Abstract
A 15-year-old female gray Appaloosa horse was presented with history of a mass over the right dorsal commissure of vulva for the past 7 months. Based on histopathological examination, and positive staining with factor VIII–related antigen, vimentin, and Verhoeff–van Gieson stain, the vulvar mass was diagnosed as hemangiosarcoma with marked solar elastosis.
Hemangiosarcoma is a malignant tumor of vascular endothelial cells. The cause of most hemangiosarcomas is unknown. It has been reported that chronic solar damage on lightly pigmented, sparsely haired dogs may contribute to development of cutaneous hemangiomas and hemangiosarcomas. 9 Hemangiosarcomas of the vulva and vestibulovagina are rarely reported in domestic animals. There is a single case report of vulvar hemangiosarcoma in a dog. 24 However, vulvar hemangiosarcoma has never been documented in the horse. The current report describes the histopathological features of vulvar hemangiosarcoma with solar elastosis in a horse.
A 15-year-old female, gray, pleasure/barrel Appaloosa racehorse was presented to the equine medicine service of Louisiana State University (LSU) Veterinary Teaching Hospital and Clinics (Baton Rouge, Louisiana) with history of a mass on the right dorsal commissure of vulva. The vulvar mass was present for approximately 7 months, and the size of the mass had progressed from thumb-sized to approximately 6 cm in diameter. The horse was confined in a pasture throughout the year. On physical examination, the horse was bright, alert, and responsive, and had all the vital parameters within normal limits. There was no evidence of vaginal discharge or hemorrhages. Clinically, a presumptive diagnosis of squamous cell carcinoma was made. The mass was surgically excised with wide margins, and tissue samples were submitted to Louisiana Animal Disease Diagnostic Laboratory (Baton Rouge, LA) for histopathological examination. Grossly, the vulvar mass (6 cm × 4 cm × 2 cm) was slightly firm, unencapsulated, ulcerated, and dark red with multifocal areas of necrosis (Fig. 1A). The tissue sections from the vulvar mass were fixed in 10% neutral buffered formalin, routinely processed, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin.
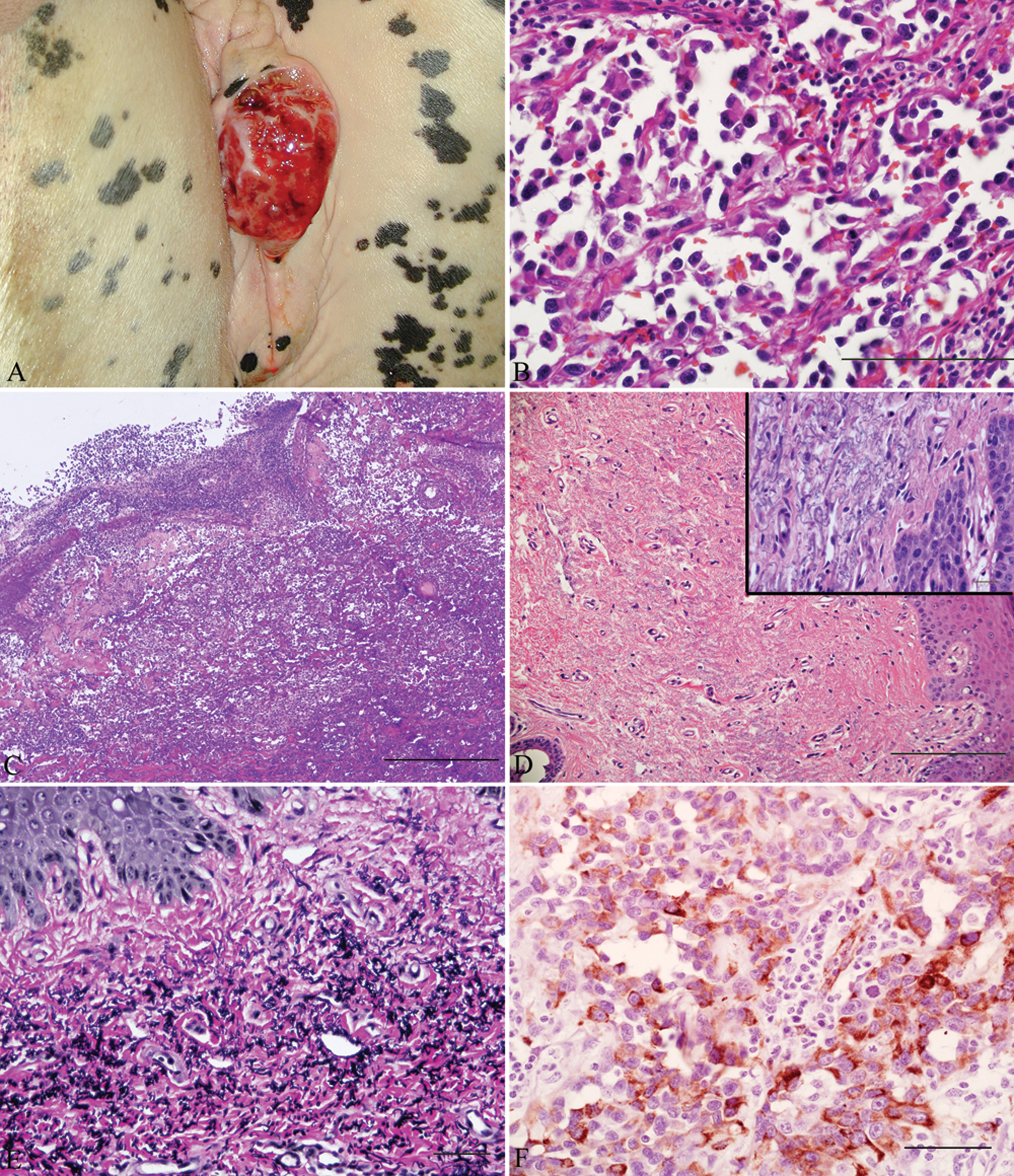
Vulvar mass; horse. A, dark red, ulcerated mass with multifocal areas of necrosis on right dorsal commissure of vulva. B, the neoplasm is composed of variably sized vascular spaces lined by pleomorphic epithelioid cells containing small numbers of erythrocytes. Hematoxylin and eosin (HE) stain. Bar = 100 µm. C, multifocal areas of epidermal ulceration and presence of abundant serofibrinous material admixed with numerous neutrophils, small numbers of lymphocytes, plasma cells, and macrophages, and moderate amounts of cellular debris. HE stain. Bar = 500 µm. D, superficial dermis adjacent to the neoplasm containing moderate amounts of wavy, basophilic, fragmented, or tangled fibers. HE stain. Bar = 200 µm. Inset: Higher magnification of tangled fibers. Bar = 20 µm. E, positive histochemical staining demonstrating band of dark tangled elastic fibers in the superficial dermis. Verhoeff–van Gieson stain. Bar = 50 µm. F, immunohistochemical staining with positive labeling of the population of neoplastic cells with factor VIII–related antigen. Immunoperoxidase with 3,-3’-diaminobenzidine with Mayer hematoxylin counterstain. Bar = 100 µm.
Histologically, the vulvar mass was an unencapsulated, poorly defined, densely cellular, infiltrative neoplasm composed of variably sized and shaped vascular spaces and occasionally solid nests supported by a fine fibrovascular stroma. The vascular spaces were lined by plump neoplastic endothelial cells with distinct cell borders and large amounts of eosinophilic cytoplasm, oval to elongate hyperchromatic nuclei with variably stippled chromatin, and occasionally one prominent magenta nucleolus. Vascular spaces rarely contained small numbers of erythrocytes (Fig. 1B). Anisocytosis was moderate, and anisokaryosis was marked. Mitoses were 7 per 10 high power fields with the presence of occasional bizarre mitotic figures. The neoplasm was multifocally infiltrated by moderate numbers of lymphocytes, plasma cells, and neutrophils, and had multifocal areas of necrosis. The neoplasm occasionally contained few lymphoid aggregates. The epidermis was multifocally ulcerated and contained abundant serofibrinous material admixed with numerous neutrophils (degenerate and viable), small numbers of lymphocytes, plasma cells, and macrophages, and moderate amounts of cellular debris (Fig. 1C). Neoplastic cells extended to the deep surgical margins. The superficial dermis adjacent to the neoplasm multifocally contained moderate amounts of wavy, basophilic, fragmented, or tangled fibers (Fig. 1D) that stained black with Verhoeff–van Gieson stain (solar elastosis; Fig. 1E). The solar elastosis was present diffusely over the mass. Immunohistochemical staining of formalin-fixed, paraffin-embedded tissue sections was performed for Von Willebrand factor or factor VIII–related antigen (rabbit polyclonal antibody a ) and vimentin (mouse monoclonal antibody a ) using streptavidin–biotin–peroxidase method. a The performance of positive and negative controls was within normal limits. Immunohistochemical staining for factor VIII–related antigen has been used to identify vascular neoplasms in horses.5,14 Immunohistochemical examination of the neoplastic cells showed strong positive staining for factor VIII–related antigen (Fig. 1F) and vimentin. The histomorphological features of the neoplastic cells and immunohistochemical and histochemical stains in the current case were indicative of vulvar hemangiosarcoma with solar elastosis.
Neoplasms of vascular origin are uncommon in horses. Although rare in horses, they have been reported to occur in the subcutis, 10 pericardium, 1 third eyelid, 5 pelvis, 14 vertebra, 12 and brain. 16 Hemangiosarcomas occurred in middle-aged horses with no apparent sex predilection, with the musculoskeletal and respiratory systems most frequently affected. 22 Cutaneous hemangiosarcoma has been reported more often in young horses.3,11 In a retrospective study in the Pacific Northwest region of the United States, the overall incidence of equine cutaneous neoplasia submissions during a 3.5-year period revealed that equine sarcoid (51.4%) and squamous cell carcinoma (SCC; 18.3%) were the most prevalent, and hemangiosarcoma (0.7%) was the least prevalent tumor. 25 Increased exposure to solar radiation was suggested as a possible cause for squamous cell carcinoma in the Pacific Northwest. 25 Cutaneous hemangiosarcomas were recorded in horses with an age of 3–20 years, which was consistent with the current report. Another survey found only one hemangioma in the neck of a horse in 2,250 equine necropsies and biopsies (Kerr KM, Alden CL: 1974, Equine neoplasia—a ten year survey. In: Proceedings of the Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians, pp. 183–187). The most reported equine perineal neoplasms include SCC, lymphoma, sarcoid, and melanoma. Squamous cell carcinomas most frequently involve the mucocutaneous junctions of the eyelids and less commonly develop on the face, perineum, and external genitalia. 6 Such tumors are usually locally invasive, and metastasis to regional lymph nodes has been reported in poorly differentiated carcinoma. 4 The etiopathogenesis of SCC in mammals is unclear. Various studies in horses, cattle, and dogs indicate that prolonged exposure of nonpigmented or sparsely haired skin to ultraviolet (UV) light, 6 and the presence of papilloma viruses, 23 mutant p53 protein, 21 upregulation of COX-2, 17 and specific cellular inflammatory responses 19 have carcinogenetic potential.
Solar-induced cutaneous hemangiomas and hemangiosarcomas have been reported uncommonly in perineal skin of dogs.7,15 However, there are no reports of perineal hemangiosarcoma in the horse. In horses with SCC, the influence of UV radiation has been suggested pathologically as actinic changes characterized by basophilic, irregular thickened elastic fibers in the dermis. 2 The presence of severe solar elastosis in the present case suggests that solar radiation could have played a role in development of vulvar hemangiosarcoma. The farm management could have contributed to the development of the tumor because the horse was maintained on pasture 24 hr a day, all year round, for most of her life. The other possible explanation is that expansion of the vulvar mass resulted in more solar exposure to the vulva and led to actinic keratosis over the mass. However, these assumptions are merely speculative due to paucity of literature, and further investigation of more cases of equine cutaneous hemangiosarcoma with solar elastosis is required to confirm the solar radiation as a possible cause.
Completeness of excision of nonvisceral hemangiosarcoma was the most important factor to predict clinical outcome in affected dogs and cats. 18 The prognosis for most cutaneous SCC is good if treatment is initiated before extensive tissue invasion or metastasis has occurred. 4 Another study revealed that SCC with solar elastosis in nonpigmented epithelium of the horse has more favorable prognosis following complete surgical excision. 2 Ocular hemangiosarcoma in horses was reported to be a very aggressive tumor that recurred and metastasized after surgical excision. 8 Similarly, ocular SCCs are generally aggressive and malignant with high rates of infiltration and recurrence. The rate of metastasis ranged from 6% to 18%.13,20 In contrast, no recurrence was noted for epithelioid hemangiosarcoma on the ventral neck of a horse. 26 No complications or recurrence were reported 6 months after surgical excision of the vulvar epithelioid hemangiosarcoma in the current case. Based on the findings presented herein, hemangiosarcoma should be included as a possible differential for equine perineal neoplasms. Definitive antemortem diagnosis can be obtained by biopsy. Monitoring of more cases is warranted to characterize the behavior of vulvar epithelioid hemangiosarcoma with solar elastosis although no recurrence has been reported 6 months after surgical excision in the present case.
Footnotes
Acknowledgements
The authors would like to thank Dr. Rebecca McConnico and Daniel J. Burba for providing the gross photographs.
a.
Rabbit monoclonal antibody (code no. A0082), mouse monoclonal antibody (code no. M0725), EnVision+ System-HRP labeled polymer; Dako North America Inc., Carpinteria, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
