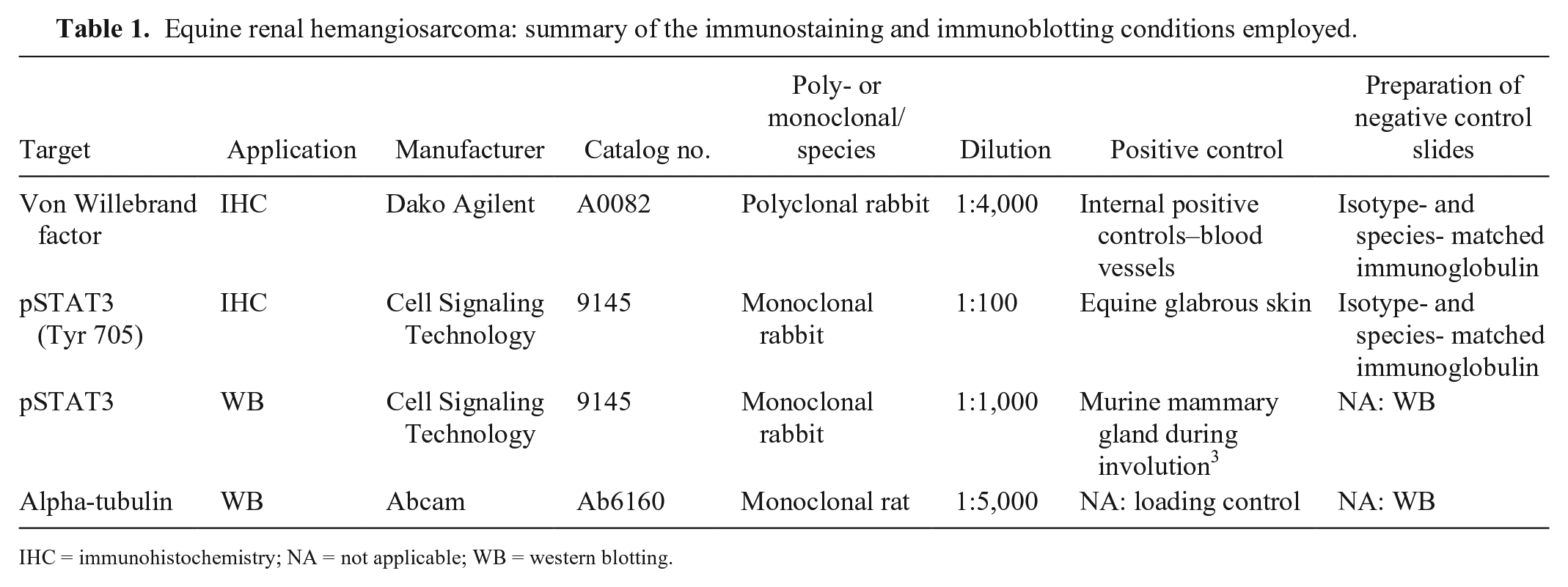
Abstract
Hemangiosarcoma is an uncommon tumor in horses. We characterized 3 cases of equine renal hemangiosarcoma, focusing on clinical and pathologic features, and describe occurrence of the epithelioid variant of hemangiosarcoma in one of these cases. Nuclear expression of phosphorylated STAT3 (pSTAT3) was assessed to analyze potential inappropriate STAT3 activation as a component of tumor pathogenesis. Clinical signs in the 3 horses included insidious weight loss, followed in one case by serosanguineous nasal discharge and terminal epistaxis, and nonspecific signs of abdominal pain. Two of the hemangiosarcomas had a classical histopathologic appearance; in the other, neoplastic cells were polygonal and were arranged in densely packed sheets, resembling the epithelioid variant. Cross-reactivity of a pSTAT3 antibody was established by demonstration of pSTAT3 expression in the epithelium of glabrous skin by immunoblotting and immunohistochemistry. In the epithelioid hemangiosarcoma, ~40% of neoplastic cells exhibited nuclear pSTAT3 expression, but in the other 2 cases, expression was weak and variable in the neoplastic population, although stromal cell pSTAT3 activity was evident in pulmonary metastases in one case.
Equine hemangiosarcomas are uncommon, and may appear as solitary masses exhibiting various degrees of local invasion, or as masses affecting multiple organs.20,22 An epithelioid variant of hemangiosarcoma, rarely observed in horses, is characterized by neoplastic cells with epithelioid morphology, sporadic occurrence of cytoplasmic vacuoles, and variable growth patterns.21,24 Examples of this epithelioid subset have been recorded in various sites in the horse, including the deep dermis of the ventral neck, 24 ocular tissues, 1 the nasal cavity and submandibular lymph nodes, 5 and the vulval commissure. 10
There is relative paucity of information in the literature concerning the clinical presentation and pathologic features of renal hemangiosarcomas in horses. In a study of 35 cases of equine disseminated hemangiosarcoma, only one patient was confirmed as having a primary tumor in the kidney, although 9 of the subjects had renal involvement. 20 Other reports have documented single animals with renal metastasis, or renal involvement with an unidentified primary tumor site.4,8,14
Signal transducers and activators of transcription (STATs) are key transcription factors that coordinate critical processes such as cell division, differentiation, and survival. Aberrantly enhanced or uncontrolled signaling from specific STATs, in particular STAT3 and STAT5, can have a directly oncogenic effect through promotion of cellular proliferation and prevention of cell death. 2 STATs are indirectly activated by cytokines; constitutive activation of STAT3 may be secondary to activation of tyrosine kinases. One mechanism of such tyrosine kinase activation is via paracrine or autocrine sources of inappropriate cytokine signaling. 2 We have previously described nuclear localization of STAT3 in equine mammary tumors, 12 and STAT3 expression has been documented in canine hemangiosarcomas. 19 We therefore hypothesized that STAT3 activity may be important in the pathogenesis of equine hemangiosarcomas, and particularly in those arising in the kidney. STAT3 is generally activated by transient phosphorylation, most usually on tyrosine residues, before dimerization and nuclear translocation. 7 Consequently nuclear localization, or specific detection of phosphorylated STAT3 (pSTAT3), may indicate transcriptional activity.
We characterized 3 cases of equine renal hemangiosarcoma, focusing on clinical and pathologic features, and describe occurrence of the epithelioid variant of hemangiosarcoma in one of these cases. We assessed nuclear expression of pSTAT3 to explore the potential contribution of aberrant STAT3 signaling to tumor development.
The first case was presented to the University of Cambridge Equine Hospital (Cambridge, UK) and was subsequently examined postmortem. No previous cases of equine renal hemangiosarcoma were recorded in this institution’s anatomic pathology database. Two further cases were retrospectively identified from a search of the histopathology and autopsy archives for 2006–2016 at Rossdales Laboratories (Suffolk, UK). For each case, data regarding signalment, history, clinical presentation, and pathologic findings, were collated. At the time of original diagnosis, tissues were fixed in 10% neutral-buffered formalin, processed, sectioned, and stained with hematoxylin and eosin following standard histologic protocols. Immunohistochemistry (IHC) for von Willebrand factor (vWF; Dako Pathology/Agilent Technologies LDA UK, Cheadle, Cheshire, UK; dilution 1:4,000) was performed at the time of diagnosis for 2 cases and retrospectively for the remaining case.
Western blotting was carried out to corroborate species cross-reactivity for the pSTAT3 antibody used in our study and was performed on glabrous skin collected from a surgical biopsy sample, from an equine phallectomy, submitted to the University of Cambridge, Department of Veterinary Medicine, anatomic pathology service. Upon receipt of the biopsy sample, a small piece of macroscopically normal penile skin was immediately frozen at −80°C. Western blotting for pSTAT3 (Cell Signaling Technology, Danvers, MA) and alpha-tubulin (as a loading control; Abcam United Kingdom, Cambridge, UK) was performed following standard protocols for protein extraction and immunoblotting. 15 Murine mammary gland sampled during involution was used as a positive control for western blotting. 3
IHC for pSTAT3 (Cell Signaling Technology; dilution 1:100) was performed specifically for our study. Immunohistochemical staining followed a routine protocol using an automated IHC system as described previously 11 (Table 1). Reticulin staining, to specifically characterize the arrangement of cells in the epithelioid variant of the tumor, 24 was undertaken on horse 2, and followed a standard protocol. All histologic slides were reviewed by 2 pathologists (K Hughes and AK Foote).
Equine renal hemangiosarcoma: summary of the immunostaining and immunoblotting conditions employed.
IHC = immunohistochemistry; NA = not applicable; WB = western blotting.
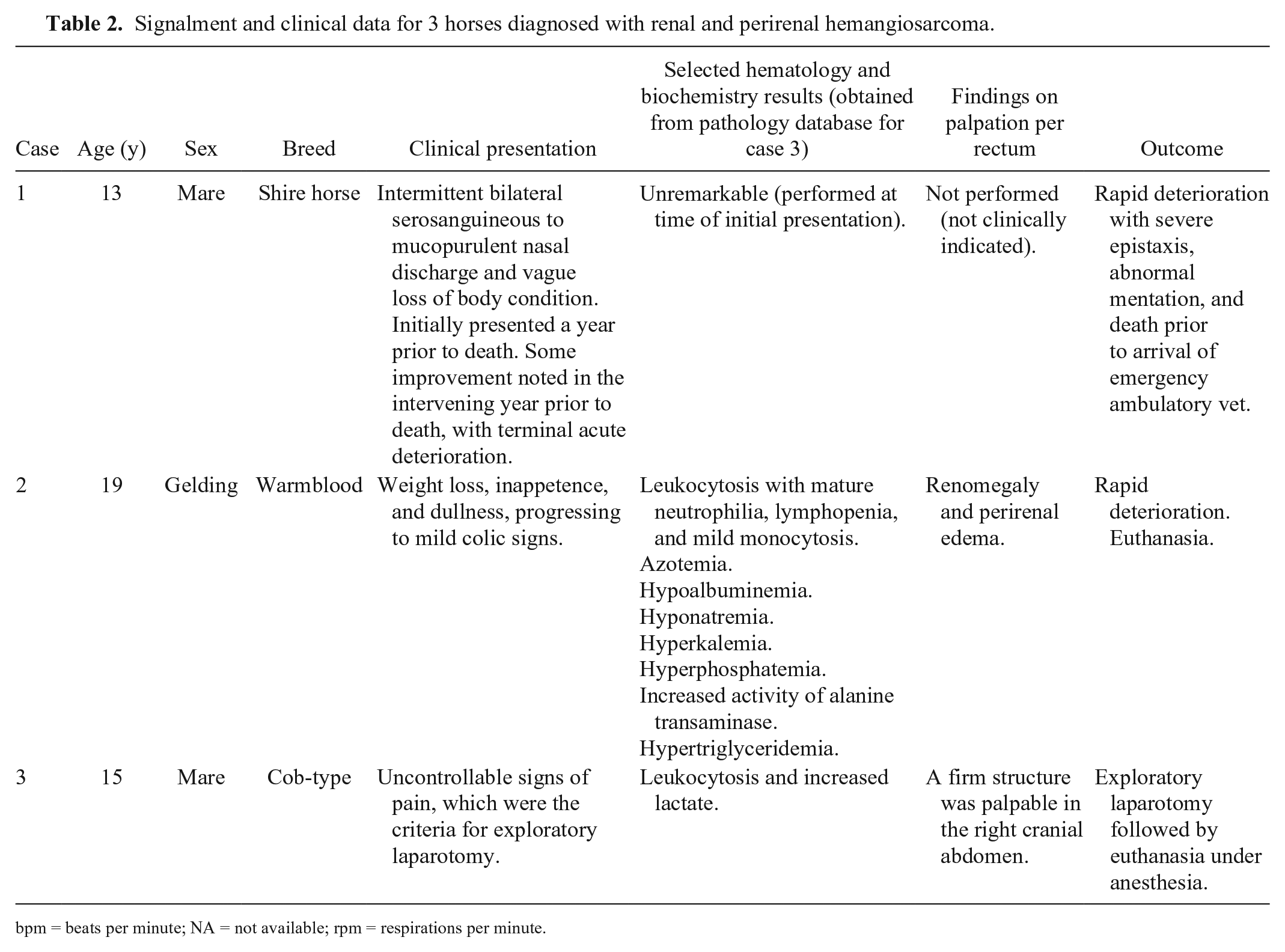
The mean and median ages of the horses were 15.7 and 15 y, respectively (Table 2). The clinical presentation and clinical findings in the patients varied, although some features were common to more than one case. Horse 1 exhibited nonspecific weight loss and intermittent serosanguineous-to-mucopurulent nasal discharge over the course of a year, with a terminal acute episode of severe epistaxis.
Signalment and clinical data for 3 horses diagnosed with renal and perirenal hemangiosarcoma.
bpm = beats per minute; NA = not available; rpm = respirations per minute.
Horse 2 also exhibited weight loss, nonspecific dullness, and discomfort. The horse had a “pot-bellied” appearance, and ascites was suspected. Abdominocentesis revealed clear and colorless fluid, with total protein <2.0 g/dL, a few normal neutrophils, and no atypical cells. Accumulations of subcutaneous edema were noted during the clinical examination.
Horse 3 had signs of abdominal discomfort refractory to routine analgesia, an elevated heart rate and elevated respiratory rate leading to a clinical suspicion of an intra-abdominal lesion. Exploratory laparotomy revealed hemorrhagic peritoneal fluid and hematomas of various sizes. A 300 × 250 × 150 mm, firm mass, with the texture and appearance of a large hematoma, was present in the right dorsal aspect of the abdomen. The owner elected to euthanize the patient prior to recovery from general anesthesia.
Hematology and biochemistry findings were varied, with unremarkable results for horse 1, biochemistry consistent with renal failure in horse 2, and leukocytosis and elevated lactate in horse 3.
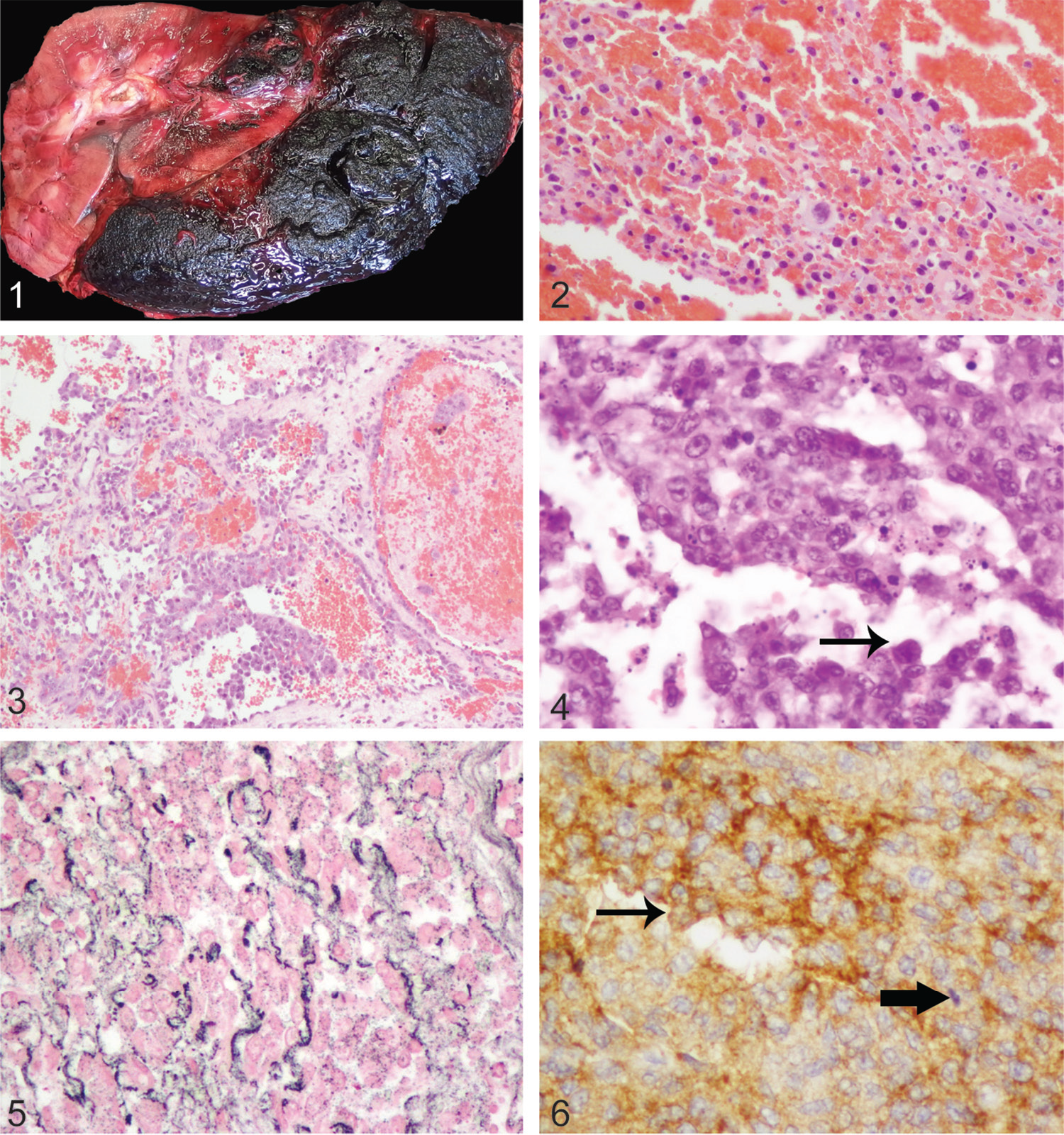
Postmortem examination of horses 1 and 3 was carried out by a pathologist at a pathology facility, whereas postmortem examination of horse 2 was conducted under field conditions by an equine veterinarian. The left kidney of horse 1 was found to be expanded by a 360 × 220 × 140 mm, dark-red to black, friable mass that extended beyond the renal capsule (Fig. 1), and the lung had myriad 3–10 mm diameter, moderately firm, moderately well-demarcated but unencapsulated dark-red foci (Supplementary Fig. 1). The gross diagnosis was primary hemangiosarcoma of the left kidney with pulmonary metastases.
Equine renal hemangiosarcoma.
Postmortem examination for horse 2 revealed >20 L of clear, colorless abdominal fluid (interpreted as ascites), perirenal edema, and irregular enlargement of the left kidney. No other abnormalities were noted. The presumptive gross diagnosis made by the attending veterinarian was a renal tumor, and samples were collected for histopathology.
Examination of horse 3 revealed a poorly demarcated, cavitated mass infiltrating the left ilium, which extended into the periosteal tissue and adjacent muscle. Variably sized, clearly delineated, unencapsulated, dark-red masses with capsular and perirenal extension were present in the kidneys bilaterally, and were considered to have led to a large right perirenal hematoma and hemoabdomen (Supplementary Fig. 2). Similar smaller masses were present multifocally in the sublumbar lymph node, liver, and lung, although the pulmonary masses were considerably less numerous than in horse 1. The gross diagnosis was primary hemangiosarcoma of the left ilium with metastases to the sublumbar lymph node, liver, kidney, and lung.
In horses 1 and 3, tumor masses were typical of hemangiosarcoma, 14 with bundles and streams of spindle-shaped to polygonal neoplastic cells forming variably distinct blood-filled spaces and clefts, interspersed with solid cellular foci (Figs. 2, 3; Supplementary Figs. 3, 4). Neoplastic cells exhibited a variable degree of punctate cytoplasmic immunohistochemical staining for vWF. In the primary renal mass in horse 1, there was also considerable hemorrhage, fibrin, and necrosis, which necessitated examination of a large number of histologic sections prior to identification of small foci of neoplastic cells.
In the perirenal tissue collected postmortem from horse 2 by the referring veterinarian, the neoplastic cells in a locally extensive focus had an arrangement similar to those already described. However, in ~60% of the tissue examined, the neoplastic cells were more closely packed in dense sheets, and indistinct clusters and cords (Fig. 4). Reticulin staining demonstrated that multifocally the cells were aligned upon a basal lamina, and hence the cords formed vague slit-like spaces (Fig. 5). The neoplastic cells were medium-sized, and had indistinct cell borders with a small-to-moderate amount of pale eosinophilic cytoplasm, which occasionally exhibited one or more vacuoles, most frequently smaller than the nucleus. Nuclei were round-to-oval and were prominent, with coarsely stippled to vesicular chromatin and most frequently one large, prominent, basophilic nucleolus. The mitotic rate was high, with 104 mitoses in 10 high-power fields. Large numbers of lymphocytes were present multifocally and there were also areas of necrosis. The cells exhibited moderate-to-marked intensity, diffuse, cytoplasmic staining for vWF (Fig. 6). The mass was diagnosed as an epithelioid hemangiosarcoma.
Human epidermis has been shown to express high levels of STAT3 17 and therefore the epidermis of equine glabrous skin was used to assess cross-reactivity of the pSTAT3 antibody used in our study with equine tissue. High levels of expression of epithelial pSTAT3 in equine glabrous skin were noted, a clear nuclear staining pattern was observed, and not every nucleus was positive, suggesting specificity of staining (Fig. 7). Immunoblotting for pSTAT3 on glabrous skin collected from adjacent to the histologic section revealed an appropriately sized band (Fig. 8), further corroborating cross-reactivity of the antibody.
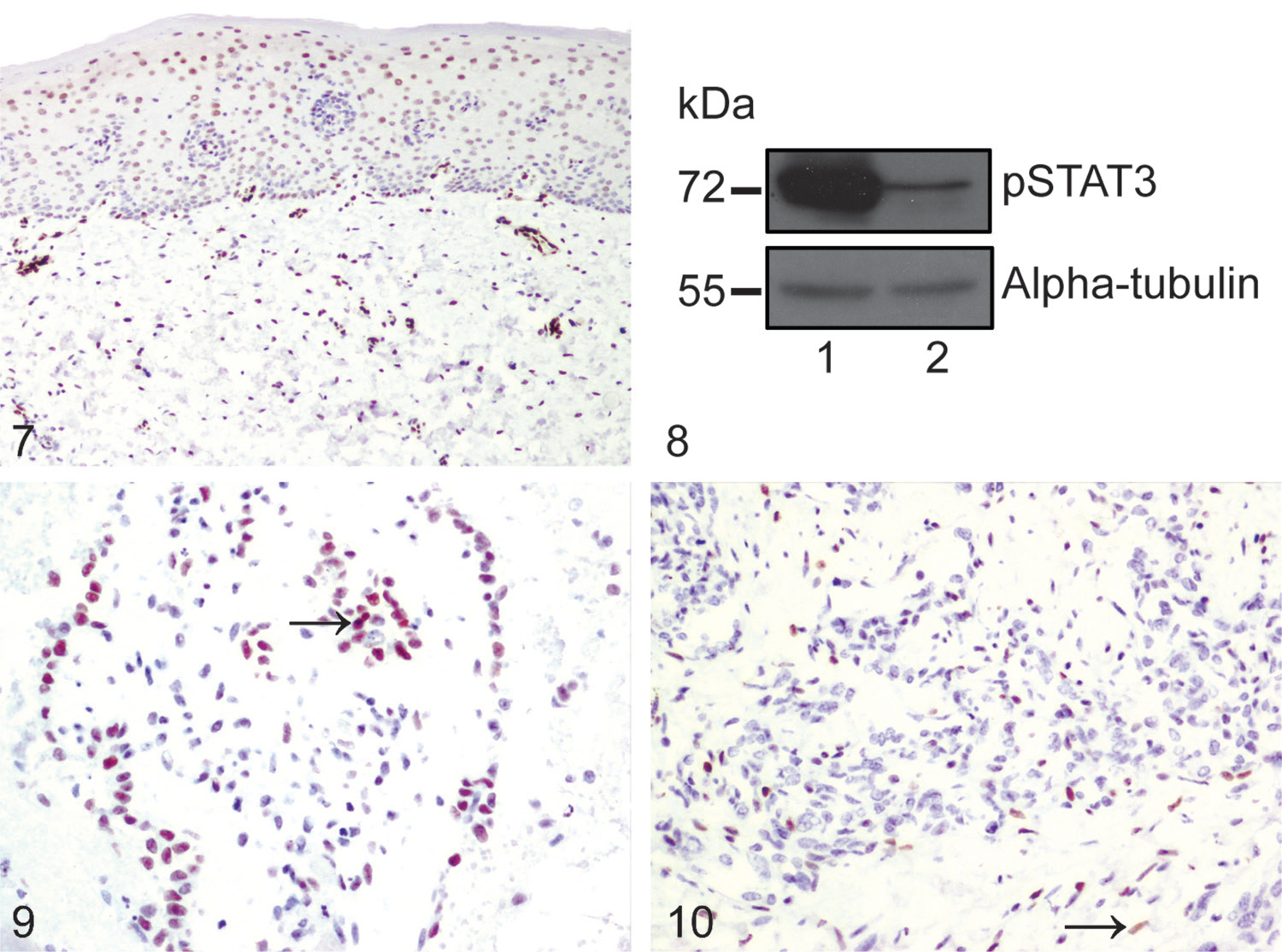
Phosphorylated STAT3 (pSTAT3) expression in equine glabrous skin, renal hemangiosarcoma, and pulmonary metastases. For Figures 7, 9, and 10, immunohistochemical staining for pSTAT3 with hematoxylin counterstain.
In horse 1, very few neoplastic cells within the renal mass or pulmonary metastases exhibited positive nuclear staining for pSTAT3, although occasional neoplastic cells had weak-positive nuclear staining. In horse 2, ~40% of the neoplastic cells in the foci exhibiting typical histopathologic features of hemangiosarcoma had moderate-to-intense nuclear staining for pSTAT3 (Fig. 9). In horse 3, most of the neoplastic cells in the kidney were negative for pSTAT3; scattered cells in the lungs exhibited moderate nuclear immunoreactivity, but most of these were considered to be stromal cells rather than neoplastic cells based on morphology alone (Fig. 10).
Clinical diagnosis of hemangiosarcoma in the horse can be difficult,20,22 and it is noteworthy that the 3 cases described herein were diagnosed postmortem, consistent with previous assertions that this tumor type has a low rate of antemortem diagnosis.4,20 The clinical characteristics of the cases documented here varied, although some similarities were noted. In horses 1 and 2, weight loss formed an important facet of the clinical picture at initial presentation, and this emphasizes that abdominal neoplasia should be considered as a clinical differential diagnosis for weight loss when other more likely possibilities have been ruled out. 6 Others have suggested that a comprehensive diagnostic investigation of a horse with weight loss should include abdominocentesis with cytology, 6 but our finding that abdominal fluid from horse 2 did not reveal atypical cells is consistent with other authors’ suggestions that, in the case of abdominal, or specifically renal, hemangiosarcoma, cytologic examination of peritoneal fluid may be unrewarding. 22
In 2 of our 3 cases, the final diagnosis included a primary (horse 1) or metastatic (horse 3) renal hemangiosarcoma with pulmonary metastasis. Horse 1 had epistaxis, which is similar to a previous report, 14 suggesting that hemangiosarcoma with metastatic spread to the lungs should be considered as a possible etiology for epistaxis, again when other more likely causes have been excluded.
In horse 3, the site of the primary neoplasm was considered to be the left ilium, with marked local invasion of the surrounding muscles apparent at postmortem examination. Hemangiosarcoma of the equine pelvis has been reported previously, although interestingly, in that case there was no evidence of metastatic spread in spite of extensive local infiltration. 16 In our case, there were widespread metastases including to the kidneys, bilaterally, and the liver and lungs. In horse 3, the primary mass was identified on the basis of size and local infiltration, as has been described in a previous study. 20
In horse 1, most of the renal histologic sections comprised very extensive hemorrhage, fibrin, and necrosis, and identification of small foci of viable neoplastic cells was only achieved following examination of a large number of sections. Similarly, other authors describing hemangiosarcoma of the intercostal muscles and ribs in a horse strongly suspected renal involvement, but only noted hemorrhage and necrosis in renal sections. 23 Although we used IHC staining for vWF to confirm the histologic diagnosis, CD31 may also be used as an IHC marker for hemangiosarcomas, and has been used in previous studies.1,24
Horse 2 had an epithelioid variant of hemangiosarcoma, a specific histopathologic pattern, which is established in the veterinary literature as a counterpart of the human epithelioid angiosarcoma.9,24 Angiosarcoma is a highly malignant neoplasm originating from vascular endothelium, and may arise as a primary tumor of the kidney. 18 Epithelioid variants of hemangiosarcoma in animals 24 and epithelioid angiosarcoma in humans 13 have plump, epithelioid cells forming small channels, which may contain erythrocytes. Our case was similar to previously described examples in that the cells had a very high mitotic rate, formed small bulging projections that impinged upon the spaces formed by the tissue clefts (“hobnail” projections), and occasionally had cytoplasmic vacuolation. It has been suggested previously that epithelioid hemangiosarcoma is notable as it may present a diagnostic challenge, 24 and here we record that renal hemangiosarcoma may exhibit this pattern in horses. Our example also had foci with a more typical histopathologic pattern of hemangiosarcoma, as suggested previously. 9
We hypothesized that STAT3 signaling may be activated inappropriately in equine hemangiosarcoma. We demonstrated that, as in human subjects, the epidermis of equine skin exhibits high levels of nuclear pSTAT3 expression and that the antibody used in our study detects a protein band of expected size by western blotting. One limitation of this study is that it would have been optimal to use western blotting, in addition to IHC, to analyze protein expression patterns of the renal hemangiosarcomas described herein. However, unfortunately, frozen fresh tissue specimens were not available from these cases, and excess tissue from a phallectomy, submitted to the surgical biopsy service, was considered the best available alternative to assess antibody cross-reactivity. 17
In spite of our confidence in the specificity of the IHC staining for pSTAT3, our results are not supportive of STAT3 signaling being a key driver of neoplastic transformation in this tumor type given that pSTAT3 staining was variable, and weak in 2 of the 3 cases. Variability of staining between cases may be related to differences in length of time of tissue fixation and thus variability in effectiveness of antigen retrieval, in addition to potential genuine differences in levels of expression of pSTAT3 between patients. Interestingly, ~40% of the neoplastic cells in horse 2 exhibited moderate-to-intense nuclear staining for pSTAT3, suggesting that, in some equine hemangiosarcomas, STAT3 signaling may play a role. Furthermore, in pulmonary metastatic foci in horse 3, scattered cells with morphology and location suggestive of stromal cells exhibited weak nuclear pSTAT3 expression, indicating that STAT3 signaling pathways may be active in the tumor microenvironment in some cases. This hypothesis is consistent with the suggestion that STAT3 has multiple biological functions in tumorigenesis. 25 Further investigation of this suggestion would require analysis of a larger sample set, using immunofluorescence to dual stain for pSTAT3 and a neoplastic cell marker to prove whether these cells were neoplastic or were, indeed, stromal cells.
Given our small number of cases, our discussion of the role of STAT3 signaling in equine renal hemangiosarcomas is speculative. Investigation of the pathogenesis of less common tumors requires coordinated multi-center studies. In addition, as we have demonstrated here in the context of antibody validation, prospective collection of frozen tissue in addition to formalin-fixed tissue samples facilitates employment of a wider range of molecular investigative techniques and will likely prove crucial in future analyses.
Footnotes
Acknowledgements
The data detailed in this manuscript formed the basis of a platform presentation at the 2016 ACVP Annual Meeting, New Orleans, LA, Dec 3–7, 2016. We thank the referring veterinary surgeons for submission of these cases. We gratefully acknowledge the technical assistance of V. Owenson and L. Grimson of the Department of Veterinary Medicine, University of Cambridge, in the preparation of tissue sections and for oversight of automated immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Research histopathology costs for pSTAT3 immunohistochemistry were supported by a 2015 Zoetis Clinical Research Prize awarded to KH. Publication costs were supported by the Queen’s Veterinary School Hospital, University of Cambridge, and Rossdales Laboratories.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
