Abstract
In albino rats, spontaneous occurrence of melanocytic tumors is rare, with diagnosis difficult. This study evaluated immunoreactivity for PNL2 in normal and neoplastic melanocytes in formalin-fixed and paraffin-embedded tissues of albino rats. The samples consisted of 11 (1.57%) amelanotic melanomas in 700 rats (2 studies), 23 non-melanocytic tumors, and a wide variety of normal tissues. In normal albino rats, PNL2 stained the melanocytes in the iris and choroid of the eyeball and the hair bulb and basal cell layers of the epidermis of the whole body. In amelanotic melanoma, the tumor cells consisted of spindle cells with eosinophilic cytoplasm without melanin granules. PNL2 consistently stained cytoplasm in all amelanotic melanoma cells. In contrast, the nonmelanocytic tumor cells were not labeled. Electron microscopically, neoplastic, and normal melanocytes showed numerous cytoplasmic premelanosomes (stage II melanosome). In conclusion, PNL2 is direct against a fixative-and decalcific-resistant melanocyte-associated antigen, and has high specificity against normal and neoplastic melanocytes of albino rats.
Keywords
In albino rats, spontaneous occurrences of melanocytic tumors are rare. 4– 7, 12– 14 Pigment-containing melanomas such as those seen in Brown Norway (melanin pigmented) rats are easily diagnosed via histology, 5 but diagnosis becomes difficult without pigment production. 3 Diagnosis of amelanotic melanoma needs further investigation by histochemical, immunohistochemical, or ultrastructural studies. 4 However, even using these methods, only a few cases tend to be successfully diagnosed. 10
Recently, a monoclonal antibody named PNL2 was introduced as a new human melanocyte differentiation marker. 1, 2, 9 Most melanomas in albino rats are positive for vimentin and S100 and negative for cytokeratin. 6, 7, 12, 14 However, these markers are not specific and may be found in a variety of tissues. 10 There is no effective antibody to demonstrate a melanocyte of albino rat. Therefore, the immunoreactivity for PNL2 in normal and neoplastic melanocytes in formalin-fixed and paraffin-embedded tissues of albino rats was evaluated.
The samples consisted of 11 (1.57%) amelanotic melanomas in 700 rats (2 studies) (Table 1) and 23 non-melanocytic tumors found in routine diagnostic histopathologic examinations, and a wide variety of normal tissues obtained from 2 normal adult albino rats. The rats were supplied by Charles River Japan, Inc. (Kanagawa, Japan). The animals were housed and euthanatized in accordance with the guidelines for the care and use of laboratory animals prepared by the Japanese Association for Laboratory Animal Science and the guidelines of our institution. At necropsy, all tissues were fixed in 10% neutral-buffered formalin, and eyeballs were placed in Davidson fixative. Bone tissues were also fixed in 10% neutral-buffered formalin and then soaked in a decalcifying solution (15% formic acid and 15% formaldehyde in distilled water) for 1 week. Then, these tissues were embedded in paraffin, stained with hematoxylin and eosin, and the Schmorl method and Masson-Fontana's technique for melanin detection were applied.
Summary of animals' strain, sex, affected site, age, and number of mitoses, and immunohistochemical and electron microscopic study of amelanotic melanoma in albino rats.
Total mitoses in 10 high-powered (x 400) fields.
Confirmation of premelanosome by TEM; + = positive; NE = not examined; TEM = transmission electron microscopy.
The immunostaining procedure was performed using a labeled streptavidin–biotin system (LSAB2 System-HRP, Dako Japan Ltd., Kyoto, Japan). Details for the primary antibodies are given in Table 2. Regarding the antigen retrieval, the slides were transferred to 0.5% Immunosaver (Nisshin EM Corp., Tokyo, Japan) 8 and heated for 30 minutes at 95°C using a 400W-MW processor (MI-77, Azumaya Corp., Tokyo, Japan) in the microwave treatment for PNL2, or heated for 40 minutes at 121°C using an autoclave (Tommy Seiko Corp., Tokyo, Japan) in the autoclave treatment for Melan A. The slides were incubated with 2% proteinase K (Dako Japan Ltd., Kyoto, Japan) in Tris buffered solution (TBS) for 7 minutes at room temperature in the proteinase treatment for S100. The sections were incubated for 60 minutes at room temperature with the primary antibodies. Subsequently, the remaining methods were applied according to the supplier's instructions. Because canine cutaneous benign melanoma contains numerous melanin granules positive for PNL2, it was used as the positive control. For the negative controls, the primary antibodies were omitted.
Clone, supplier, and optimal dilution for each of the primary antibodies.
Mab = monoclonal antibody; Pab = polyclonal antibody.
All antibodies were diluted in TBS; TBS = Tris buffered solution.
For the electron microscopic examination, blocks (1 mm3) of the formalin-fixed samples or paraffin-embedded samples, which were deparafinized in xylene and ethanol solution from amelanotic melanoma, and eyelid of albino rats were postfixed with 1% phosphate-buffered osmium tetroxide. Then, the samples were routinely processed and embedded in resin. Ultrathin sections were stained and examined using a Hitachi H-7600 transmission electron microscope (Tokyo, Japan).
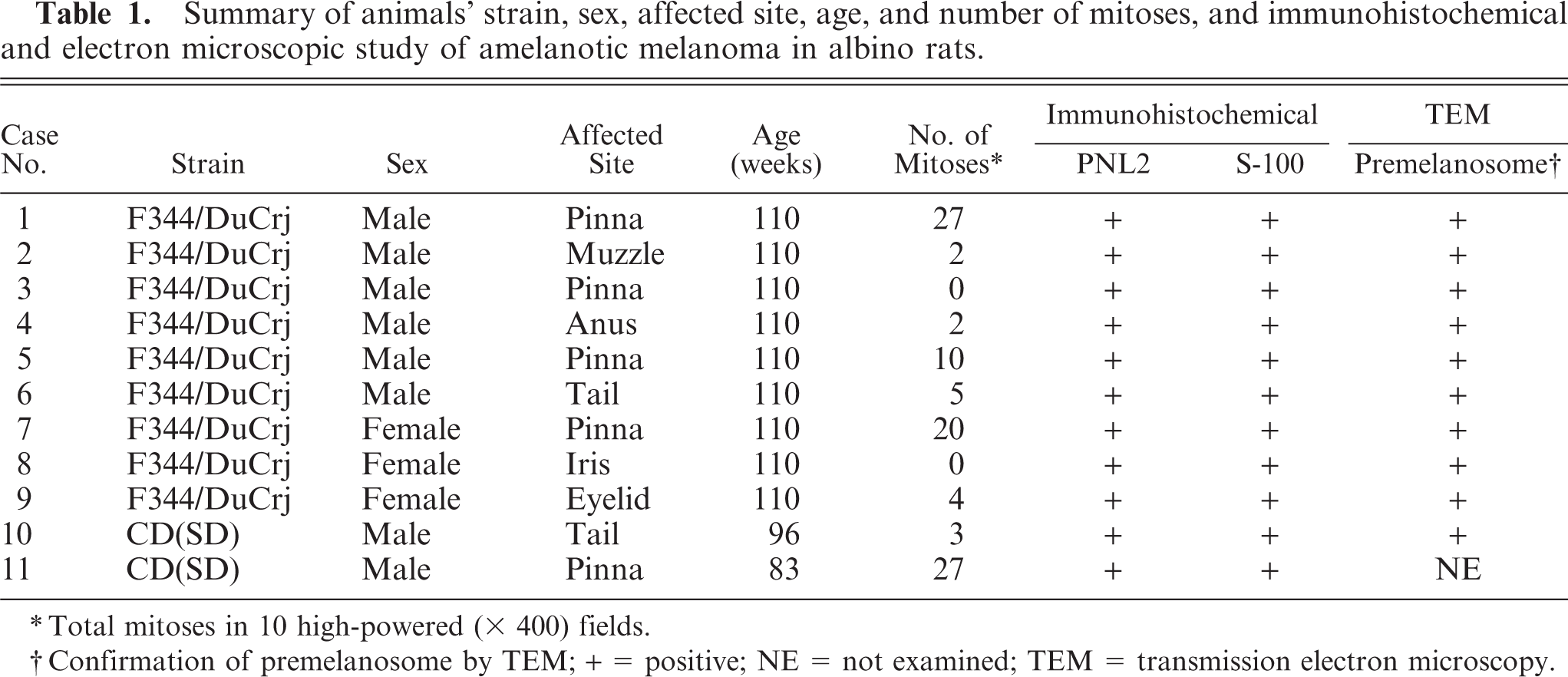
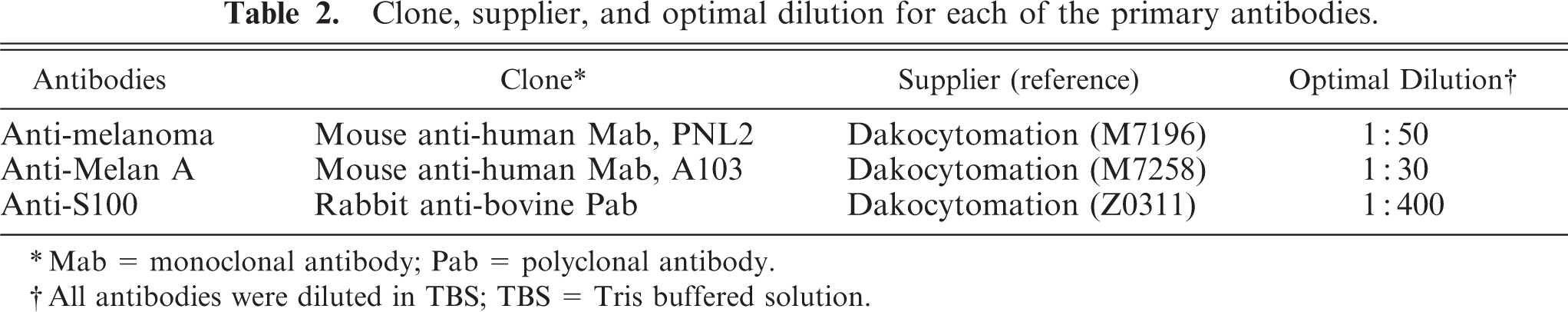
PNL2 stained a large number of melanocytes in the iris (Fig. 1) and choroid of the eyeball, eyelid (palpebral conjunctiva), anus (Fig. 2), the area of the scrotum with little hair, and the rhinarium of the muzzle. Furthermore, it gave a slight immunostaining of the hair bulb and basal cell layer of the epidermis of the whole body. The staining was granular and cytoplasmic and highlighted the dendritic processes. Electron microscopically, melanocytes of the palpebral conjunctiva were located in the basal cell layer of the stratified squamous epithelium and had a large number of cytoplasmic premelanosomes. This site corresponded to the localization of the PNL2-positive melanocytes.
Iris; albino rat. The PNL2 immunostaining is limited to the iris cells. Co, cornea; L, lens. LSB method counterstained with Mayer's hematoxylin. Bar = 63 μm.
Melanocytes in the basal cell layers of the stratified squamous epithelium of the anus. LSB method counterstained with Mayer's hematoxylin. Bar = 42 μm.
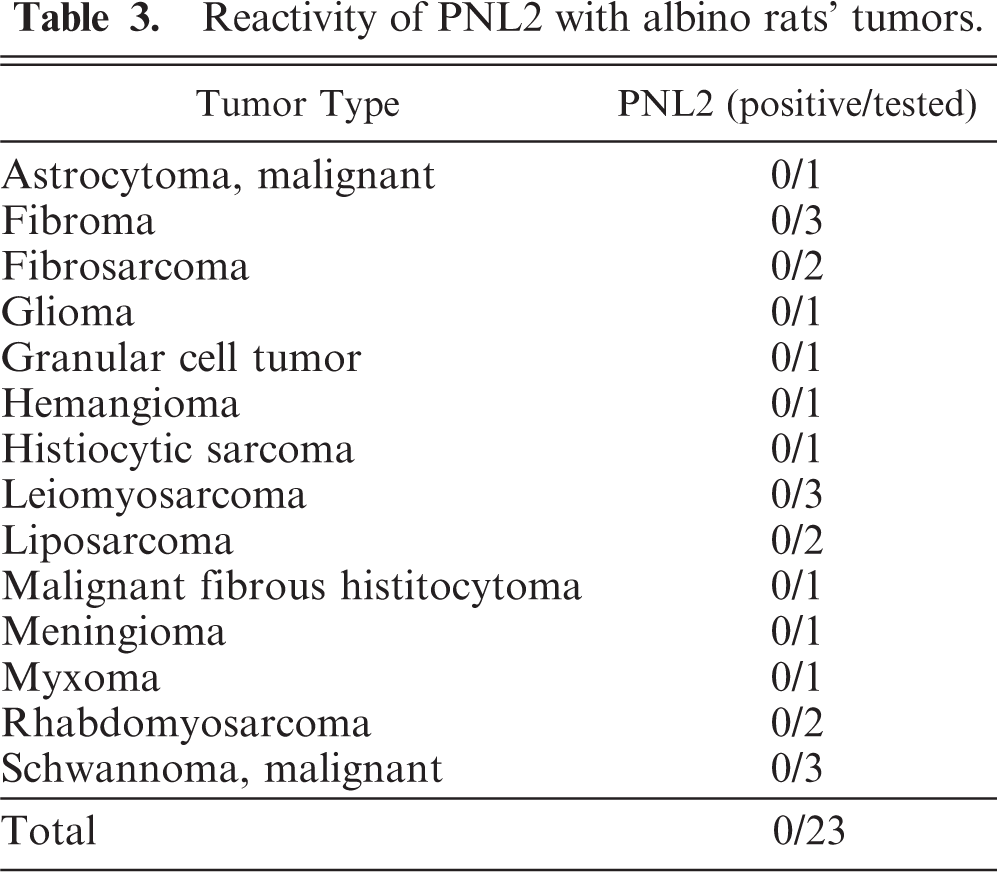
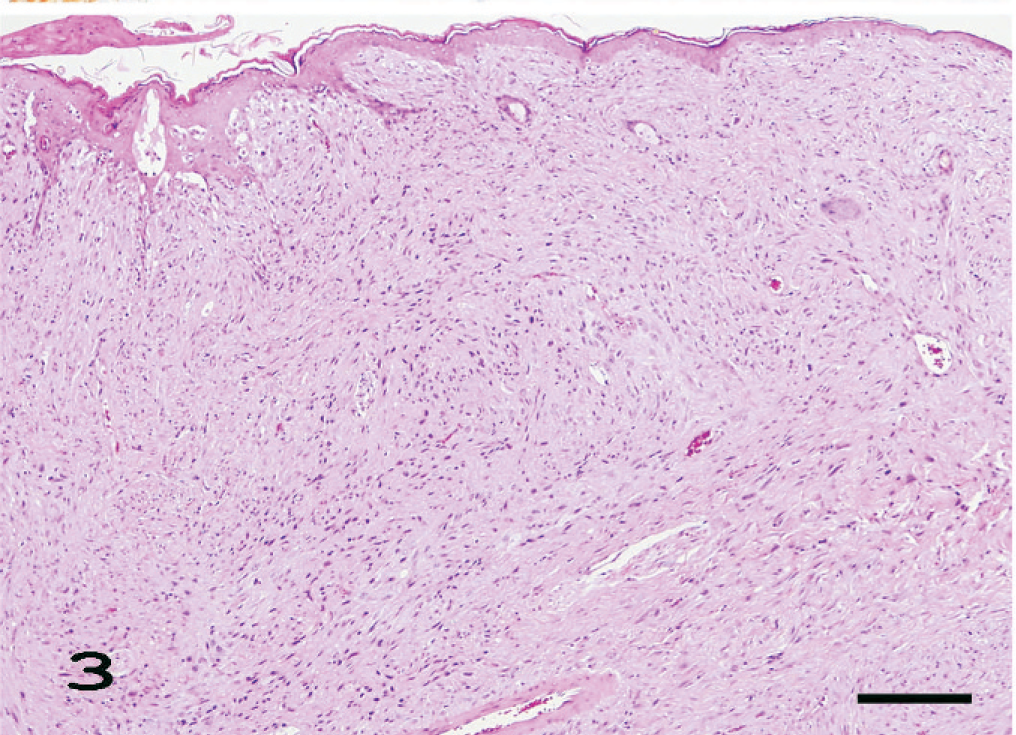
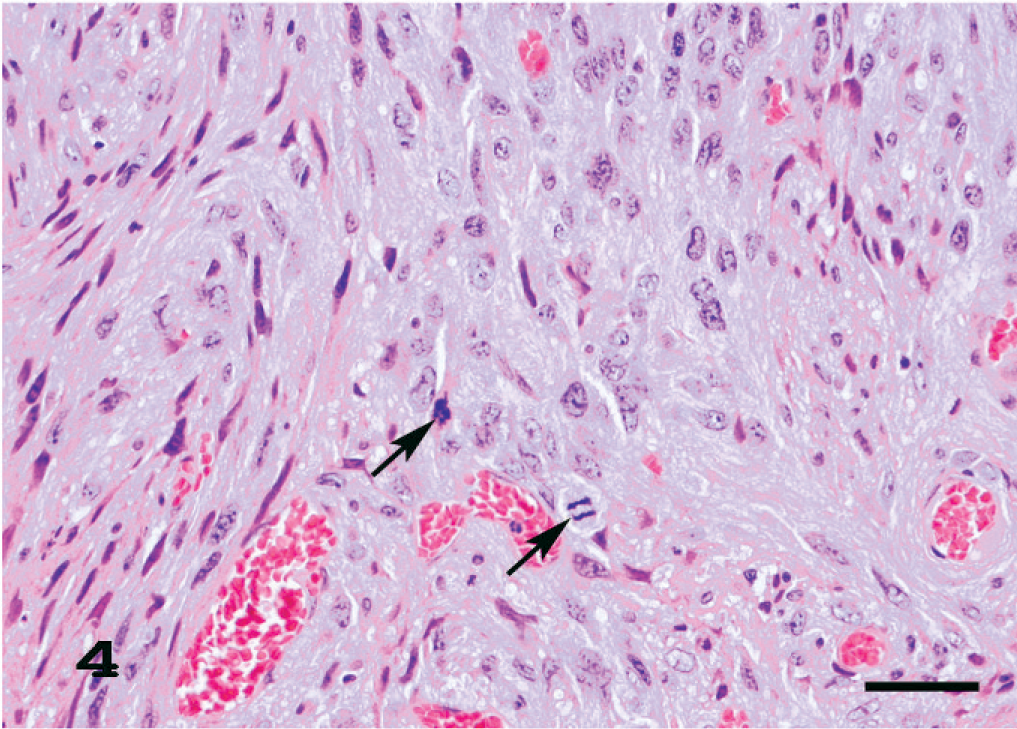
Eleven amelanotic melanomas occurred in the pinna, muzzle, anus, tail, iris, and eyelid (Table 1). Each animal had a single, firm, and tan or white tumor mass that sporadically ulcerated in the case of skin tumors. Histologically, the tumors consisted of spindle cells with abundant pale eosinophilic cytoplasms containing no melanin granules. The spindle cells had indistinct cell borders and were arranged in an interlacing fascicular pattern (Fig. 3). Mildly anaplastic spindle cells were seen in case Nos. 1, 7, and 11 (Fig. 4). These tumor cells had eosinophilic cytoplasms and nuclei in various sizes and shapes with a large number of mitotic figures. These cases had more than 20 mitotic figures in 10 high-powered fields (× 400), while case Nos. 2 to 6 and 8 to 10 had fewer (0–10) mitotic figures (Table 1). Immunohistochemically, tumor cells exhibited moderate and consistent cytoplasmic staining for PNL2 (Fig. 5, Table 1). This staining was granular with no background staining. PNL2 staining was not altered by the formic acid–based decalcification. By contrast, the non-melanocytic tumor cells were found to be uniformly negative for PNL2 (Table 3), and various inflammatory cells were immunonegative (Fig. 5). Canine melanoma cells as the positive control consistently stained not only numerous melanin granules, but also spindle tumor cells with no melanin granules. None of the negative controls were stained. Tumor cells of all cases were positive for S100 protein (Table 1) but negative for Melan A and were not stained by the Schmorl method or the Masson-Fontana's technique for melanin. Electron microscopy showed that all cases except case No. 11 were detected with premelanosome (stage II melanosome) in melanocytes (Fig. 6).
Reactivity of PNL2 with albino rats' tumors.
Amelanotic melanoma of the ear; case No. 1. A dermal nodule is composed predominantly of tumor cells. These tumor cells are arranged in an interlacing fascicular pattern. HE. Bar = 31 μm.
Amelanotic melanoma of the ear. Higher magnification of Fig. 3. Mildly anaplastic spindle cells are seen. These tumor cells have prominently eosinophilic cytoplasm and nuclei with greater variation in size and shape and large number of mitotic figures (arrows). HE. Bar = 44 μm.
Amelanotic melanoma of the ear; case No. 1. PNL2 consistently gives a moderate cytoplasmic staining of most of the tumor cells. This staining is granular with no background staining. LSB method counterstained with Mayer's hematoxylin. Bar = 42 μm. Inset. Higher magnification of PNL2-negative inflammatory cells. LSB method counterstained with Mayer's hematoxylin. Bar = 9.5 μm.
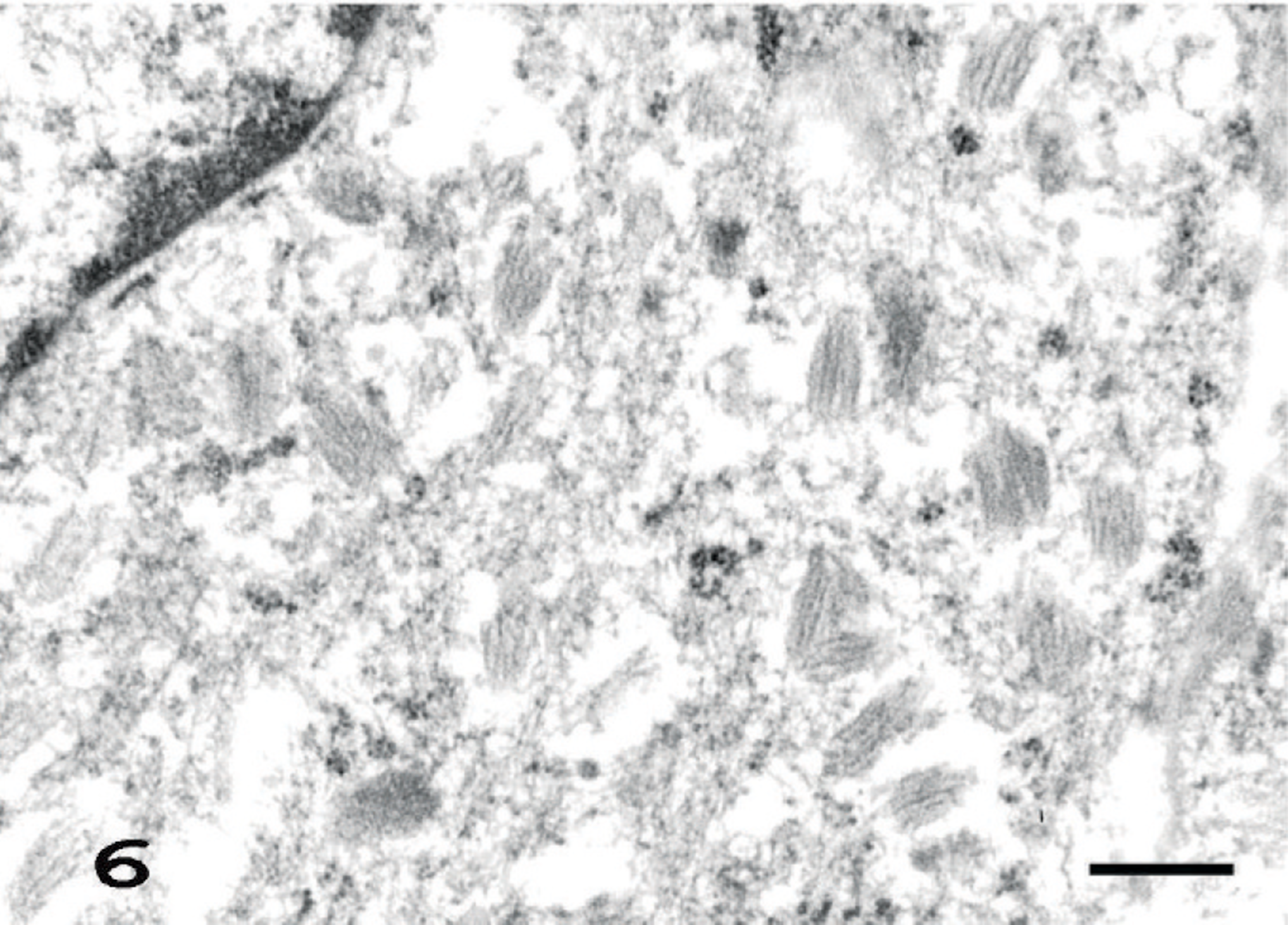
Amelanotic melanoma of the tail; case No. 6. Note a large number of premelanosomes (stage II melanosome) in cytoplasm of melanoma cell. Uranyl acetate and lead citrate. Bar = 37.5 nm.
In human tissues, anti-melanoma PNL2 antibody acts on normal melanocytes and melanocytic tumor cells, 1, 2, 9 and labels neoplastic melanocytes in a fashion similar to Melan A and HMB-45 antibodies. 1, 9 However, immunohistochemical analysis of PNL2, HMB-45, and Melan A in albino rats has not previously been reported. This study was conducted to screen various types of rat tumors with the anti-melanoma antibodies, PNL2 and Melan A. As a result, we clearly demonstrated frequent and intense PNL2 expression in both normal and neoplastic melanocytes of albino rats. Moreover, PNL2 is suitable anti-melanocyte antibody for formalin-fixed and paraffin-embedded tissues, and this staining is not altered by a decalcification. Furthermore, though the main disadvantage of using PNL2 in humans is its reactivity with granulocytes, 1, 2, 9 positive staining by PNL2 in albino rats is restricted to melanocytes, and various inflammatory cells and normal tissue cells are immunonegative. Because of its high sensitivity for melanocytes in albino rats, PNL2 will likely become a useful reagent for the diagnosis of amelanotic melanoma in albino rats.
Electron microscopically, most premelanosomes observed in the neoplastic and normal melanocytes of albino rats were stage II melanosomes. According to the general agreement, stage II melanosomes contain fibrous striations but no pigment, stage III melanosomes have some melanin along their striations, while those at stage IV are filled with melanin. 3 Stage III melanosomes with a minimal amount of melanin content are also known to occur in albino rats. 3 However, the presence of stage III melanosomes could not be confirmed in this study. The Masson-Fontana's technique did not prove the presence of stage II melanosomes, suggesting that these melanosomes are too immature for positive staining. 11 In this study, all special staining results were also negative. Results from this study lead to the conjecture that the antigens recognized by PNL2 exist in stage I and II melanosomes or in intracellular structures.
PNL2 is not only an excellent marker for normal melanocyte and nonanaplastic melanoma having a few mitotic figures, but also recognizes anaplastic melanoma that have a large number of mitotic figures. However, in humans, some melanomas are usually negative for HMB-45 and Melan A. 1 Larger numbers of melanocyte-related tumors of the albino rats are necessary to examine the specificity of the antibody.
We demonstrated the distribution of PNL2-positive melanocytes in albino rats. In summary, all cases diagnosed as melanoma were detected in premelanosome (stage II melanosome) and were PNL2-positive melanoma; all cases of nonmelanocytic tumor were PNL2 negative; and, melanocytes in normal albino rats that were detected in premelanosome (stage II melanosome) were PNL2 positive. Large numbers of PNL2 positive melanocytes recognized in this study, except for the muzzle, are in predilection sites of melanoma in rats. 6, 7, 12– 14 Melanoma often occurs at these sites with a large distribution of melanocytes. However, while the pinna and tail are also predilection sites of melanoma in rats, 14 there were few melanocytes in these sites. To resolve this contradiction, more detailed study concerning the distribution of PNL2-positive melanocytes is necessary.
In addition to humans, PNL2 stained numerous melanocytes in the C57/Bl6, nude, and Balb/c mice. 9 However, reacting to the other animals has not been reported. The precise nature of the epitope recognized by PNL2 is currently still unknown. 2, 9 The findings in the present study showed the possibility to examine the antigen detection site of PNL2 using melanocytes of humans, mice, and even albino rats. Also, PNL2 may also be of value for identifying melanocytic neoplasms in other species, such as canines. The antigen recognized by this antibody remains to be fully characterized.
Footnotes
Acknowledgements
We thank Takayoshi Ito, Masayo Onozawa, Chie Shiromaru, Misako Morino, Tomomi Sakuma, Kiyoe Hosoya, Yuuki Ikeda, and Miki Kozasa for excellent technical support and Steve Yamakami and Azumi Takahashi for language editing. We thank Yuki Kamihara from Dako Japan Inc. for her advice and support.
