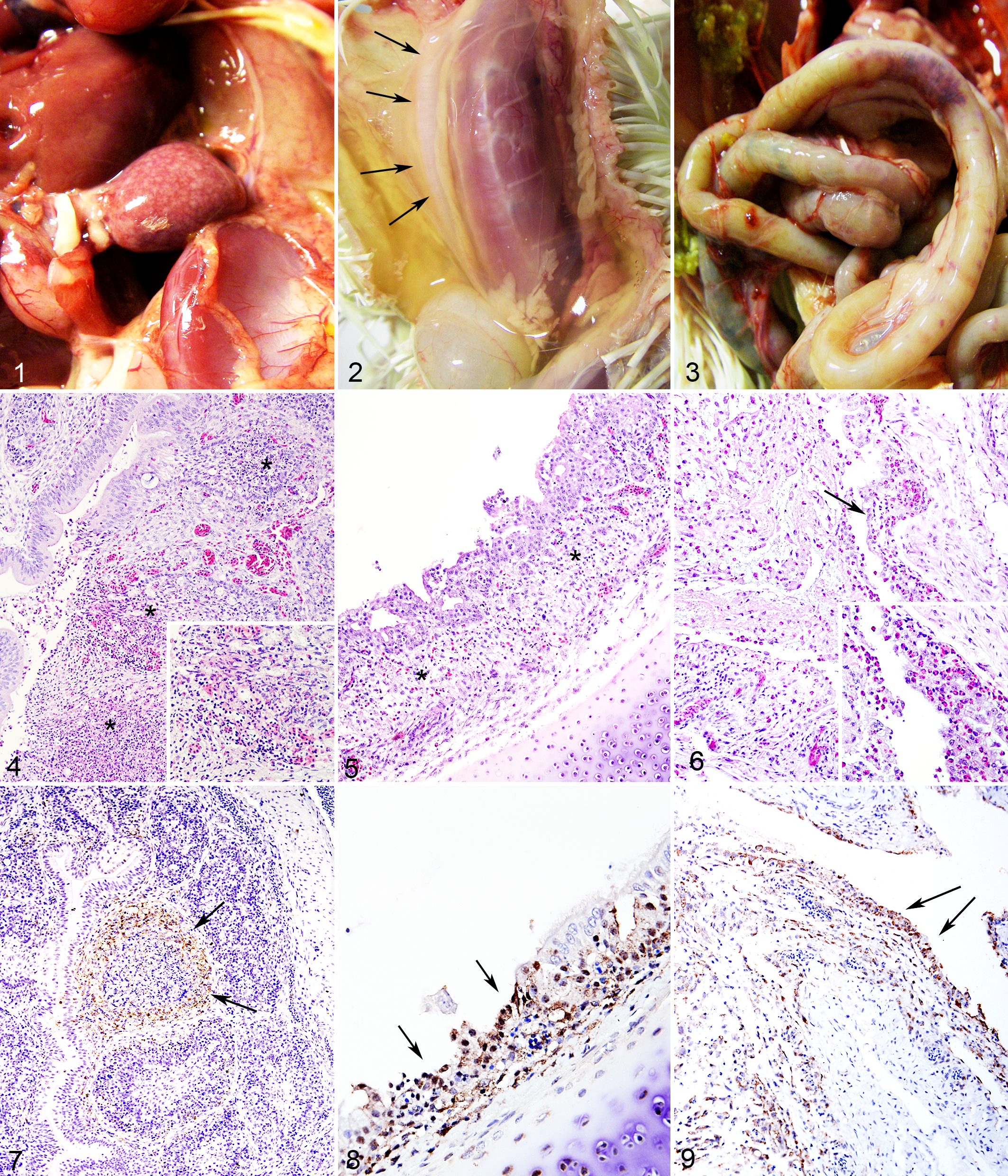
Four-week-old White Leghorn chickens infected with either Kvuzat/13 or Karachi/07 Newcastle disease virus (NDV) strain.
Detailed scoring of microscopic lesions and immunohistochemical signal in selected organs is presented in Table 1. Histologically, the type and severity of lesions caused by the 2 strains were similar. The most severe lesions were present in the lymphoid organs (eg, bursa; Fig. 4) and mucosa-associated lymphoid tissues, such as those in the conjunctiva (site of inoculum), laryngeal, and cecal tonsils. Between days 4 and 5 pi, other lesions consisted of ulceration of tracheal epithelium (Fig. 5), necrosis of bronchus-associated lymphoid tissues (BALT) throughout the lungs, multifocal heterophilic air sacculitis (Fig. 6), multifocal necrosis of the Harderian glands and reticular stroma of the bone marrow, and multifocal renal tubular necrosis. In the brain, there was multifocal moderate infiltration of lymphocytes and plasma cells in the choroid plexuses at days 4 and 5 pi. In the Karachi/07-infected birds, at day 5 pi, there was multifocal necrotizing myocarditis characterized by loss of myocardiocytes and accumulation of macrophages. There were no lesions in any of the control birds (Suppl. Figs. 1–3).
Labeling for NDV NP was mainly cytoplasmic and finely to coarsely granular, although extracellular staining was observed in areas of necrosis. Birds infected with Kvuzat/13 and Karachi/07 strains had 24 of 26 and 26 of 26 positive tissues for NDV NP at least at 1 time point, respectively. The most extensive and intense signal was observed in the eyelids, lymphoid organs (spleen, thymus, bursa [Fig. 7]), and mucosa-associated lymphoid tissues, especially cecal tonsils and laryngeal tonsils. Intense immunolabeling was also observed in the trachea (Fig. 8), BALTs surrounding bronchioles in the lungs, air sacs (Fig. 9), turbinates, renal tubular epithelial cells, pancreas, bone marrow, and scattered neurons in the brain. In Karachi/07-infected birds at day 5 pi, immunolabeling was observed within large cells (consistent with macrophages) infiltrating areas of myocardial necrosis. There was no immunohistochemical signal in any tissue from the control birds (Suppl. Figs. 4–6). Titration of oral and cloacal secretions revealed that Kvuzat/13- and Karachi/07-infected birds shed virus at high titers at days 2, 4, and 5 pi (Table 2). Highest titers were obtained in swabs from oral secretion, compared with cloacal swabs.
Virus Isolation and Titration of Oral and Cloacal Swab Samples.a
Abbreviations: C, cloacal; dpi, days postinoculation; ne, negative swabs; O, oral.
aTiters are expressed as embryo infectious dose 50% per 0.1 ml. Number in parentheses indicates the number of eggs positive by virus isolation. Dashes (—) indicate data not collected, either because no sampling was scheduled or because no birds remained in the group.
NDV strains of subgenotypes VIIi and XIIIb have been emerging recently in the Middle East and Pakistan. In particular, strains of subgenotype VIIi have been considered the cause of a possible fifth ND panzootic. 9 In the present study, one representative each of subgenotypes VIIi and XIIIb was characterized by detailed clinicopathologic assessment.
ICPI scores confirmed that both strains are highly virulent. 10 Pathogenesis experiments showed that both strains are highly pathogenic for chickens, with rapid course of the disease and death of all birds by days 4 and 5 pi. The time frame of clinical signs and the type of gross lesions observed in this study are consistent with what was observed in previous pathogenesis experiments conducted with similar methodology. 3 Histopathology and immunohistochemistry revealed that both tested strains could cause lesions and replicate in multiple organs, suggesting a very broad tissue tropism. Besides the lesions typical of ND observed in the lymphoid organs and intestine, severe necrotic lesions were also present in the air sacs, laryngeal tonsils, tracheal epithelium, BALTs, Harderian glands, and renal tubules. These lesions, especially those involving the respiratory tract, are considerably more severe than those previously reported in similar experiments. 3 The extensive immunolabeling in the air sacs, laryngeal tonsils, trachea, and, to a lesser extent, Harderian glands and nasal turbinates reflects the high magnitude of virus shedding from oral secretions. Similarly, the severe lesions and extensive virus replication observed in the intestine may account for the high levels of virus shedding in cloacal secretions. In the kidneys, multifocal necrosis and immunolabeling for NDV NP were multifocally observed in tubules, supporting the notion that virus shedding might occur through the urine.
In conclusion, the 2 NDV strains representative of novel subgenotypes VIIi and XIIIb displayed marked virulence and broad tissue tropism and were shed in large amounts from oral and cloacal secretions. These data underscore the high pathogenetic potential and replicative fitness of these strains and are in agreement with the epidemiologic evidence suggesting that these subgenotypes (especially VIIi 9 ) may spread quickly across geographic regions and display panzootic characteristics.
Footnotes
Acknowledgements
We acknowledge Tim Olivier, Dawn Williams-Coplin, and Jian Zhang for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by USDA-ARS CRIS 6040-32000-064.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
