Abstract
CD31 immunoreactivity has been reported in human nonendothelial tumors of both epithelial and mesenchymal origin. This study examined CD31 immunoreactivity of 347 formalin-fixed, paraffin-embedded normal, nonneoplastic, and neoplastic canine tissues. CD31 expression was considered positive if at least 10% of the cell population had membranous reactivity. Labeling with the CD31 antibody (clone JC/70A) was observed in 16 samples of normal organs (liver, kidney, lymph node), 6 of 6 specimens of hepatic nodular hyperplasia, 3 of 3 hepatic regenerative nodules, 1 of 4 anal sac carcinomas, 6 of 6 hemangiosarcomas, 18 of 20 hepatocellular carcinomas, 1 of 6 mammary carcinomas, 3 of 5 plasmacytomas, 18 of 53 renal cell carcinomas, and 1 of 5 cutaneous histiocytomas. CD31 expression did not correlate with case outcome in hepatocellular or renal cell carcinomas. Although distinguishing hemangiosarcoma from other neoplasms is typically straightforward, pathologists should be aware of potential cross-reactivity when relying on CD31 immunohistochemistry for diagnosis, particularly in small biopsy samples or when faced with an epithelioid or poorly differentiated vascular neoplasm.
CD31 (platelet-endothelial cell adhesion molecule 1 [PECAM-1]) is a transmembrane glycoprotein with various functions in multiple physiologic and pathologic pathways, including leukocyte detachment, platelet activation, T-cell activation, angiogenesis, atherosclerosis, and maintenance of vascular integrity. 5,24,25,33,45 CD31 also has immunoregulatory roles in endothelial and hematopoietic cells. 42
Originally, CD31 was considered a very specific marker for endothelial cells and their tumors; however, CD31 is also expressed in platelets, monocytes, neutrophils, megakaryocytes, plasma cells, macrophages, and some lymphocytes. 18,26,31,36 Using a different CD31 antibody than the one evaluated in the current study, Fernández et al. 13 demonstrated expression of this molecule in a variety of human epithelial and mesenchymal cells using frozen tissues.
In a previous study, 32 we observed distinct labeling of tubular epithelial cells in normal kidney with an antibody to CD31. In the current study, the immunohistochemical expression of CD31 was evaluated in normal and lesional canine tissues.
Materials and Methods
A total of 347 specimens were examined, including 131 normal tissues (sections of adrenal gland, cerebrum, heart, small and large intestine, kidney, liver, lung, lymph node, mammary gland, ovary, pancreas, parathyroid gland, pituitary gland, prostate, salivary gland, skin, spleen, stomach, testis, thyroid, tonsil, urinary bladder, and uterus), 10 nonneoplastic lesions (hepatic nodular hyperplasia, regenerative hepatic nodules, and parathyroid hyperplasia), and 206 neoplastic tissues. Neoplastic tissues included benign epithelial tumors (9), endocrine/neuroendocrine tumors (4), carcinomas (141), sarcomas (12), melanomas (5), lymphomas (8), plasmacytomas (5), cutaneous histiocytomas (5), testicular and ovarian tumors (13), thymoma (1), and mesothelioma (3). All specimens had been fixed in neutral buffered formalin and paraffin embedded.
Immunohistochemistry
Immunohistochemistry for CD31 was applied to each of the above-mentioned specimens. The monoclonal anti-CD31 antibody (clone JC/70A, Dako-Agilent, Carpinteria, CA) recognizes a fixation-resistant epitope in endothelial cells. 31 The slides were immersed in Reveal (Biocare Medical, Concord, CA) antigen retrieval solution in a decloaking chamber (Biocare) for 30 seconds at 125°C and 18 to 24 PSI. The primary antibody was diluted at 1/100 using Da Vinci diluent (Biocare Medical) and incubated for 60 minutes. An immunoperoxidase, polymer-based detection system was used (PromARK Mouse-on-Canine HRP-Polymer, Biocare Medical). The chromogen was diaminobenzidine (DAB). All incubations were at room temperature. Randomly selected sections of test samples were treated with Biocare Medical polymer negative control serum in place of the primary antibody. The target cell population was considered positive if at least 10% of the cells had a distinct membranous labeling for CD31. The number of positive cells was scored semiquantitatively in the hepatic and renal lesions and plasmacytomas as follows: score 1 (10%-20% positive cells), 2 (21%-40%), 3 (41%-60%), and 4 (>60%). The intensity of the reaction was subjectively graded as weak, moderate, or strong based on the target area showing the highest intensity in each sample.
Results
Immunohistochemistry: Normal Tissues
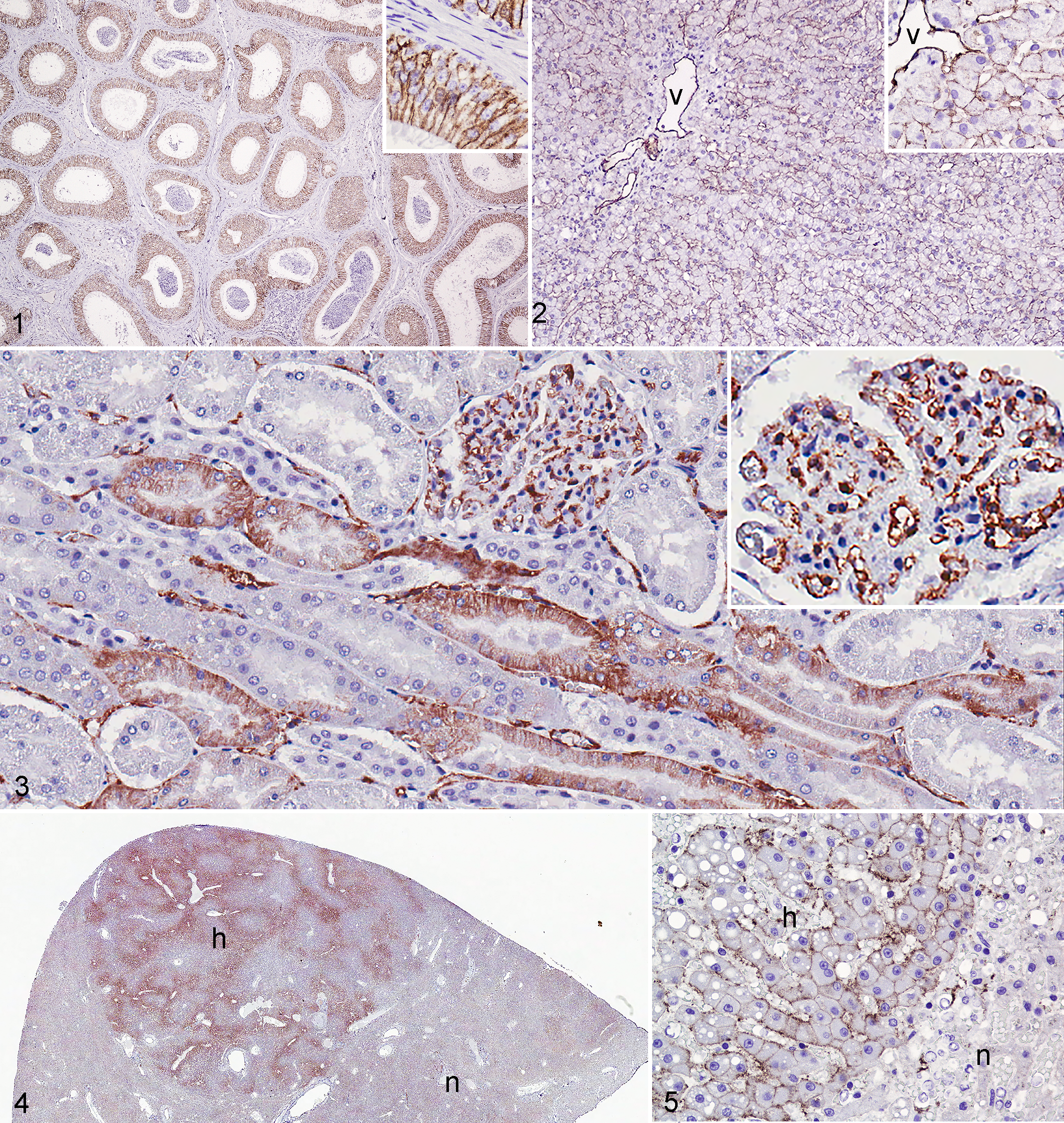
CD31 labeling of vascular endothelium served as an internal positive control. In addition, there was moderate to strong membranous labeling of deferent ductal and epididymal epithelium (Fig. 1) as well as of the lining of lymph node sinuses. Three of 5 normal hepatic specimens had weak to strong membranous labeling of hepatocytes (in 1 case, this was patchy) but no distinct labeling of sinusoidal lining cells (Fig. 2). Sixty-one percent (25/41) of normal renal tissue samples had patchy membranous to cytoplasmic labeling of cortical tubular epithelial cells (Fig. 3).
Immunohistochemistry: Nonneoplastic Lesions
Hepatic Nodular Hyperplasia
The number of CD31-positive cells was variable (1 case with score 1; 2 with score 3; 3 with score 4). All 6 hyperplastic nodules had weak to strong (in 1 case) membranous labeling that outlined hepatic plates (Figs. 4 and 5). Labeling was diffuse or concentrated in centrilobular and periportal areas. Labeling intensity was, in some cases, similar to that in adjacent normal hepatocytes, but in other cases, normal hepatocytes were negative. CD31 labeling intensity in hepatocytes of hyperplastic nodules was moderate to strong. In hepatic cirrhosis, the hepatocytes in regenerative nodules had strong membranous labeling in all 3 cases; this was diffuse in 2 cases and patchy in the third case (2 with a score of 4 and 1 with a score of 3; Fig. 6). Sinusoidal lining cells within regenerative nodules were negative, but newly formed vessels in areas of fibrosis surrounding the nodules were positive.
Immunohistochemistry: Neoplastic Lesions
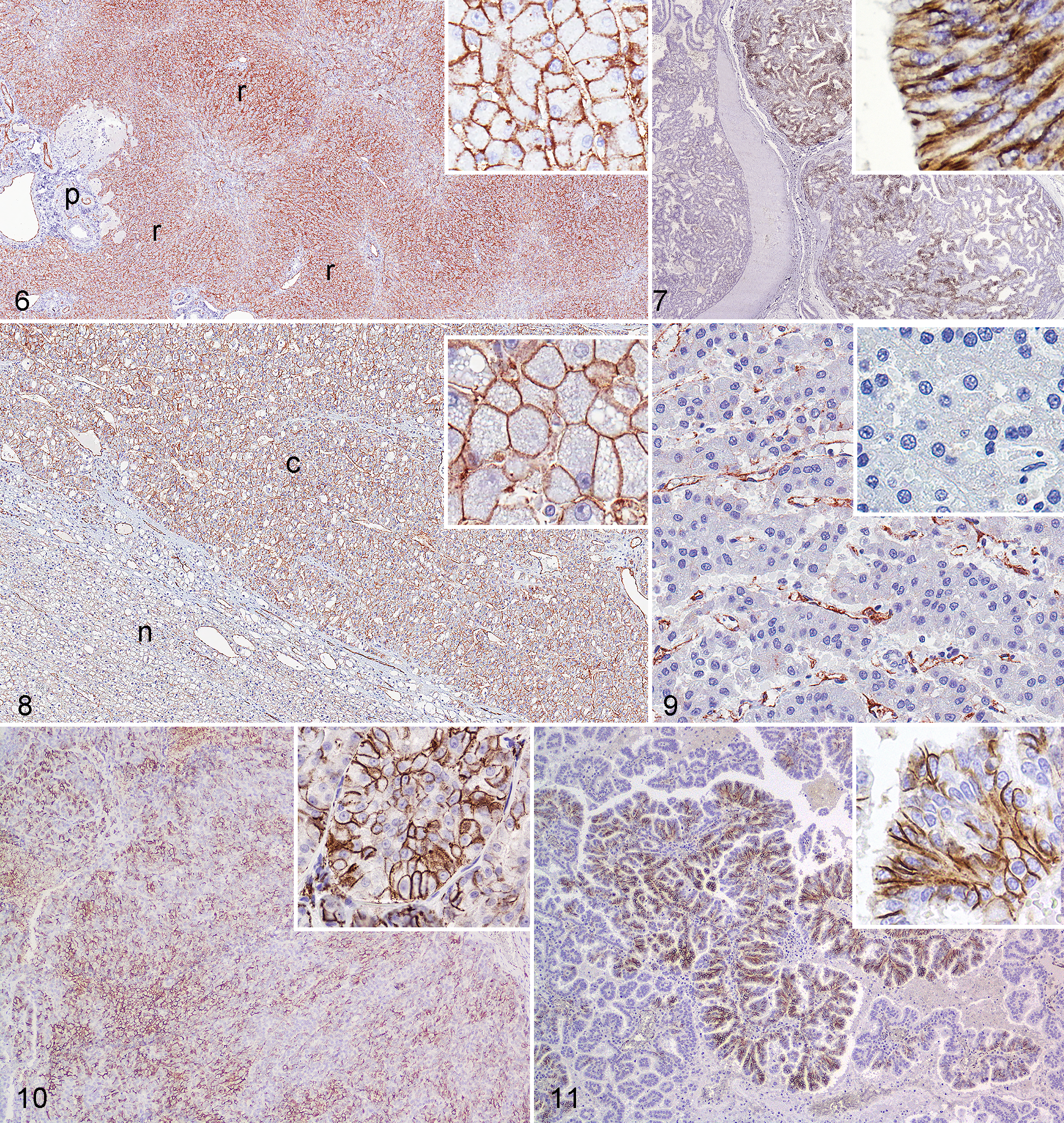
Forty-two carcinomas and 6 sarcomas were positive for CD31 (Table 1). One of 4 cases of carcinoma of the apocrine glands of the anal sac had patchy strong membranous labeling (Fig. 7). Overall, 18 of 20 hepatocellular carcinomas (HCC) expressed CD31. Membranous labeling of neoplastic hepatocytes was weak to strong and patchy to diffuse in 12 of the 20 HCC (2 with score 1, 1 with score 3, 9 with score 4; Fig. 8). Variable labeling of intratumoral sinusoidal lining cells was observed in 11 of 20 HCC (Fig. 9). Labeling of adjacent nonneoplastic hepatic parenchyma was weaker than in neoplastic tissue in the few cases available for comparison. Fourteen of 43 (33%) primary renal cell carcinomas (RCC) and 4 of 10 (40%) metastatic RCC also expressed CD31. The number of positive cells and intensity was slightly higher in primary than in metastatic RCC (7 with score 1, 6 with score 3, 1 with score 2, 4 with score 4). All but 3 of the positive RCC also had CD31 immunoreactivity in adjacent nonneoplastic kidney. Differences in CD31 expression among histologic types of HCC and RCC were not apparent (Figs. 10 and 11). One of 6 mammary carcinomas had patchy, membranous, and moderate to strong labeling of all ductal epithelial cells or of scattered cells in a duct. Plasmacytomas (3/5) had weak to moderate and patchy, membranous labeling. One of 5 cutaneous histiocytomas had weak and patchy membranous labeling. Other epithelial neoplasms examined did not express CD31. The only sarcoma positive for CD31 was hemangiosarcoma (6/6).
CD31 Immunoreactivity in Pathologic Specimens From Dogs.a
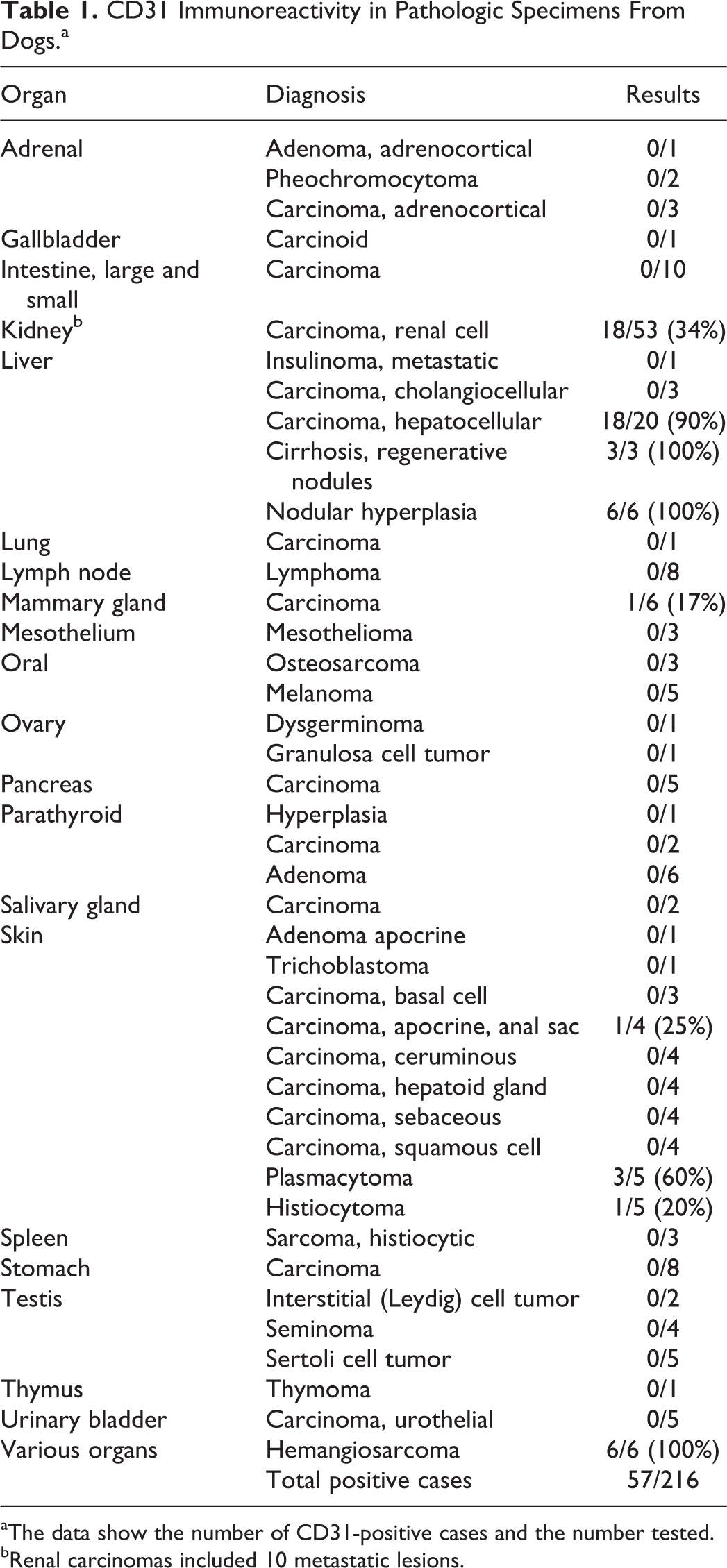
aThe data show the number of CD31-positive cases and the number tested.
bRenal carcinomas included 10 metastatic lesions.
Discussion
In this study, we evaluated the immunohistochemical expression of CD31 in nonendothelial canine tumors. CD31 detection has been documented in human endothelial tumors of blood vessel origin 12,27,29 and less commonly in tumors of lymphatic origin. 22,27,31,44 Although CD31 was initially considered highly specific for human vascular tumors, 11 various proportions of nonendothelial tumors also express this protein. Miettinen et al. 27 reported weak labeling of one leiomyosarcoma and several colonic carcinomas and mesotheliomas, whereas others did not find CD31 immunoreactivity in nonendothelial mesenchymal neoplasms. 10 CD31 has been detected in dendritic cell neoplasms, different types of lymphomas, and extranodal histiocytic sarcomas. 20,28,37 Evaluation of CD31 reactivity in carcinomas has generated particular interest, with labeling of some salivary gland, thyroid, cutaneous, and gastrointestinal carcinomas. 9 Although breast carcinomas did not express CD31 in that study, 9 others have reported CD31 immunoreactivity in breast cancer, particularly ductal carcinoma. 1,3,4,30,35,38
CD31 monoclonal antibody, clone JC/70A, is currently used in a variety of animal species to evaluate the vascular bed in normal and pathological conditions, including endothelial neoplasms of blood and lymphatic vessels. 2,14,15,21,34,43 In the current study, CD31-positive specimens included normal organs (liver, kidney, lymph node), 6 of 6 hepatic nodular hyperplasia, 3 of 3 hepatic regenerative nodules, 1 of 4 anal sac apocrine carcinomas, 6 of 6 hemangiosarcomas, 18 of 20 hepatocellular carcinomas, 1 of 5 mammary carcinomas, 3 of 5 plasmacytomas, 18 of 53 renal cell carcinomas, and 1 of 5 cutaneous histiocytomas.
The significance of CD31 expression in normal and lesional tissues in dogs is not clear. It has been suggested that CD31 immunoreactivity in human epithelial neoplasms or epithelial tissues is the result of partial cross-reaction of the antibody with related cell adhesion molecules 41 or abnormal expression of CD31. 9 The second possibility would explain the increased expression of this marker in pathologic liver tissue (eg, regenerative nodules in cirrhosis and hepatocellular carcinoma), but we did not find a consistent expression in the cases examined. CD31 contributes to cellular differentiation and migration in physiologic and pathologic states. 1,35 In human breast carcinoma, CD31 expression in neoplastic cells correlates with poor overall and metastasis-free survival in patients with no nodal metastasis. 4 In the current series, patchy CD31 reactivity was detected in only 1 of 6 canine mammary carcinomas. Based on the limited number of cases studied, the significance of this finding is unknown.
In the current study, CD31 immunoreactivity was common in normal and lesional canine liver and kidney. Normal, hyperplastic, and regenerative nodules and neoplastic hepatocytes expressed CD31 in various percentages and intensities, typically with higher intensity in neoplastic tissues than in adjacent parenchyma. CD31 was expressed in sinusoidal cells in HCC but not in normal canine liver or nonneoplastic liver lesions. In humans, CD31 immunoreactivity is not observed in normal hepatic parenchyma, and its expression in the sinusoidal endothelium is controversial. 6 –8,16,17,23,40 However, the sinusoids of hepatic regenerative nodules—especially in dysplastic nodules and hepatocellular carcinoma—undergo a process called capillarization, in which sinusoidal cells develop structural and molecular changes typical of endothelial cells such as loss or reduction of cytoplasmic fenestrations, deposition of basement membrane, and expression of adhesion molecules including CD31. 6,16,19,39 Although not detected in all studies, 17,39,46 this CD31 expression by sinusoidal lining cells might be the result of neovascularization of cancerous parenchyma (tumor angiogenesis) or abnormal differentiation of pre-existing sinusoids. 16,39 One reason for such differences among studies is the type of CD31 antibody used, although clone JC/70A was used in most studies. 4,6,8,13,43,44
With the exception of sinusoidal capillarization in hepatic lesions, CD31 expression has not been reported in human hepatocellular or renal cell neoplasms. In the current study, CD31 was expressed in a proportion of various normal, nonneoplastic, and neoplastic canine tissues. The neoplasms that most commonly expressed CD31 were hepatocellular carcinoma and renal cell carcinoma, and a correlation between immunoreactivity and case outcome was not apparent. Although the histologic distinction of HCC or RCC from a vascular neoplasm is generally straightforward, pathologists should be aware of CD31 cross-reactivity, especially if the epithelioid variant of hemangiosarcoma is a differential diagnosis or if the sample size is small.
Footnotes
Acknowledgments
We thank the histology technicians, pathologists, and pathology residents at the Indiana Animal Disease Diagnostic Laboratory and Dr Tyler Peat for his input in the early stages of this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
