Abstract
Epithelioid hemangiosarcoma (EH), a rare histological variant of hemangiosarcoma, is reported in various animal species, including humans, dogs, cows, horses, and cats. Epithelioid hemangiosarcomas are composed of highly pleomorphic epithelioid cells arranged in cords, islands, nests, or solid cellular areas, similar to epithelial neoplasms. Moreover, in humans, approximately 50% of EHs have cytoplasmic immunolabeling for cytokeratin AE1/AE3 (CK AE1/AE3), making it challenging to distinguish them from carcinomas. This retrospective study assessed the CK AE1/AE3 immunolabeling in canine EH cases from 5 veterinary institutions. Immunohistochemistry for CD31 and CK AE1/AE3 was performed on 30 cases. CK AE1/AE3 immunolabeling was detected in 43% (13/30) of cases, with cytoplasmic labeling ranging from 5% to 100% of neoplastic cells. All tumors consistently had membranous immunolabeling for CD31. The CK AE1/AE3 immunolabeling pattern in canine EHs closely resembled those documented in humans, indicating a similar diagnostic challenge. Therefore, it is recommended to include a vascular immunohistochemistry marker, such as CD31, whenever EH is suspected, particularly in small incisional cutaneous and subcutaneous biopsies.
Hemangiosarcoma is a malignant neoplasm that originates from the vascular endothelial precursor cells derived from the bone marrow and has been reported in several animal species, including humans, dogs, cows, horses, and cats.8,16,19 Hemangiosarcomas are histologically classified as well-differentiated or conventional, kaposiform, anaplastic, and epithelioid.7,14 Epithelioid hemangiosarcoma (EH) is commonly reported in dogs, with sporadic reports in cows, horses, and cats.14,20
Epithelioid hemangiosarcoma is composed of highly pleomorphic epithelioid cells arranged in cords, islands, nests, or solid cellular areas. Neoplastic cells rarely form vascular clefts or spaces, thereby resembling neoplasms of epithelial cell origin, such as anaplastic or undifferentiated carcinomas. Immunolabeling for endothelial cell markers, including CD31 and factor VIII–related antigen, can confirm the vascular origin of neoplastic cells in EHs. 7 However, histological features of EH frequently preclude a straightforward diagnosis on routine evaluation, especially when unaccompanied by immunolabeling for vascular markers.1,7,14,20 This can be further complicated by the fact that cytokeratin (CK) immunolabeling has been documented in epithelioid angiosarcomas in humans. However, pan-CK expression in EHs in veterinary species has not been documented to date. 7
In this retrospective study, we present findings on EHs in dogs retrieved from the diagnostic pathology submissions across 5 institutions. Our investigation focused on assessing cytoplasmic immunolabeling of CK AE1/AE3, considering both the percentage and intensity of immunolabeled cells. In addition, we conducted a comparative analysis of CK AE1/AE3 immunolabeling between conventional hemangiosarcomas and EHs to discern potential distinctions in expression patterns.
Cases were selected from biopsy and autopsy specimens from dogs submitted for pathological analysis across 5 institutions from 2009 to 2023 (Table 1). The inclusion criteria included cases with a morphological diagnosis of “epithelioid” and “hemangiosarcoma” in dogs, at any anatomical location, using the definition available in the literature. 14 Ten additional cases of conventional hemangiosarcoma were selected from the Histology Laboratory archive at Auburn University to be used as controls (Supplemental Table S1).
Signalment, anatomical location, immunohistochemical profile, and histological features and of epithelioid hemangiosarcomas in dogs.
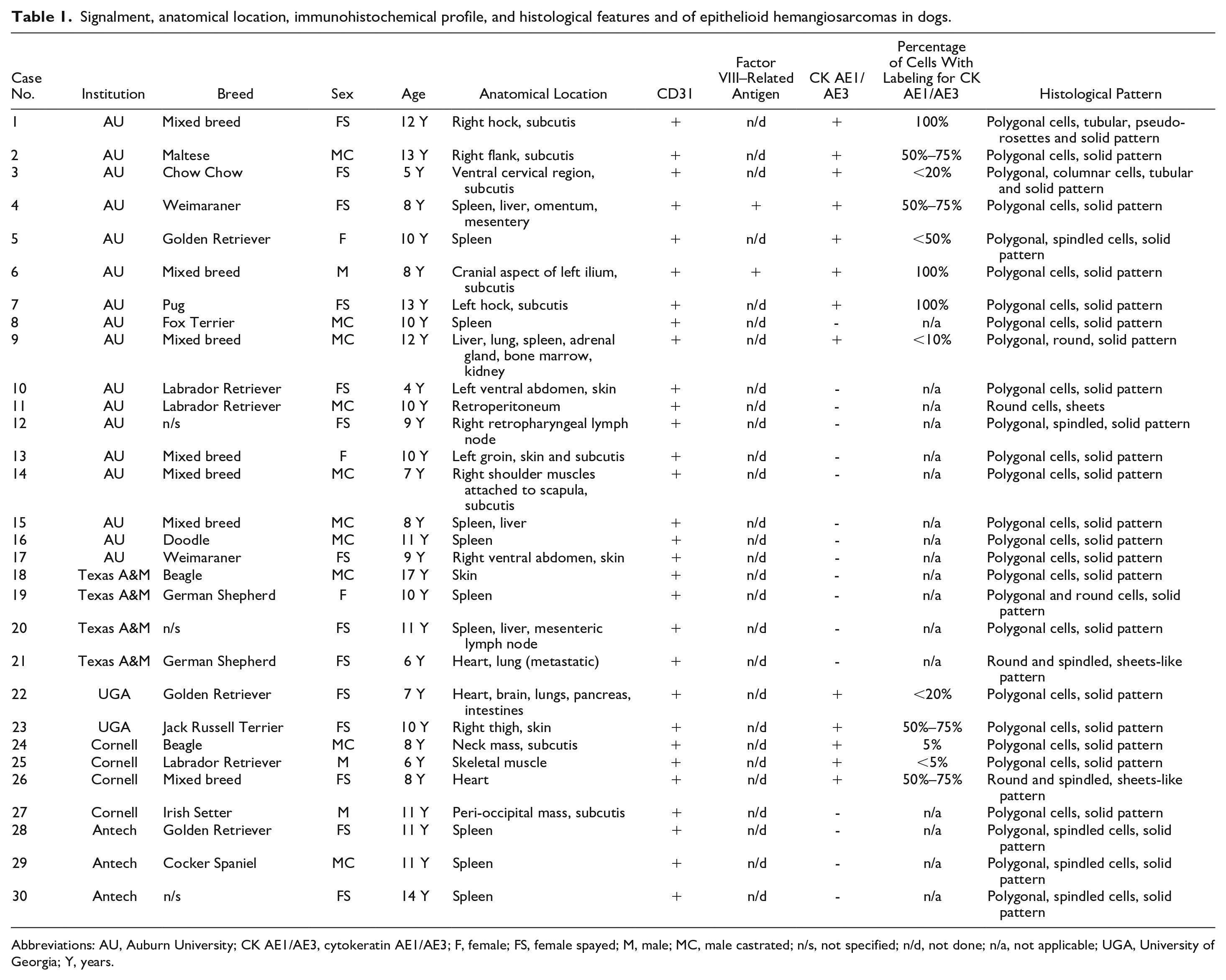
Abbreviations: AU, Auburn University; CK AE1/AE3, cytokeratin AE1/AE3; F, female; FS, female spayed; M, male; MC, male castrated; n/s, not specified; n/d, not done; n/a, not applicable; UGA, University of Georgia; Y, years.
Formalin-fixed paraffin-embedded tissues underwent immunohistochemical analysis with primary antibodies against CK AE1/AE3 (mouse monoclonal, anti-human AE1/AE3, prediluted, clone AE1/AE3, catalog no 901-011-091620; BioCare Medical, 60 Berry Dr Pacheco, CA 94553, US) and CD31 (mouse monoclonal, anti-human CD31, clone JC70A, catalog no GA610, Agilent Dako Omnis, Santa Clara, CA 95051, US). The BioCare Medical IntelliPATH FLX Automated Immunohistochemistry Staining System was employed following the manufacturer’s guidelines. CK AE1/AE3 was used in its prediluted form, while CD31 was applied at a 1:200 dilution in DaVinci Green Diluent (catalog no 901-PD900-070517; BioCare Medical). Tissue sections, 4-µm thick, were deparaffinized with xylene (catalog no XY004; Cancer Diagnostics Inc, Durham, NC 27703, US). Heat-induced epitope retrieval was conducted for 16 minutes at 110°C using citrate buffer pH 6.0 (Reveal Decloaker 10×, catalog no 901-RV1000X-071117; BioCare Medical). Endogenous peroxidase activity was blocked with a 3% peroxide solution for 5 minutes (catalog no 901-PX968-071417; BioCare Medical). A blocking reagent (serum-free casein block) was applied to the sections and incubated at room temperature for 5 minutes (Background Punisher, catalog no 901-BP974-081417; BioCare Medical). Primary antibodies were applied to the slides for 30 minutes. Subsequently, the horseradish peroxidase–labeled goat anti-mouse micro-polymer secondary antibody (MACH 2 Mouse HRP-Polymer, catalog no 901-MHRP520-112017; BioCare Medical) IgG reagent was applied for 10 minutes. Tissues were developed with 3, 3-diaminobenzidine chromogen (catalog no 901-BDB2004-082417; BioCare Medical) for 5 minutes. Slides were counterstained with hematoxylin (catalog no 901-CATHE-081117; BioCare Medical) for 1 minute, followed by dehydration, clearing, and mounting. Sections of haired skin from the respective species were used as CK AE1/AE3-positive controls. Vascular neoplasms, including hemangiosarcomas and hemangiomas from the respective species, were used as CD31-positive controls. Avidin-biotin blocking procedures are eliminated when using the MACH 2 conjugated secondary antibodies, to eliminate the requirement for negative reagent controls in immunohistochemistry. In addition, nonvascular tissues within the examined sections were assessed as internal negative controls. Immunolabeling distribution was further categorized in percentages (0%–100%) based on the percent area immunolabeled as assessed by direct visualization in 10 standardized high-power (400×) fields (2.37 mm2). The evaluation was performed individually by 2 board-certified anatomical pathologists (D.F.B.M. and M.S.) and further compared to reach a consensus.
A total of 30 cases were retrieved (Table 1), consisting of 14 spayed females (47%), 10 neutered males (33%), 3 intact females (10%), and 3 intact males (10%). The mean age of dogs was 9.7 years (range: 4–17 years, median: 10 years), with 18 dogs between 10 and 17 years and 13 dogs between 4 and 9 years. The most commonly represented breeds were the Golden Retriever (n = 3) and Labrador Retriever (n = 3), followed by German Shepherd (n = 2), Beagle (n = 2), and Weimaraner (n = 2). In addition, there were 7 mixed-breed dogs, and in 3 cases, the breed was not specified. The most common anatomical location in dogs was the spleen (n = 11) as a solitary lesion (n = 7) or part of widespread or systemic disease (n = 4). Eight dogs had a primary subcutaneous lesion, and 5 dogs had a primary cutaneous lesion. Three dogs had EHs in the heart as a solitary lesion (n = 1) or as a part of widespread hemangiosarcoma affecting the lungs, brain, pancreas, and/or intestine (n = 2). Other anatomical locations included skeletal muscle (n = 1), retroperitoneum (n = 1), and right retropharyngeal lymph node (n = 1). In our study, 50% of the EHs originated from visceral sites (spleen, widespread, or lymph node) and 50% arose from soft tissues (cutaneous, subcutaneous, muscular, and retroperitoneal). These results are consistent with existing literature, which identifies the spleen, subcutis, skeletal muscle, and retroperitoneum as the most frequent location for EHs. 14 Positive CK AE1/AE3 immunolabeling was present in 75% of subcutaneous EHs (6/8), 27% of splenic EHs (3/11), 66% of cardiac EHs (2/3), 100% of skeletal muscle EHs (1/1), and 20% of cutaneous EHs (1/5). Interestingly, 62% (8/13) of CK AE1/AE3 immunolabeled cases were from the soft tissues (subcutis, skin, and skeletal muscle) while the remaining cases were identified in visceral organs.
Histologically, neoplastic cells predominantly displayed a polygonal morphology, forming a solid pattern (n = 18; cases 2, 4, 6–8, 10, 13–18, 20, 22–25, and 27). In addition, some cases contained a mixture of different cellular morphologies, specifically polygonal and spindle cells forming a solid pattern (n = 5, cases 5, 12, and 28–30) (Table 1, Fig. 1a–f). Polygonal cells had eosinophilic cytoplasm with indistinct cell borders and paracentral nuclei. Spindle cells had scant to moderate fibrillar cytoplasm with distinct cell borders. In the solid histological pattern, vascular cleft formation was minimal to absent. Mixed growth arrangement patterns were observed in 4 cases (cases 9, 19, 21, and 26). Two cases displayed tubular and pseudorosette formations (cases 1 and 3), while one case (case 11) had a round cell morphology with neoplastic cells arranged in a sheet-like pattern. In the tubular pattern, tall columnar or polygonal cells were arranged in tubule-like structures surrounding blood-filled lumens.
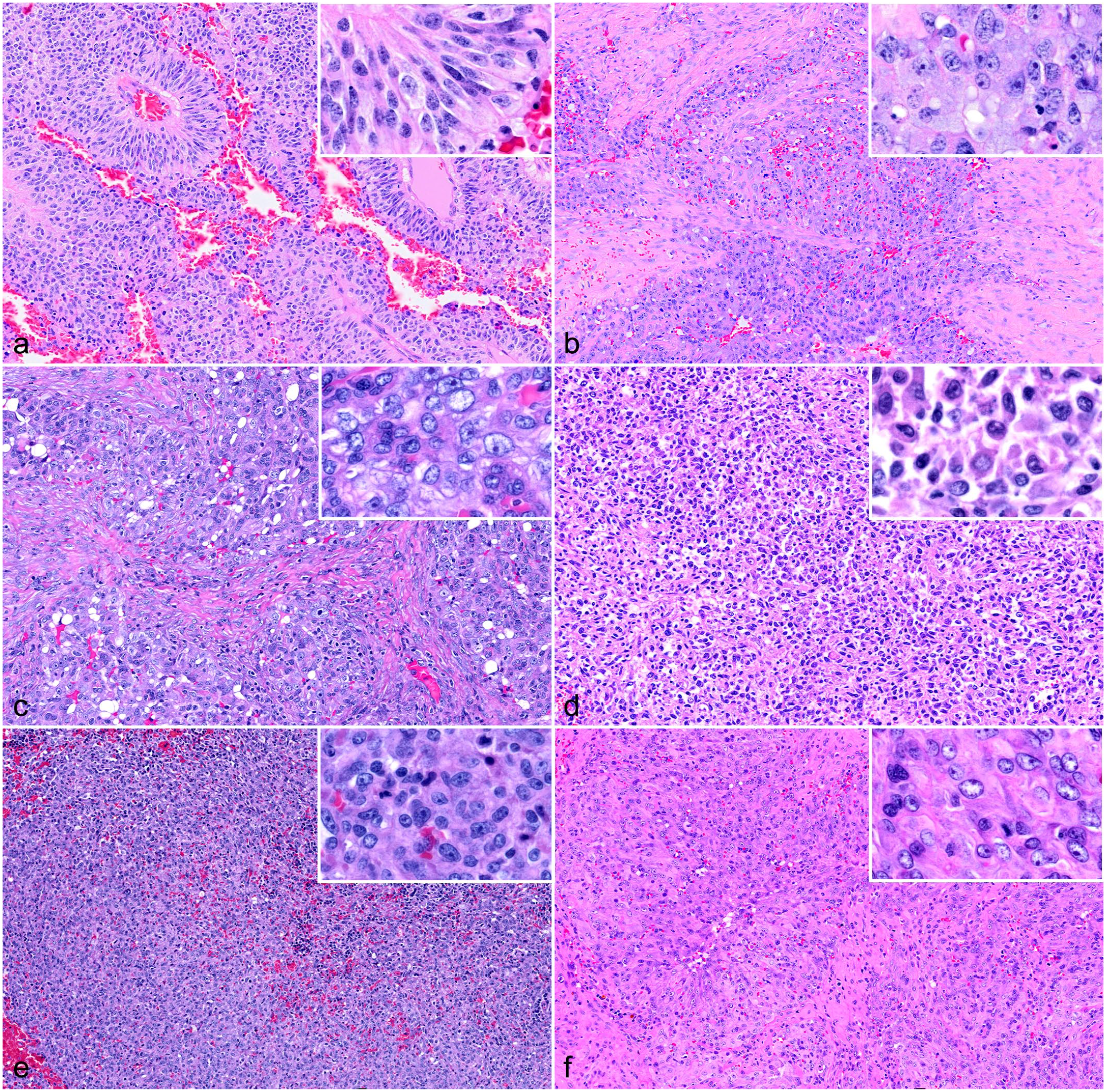
Histopathological features of epithelioid hemangiosarcoma in dogs. Hematoxylin and eosin. (a) Ventral cervical region, subcutis. Neoplastic cells are polygonal and columnar and are arranged in pseudorosette structures. Case 3. (b) Right flank, subcutis. Polygonal cells form a solid pattern with minimal small vascular slits. Case 2. (c) Cutaneous mass. Polygonal neoplastic endothelial cells arranged in a solid pattern with minimal small vascular slits. Case 18. (d) Heart mass. Round and spindled neoplastic cells arranged in a sheet-like pattern. Case 21. (e) Splenic mass. Polygonal cells form a solid pattern with minimal small vascular slits. Case 20. (f) Splenic mass. Polygonal neoplastic endothelial cells forming a solid pattern. Case 8. Insets highlight tumor cell morphologies.
Cytoplasmic immunolabeling with CK AE1/AE3 was observed in 13 cases (43%) (Table 1; Fig. 2; Supplemental Fig. S1a, b). The proportion of immunolabeled cells varied among cases, ranging from 5% to 100%. Three cases (23%; cases 1, 6, and 7) had 100% cytoplasmic immunolabeling in polygonal cells arranged in tubular and solid histological patterns. In addition, 4 cases (31%; cases 2, 4, 23, and 26) had 50% to 75% cytoplasmic immunolabeling in polygonal, round, and spindle cells arranged in solid and sheet-like patterns. Finally, a total of 6 cases (46%; cases 3, 5,9, 22, 24, and 25) had <50% of cytoplasmic immunolabeling in polygonal, columnar, round, and spindle cells arranged in tubular and solid patterns (Fig. 2a–f). All EH cases and selected cases of conventional hemangiosarcomas had 100% immunolabeling for CD31, confirming the endothelial origin of neoplastic cells and supporting the diagnosis of hemangiosarcoma (Table 1 and Supplemental Table S1) (Supplemental Fig. S1). CK AE1/AE3 immunolabeling was not observed in canine conventional hemangiosarcomas.
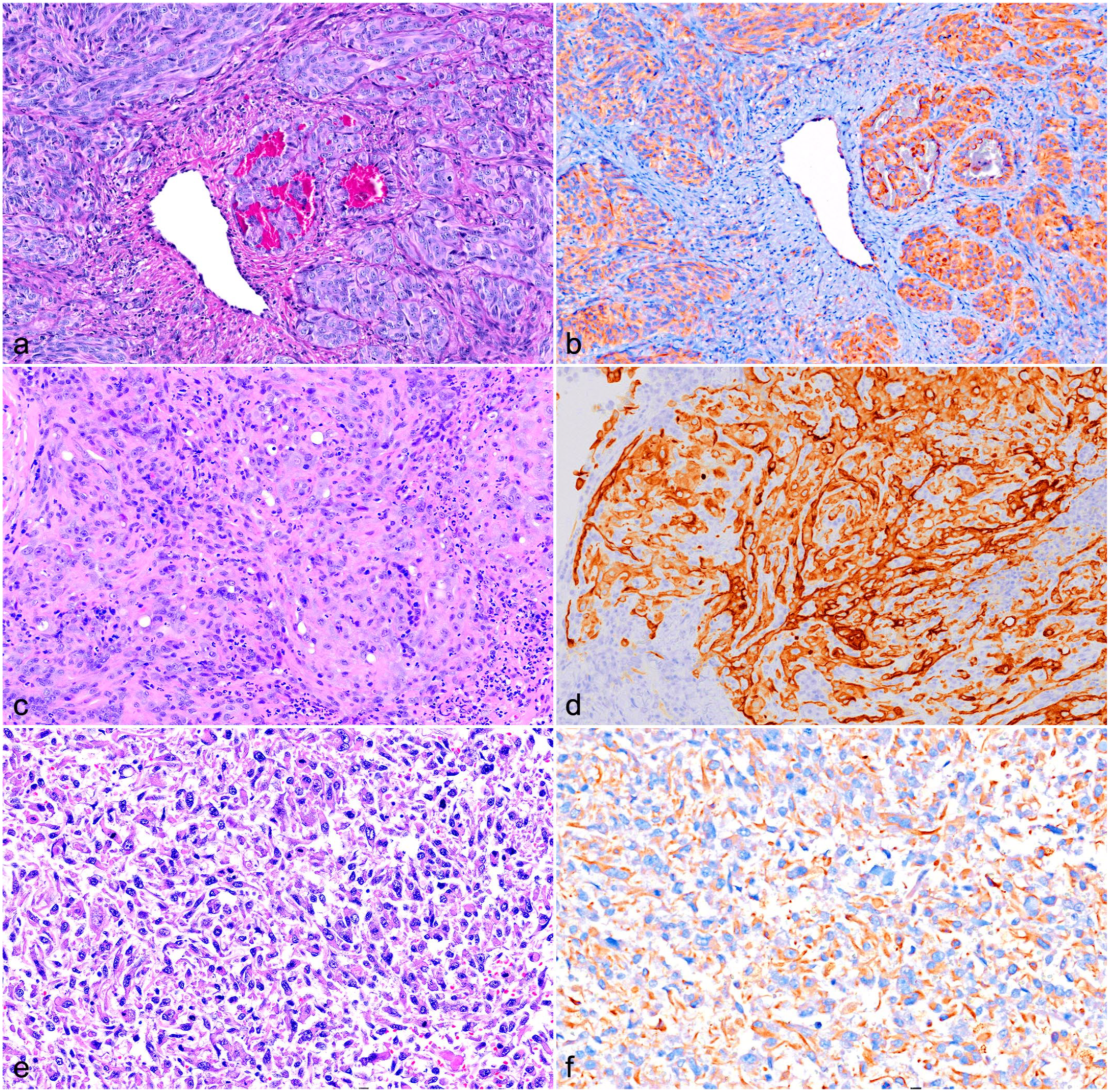
Epithelioid hemangiosarcoma, dog. (a) Right hock, subcutis, case 1. Hematoxylin and eosin (HE). (b) Strong cytoplasmic immunolabeling in 100% of neoplastic cells. Case 1 Immunohistochemistry (IHC) for cytokeratin AE1/AE3 (AE1/AE3). (c) Left hock, subcutis, case 7. HE. (d) Strong cytoplasmic immunolabeling in 100% of neoplastic cells. Case 7. IHC for AE1/AE3. (e) Heart mass, case 26. HE. (f) Strong cytoplasmic immunolabeling in >50% of neoplastic cells. Case 26. IHC for AE1/AE3.
In our study, CK AE1/AE3 immunolabeling was detected in 43% of EHs (13/30), a finding that starkly contrasts with existing veterinary literature, where CK AE1/AE3 immunolabeling in EHs has not been previously documented.1,2,4,5,12,13,15,18,20 However, these findings align with the CK expression patterns observed in human angiosarcomas. 6 In a comprehensive study comprising 18 human epithelioid angiosarcomas, approximately 50% of the tumors showed positive immunolabeling for one of the CK isoforms. Specifically, CK8 and CK18 were detected in 50% of cases, CK7 in 35%, and CK19 in only one case. 11 All cases lacked immunolabeling for CK14 and CK34βE12. 11 Similarly, 2 other case series reported CK AE1/AE3 and CAM 5.2 immunolabeling in 67% of primary cutaneous epithelioid angiosarcomas and 100% of thyroid epithelioid angiosarcomas, respectively.10,17
In contrast to our findings, nonepithelioid vascular tumors in humans can express CK18, CK8, and CK7. 11 None of the 10 conventional hemangiosarcoma cases used as controls in our study had immunolabeling for CK AE1/AE3. This discrepancy may be attributed to the different specificities of primary antibodies. In our study, the CK expression was evaluated using the monoclonal mouse CK AE1/AE3 antibody, which detects a mix of low-molecular-weight and high-molecular-weight CKs. Specifically, the AE1 antibody recognizes high-molecular-weight CKs 10, 13, 14, 15, and 16 and low-molecular-weight CK19. The AE3 antibody recognizes high-molecular-weight CKs 1, 2, 3, 4, 5, and 6 and low-molecular-weight CK7 and CK8.3,9 However, CK17 and CK18 are not recognized by this antibody.3,9 Cytokeratin isoforms commonly expressed in human epithelioid angiosarcomas include CK8, CK18, and less frequently CK7 and CK19, all of which fall within the range of the AE1/AE3 antibody spectrum, except for CK18. CK18, the predominant isoform detected in human nonepithelioid vascular tumors, is outside the spectrum recognized by our primary antibody. This could explain the absence of CK AE1/AE3 immunolabeling in conventional hemangiosarcomas in dogs.
In human studies, CK7 and CK18 immunolabeling has also been observed in normal endothelial cells within veins, venules, and lymphatics in the skin, subcutis, gastrointestinal, and reproductive tract. 11 However, there are no data on CK isoforms expressed in normal endothelial cells in veterinary species. Therefore, systematic studies are critically needed to elucidate the expression profile of different CK isoforms in the normal endothelium of domestic animals. Such studies could provide valuable insights into the expected patterns of CK immunolabeling in various vascular neoplasms.
In conclusion, 43% (13/30) of canine EHs exhibited CK AE1/AE3 immunolabeling, mirroring patterns observed in human cases. Anatomical pathologists should be aware of this feature to avoid misdiagnosing a carcinoma (primary or secondary) instead of recognizing it as a vascular tumor. Diagnosing EH in small incisional biopsies, particularly from cutaneous and subcutaneous tissues, can be challenging due to cellular pleomorphism, with differential diagnoses including undifferentiated carcinoma, epithelioid hemangioendothelioma, and epithelioid amelanotic melanoma. To prevent diagnostic errors, it is crucial to include specific vascular immunohistochemical markers, such as CD31, in addition to CK AE1/AE3 when EH is suspected.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241273268 – Supplemental material for Cytokeratin AE1/AE3 immunolabeling in epithelioid hemangiosarcoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858241273268 for Cytokeratin AE1/AE3 immunolabeling in epithelioid hemangiosarcoma by Daniel Felipe Barrantes Murillo, Tatiane Terumi Negrão Watanabe, Dominique J. Wiener, Andrew Miller, Daniel R. Rissi and Maninder Sandey in Veterinary Pathology
Footnotes
Acknowledgements
We thank Lisa Parsons, Lisa Jolly, and Keisha Snerling from the Histology Laboratory of Auburn University for technical assistance and slide preparation for the immunohistochemistry procedures.
Author Contributions
M.S. designed the study and D.F.B.M. contributed to the experimental design and supervision of the study. D.F.B.M., T.T.N.W., D.J.W., A.M., D.R.R., and M.S. collected the data, and performed the histopathological evaluations. M.S. and D.F.B.M. performed the immunohistochemical evaluation and data analysis. D.F.B.M. and M.S. wrote the manuscript with the collaboration of all the authors. All the authors reviewed and critically edited the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.T.N.W. is employed by Antech Diagnostics and Mars Petcare Science & Diagnostics. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
