Abstract
In felines, ocular and nonocular melanomas are uncommon tumors that represent a diagnostic challenge for pathologists, especially when amelanotic. To date, the immunohistochemical diagnostic panel in cats is based on specific melanocytic markers (Melan-A and PNL2) and a nonspecific but sensitive marker (S100). In human medicine, SOX-10 is reported to be a sensitive antibody for the detection of melanoma micrometastasis in the lymph node. TRP-1, an enzyme involved in melanogenesis, has recently been used in humans and dogs as a specific melanocyte marker. The aim of this study was to evaluate the cross-reactivity and the expression of SOX-10 and TRP-1 antibodies in feline normal tissue and melanocytic tumors. Thirty-one cases of ocular, cutaneous, and oral melanomas were retrospectively evaluated and confirmed by histopathological examination and by immunolabeling with Melan-A and/or PNL2. SOX-10 nuclear expression in normal tissues was localized in epidermal, subepidermal, hair bulb, and iridal stromal melanocytes and dermal nerves. In melanomas, nuclear expression of SOX-10 was detected in ocular (11/12; 92%), oral (6/7; 86%), and cutaneous sites (12/12; 100%). TRP-1 cytoplasmic immunolabeling in normal tissue was observed in epidermal and bulbar melanocytes and in the lining pigmented epithelium of the iris and in its stroma. Its expression was positively correlated to the degree of pigmentation in the tumor and was observed in 75% of ocular (9/12), 43% of oral (3/7), and 33% of cutaneous melanomas (4/12). This study demonstrated the cross-reactivity of SOX-10 and TRP-1 antibodies in feline non-neoplastic melanocytes and their expression in ocular and nonocular melanomas.
In felines, melanocytic tumors are considered an uncommon neoplasm, when compared with dogs and humans. The eye is the most common site of origin, particularly in the iris, where the tumor usually shows a diffuse growth pattern (feline diffuse iris melanoma), followed by skin and oral mucosa. 12 Melanoma is derived from melanocytes that originate from neuroectodermal crest and maintain the ability to show extreme morphological variability. 21 In cases of complete melanin absence, melanoma represents a diagnostic challenge for pathologists due to the overlapping of histopathological features with other tumors, particularly soft tissue sarcomas. 18
Melan-A, PNL2, and S100 are well-known and the most commonly used immunohistochemical markers used to diagnose melanoma, but their sensitivity and specificity are still suboptimal in both feline and canine species.18,20
Sry-related HMg-box gene 10 (SOX-10) is a transcription factor that plays an important role in neural crest differentiation and the development of embryonal tissues. This protein is expressed in neuroectodermal cells, such as normal and neoplastic melanocytes and Schwann cells, but can be also variably detected in myoepithelium, ganglia, and mammary epithelium.10,11 In dogs, SOX-10 has been used to differentiate peripheral nerve sheath tumors from those with a perivascular wall origin. 19 In a recent study, SOX-10 was evaluated by immunohistochemistry and molecular analysis (quantitative real-time polymerase chain reaction [PCR]) to discriminate between canine oral melanoma and soft tissue sarcoma. SOX-10 showed a 100% sensitivity with both techniques and 90% and 85% specificity with immunohistochemistry and quantitative real-time PCR, respectively, for the detection of melanocytic neoplasms. 23 In human medicine, SOX-10 is mainly used as an immunohistochemical marker to detect melanoma micrometastases in lymph nodes due to its high sensitivity. Notably, the nuclear expression of SOX-10 facilitates its identification within tissues and avoids confusion with melanophages.7,26
Tyrosinase-related protein 1 (TRP-1) is an enzyme contained within melanosomes involved in the melanin biosynthetic pathway and is recognized as one of the most abundant proteins in normal and neoplastic melanocytes. 24 Despite its specificity for the melanocytic line, it can be occasionally detected in mammary and prostatic tumors. 1 In human medicine, TRP-1 expression in melanomas is significantly associated with disease-free survival time. 8 Its expression has also been demonstrated in canine melanoma where it showed a high specificity, while being less sensitive than PNL2 and Melan-A. 20
Currently, there are no data available on the expression of SOX-10 and TRP-1 in the feline species, thus the aims of this study were to validate the expression of SOX-10 and TRP-1 in both feline normal and neoplastic tissue from skin, oral mucosa, and eyes and to explore whether their inclusion in a wider immunohistochemistry panel could increase the diagnostic sensitivity for the detection of melanocytic tumors in cats.
Materials and Methods
Case Selection
Feline cases with a histological diagnosis of melanoma were retrospectively selected from the database of the Department of Veterinary Medicine (Pathology section) of the University of Perugia (Italy), in a period between 2005 and 2018; moreover, biopsies in which a malignant melanocytic tumor was listed in the differential diagnosis were included. To be included in the study, each case had to meet the following criteria: (1) presence of malignancy features (ie, loss of pigmentation, cellular atypia, and mitotic count); (2) absence of artifacts (ie, incomplete fixation or electrosurgical polarization of cells); (3) presence of at least 1 cm of neoplastic surface on cut section; and (4) immunohistochemical positivity of neoplastic cells for either Melan-A or PNL2. Normal control tissues were from haired skin, oral mucosa, and eye, from 5 domestic shorthair cats undergoing necropsy for causes unrelated to cutaneous, oral, and ocular disease (ie, trauma). Sampling was performed after obtaining owner’s consent.
Histology
For each selected case of melanoma, we evaluated:
predominant histotype (epithelioid, spindle, balloon, signet ring); in cases with presence of 2 or more histotypes, neoplasms were classified as mixed;
degree of pigmentation (amelanotic = 0, mild = 1, moderate = 2, marked = 3), considered as the quantity of melanin pigment present in the cytoplasm of neoplastic cells, independently from the percentage of pigmented cells;
extension of pigmentation calculated as percentage of neoplastic cells that contained melanin pigment (0, <50%, >50%);
nuclear atypia (quantified on a 1%–100% scale), as previously described for canine melanocytic neoplasms; 22
cellular pleomorphism (mild, when <10% atypical cells = 1; moderate, between 10% and 50% atypical cells = 2; severe, when >50% atypical cells = 3) evaluated on the total neoplastic population;
mitotic count; assessed in ten 400x fields with an FN 22 ocular (2.37 mm2 area), counting positive cells in “hot spots”; 9
percent of intratumoral necrosis;
presence/absence of junctional activity, ulceration, and intravascular invasion.
Heavily pigmented tumors were bleached as previously described. 14
Normal samples were examined on hematoxylin and eosin-stained sections to assess the absence of inflammatory and/or neoplastic lesions in order to include the sample as a control in the study.
Antibodies Selection
Considering that specific antibodies generated to cross-react with feline tissues are not commercially available, it was necessary to select SOX-10 and TRP-1 antibodies from the available products designed for other species (ie, humans, mice, dogs). The selection was based on the result of an in silico analysis, which was obtained from the alignment of the reported amino acid sequences of the human (the target species of the antibodies used in this study) and feline proteins (www.uniprot.org). In particular, the residues used as targets to design the antibodies were taken in consideration. Moreover, the expression of SOX-10 and TRP-1 in normal skin and oral mucosa was compared to what is reported for the same antibodies in canine tissues.
Immunohistochemistry
Five-micrometer sections from each formalin-fixed paraffin-embedded block were cut and mounted on poly-
Panel of primary antibodies applied on healthy skin and melanomas of cats.

Abbreviations: mAb, monoclonal antibody; pAb, polyclonal antibody.
The immunolabeling evaluation in melanomas for each antibody was based on presence/absence, intensity of the signal (absent = 0, mild = 1, moderate = 2, marked = 3), and percentage of positive cells on the total tumor area. The expected signal was nuclear for SOX-10 antibody and cytoplasmic for TRP-1. In normal tissues, the presence of positive cells was assessed throughout the tissue.
Statistical Analysis
As a consequence of the small number of cases and the nonuniform distribution of data (graphic tests), nonparametric tests were performed to verify the experimental hypotheses, such as Kruskal–Wallis test and Spearman correlation coefficient.
For the statistical analysis, the histological and immunohistochemical data were analyzed by IBM SPSS statistics version 20 software (Statistical Package for Social Science, New York, USA). The relationships between the following variables were investigated: tumor location, percentage of pigmentation, histotype, mitotic count, nuclear atypia and pleomorphism, and intensity and percentage of positive neoplastic cells. A P value ≤.05 was considered significant.
Results
Population Study
Thirty-one cases matched the inclusion criteria, and a melanocytic origin was confirmed by immunoreactivity for Melan-A and/or PNL2. Some of the cases presented in this publication are part of a previously published case series. 15 For each case, a hematoxylin and eosin-stained section and formalin-fixed paraffin-embedded tissue for further immunohistochemical investigation were available. Therefore, the definitive case series of this study was represented by 31 melanomas with ocular (12/31, 39%), oral/perioral (7/31, 23%) and cutaneous (12/31, 39%) origins.
At the time of diagnosis, the median age of the population was 10.6 years (interquartile range, 10–13.25 years), ranging between 2 and 17 years, with most of the cats being 10 years of age or older. In 3 cases, the age was not recorded among anamnestic data. Of the 31 cats, 15 (83%) were domestic shorthair and 3 were (17%) Persian. The breed was unknown in 13 cases. Overall, there were 13 males (42%) and 15 females (48%); in 3 cases, the sex was not specified. Data regarding the signalment and specific tumor location for each case are reported in Supplemental Table S1. No associations between the age, sex, and breed were found.
Antibody Selection and Immunoreactivity of Normal Tissue
The protein BLAST analysis for SOX-10 revealed a 76.6% identity between the whole human and feline protein sequences. According to the datasheet of the antibody used in this study, the antibody is specific for a human epitope mapped between amino acids 2 and 29 at the N-terminus of SOX-10, which showed 100% identity with the feline amino acid sequence (Table 2). For TRP-1, the overall identity between feline and human proteins was 88.6%. The antibody used in this study was developed with a synthetic peptide located between the amino acids 396 and 445 of the human TPR-1 protein (P17643, NP_000541); this region showed an identity of 97.9% compared with the feline amino acid sequence (Table 2).
Amino acid sequences alignment of SOX-10 and TRP-1 between human (Homo sapiens) and domestic cat (Felis catus).
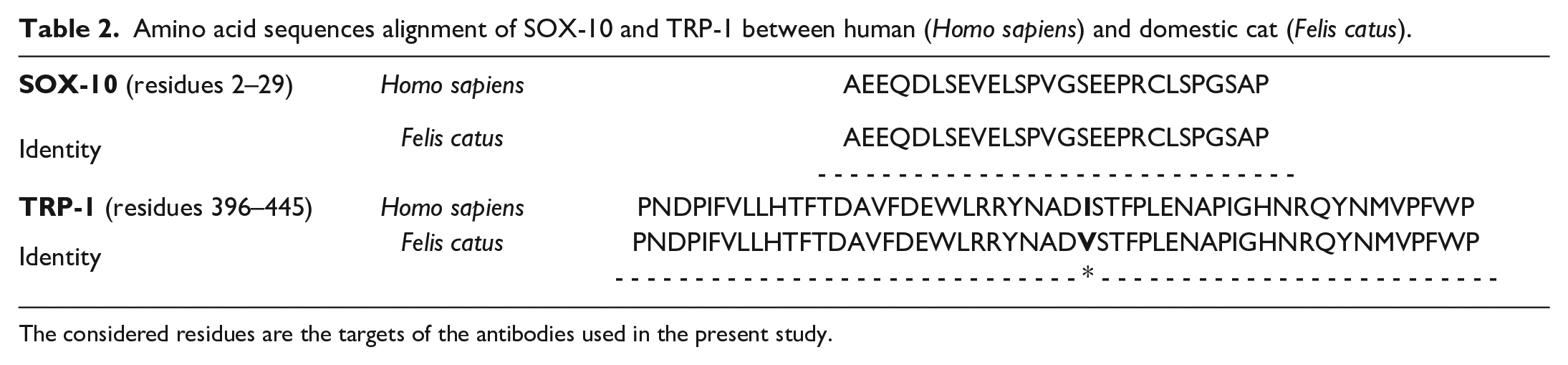
The considered residues are the targets of the antibodies used in the present study.
SOX-10 immunolabeled in the nucleus of intraepidermal, rare dermal (subepidermal), and hair bulb melanocytes in the different samples from haired skin (Fig. 1a, b). Similarly, immunolabeled intraepithelial cells were occasionally seen in some samples of oral mucosa. The distribution of these cells seemed to be uneven, usually in association with areas of macroscopic and histological pigmentation of the mucosal epithelium. Nerve structures present in the dermis and in the submucosa also showed cytoplasmic immunolabeling. In the iris, numerous immunolabeled nuclei of melanocytes, characterized by a moderate cytoplasmic pigmentation and residing in the stroma, were observed (Fig. 1c). Nonspecific immunolabeling/background was absent.
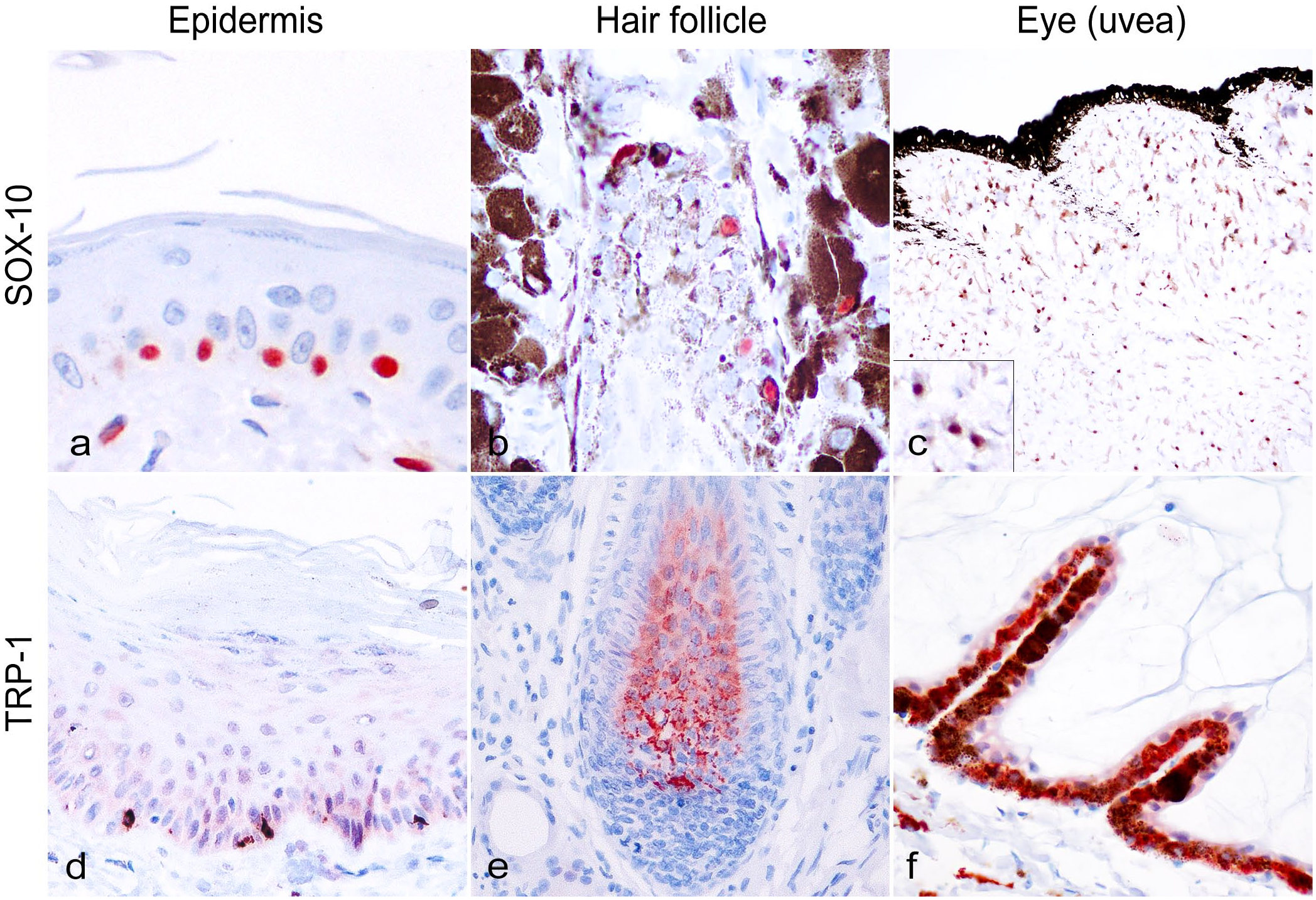
Immunohistochemistry (IHC) for (a–c) Sry-related HMg-Box gene 10 (SOX-10) and (d–f) tyrosinase-related protein-1 (TRP-1) in normal feline tissues. Cat. (a) Epidermis. Scattered SOX-10 immunolabeled melanocytes in the basal layer of the epidermis. SOX-10 IHC. (b) Hair follicle. SOX-10 immunolabeling in melanocytic cells in the hair bulb. SOX-10 IHC. (c) Eye (uvea). Disseminated SOX-10 immunolabeling in melanocytes in the iris stroma. Inset: higher magnification. SOX-10 IHC. (d) Epidermis. TRP-1 immunolabeling in sparse basal dendritic melanocytic cells. TRP-1 IHC. (e) Hair follicle. Numerous melanocytes with cytoplasmic TRP-1 immunolabeling in hair bulb. TRP-1 IHC. (f) Eye. Diffuse TRP-1 immunolabeling in the cytoplasm of iris pigmented epithelium. TRP-1 IHC.
In both cutaneous and oral samples, TRP-1 immunolabeling was seen in non-neoplastic melanocytes within the basal layer of the epidermis (Fig. 1d), the hair bulbs (Fig. 1e), and the basal mucosal epithelium. In the iris, immunolabeling was detected in the lining of pigmented epithelium and in melanocytes residing in the stroma (Fig. 1f).
Histological Features of Melanocytic Tumors
The histopathological features evaluated for each case are reported in Supplemental Table S2.
In 12 of ocular melanomas, 8 (67%) were epithelioid histotype, 2 (17%) were spindle, and 2 (17%) were mixed (epithelioid, balloniform, and/or spindle). In the 7 cases of oral neoplasms, 4 (57%) were spindle and 3 (43%) were epithelioid pattern. In cutaneous tumors, 4 (33%) cases were epithelioid histotype, 4 (33%) were spindloid, 2 (17%) were signet ring, and 2 (17%) were mixed (epithelioid, spindloid, and/or balloniform). The signet-ring pattern was not observed in ocular or oral neoplasms.
Nuclear atypia and cellular pleomorphism showed heterogeneous results in all neoplastic sites, with a minimum value of 10%, a maximum of 90%, and an average of 50% of nuclear atypia. The mitotic count of the cases evaluated ranged from 0 to 69, with a median of 23 (interquartile range, 9–51). Specifically, the average mitotic count in ocular melanomas was 24.4, in oral melanomas 34.25, and in cutaneous melanomas 26.6. The degree of pigmentation and the percentage of pigmented cells in oral/perioral neoplasms was low, with only 3 (43%) pigmented tumors characterized by mild to moderate quantities of melanin. In these cases, extension of pigmentation did not exceed 50% of the total surface. Among the ocular neoplasms, no amelanotic tumors were present. In cutaneous melanomas, the pigmentation was more heterogeneous, with 2 (17%) amelanotic tumors; pigmented neoplasms had moderate to marked degrees of pigmentation. All neoplasms with greater than 50% pigmented cells (3/12, 25%) had an epithelioid histotype.
At the oral/perioral level, junctional activity was identified in 3 cases out of 5 cases (60%), while intraepithelial cell nests were not observed. Similarly, in the skin, junctional activity was seen in 9 out of 11 (82%) and intraepithelial cells only in 2 cases (18%). In 2 cases from oral mucosa and 1 from skin, the mucosa was extensively ulcerated, hence junctional activity could not be assessed.
Immunohistochemical Expression of Markers in Melanocytic Tumors
The immunohistochemical findings for each case are reported in Supplemental Table S3.
All ocular and nonocular melanomas were positive for S100 (100%; Fig. 2a–c). Melan-A showed positive immunolabeling in 100% of ocular (Fig. 2d), 100% of oral (Fig. 2e), and 92% of cutaneous melanomas (Fig. 2f). PNL2 was instead observed mainly in ocular tumors (11/12; 92%; Fig. 2g) compared with only 3/7 oral/perioral melanomas (43%; Fig. 2h) and 6/12 cutaneous melanomas (50%; Fig. 2i). Regarding the distribution in the cells, immunolabeling for S100 was localized in the nucleus and/or cytoplasm, while Melan-A and PNL2 were limited to the cytoplasm, as expected. PNL2 expression was significantly correlated with the ocular site (P = .006) and with highly pigmented neoplasms (P < .01). Both PNL2 and Melan-A were more commonly expressed in neoplasms with an epithelioid histotype (P < .05).
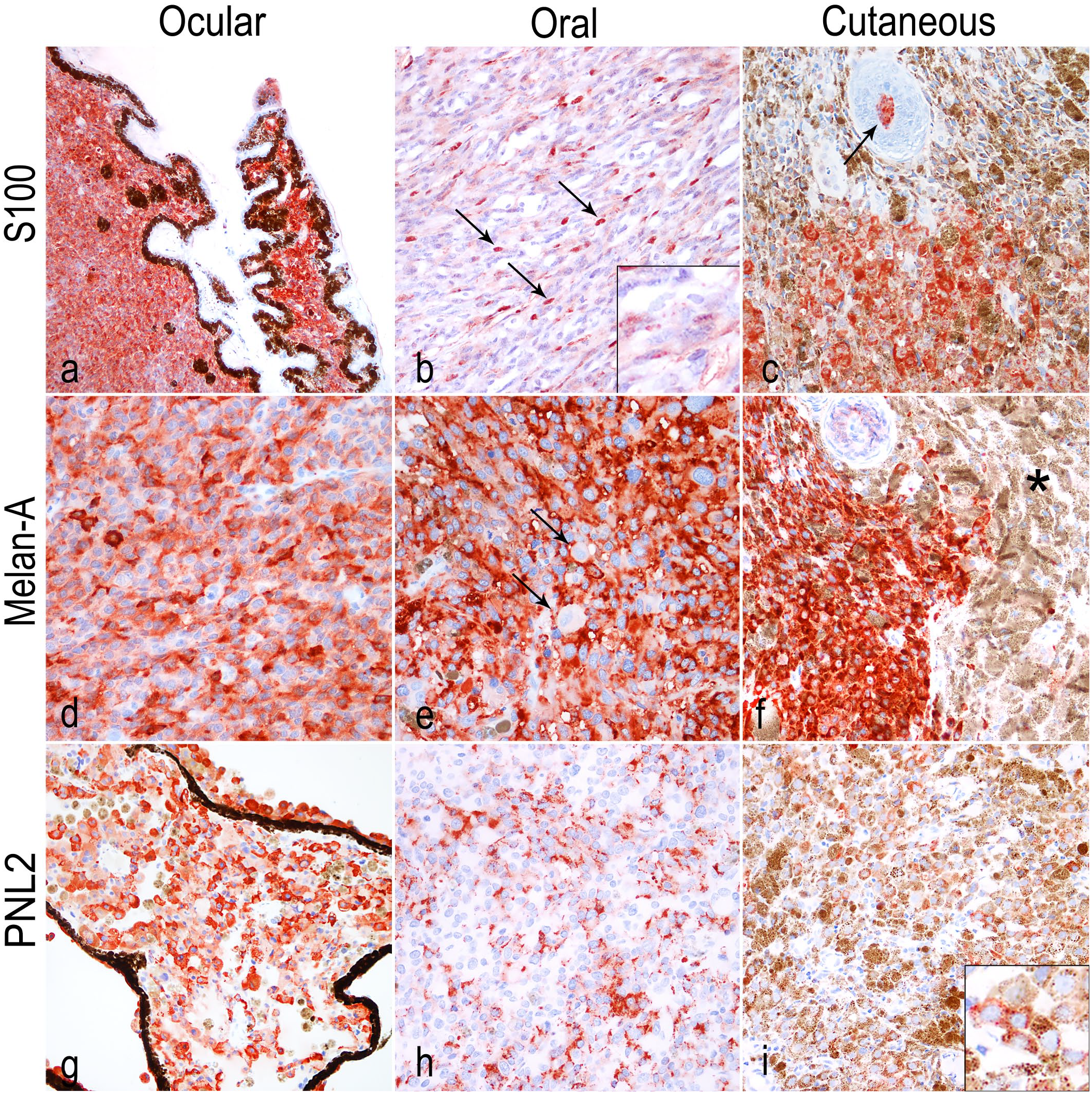
Cat. (a) Ocular melanoma. Strong immunolabeling for S100. S100 immunohistochemistry (IHC). (b) Oral melanoma. Cytoplasmic and nuclear immunolabeling of neoplastic cells. Strong nuclear (arrows) and weak cytoplasmic immunolabeling for S100 (inset). S100 IHC. (c) Cutaneous melanoma. S100 immunolabeled cells in the hair bulb trapped within the tumor (arrow). S100 IHC. (d) Ocular melanoma. Immunolabeling for Melan-A is mostly diffuse, nevertheless, intratumoral variability is frequently observed. Melan-A IHC. (e) Oral melanoma. Diffuse cytoplasmic immunolabeling for Melan-A. Cells characterized by karyomegaly and higher nuclear atypia often show weaker cytoplasmic immunolabeling (arrows). Melan-A IHC. (f) Cutaneous melanoma. Melan-A immunolabeling is limited to the neoplastic population of melanocytes, whereas the melanin-laden macrophages on the right (asterisk) are negative. Melan-A IHC. (g) Ocular melanoma. As for Melan-A, PNL2 shows intratumoral variability in numerous cases. PNL2 IHC. (h) Oral melanoma. Immunolabeling for PNL2 displayed an uneven distribution in the cytoplasm. (i) Cutaneous melanoma. PNL2 expression in a heavily pigmented tumor (detail in the inset).
SOX-10
Nuclear SOX-10 expression was detected in cutaneous, ocular, and oral melanomas (100%, 92%, and 86%, respectively; Fig. 3a–c). In all the positive samples, the intensity of the immunolabeling varied from mild to marked, and the expression distribution pattern was widespread within the neoplasm. Positivity was observed in all of the neoplastic histotypes and, given its nuclear expression, it was possible to evaluate the immunolabeling even in neoplasms with a high degree of pigmentation, with no need to perform bleaching. SOX-10 was positively associated with PNL2 (P < .05, ρ = 0.538) and Melan-A expression (P < .01, ρ = 0.559). Both correlations are considered moderately strong.
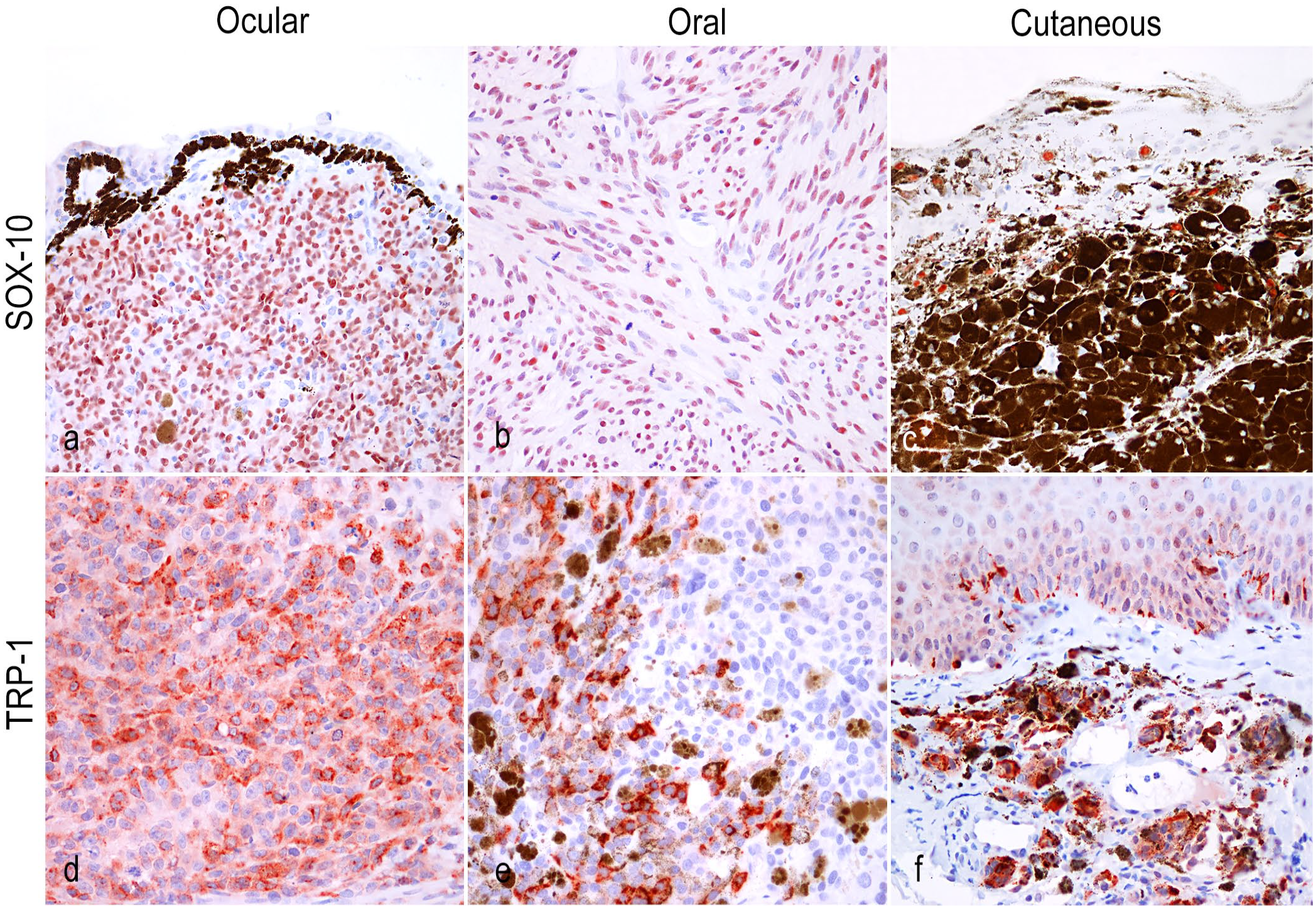
Cat. (a) Ocular melanoma. Moderate nuclear immunolabeling for SOX-10 (about 90% of neoplastic cells). SOX-10 immunohistochemistry (IHC). (b) Oral melanoma. Spindle cells, in amelanotic melanomas with both epithelioid and spindle cells, are often negative for Melan-A and PNL2, whereas SOX-10 shows variable immunolabeling in nuclei. SOX-10 IHC. (c) Cutaneous melanoma. Heavily pigmented cutaneous melanomas immunolabeled with SOX-10 using a red chromogen. Immunolabeled nuclei are visible in the melanin-laden neoplastic cells and in normal melanocytes among keratinocytes. SOX-10 IHC. (d) Ocular melanoma. TRP-1 is variably expressed in neoplastic melanocytes, similar to Melan-A and PNL2. TRP-1 IHC. (e) Oral melanoma. TRP-1-positive cells are admixed with negative population, often near melanophages. TRP-1 IHC. (f) Cutaneous melanoma. TRP-1 immunolabled neoplastic melanocytes arranged in nests in the dermis and normal intraepidermal melanocytes. TRP-1 IHC.
TRP-1
In melanomas, expression was observed in 75% of ocular (9/12; Fig. 3d), in 29% of oral (2/7; Fig. 3e), and in 36% of the skin melanomas (4/11; Fig. 3f). For case 21, there was no formalin-fixed paraffin-embedded tissue available for the immunohistochemical investigation. Immunolabeling was observed as a granular cytoplasmic reaction. The percentage of positive neoplastic cells was in most cases less than or equal to 10% (12/15, 80%); while only 3 ocular cases had 15%, 50%, and 80% immunolabeling. Although the expression was maintained only by a small percentage of tumor cells in some of the tumors examined, the intensity was always evaluated as moderate or marked. As for PNL2, TRP-1 immunolabeling was correlated with a pigmentation percentage greater than 50% (P < .01). Moreover, it was significantly associated with PNL2 (P < .001, ρ = 0.599) and Melan-A expression (P < .01, ρ = 0.489); the correlation of the variables is moderately strong.
Discussion
Melanocytic tumors are uncommon in cats compared with dogs and humans, with the exception of feline diffuse iris melanoma. Due to highly morphologic variability, occasional absence of pigmentation, and morphological overlapping with other tumors, histological diagnosis of melanoma is not always straightforward for the pathologist. Furthermore, the immunohistochemical markers routinely used (Melan-A, PNL2, and S100) do not have 100% specificities and sensitivities. In humans and dogs, SOX-10 and TRP-1 have been tested and used to implement the routine immunohistochemical panel, but these markers have never been assessed in cats.2,11,20 Thus, the aims of this study were to demonstrate immunoreaction of these new primary antibodies in non-neoplastic feline melanocytes and to evaluate these antibodies for the diagnosis of feline melanoma.
In silico alignments of protein sequences is a method used to provide information on the likelihood cross-species immunoreactivity of an antibody; for instance, this approach was used in evaluating an anti-human CD30 antibody in feline tissues and lymphoma.4,5 Although the amino acid identity of human and feline SOX-10 and TRP-1 was 76.6% and 88.6%, respectively, the identity between antibodies’ epitopes and the target complementary sequences was 100% and 97.7%, respectively; therefore, a cross-reactivity of the antibodies with the feline proteins is to be expected, as demonstrated by our results. SOX-10 expression was characterized by a strong nuclear reaction in cutaneous and mucosal intraepithelial melanocytes, hair bulb melanocytes, uveal melanocytes, and somatic nerves, as previously reported in humans. 10 In contrast to other specific melanocytic markers with cytoplasmic staining patterns, the nuclear immunolabeling of SOX-10 allows for an easy identification of positive cells by the pathologist, even in highly pigmented neoplasms. Indeed, in human medicine, it is considered a reliable marker for the identification of metastatic melanoma in sentinel lymph nodes. 26 On the other hand, TRP-1 immunoreactivity was localized not only in the cytoplasm of cutaneous and iridal melanocytes but also in the lining pigmented epithelium of iris; the latter result could be explained by the presence of this enzyme within melanosomes, which can be present also in the cytoplasm of other cells as well as in melanocytes (ie, keratinocytes).2,3 The distribution of intraepithelial melanocytes, evaluated by SOX-10 and TRP-1 immunolabeling, seems to be uneven, similar to what was recently reported in dogs. 15 On the contrary, this is the first preliminary description of the expression of these markers in the feline eye. Still, further studies on feline melanocytes should be performed on a larger group of animals to confirm our results and evaluate feline melanocyte density.
The second aim of the study was to evaluate the expression of SOX-10 and TRP-1 in cutaneous, oral, and ocular feline melanomas and to assess whether these markers could perform better than Melan-A and PNL2. Considering the previous studies on feline melanomas, only a few showed a diagnosis of amelanotic melanoma based on immunohistochemical examination with specific markers; in others, diagnosis was based only on S100 expression, known as very sensitive but nonspecific marker for melanocytic cells.6,16,17 For this reason, data and associations reported in the articles are questionable, and poorly differentiated melanomas may not have been included on the basis of histopathological examination alone. Conversely, soft tissue sarcomas could be erroneously diagnosed as melanoma if only S100 immunoreactivity was used. 18 In our study, the inclusion criterion was that melanomas showed expression of at least one of the 2 specific markers for melanocytic origin based on validated markers identified in the literature. The positivity for S100 in this case showed that the sample was suitable for immunohistochemical investigation, excluding problems such as overfixation in formalin, which can compromise results in some cases.
Currently, there are no antibodies with a concurrent high sensitivity and specificity for the detection of melanocytic neoplasms. However, in dogs, an antibody cocktail that includes Melan-A, PNL2, TRP-1, TYRP-2, and S100 is used, which has shown good results in the diagnosis of melanomas. 20 On the other hand, for cats, there are no published studies on the use of the same cocktail. Until a decade ago, the only validated markers for the confirmation of melanocytic origin in feline neoplasms were Melan-A and S100, limiting the diagnostic power of veterinary pathologists, due to their variable sensitivity and poor specificity, respectively. 16 More recently, PNL2 has been added to the feline panel and has shown an excellent sensitivity in both ocular and nonocular melanocytic tumors.13,25
In our study, Melan-A was found to be a sensitive marker for all sites, while PNL2 showed poor sensitivity in the case of oral and skin neoplasms, converse to what has been described in the literature.13,25 The use of the markers selected in our series demonstrated a high sensitivity of SOX-10 for melanocytic neoplasms regardless of their site of development, while TRP-1 showed a higher sensitivity in ocular tumors and neoplasms characterized by greater than 50% pigmentation, similar to what was observed for PNL2. 15 Furthermore, in contrast to SOX-10, which resulted in immunolabeling in greater than 50% of neoplastic nuclei in most cases, TRP-1 was positive in less than 10% of tumor cells, with a degree of intensity ranging from moderate to marked. In the case of SOX-10, differences in the intensity of nuclear immunolabeling could be associated with the variable chromatin patterns of the neoplastic cells (ie, vesicular chromatin would account for a faint immunolabeling). With both tested markers, statistical analysis highlighted a significant association between their expression with Melan-A and PNL2.
From the emerging data, it is possible to hypothesize that TRP-1, being an enzyme involved in the process of melanogenesis, is expressed in more differentiated neoplasms (pigmented and exhibiting expression of Melan-A and PNL2), which are less likely to cause a dilemma in the diagnostic routine. As for SOX-10, this proved to be an excellent marker with a positive expression in melanomas comparable to S100. Nonetheless, the specificity of this antibody should be explored with further studies on different types of tumors, as expression of this protein has been observed in human myoepithelial, nervous, and occasional epithelial tumors. 11
Regarding oral melanoma, it is necessary to consider as possible differential diagnoses with SOX-10 expressing tumors originating from the myoepithelium of the salivary glands and peripheral nerve sheaths, both of which are difficult to distinguish on histopathological examination based on the morphology alone. 11 Consequently, despite expression in other types of neoplasms, SOX-10 could be included in a larger antibody panel, in association with other specific markers for melanocytic lineages, in order to increase the sensitivity in the diagnosis of melanoma.
Since the follow-up of the patients were not available, it was not possible to investigate a possible association between the variation of the expression of TRP-1 and SOX-10 with the biological behavior of the neoplasms, as already demonstrated for PNL2 immunopositivity in diffuse iris melanoma. 25
This is the first study to assess and validate the cross-reactivity of SOX-10 and TRP-1 in normal feline tissues and melanomas from different sites. In particular, nuclear immunolabeling of SOX-10 has shown to simplify the identification of melanocytes even in highly pigmented neoplasms. For both antibodies, no increase in sensitivity and specificity when combined with Melan-A, PNL2, and S100 was observed. Prospective studies on higher number of melanomas from different sites with clinical follow-up are recommended to evaluate a possible association between expression of these markers and the biological behavior of the neoplasm.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241244850 – Supplemental material for SOX-10 and TRP-1 expression in feline ocular and nonocular melanomas
Supplemental material, sj-pdf-1-vet-10.1177_03009858241244850 for SOX-10 and TRP-1 expression in feline ocular and nonocular melanomas by Margherita Orlandi, Ilaria Porcellato, Monica Sforna, Adriana Lo Giudice, Giuseppe Giglia, Luca Mechelli and Chiara Brachelente in Veterinary Pathology
Footnotes
Acknowledgements
We thank Sara Leto, Valeria Migni, Luca Stefanelli, and Giampaolo Ceccarani for their technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
