Abstract
Lentogenic Newcastle disease viruses (NDVs), circulating among waterfowl, have the potential to become highly pathogenic by replication in chickens. The pathological studies that compare NDV infections in chickens and waterfowl are rare. The virulent 9a5b mutant NDV isolate was generated by passaging the lentogenic Goose/Alaska/415/91 NDV isolate in chickens. The pathogenesis of the virulent 9a5b mutant isolate is unknown in both chickens and waterfowl. In this study, the virulent 9a5b mutant NDV isolate was inoculated intranasally in 32-day-old specific pathogen-free white Leghorn chickens and Japanese commercial ducks. Unlike ducks, which remained clinically normal throughout the study, chickens had depression, gasping, oral discharges, and greenish-white soft feces. Gross and histologic lesion patterns as well as viral replication supported the differing clinical outcome. Ducks had slight inflammation mainly in respiratory and digestive tracts, whereas slight nonpurulent encephalitis, necrotizing pancreatitis, tubulointerstitial nephritis, and mild inflammation in respiratory and digestive tracts were detected in chickens. In agreement, interferon-beta (IFN-β)–immunopositive signals were more intense in lung tissue of ducks than that of chickens, and NDV replications were detected intensively in chicken tissues. These results suggest that the 9a5b mutant NDV isolate is more virulent in chickens, and slight histological lesions were induced in ducks even with virulent NDVs.
Newcastle disease (ND) is a highly contagious disease affecting many domestic and wild avian species, causing serious economic losses to the poultry industry worldwide. 4 ND is caused by ND virus (NDV), synonymous with avian paramyxovirus–1 (APMV-1). 2,3 NDV is an enveloped virus containing a negative-sense, single-stranded RNA genome, which encodes for 6 structural and 2 nonstructural proteins. 11,21 NDVs have 3 major pathotypes: lentogenic (low virulence), mesogenic (moderate virulence), and velogenic (high virulence). In chickens, lentogenic strains produce mild or inapparent respiratory infections. Mesogenic strains are associated with low mortality, acute respiratory disease, and neurologic signs in some birds. The velogenic strains are further divided into neurotropic velogenic NDVs (causing respiratory and neurologic signs with high mortality) and viscerotropic velogenic NDVs (causing acute lethal infections with necro-hemorrhagic lesions that are most obvious in the gastrointestinal tract). 3
The virulent 9a5b NDV mutant isolate was generated from the lentogenic waterfowl Goose/Alaska/415/91 (original) isolate by 9 consecutive passages in chicken air sacs, followed by 5 passages in chicken brain. Goose/Alaska/415/91 strain has been classified phylogenetically into class I lineage 6, which includes viruses from wild waterfowl and US live-bird markets. 37 Although the original isolate has an intracerebral pathogenicity index (ICPI) equal to 0 and an avirulent fusion (F) gene cleavage site, the 9a5b NDV isolate has an ICPI equal to 1.88 and a virulent F gene cleavage site. 32
Nonvirulent NDVs have the typical avirulent cleavage motif, 112R/G-R/ K-Q-G-R116, with a leucine (L117) at the N terminus of the F protein after cleavage (F1), which is only susceptible to trypsin-like enzymes found in limited tissues such as those in respiratory and digestive tracts, causing localized infection. On the other hand, the virulent NDVs have a virulent cleavage motif, 112R/K-R-Q-R/K-R116, and phenylalanine (F117) at the N terminus of F1, which enables them to infect the host systemically by acquiring susceptibility to furin or other ubiquitous intracellular host cell proteases. 10,12 –14,18,27,30 –32,42
Moreover, a genetic comparison between the original and 9a5b isolates demonstrated 3–amino acid substitutions in the hemagglutinin-neuraminidase (HN) proteins, changing the inactive HN0 precursor (found in avirulent viruses) to a biologically active HN protein (found in virulent viruses and is responsible for the attachment of virus particles to the host cell and promotes fusion activity of the F protein). 16,24,29,36,38
Among poultry, chickens are the most susceptible to NDV infection, whereas ducks and geese are the least susceptible. 17 Furthermore, wild birds are considered to be the natural reservoir of NDV. 43 Avirulent viruses, maintained in wild waterfowl in nature and bearing the consensus avirulent-type sequence, have the potential to become velogenic after transmission and circulation in chicken populations. 32 Therefore, investigating the differences in the pathogenesis of NDV infection between chickens and waterfowl can help us to determine why waterfowl is a suitable reservoir for these viruses and why ND outbreaks occur in chickens rather than in waterfowl.
This study was initiated to compare the clinical signs, lesions, and viral distribution in specific pathogen-free (SPF) chickens and commercial ducks infected with the mutant 9a5b NDV isolate. This isolate was chosen because (1) the original isolate was isolated from migratory waterfowl and was lentogenic; (2) mutation in its genome occurred by serial passages in chickens, which changed it to a virulent isolate; (3) and the pathogenicity index was previously determined before and after mutation. The data reported here provide, to the best of the authors’ knowledge, the first histopathological comparative study between chickens and ducks after experimental NDV infection.
Materials and Methods
Virus
The 9a5b NDV mutant isolate was obtained from the Department of Public Health, Tottori University, Japan. 32 After propagation in SPF eggs, 0.1 ml of the viral suspension, which contained 108.75 egg infective dose (EID)50, was inoculated intranasally in both chickens and ducks.
Chickens and Ducks
Twenty-three 32-day-old male white Leghorn SPF chickens and twenty-three 32-day-old male Japanese commercial ducks were obtained from the Nippon Institute for Biological Science and divided into 4 groups. The first (n = 5) and the second (n = 18) groups comprised control and infected SPF chickens, respectively. The third (n = 5) and the fourth (n = 18) groups comprised control and infected Japanese commercial ducks, respectively. All groups were reared separately in bird-bred isolators in biohazard rooms and given water and food ad libitum and monitored clinically.
After a 1-week acclimatization period, the infected groups were inoculated intranasally with the virus. Three birds from each infected group were slaughtered on the 1st, 2nd, 4th, 6th, 8th, and 10th day postinoculation (dpi). Control birds were slaughtered on the last day of the experiment (10th dpi). To reduce the pain, the birds were slaughtered under Halothane inhalation anesthesia. The ethics committee on animal experiments from Tottori University approved the experimental protocol, and the experiments were carried out in accordance with the guidelines for animal experiments in the same facility.
Necropsy Examination and Sampling
Necropsy examination was performed immediately after the birds were killed. The brain, eyelid, nostrils, larynx, trachea, lung, air sac, heart, esophagus, proventriculus, gizzard, duodenum, jejunum, ileum, cecum, colon, liver, pancreas, and kidney were collected and fixed in 10% neutral buffered formalin (NBF). Tracheal samples were taken and fixed in 2.5% glutaraldehyde for scanning electron microscopy (SEM) investigation.
Histopathology and Immunohistochemistry
After 72 hours of fixation in NBF, samples from the above-mentioned organs were dehydrated, embedded in paraffin wax, and sectioned for hematoxylin and eosin (HE) staining (3-μm-thick sections) or immunohistochemistry (IHC; 5-μm-thick sections on positively charged slides). To detect the nucleoprotein (NP) of NDV by IHC, we followed the method described in Kommers et al 19 for making the primary antibody. Briefly, a peptide antigen was used to produce antisera with the sequence TAYETADESETRRIC. This sequence represents residues 181 to 194 of the NP protein with a C addition for coupling. The peptide was coupled to keyhole limpet hemocyanin and used to immunize a rabbit by the standard procedures. The immunoglobulin G fraction was purified by affinity-column chromatography with the coupled peptide (Hokudo Bioscience, Tokyo, Japan). For IHC staining, the following protocol was used: after deparaffinization, tissue sections were treated with 3% H2O2 for 15 minutes at room temperature (RT) for endogenous peroxidase inactivation and then subjected to antigen retrieval by microwaving for 10 minutes at full power in citrate buffer (pH 5.4), followed by blocking with 10% normal goat serum (NGS) for 5 minutes in the microwave. Tissues were incubated with the primary antibody (X8000) overnight at 4°C. After stringent washing, sections were incubated with a labeled polymer (ChemMate Dako EnVision/HRP [DAP]; Dako, Carpinteria, CA) for 30 minutes at RT and then subjected to stringent washing again. The substrate was diaminobenzidine (DAB; Dako), and the sections were stained with hematoxylin and covered with DPX mounting media (Sigma Life Science, Steinheim, Germany).
To detect interferon-beta (IFN-β) in lung tissues by IHC, we followed the above-mentioned protocol with the following modification: microwaving antigen retrieval was done for 20 minutes before H2O2 treatment, primary antibody (X400) was polyclonal rabbit anti-human IFN Beta (AbD Serotec, Kidlington, UK), and a weak wash was applied with this antibody.
Scanning Electron Microscopy
Tracheal samples were processed for SEM by the standard method. Briefly, tissue specimens were fixed in 2.5% glutaraldehyde in 0.1M phosphate buffer (PB; pH 7.3) at 4°C overnight, washed in PB, postfixed in 1% osmium tetraoxide (pH 7.4) for 1 hour at RT, washed in PB, and dehydrated in increasing concentrations of ethanol and finally in t-butyl alcohol. Samples were glued with carbon cement on aluminum stubs and coated with silver in a vacuum evaporator. Observation was made using an SEM (Hitachi Co. Ltd., Tokyo, Japan).
Results
Clinical Signs
Clinical signs, which appeared only in infected chickens, started from the third dpi and included a sleeping appearance and greenish-white soft feces, and they peaked at the fourth and the fifth dpi, with symptoms such as depression, gasping, and oral discharges. All of these symptoms decreased from the sixth dpi, and the chickens showed only slight depression on the last day of the experiment. No clinical signs were detected in the infected duck group or in any of the 2 control groups. No deaths were recorded in this experiment.
Gross Lesions
The infected chickens had mild congestion and hemorrhages in the nasal mucosa and lung, besides swelling of the 2 ceca. The gallbladder was swollen and engorged with bile, and the content of the digestive tracts was stained with the bile pigment. The air sacs were slightly thick and opaque. In addition, small hemorrhages were detected in the myocardium and cecal tonsils. Furthermore, the pancreatic white necrotic foci were the most prominent gross change by the 10th dpi. On the other hand, the infected ducks basically had no gross lesions, but distended ceca and engorged gallbladder were observed. In addition, the content of the digestive tracts was stained with bile pigment. No gross lesions were detected in control chickens and ducks.
Histopathology and Immunohistochemistry
Histopathological and immunohistochemical findings are summarized in Tables 1, 2, and 3. Differences between chickens and ducks infected with the 9a5b NDV isolate are described below.
Histopathological Findings in Chickens and Ducks Infected With the Mutant 9a5b Newcastle Disease Virus (NDV) Isolate

aPx–Pz means the peak period of histological findings, and P means the postinfection day.
Immunolabelingsa for Newcastle Disease Viral Nucleoprotein Antigen by Immunohistochemistry in Both Chickens and Ducks
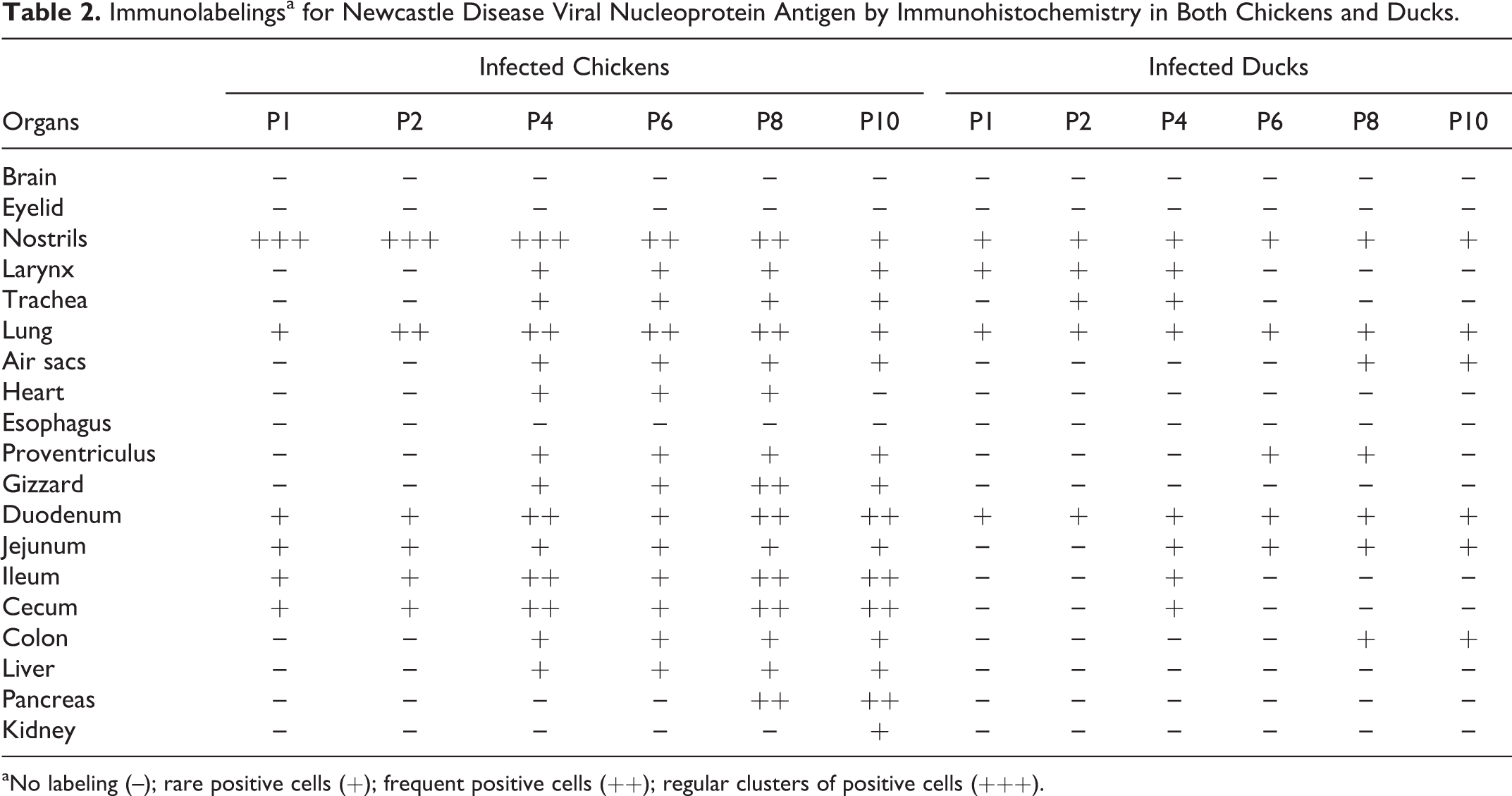
aNo labeling (–); rare positive cells (+); frequent positive cells (++); regular clusters of positive cells (+++).
Immunohistochemical Demonstration of Interferon-β in Lung Tissue at Early Stage After Infection With 9a5b Newcastle Disease Virus Isolate in Chickens and Ducksa

aNo staining (–); minimal (±); mild (+); moderate clusters of positive cells (++).
Brain
Brain lesions were observed in the infected chickens only. Slight nonsuppurative lymphocytic encephalitis was detected, and it was characterized by focal areas of microgliosis and lymphocytic perivascular cuffing in the cerebrum, cerebellum, and medulla oblongata. In addition, vacuolation and demyelination in the cerebellar white matter, as well as mild loss of Purkinje cells, were also observed. In contrast, the NDV-NP antigen could not be detected in the brain by IHC.
Conjunctiva
Although lymphocytic conjunctivitis with heterophilic infiltration was mild in chickens and slight in ducks, the NDV-NP antigen could not be detected in both.
Respiratory system
The infected chickens had moderate lymphocytic rhinitis with slight hemorrhages, deciliation, and degeneration and necrosis of epithelial cells (Fig. 1a) with associated NDV-NP–immunopositive signals in the epithelial cells and mononuclear cells at the same site (Fig. 1b). On the other hand, slight lymphocytic rhinitis with proliferation of goblet cells and heterophilic infiltration were observed in the infected ducks (Fig. 2a), and the NDV-NP–immunopositive signals detected in the epithelial cells and mononuclear cells (Fig. 2b) were less intensive than those of chickens. Moderate lymphocytic laryngitis and mild tracheitis with loss of cilia and erosion were detected in the infected chickens, whereas laryngitis and tracheitis in the infected ducks were milder than those of chickens, and hyperplasia of goblet cells and infiltration of heterophils, lymphocytes, and macrophage-like cells were prominent in laryngeal and tracheal mucosa.
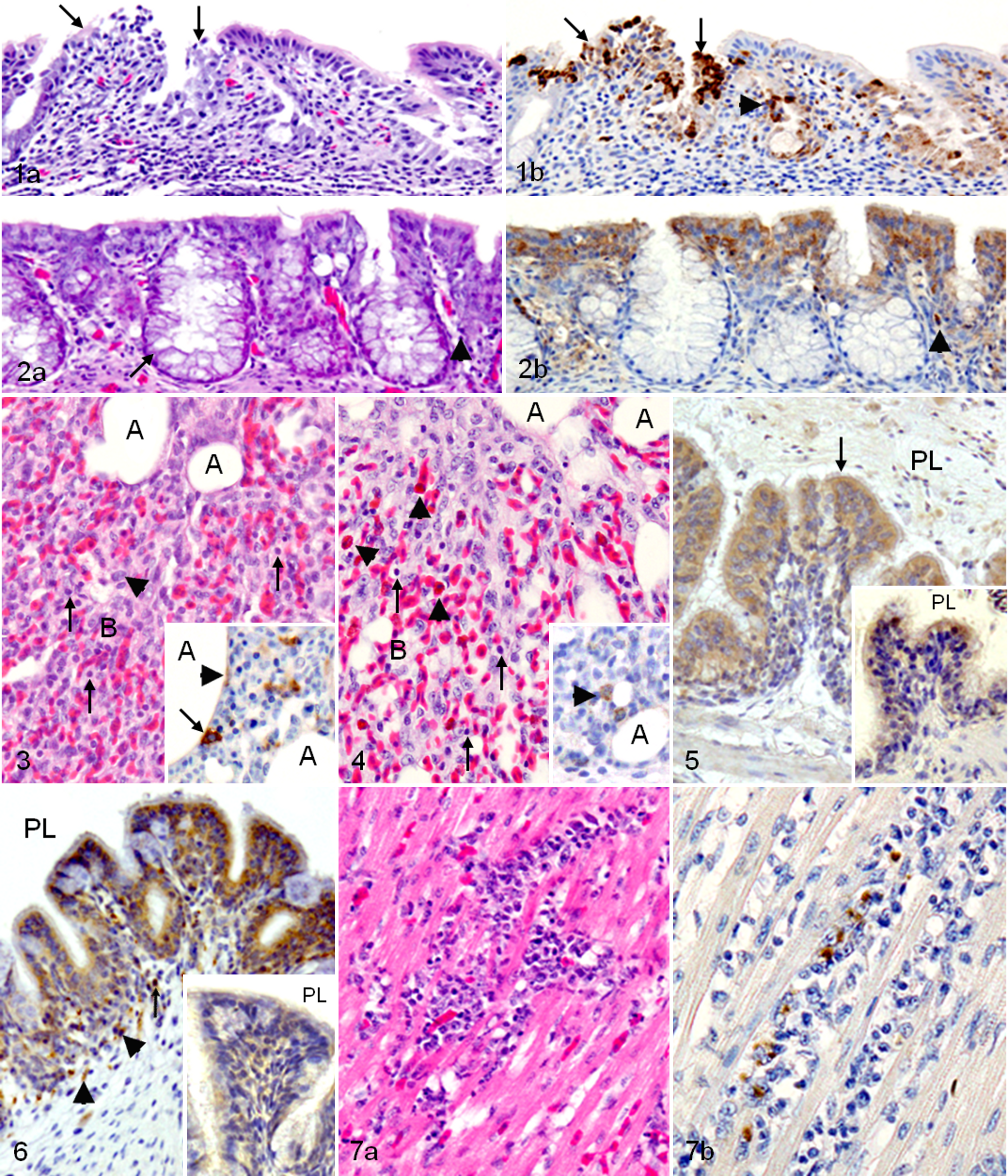
The infected chickens had moderate lymphocytic interstitial pneumonia, which was characterized by slight hemorrhages, proliferation of periparabronchial lymphoid tissues, and infiltration of macrophages, lymphoid cells, and few heterophils (Fig. 3) with associated NDV-NP antigen in macrophage-like cells and epithelial cells (inset of Fig. 3). Furthermore, moderate airsacculitis was developed at the late stage of the experiment. On the other hand, mild lymphocytic interstitial pneumonia, which was characterized by heterophilic infiltration and proliferation of periparabronchial and air passages submucosal lymphoid tissues, was developed in the infected ducks (Fig. 4). NDV-NP–positive signals were detected in macrophage-like cells and epithelial cells (inset of Fig. 4). In addition, slight airsacculitis was developed at the late stage of the infection.
Weak IFN-β–immunopositive signals were detected in the lung tissues of the infected chickens, especially in the epithelial cells of the parabronchi (Fig. 5). On the other hand, IFN-β–immunopositive staining in the infected ducks was detected in cells morphologically consistent with fibroblasts and macrophages in lung tissues, especially in the air passages subepithelial tissues and in the epithelial cells of the parabronchi (Fig. 6), and it was more intensive than those of chickens, as described in Table 3.
Heart
The heart lesion, which developed in the infected chickens only, was mild and appeared at the first dpi as scattered areas of lymphocytic myocarditis, and then degeneration of myofibers, slight hemorrhages, and heterophilic infiltration were detected later (Fig. 7a) with associated NDV-NP–immunopositive macrophages and lymphocytes in the myocardium (Fig. 7b).
Digestive tract
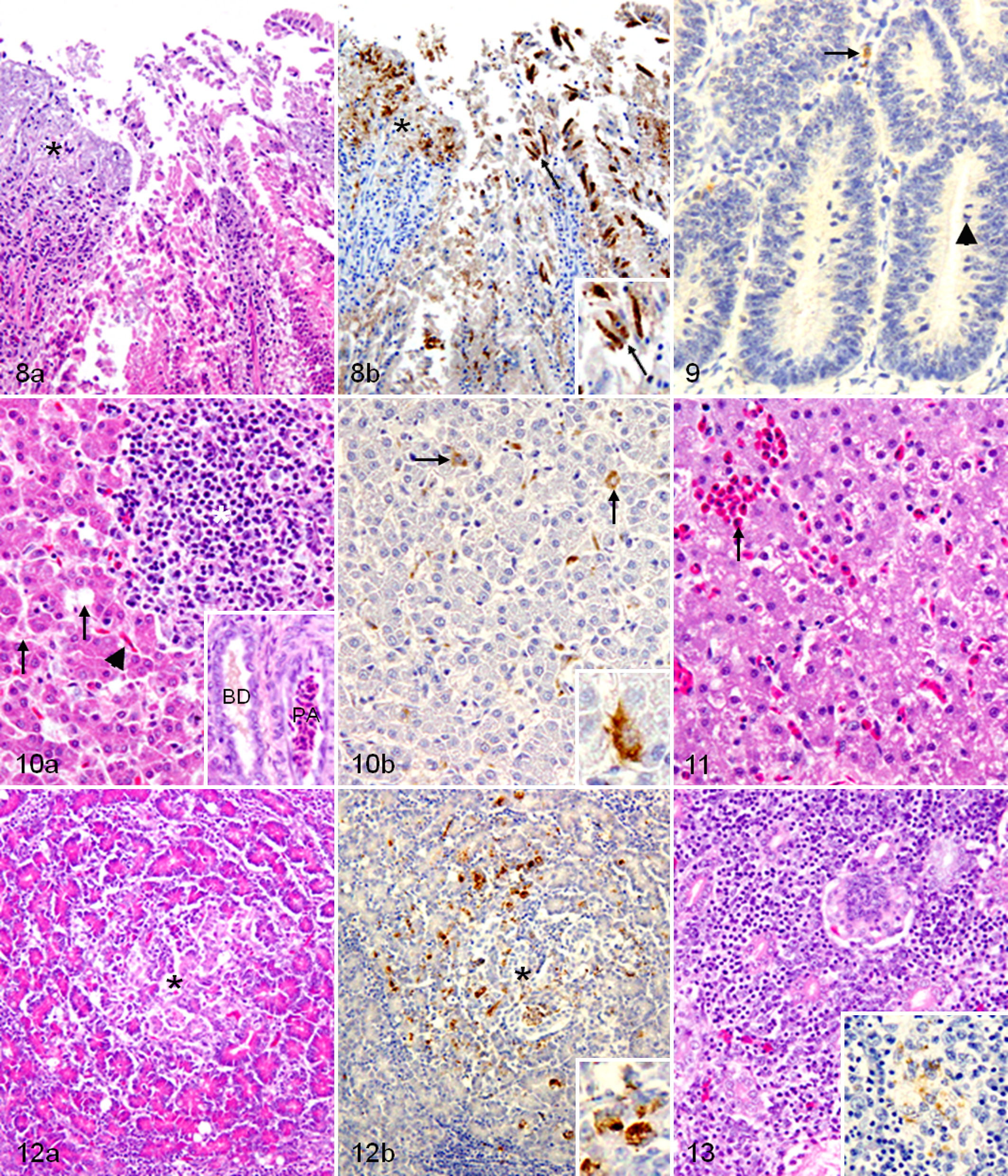
The infected chickens had slight esophagitis and mild hemorrhagic proventriculitis with infiltration of lymphocytes, macrophages, and heterophils in the mucosa and lamina propria. In addition, infiltration of macrophages and lymphocytes in the mucosa and tunic muscularis of the gizzard, mild duodenitis with scattered areas of degeneration, and necrosis in the duodenal mucosa (Fig. 8a) were detected. Furthermore, mild jejunitis, proliferation of lymphoid and goblet cells in the ileum mucosa, proliferation of cecal tonsils with slight hemorrhages, and proliferation of lymphoid tissues of the colon lamina propria were also found. NDV-NP–immunopositive signals were detected in macrophages and epithelial cells in many parts of the digestive tract (Table 2) and, as described in Fig. 8b, in the necrotic area and epithelial cells of the duodenal mucosa. On the other hand, in the infected ducks, the mitotic figures were increased in the epithelial cells of the mucosal gland throughout the digestive tract (Fig. 9). In addition, hyperplasia of goblet cells, proliferation of lymphoid cells, and heterophilic infiltration in the mucosa and submucosa of the digestive tract were observed. NDV-NP–immunopositive signals in the digestive tract of the infected ducks were less frequent and fainter than those of chickens, as described in Table 2 and as shown in macrophage-like cells between duodenal glands (Fig. 9).
Liver
The infected chickens had proliferated ectopic lymphoid tissues, and the bile canaliculi were dilated and engorged with bile (Fig. 10a) with associated NDV-NP–immunopositive macrophages and Kupffer cells (Fig. 10b). In turn, in the infected ducks, mild proliferation of ectopic lymphoid tissues and moderate heterophilic migration in hepatic sinusoids were observed (Fig. 11).
Pancreas
Only in the infected chickens was the pancreatic lesion developed. Slight necrotizing pancreatitis was observed at the first dpi and dramatically increased at the sixth dpi and did not regress until the end of the experiment (Fig. 12a), with associated NDV-NP–immunopositive signals in necrotic pancreatic acinar cells (Fig. 12b).
Kidney
Proliferations of the ectopic lymphoid tissues were observed in the infected chickens at the second dpi, whereas tubulointerstitial nephritis was detected by the sixth dpi and increased in severity until the end of the experiment (Fig. 13), with associated NDV-NP–immunopositive staining in the degenerated renal tubules (inset of Fig. 13). On the other hand, renal tissues of the infected ducks had slight proliferation of ectopic lymphoid tissue.
The histological features in both control chickens and ducks were within the normal limits, and no NDV-NP–immunopositive signals were detected in these groups.
Tracheal Ultrastructural Changes
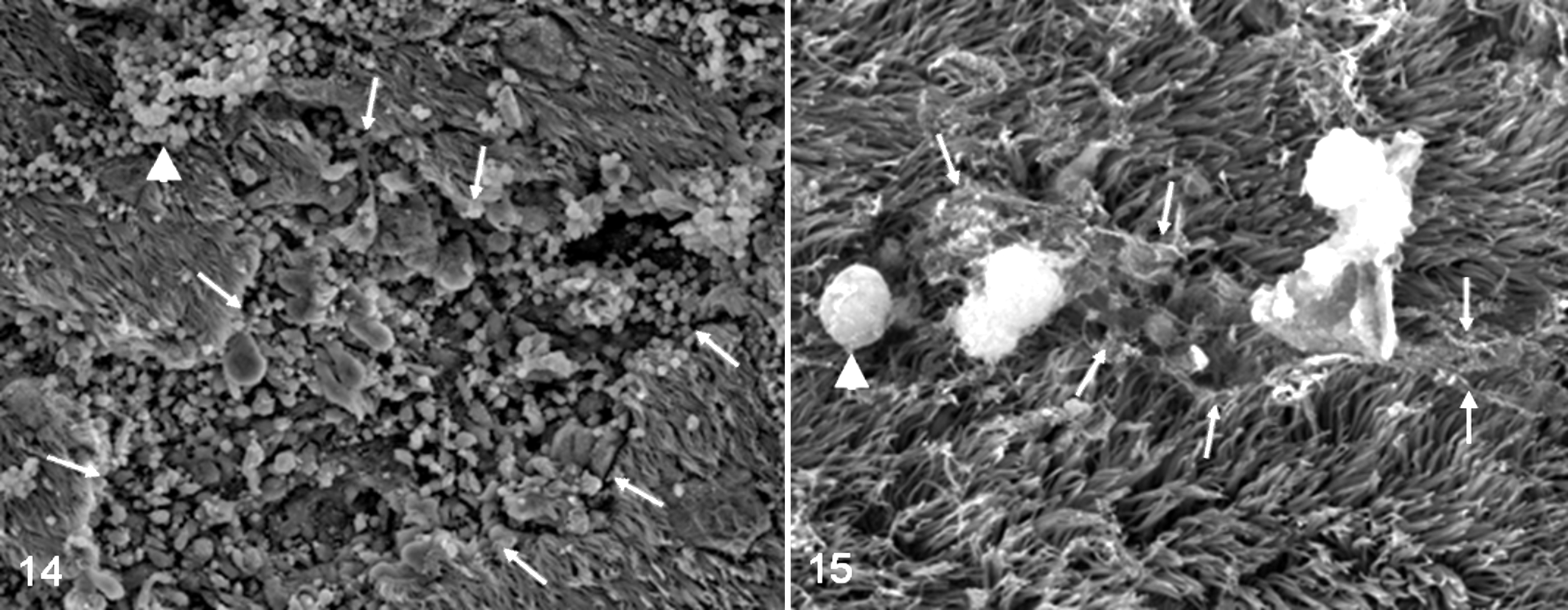
Infected chickens had excessive secretion of globular mucous particles on the epithelial surface at the first dpi. By the fourth dpi, a marked increase in the proportion of nonciliated to ciliated epithelium was observed, and disorientation of the cilia as well as adherence of the cilia to each other and to the underlying surface was seen. Deciliation of large parts of the tracheal surface and erosions in the mucosal epithelial cells were also detected (Fig. 14). In addition, scanty mucous was found on the tracheal surface by the 10th dpi. In turn, the infected ducks had mild globular mucous particles, slight disorientation of the cilia, and a fine mucous network on the cilial tips (Fig. 15), and blankets of mucous were covering the underlying epithelium. Ultrastructural features of tracheal mucosa in both control chickens and ducks were within normal limits.
Discussion
In this study, the 9a5b NDV isolate (which has a virulent F gene cleavage site and a high ICPI of 1.88) 32 induced clinical signs in the infected chickens only, and they were mild. In agreement, the chicken’s lesions were more severe than the ducks’ lesions, and the distribution and intensity of the NDV-NP–positive signals were greater in chickens than in ducks. In our previous study on the immune organs from the same experiment, the results showed that the replication and dissemination of the 9a5b NDV isolate were wider in chickens than in those of ducks and that the spleen was the most affected organ. 6
NDV virulence groups (lentogen, mesogen, and velogen) are very broad categories, and the degree of clinical disease does not always segregate with ICPI. There are cases, for example, in which a strain is considered “virulent” by ICPI but does not cause much in the way of clinical disease. 35,39,40 In addition, the pathogenic effects of NDV are not dependent on the fusion cleavage site alone. 39 As described previously, NDV isolates that had a virulent fusion protein cleavage site and high ICPI differed in their ability to cause clinical signs, in their lesions, and in their viral distribution in SPF chickens. 28,35 Moreover, chickens and ducks vary in their response to virulent NDV infection. 1
In the present study, the respiratory and digestive lesions started at an early stage (except airsacculitis) and subsided at a late stage in both chickens and ducks. On the other hand, necrotizing pancreatitis and tubulointerstitial nephritis were detected only in chickens; they dramatically increased at the late stage and did not subside until the 10th dpi. We are unaware of what the sequelae of these lesions might have been if the birds were studied for a longer period. Moreover, mild lymphocytic myocarditis and slight nonpurulent encephalitis were detected only in chickens, and these lesions were described previously. 19,20 Immunohistochemical NDV-NP–positive signals were detected in infected chickens at a late stage in the degenerated pancreatic acinar cells and degenerated renal tubules, at the middle of the experimental period in the myocardium, but not in the brain, indicating no or too low virus replication in the brain tissue to be detected by IHC in addition to the direct effect of NDV on the pancreatic acinar cells, renal epithelial cells, and myocardial fibers. This finding is consistent with a previous observation in SPF chickens infected with NDV isolates of different virulence. 8
In our previous study, the pattern of apoptosis in the spleen was distinct between chickens and ducks after 9a5b NDV infection. Although apoptotic cells were abundant in the periellipsoidal white pulp, periellipsoidal lymphoid sheath, periarteriolar lymphoid sheath, and perivenous lymphoid sheath (PVLS) of infected chickens, apoptosis was concentrated in the germinal centers (GCs) of the infected ducks. 6
Ducks in the present study had no lesions in the brain, heart, pancreas, or kidney; the NP-positive signals were not detected in these organs either. These findings indicate wider dissemination of the 9a5b isolate in tissues of chicken than in those of duck. Chickens, turkeys, and ducks vary in their response to virulent NDV infection. Although asymptomatic infection occurred in ducks and turkeys even at the highest dose (106 EID50) without any deaths noted, chickens were shown to be extremely susceptible, and all died even with a low dose (101.5 EID50). 1 In line with that, in SPF chickens, the mesogenic Anhinga NDV isolate induced eyelid edema, multifocal myofiber disruption with infiltrates of lymphocytes, and macrophages in the heart. The cerebellum had multifocal gliosis with Purkinje cell loss and epithelial necrosis in the comb as well as NDV-NP–positive signals in some organs, including brain. 20 Chickens are the most susceptible to NDV infection among poultry species, whereas ducks and geese are the least susceptible; 17 for this reason, wild birds are considered to be the natural reservoir of NDV. 43 Unfortunately, pathological studies of Newcastle disease in ducks are rare, so it is difficult to compare our results with previous data.
IFN-β appears to be protective during the early stage of influenza infection and cannot be compensated by IFN-α, and therefore superior innate immunity might protect the duck during this critical period. 7 Retinoic acid–inducible gene-like receptors (RLRs), including retinoic acid–inducible gene I (RIG-I) and melanoma differentiation–associated gene 5 (Mda-5), are cytoplasmic RNA sensors 41 that can detect influenza virus 25 and NDV, 33 leading to production of IFN-β and expression of downstream IFN-stimulated antiviral genes. 25 RLRs play central roles in viral recognition and subsequent induction of antiviral immune responses in conventional dendritic cells (cDCs), macrophages, and fibroblasts. 22,34 RIG-I is present in ducks and absent in chickens, 7 whereas the V proteins of paramyxoviruses directly interact with Mda-5 to block RLR signaling, 5 and this indicates that chickens after NDV infection cannot produce IFN-β through the RLR pathway, which gives the duck an advantage in this point. 5,7,25,33,41 In this study and in our previous study, 6 immunohistochemical IFN-β expression in lung tissues and in immune organs, respectively, was earlier and stronger in ducks compared with chickens after infection with the 9a5b NDV isolate. Therefore, ducks may have rapid and strong innate immunity against NDV infection.
In our study, histopathological lesions were more severe in infected chickens. On the other hand, heterophilic infiltration, proliferation of lymphoid tissues, and hyperplasia of goblet cells were more often observed in the infected ducks. Avian heterophils are highly phagocytic and capable of a broad spectrum of antimicrobial activity, and they form the first line of cellular defense against invading microbial pathogens in the lungs and air sacs, where resident macrophages are typically lacking. 9,15 In this study, SEM investigation of tracheal tissues revealed that 9a5b NDV infection induced rapid and abundant mucous secretion at the 1st dpi in chickens, but scanty mucous was observed at the 10th dpi, possibly indicating severe exhaustion of goblet cells, which is in line with previous studies. 23,26 In infected ducks, the mucous secretion was mildly increased from the 1st to the 10th dpi. Moreover, deciliation and mucosal destruction were greater in chickens than in ducks. Therefore, in field conditions, secondary infection may occur more easily in chickens than in ducks.
As described previously, chicken eyelid inoculation with 3 virulent NDV isolates did not cause any lesions at the first dpi. 35 In contrast, in the present study, at the first dpi, the lesions in the infected chickens were not restricted to the respiratory and digestive tracts. A few localized areas of lymphocytic myocarditis and pancreatitis were detected even though NDV-NP could not be detected by IHC in the heart and pancreas on that day (Table 2). Therefore, the early detectable lesions in our experiment may depend on the route of inoculation as well as virulence differences. Further studies to detect the virus by in situ hybridization or viral isolation and to explain the development of early lesions in the heart and pancreas are needed.
Finally, the 9a5b mutant isolate induced a mild infection in SPF chickens and asymptomatic infection in Japanese commercial ducks. Accordingly, the lesions and virus dissemination were greater in chickens than in ducks. In contrast, IFN-β immunopositivity was more intensive in lung tissue of ducks, which may indicate stronger duck innate immunity against NDV infection. Further studies to compare chickens and waterfowl are needed, which may help us to elucidate the natural mechanisms underlying waterfowl resistance to NDV infection.
Footnotes
Acknowledgements
We appreciate the financial support rendered by the Egyptian government to the first author (Anis Zaid). We are grateful to all members of the Department of Veterinary Pathology and to Ms E. Kawahara, Tottori University, for assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Egyptian government and the Laboratory of Veterinary Pathology, Tottori University, Japan.
