Abstract
Epithelioid hemangiomas, hemangioendotheliomas, and angiosarcomas are well recognized histologic variants of endothelial tumors in humans that in the past have been confused with neoplasms of epithelial or histiocytic origin. We describe 12 epithelioid endothelial vascular tumors in 8 dogs, 3 horses, and 1 cow that share microscopic features with these tumors in humans. Ten tumors were located within the dermis and subcutis, 1 in the gastrocnemius tendon, and 1 in the skeletal muscle of the thigh. Key histologic features were the plump, epithelioid appearance of neoplastic endothelial cells and occasional cytoplasmic vacuolation, which rarely contained a single erythrocyte. Patterns of epithelioid endothelial cell growth ranged from conventional vasoformative structures to glandlike acini and short ducts to solid, sheetlike arrangements. All tumors were positive for endothelial markers: 12/12 (100%) were positive for von Willebrand factor with variable intensity and distribution and 9/9 (100%) were positive for CD31. All 12 were negative for cytokeratin. We subdivided these tumors into epithelioid hemangiomas (n = 3) and hemangiosarcomas (n = 9) based on conventional morphologic criteria, growth pattern (invasive or not), and metastasis (known in 1 case at the time of biopsy). Additional follow-up information obtained by a written questionnaire was available for 6/12 (50%) animals and revealed local recurrence in 3 animals (1 epithelioid hemangioma and 2 epithelioid hemangiosarcoma) and regional metastasis in 3 animals (all epithelioid hemangiosarcomas). This series represents a novel group of vascular tumors in domestic animals of which there are only 3 previous cases reported in the veterinary literature.
Keywords
Spontaneous tumors of blood vessel endothelial cells have been described commonly in the dog, less frequently in the cat and horse, and sporadically in most other domestic species. 6 In the dog, hemangiomas are typically benign, solitary, deep dermal tumors, whereas hemangiosarcomas often present as a disseminated malignancy involving the spleen, heart and lung, liver, and soft tissues of the trunk and extremities. 3, 4 Cutaneous hemangiomas are common in the dog whereas primary canine cutaneous hemangiosarcomas are less common, and in cases of disseminated disease it is difficult to differentiate de novo from metastatic dermal hemangiosarcomas. 9 It has been suggested that dermal hemangiomas and hemangiosarcomas could be solar induced, particularly in the glaborous skin of sparsely haired dogs. 9
Histologically, endothelial tumors in animals are most commonly designated as angiomatosis, hemangiomas, and hemangiosarcomas. Several variants of hemangiomas are described, such as capillary and cavernous hemangiomas, infiltrative hemangioma, arteriovenous hemangioma, granulation tissue–type hemangioma, spindle cell hemangioma, solar-induced dermal hemangioma, angiokeratomas, and angiolipomas. 8 Recognized variants of hemangiosarcoma include solar-induced hemangiosarcoma and epithelioid (histiocytoid) hemangiosarcomas. 8 In humans, epithelioid variants of cutaneous endothelial tumors were first described in 1982 20 and now include epithelioid hemangiomas, epithelioid hemangioendotheliomas, and epithelioid angiosarcomas. 11 Because well differentiated vascular channels often seen in endothelial tumors are less conspicuous or lacking in epithelioid variants and because the cells have an epithelioid morphology, this group of neoplasms pose a diagnostic challenge. Tumors matching this histologic description have been described in veterinary textbooks as “epithelioid (histiocytoid) hemangiosarcoma” 8 or as a variant of hemangiosarcoma 7 ; however, published cases number only 3. 2, 5, 13 We report an additional 12 cases of epithelioid variants of endothelial tumors in 8 dogs, 3 horses, and 1 cow that were submitted to the Cornell University College of Veterinary Medicine between 1987 and 2005.
Materials and Methods
Clinical cases
Twelve cases of vascular endothelial tumors that showed microscopic features similar to the spectrum of human epithelioid endothelial tumors were identified by 1 author (BAS) from biopsies submitted to the Department of Biomedical Sciences, College of Veterinary Medicine, Cornell University, NY, between 1987 to 2005. Tissues were submitted as routine biopsy specimens of skin masses (10), muscle mass (1), and tendon mass (1) from 11 different veterinary hospitals and were represented by 3 species: canine (n = 8), equine (n = 3), and bovine (n = 1). Dog No. 10 was referred to the Cornell University Hospital for Animals, and slides from another veterinary pathology practice were examined as a second opinion.
A 2-page letter was sent to all 11 hospitals that had submitted cases, asking for any available follow-up information (recurrence or not, metastasis, etc.). Responses were received regarding 6 animals (Table 1).
Signalment, location, diagnosis, and clinical outcome for 12 animals with epithelioid endothelial tumors.
F = female; FN = female neutered; M = male; MN = male neutered.
Histology and immunohistochemistry
Tissues were fixed in 10% (w/v) neutral buffered formalin, embedded in paraffin, sectioned at 4 μm, and mounted on glass slides. Sections were stained with hematoxylin and eosin (HE). All cases were also stained with Gomori reticulin silver impregnation.
All cases were evaluated immunohistochemically for the expression of von Willebrand factor (vWF) (also known as Factor VIII–related antigen) and cytokeratin. Where tissue was available, the vWF stains were repeated in addition to evaluating the original vWF stains. Nine tumors, animal Nos. 1, 3, 4, 6, and 8–12, were stained for CD31 (also known as platelet/endothelial cell adhesion molecule-1 [PECAM-1]). Tissues for animal Nos. 2, 5, and 7 were no longer available. Four-micrometer-thick sections were mounted on charged microscope slides (Fisher Scientific). Immunohistochemistry was performed using the Autostainer plus (Dakocytomation) using the streptavidin–biotin immunoperoxidase technique. Briefly, the slides were dried at 60°C for 20 minutes, then deparaffinized using Xylene (Fisher Scientific) and ethanol (Pharmco). Slides were loaded into the Autostainer plus, rinsed with buffer, and placed in 3% hydrogen peroxide solution to block endogenous peroxidase activity for 5 minutes at room temperature. Cytokeratin sections were pretreated with 1°200 pepsin, and vWF sections were pretreated with 1°2,000 pronase. Nonspecific protein adhesion was blocked using normal goat serum for 5 minutes for vWF and cytokeratin and normal rabbit serum for CD31 (Zymed goat antimouse kit, goat antirabbit, and rabbit antigoat kit). The vWF (Dakocytomation) primary antibody was used at 1°1,500 dilution; the Cytokeratin AE1/AE3 (Dakocytomation) was used at 1°200 dilution, and the CD31 (Santa Cruz) primary antibody was used at 1°50 dilution, all at room temperature for 30 minutes. The secondary antibody, a biotinylated goat antimouse (vWF), biotinylated rabbit antigoat (CD31), and biotinylated goat antirabbit (cytokeratin), was incubated for 10 minutes at room temperature. The streptavidin–peroxidase conjugate was used for 10 minutes at room temperature. The chromagen, 3,3-diaminobenzidine-tetrahydrochloride (Dakocytomation) was placed on the slide for 1 minute at room temperature. Slides were counterstained using hematoxylin (Dakocytomation) for 2 minutes, washed in tap water for 2 minutes, then dehydrated using ethanol alcohol, xylene, and pro-par. Slides were cover-slipped using permount mounting media (Fisher Scientific). As a negative control, a duplicate of each section was incubated with normal mouse IgG, normal goat IgG, or normal rabbit IgG for the same time as the primary antibody used. Internal controls for cytokeratin (overlying epidermis or adnexal structures), vWF, and CD31 (normal dermal blood vessels) were examined for positive staining.
Results
The animals were 8 dogs, 3 horses, and 1 cow (Table 1). The dogs were mature (mean and median ages 10.3 and 11 years, respectively) with 5 males and 3 females. Six of the eight dogs (75%) were Golden Retrievers. The mean age of the horses was 9.6 years, the median age 12 years; all were male, and 2 were Morgans. The cow was a female Holstein-Fresian and was 7 months old. The tumor location and results of the clinical outcome post-excision are summarized in Table 1.
Histopathology
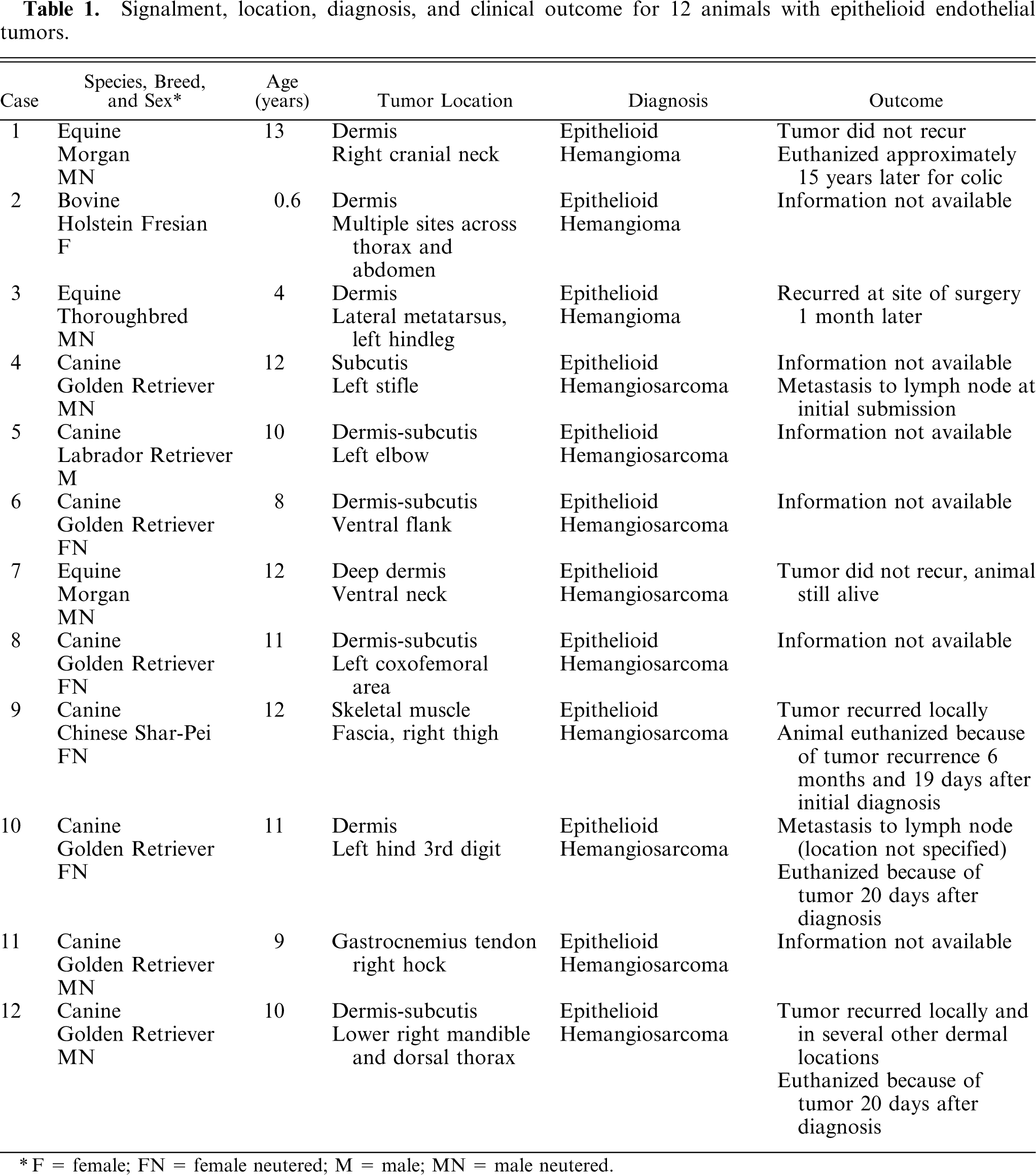
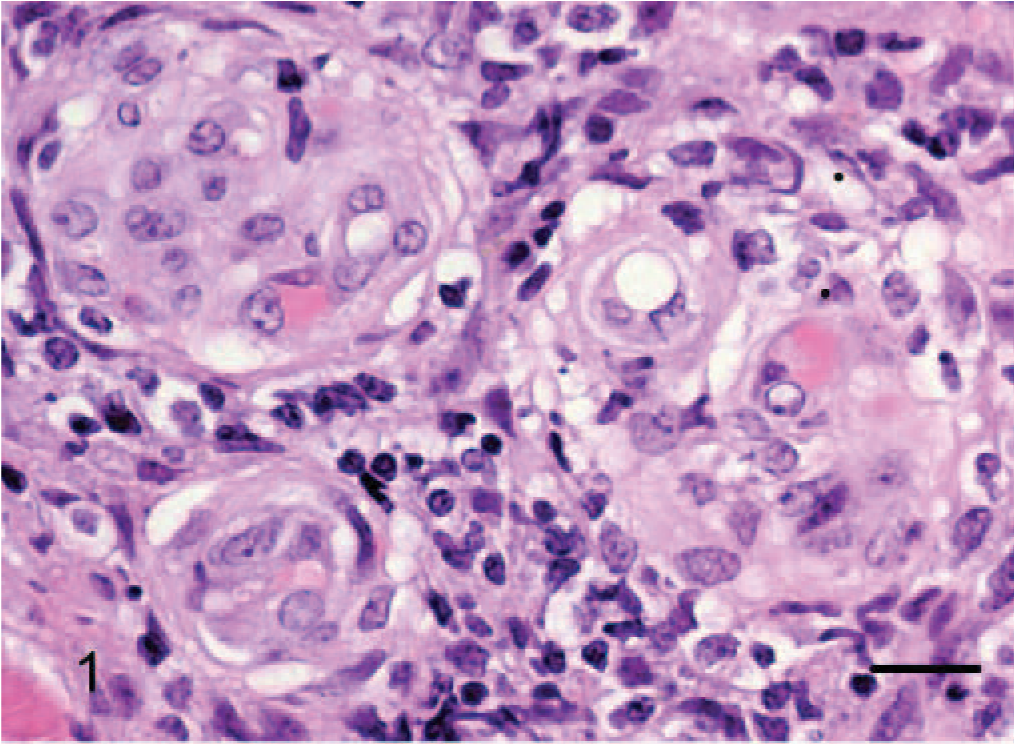
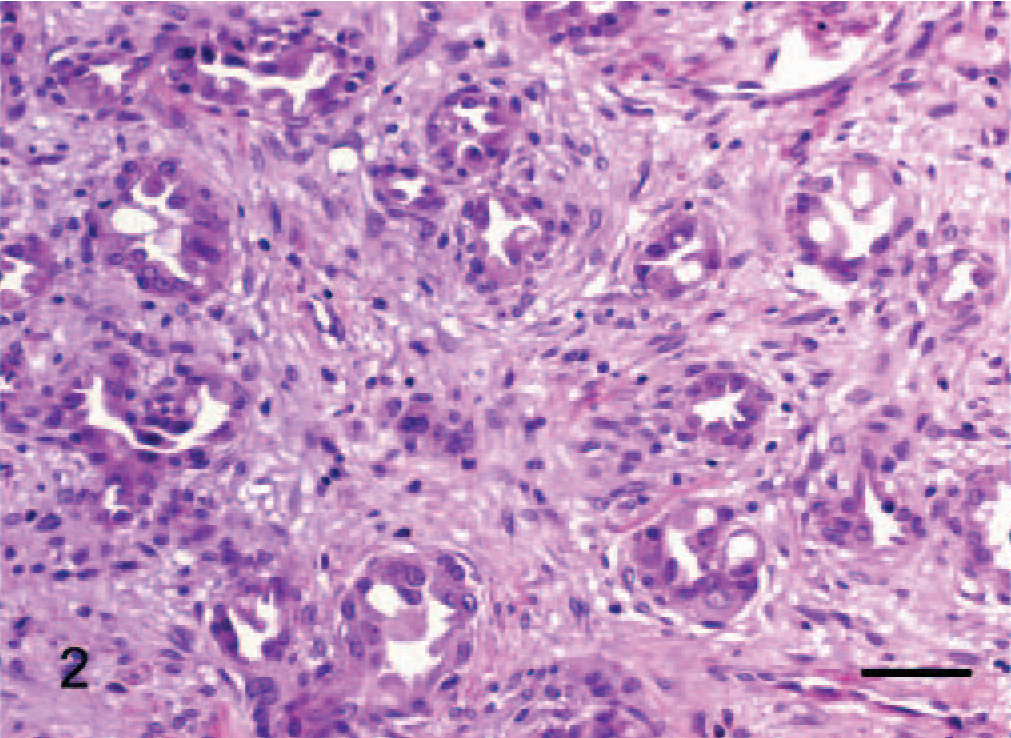
Epithelioid hemangiomas (animal Nos. 1–3), were all located within the collagenous dermis and were unencapsulated but well demarcated, multilobular to coalescing vascular tumors. Architectural arrangements were of clusters of small blood vessels, concentrically thickened by a few layers of cells (Fig. 1), which in horse No. 1 were quite reminiscent of an epithelial tumor with simple and branching tubules and micropapillae within the vascular lumen (Figs. 2, 3). Where neoplastic endothelial cells lined narrow vasoformative slits they frequently had a tall “hob-nailed” configuration. In these 3 tumors, the lumenal endothelial cells, although occasionally flattened, were often conspicuously plump with abundant pale eosinophilic cytoplasm that was occasionally vacuolated. Endothelial nuclei were generally central to paracentral and had finely stippled chromatin and commonly 1–2 small nucleoli. Cellular atypia was lacking, and no mitotic figures were noted. The nuclei of the ablumenal cells (interpreted as pericytes) varied from round to elongate (Fig. 1). In horse No. 3, plump endothelial cells formed more conventional blood vessels that were prominently surrounded and infiltrated by lymphocytes (Fig. 1); this was minimal and confined to the periphery of the tumors in the other 2 cases. The important diagnostic feature, which was most prominent in horse Nos. 1 and 3, was the occasional single, large, clear, intracytoplasmic vacuoles (Fig. 3) within the endothelial cytoplasm, which rarely contained a single erythrocyte. Large cytoplasmic vacuoles displaced the nucleus peripherally to give a “signet ring” appearance to the endothelial cells.
Dermis; horse No. 3. Epithelioid hemangioma: clusters of small blood vessels formed of plump neoplastic cells that obscure the lumen, which are surrounded by lymphocytes. HE. Bar = 24 μm.
Dermis; horse No. 1. Epithelioid hemangioma: neoplastic endothelial cells form simple and branching tubules. HE. Bar = 48 μm.
Dermis; horse No. 1. Epithelioid hemangioma: lumenal papilliferous projections of endothelial cells sometime contain intracytoplasmic vacuoles (arrows) that displace the nucleus forming a signet ring. HE. Bar = 16 μm.
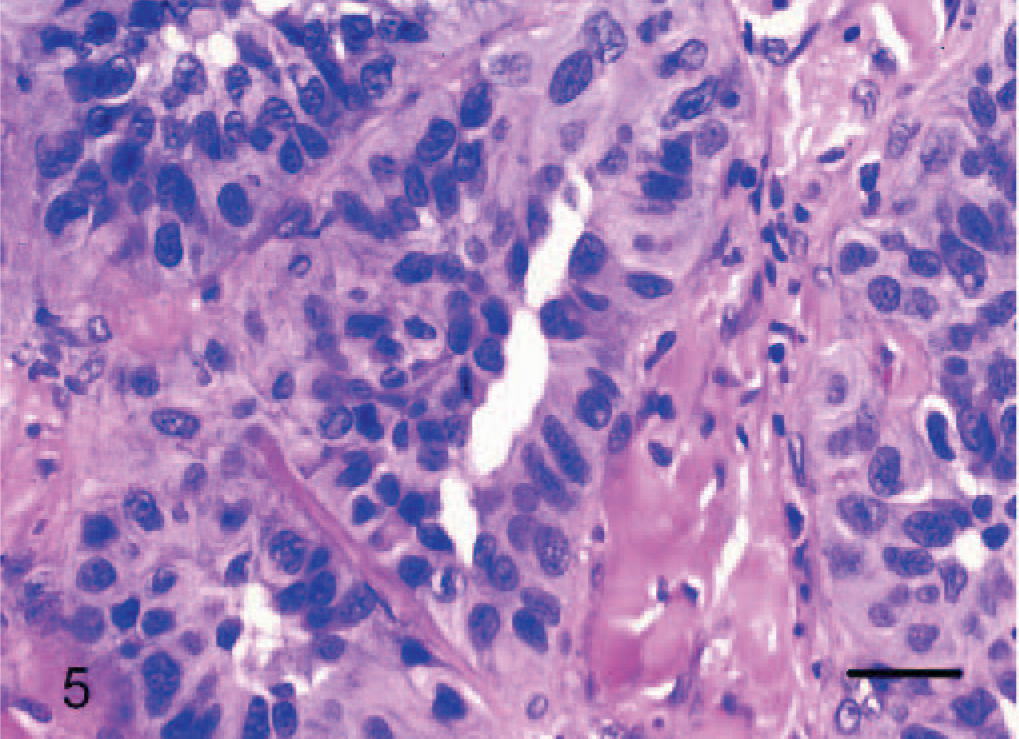
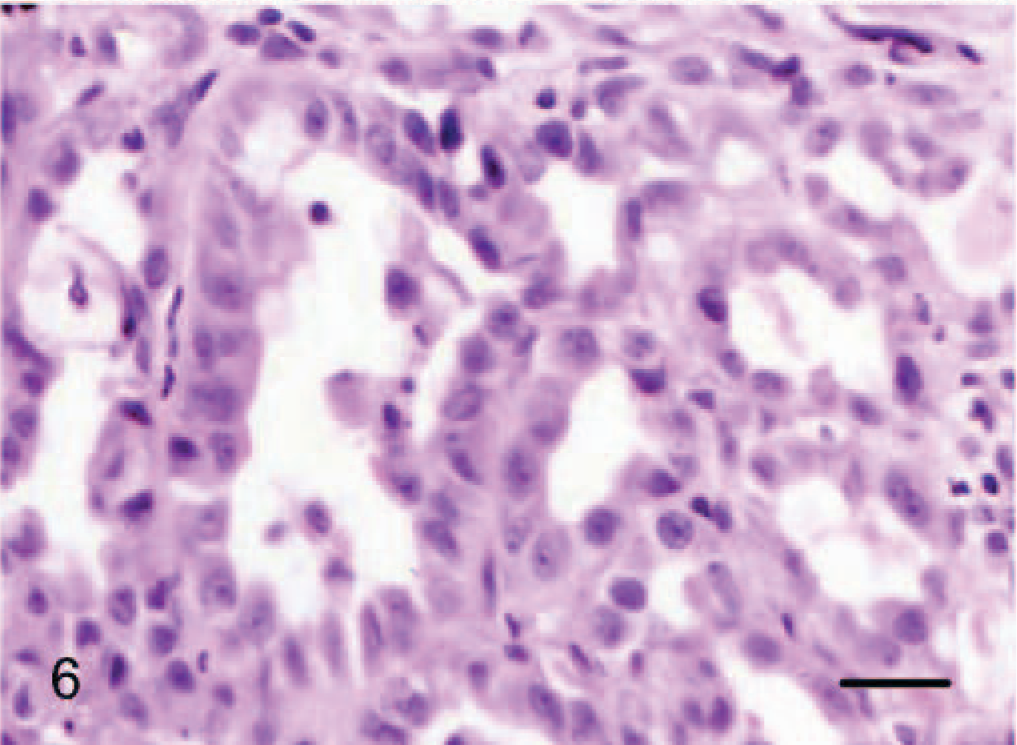
Epithelioid hemangiosarcomas (animal Nos. 4–12) were mostly situated within the deep dermis and subcutis (animal Nos. 4–8, 10, and 12), with 2 cases in the skeletal muscle of the right thigh (dog No. 9) and the right gastrocnemius tendon (dog No. 11). Tumors were unencapsulated, multilobular to coalescing, infiltrative and poorly demarcated, or well demarcated but expansile. There was generally a moderately dense collagenous stroma, and when the tumors formed lobules, these were divided by thin bands of dense collagen. Microscopic patterns of the proliferative endothelial cells varied from more conventional capillary-like to cavernous vessels, which blended into areas of branching tubular and glandlike structures (Figs. 4–6). In some areas, the endothelial cells were less epithelioid but adopted tall hobnail-like arrangements (Fig. 7). Some tumors showed mostly a glandular pattern with rare vasoformative areas (dog Nos. 4–6, 10, and 11). Four epithelioid hemangiosarcomas had predominant areas of solid, compact growth (Figs. 8–10) with smaller vascular structures (animal Nos. 7–9, and 12). Where vasoformative, neoplastic endothelial cells were spindled, whereas in epithelioid areas they were larger, predominantly polygonal, and, in the most glandlike areas, cuboidal to columnar (Figs. 5, 6). The cytoplasm was variably microvesicular with large, single, prominent vacuoles in the cytoplasm of a small percentage of cells, which occasionally contained a single erythrocyte (Fig. 10). Nuclei were round to oval with coarsely stippled to ropey chromatin and one to several, large, and prominent nucleoli. The mitotic rate was highly variable in these 9 tumors, ranging from 2 to 43 mitoses in 10-high power fields. There was variable anisokaryosis, mild in some cases (animal Nos. 4–7, 10, and 11) and moderate to marked in others (dog Nos. 8, 9, and 12) with occasional bizarre mitotic figures. Variably sized areas of coagulative necrosis were present in 6 tumors. Occasionally, there were small, dense, nodular infiltrations of lymphocytes and occasional eosinophils surrounding the tumor (dog Nos. 4, 6, and 9–12) (Fig. 10). In dog No. 5, nodules of neoplastic endothelial cells were present in the subcapsular sinus of the draining left axillary lymph node. These 9 cases, however, were viewed as sarcomas because of nuclear and cytoplasmic atypia, abnormal chromatin pattern, multiple nucleoli, areas of necrosis, variably high mitotic rate, invasive behavior, and, in 3 cases, metastasis.
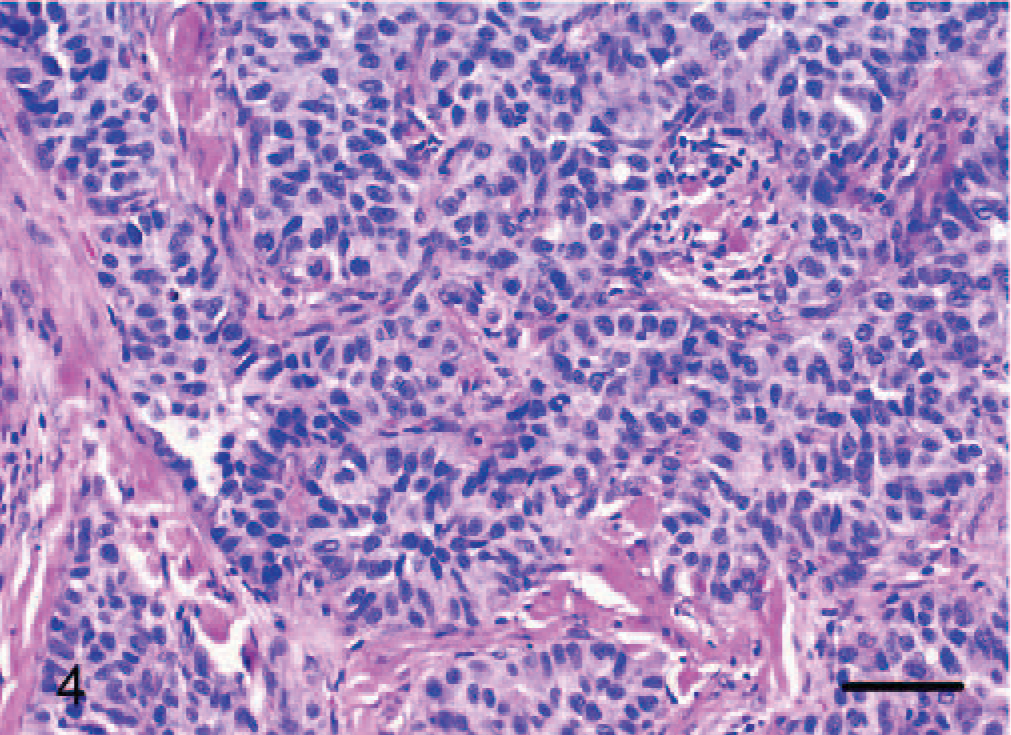
Dermis; dog No. 10. Epithelioid hemangiosarcoma: endothelial cells form solid branching tubular to glandlike structures. HE. Bar = 48 μm.
Dermis; dog No. 10. Epithelioid hemangiosarcoma: cuboidal to columnar endothelial cells line glandlike and tubular structures. HE. Bar = 24 μm.
Dermis; dog No. 4. Epithelioid hemangiosarcoma: cuboidal to low columnar endothelial cells line tubular structures with a prominent lumen. HE. Bar = 24 μm.
Dermis-panniculus; dog No. 6. Epithelioid hemangiosarcoma: may contain areas more reminiscent of conventional hemangiosarcoma, however, note the plump endothelial cells that form prominent (hobnail) lumenal projections (arrow). HE. Bar = 96 μm.
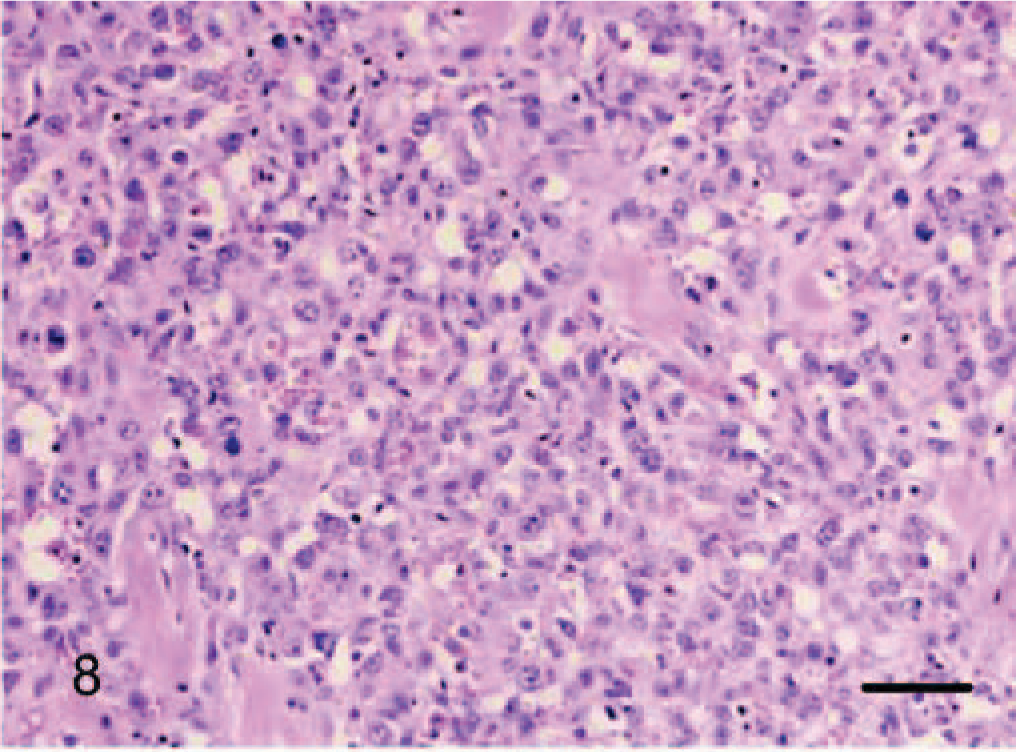
Dermis; dog No. 12. Epithelioid hemangiosarcoma: solid growth pattern with small vasoformative structures and prominent cytoplasmic vacuoles. HE. Bar = 96 μm.
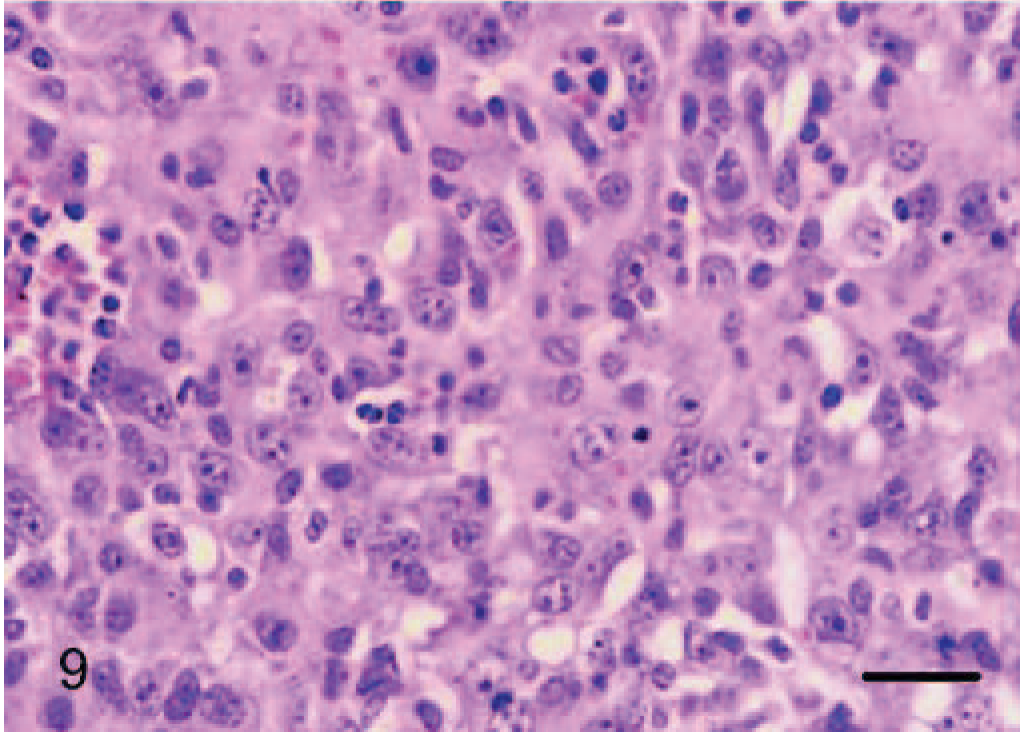
Dermis; dog No. 12. Epithelioid hemangiosarcoma: solid growth pattern with prominent cytoplasmic vacuoles. HE. Bar = 24 μm.
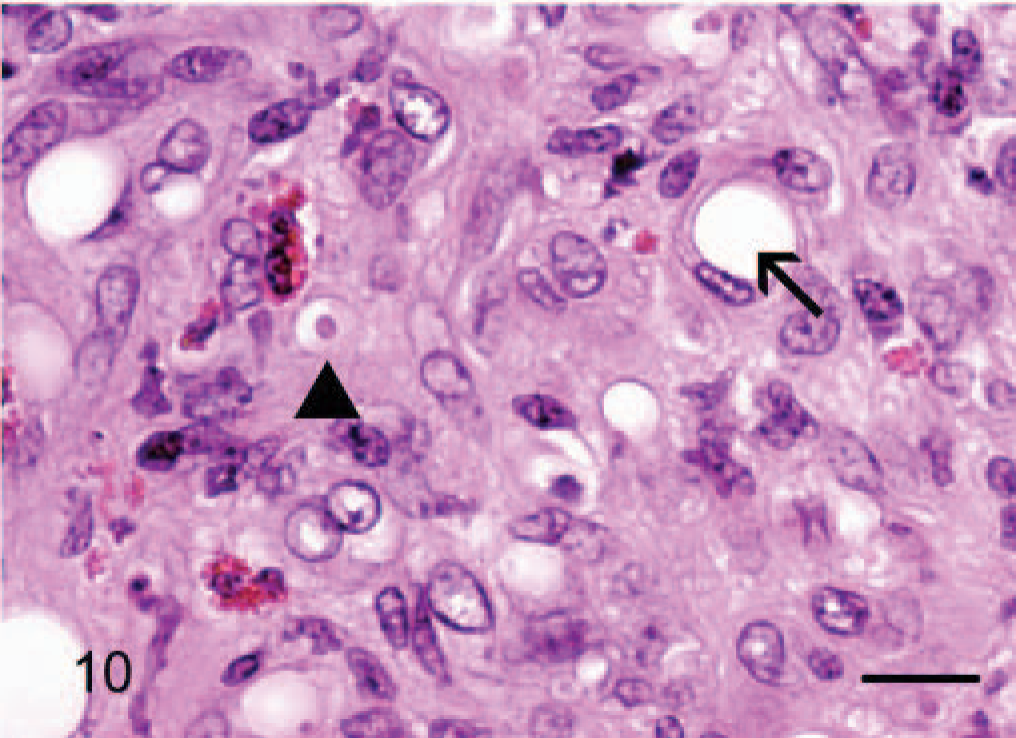
Dermis; horse No. 7. Epithelioid hemangiosarcoma: areas of solid compact pattern of growth with rare intracytoplasmic vacuoles that displace the nucleus (arrow) and rarely contain a single erythrocyte (arrowhead). There are few scattered eosinophils infiltrating the mass. HE. Bar = 24 μm.
Special stains and immunohistochemistry
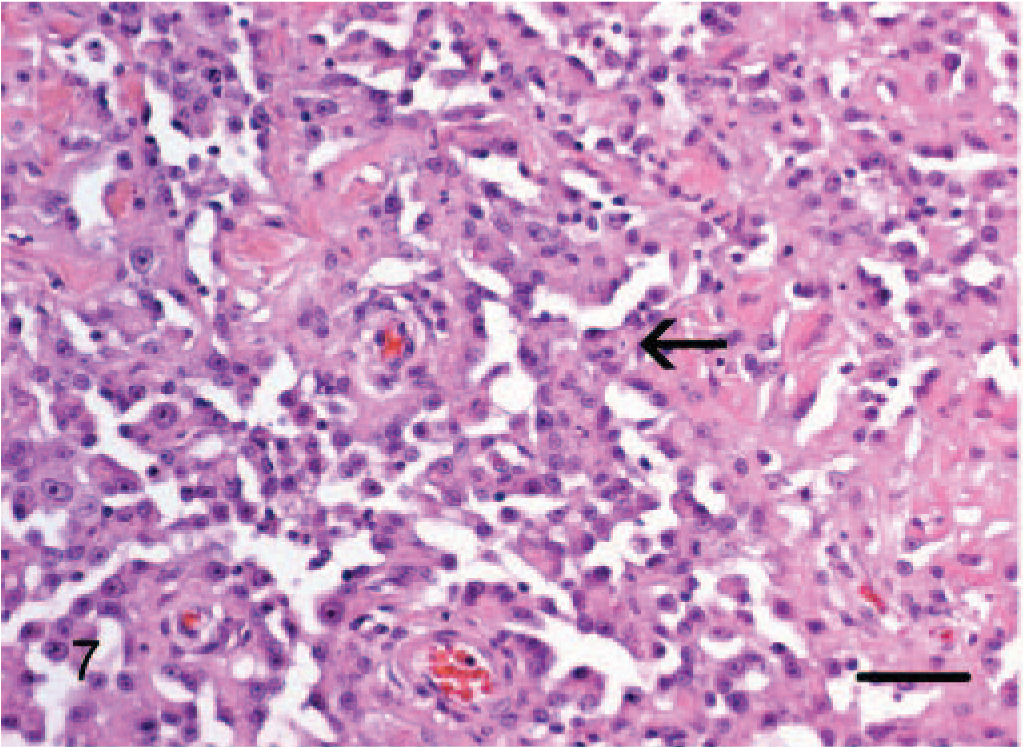
In all 12 tumors, to a variable degree, staining of a basal lamina with Gomori reticulin consistently defined a tubular network of neoplastic endothelial cells within tumor nodules. Reticulin fibers were sparser in areas where papilliferous proliferations of tumor cells extended into the lumen of tubules. Solid areas noted on H and E (Fig. 11) was consistently revealed by Gomori reticulin to be composed of tightly anastomosing tubular networks.
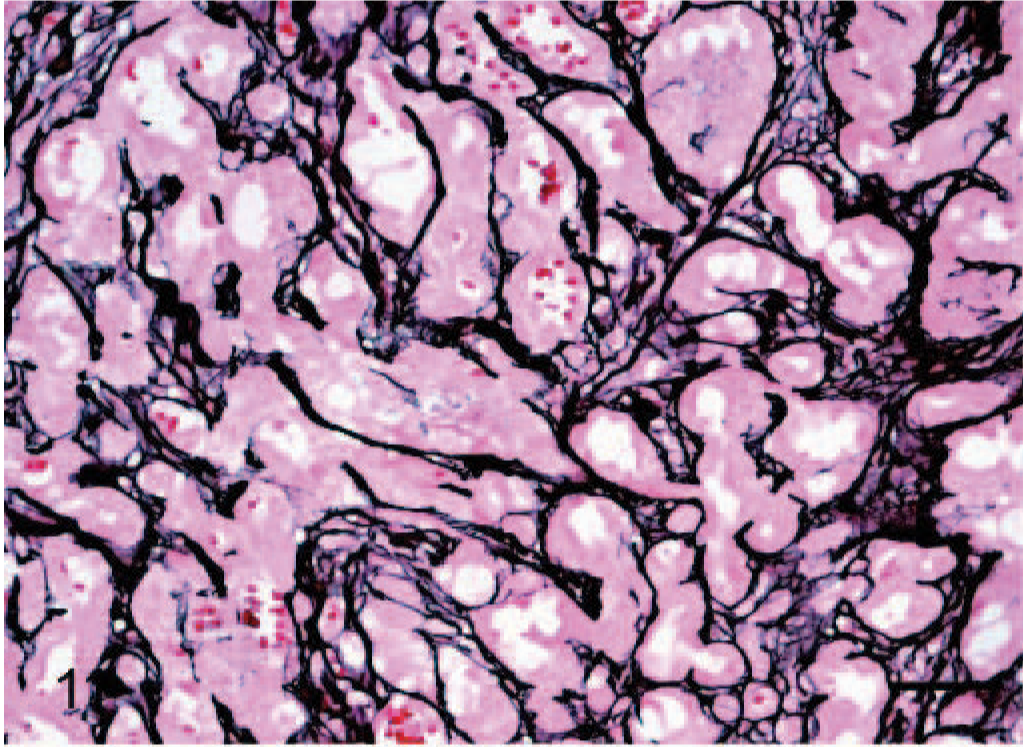
Subcutis; dog No. 4. Epithelioid hemangiosarcoma: Solid areas noted on H and E are consistently revealed to be composed of blood vessels forming a tubular network outlined by reticulin positive fibers (basement membrane). Gomori reticulin. Bar = 48 μm.
vWF staining was positive in all 12 tumors, although intensity and distribution varied significantly. In 2 tumors designated as epithelioid hemangiomas, there was strong positive cytoplasmic staining of between 80% and 90% of the tumor cells (Fig. 12). In cow No. 2, there was positive cytoplasmic staining for only 20–30% of cells, but this stain was performed in 1989 and may be inferior to the current protocol. As features of malignancy increased, there was progressively weaker staining expressed by a smaller percentage of cells. Dog No. 10, an epithelioid hemangiosarcoma, had the fewest features of malignancy and the strongest cytoplasmic staining, with approximately 90% of tumor cells staining positive (Fig. 13). Conversely, dog No. 8, also an epithelioid hemangiosarcoma, had the most features of malignancy, including a very high mitotic rate (29 in 10 high power fields) and showed weak positive staining for only 5% to 10% of tumor cells (Fig. 14).
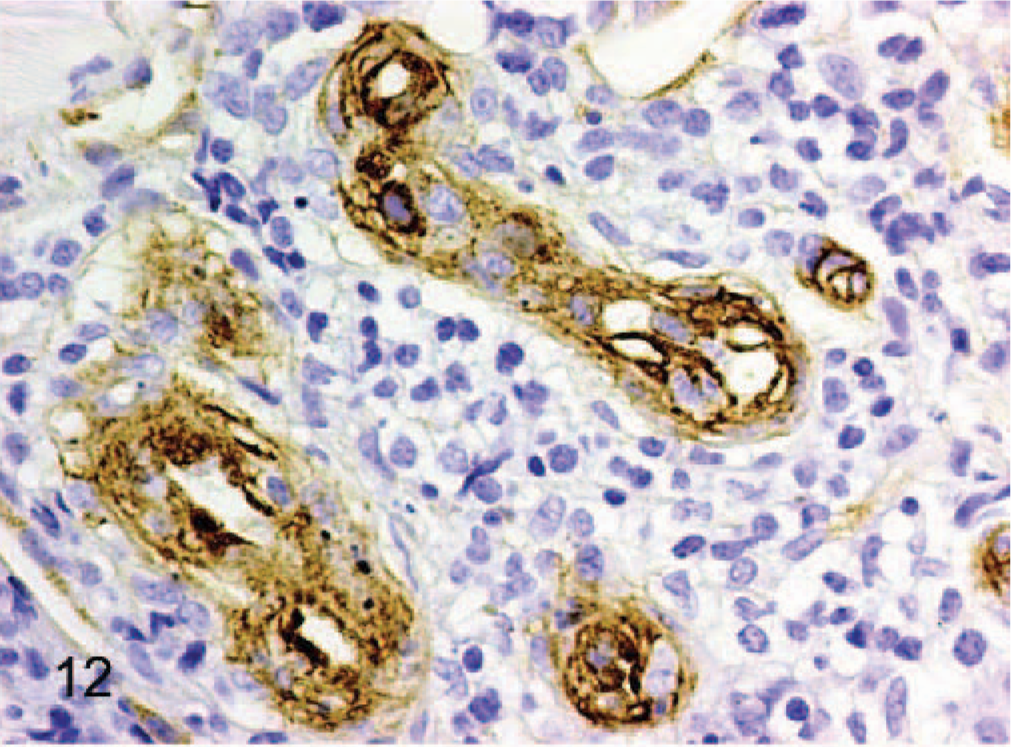
Dermis; horse No. 3. Epithelioid hemangioma: endothelial cells have strong cytoplasmic positive staining for vWF antigen, whereas intervening lymphocytes do not stain. Streptavidin–biotin immunoperoxidase method, Mayer's hematoxylin counterstain. Bar = 24 μm.
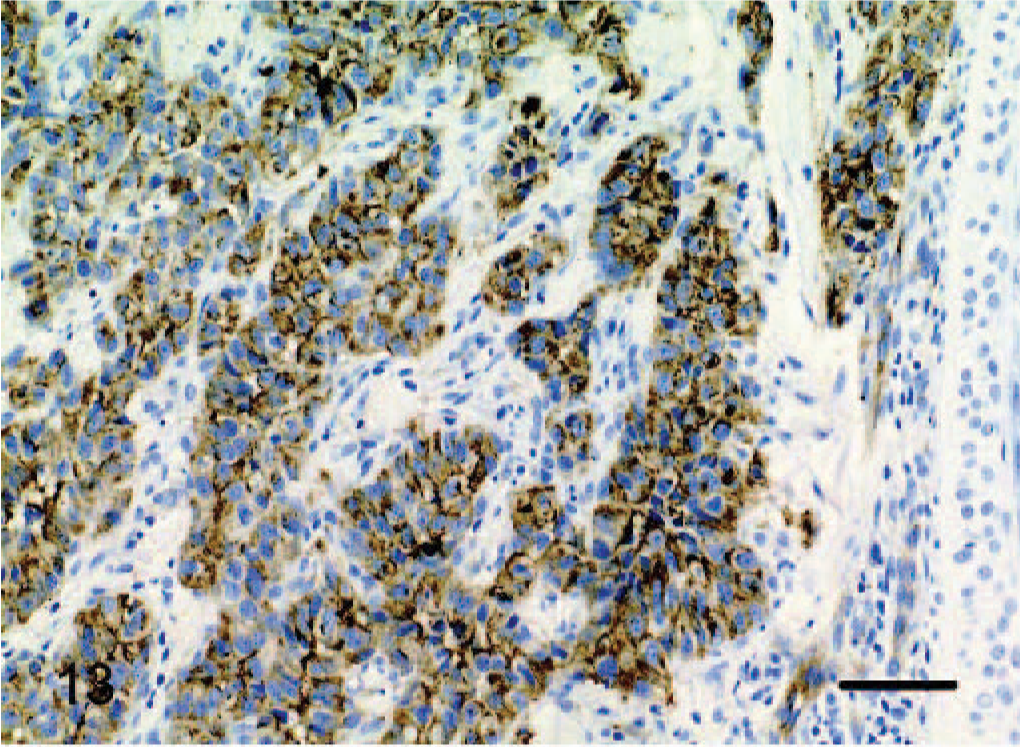
Dermis; dog No. 10. Epithelioid hemangiosarcoma, low-grade malignancy: clusters of endothelial cells have strong cytoplasmic staining for vWF antigen. Streptavidin-biotin immunoperoxidase method, Mayer's hematoxylin counterstain. Bar = 48 μm.
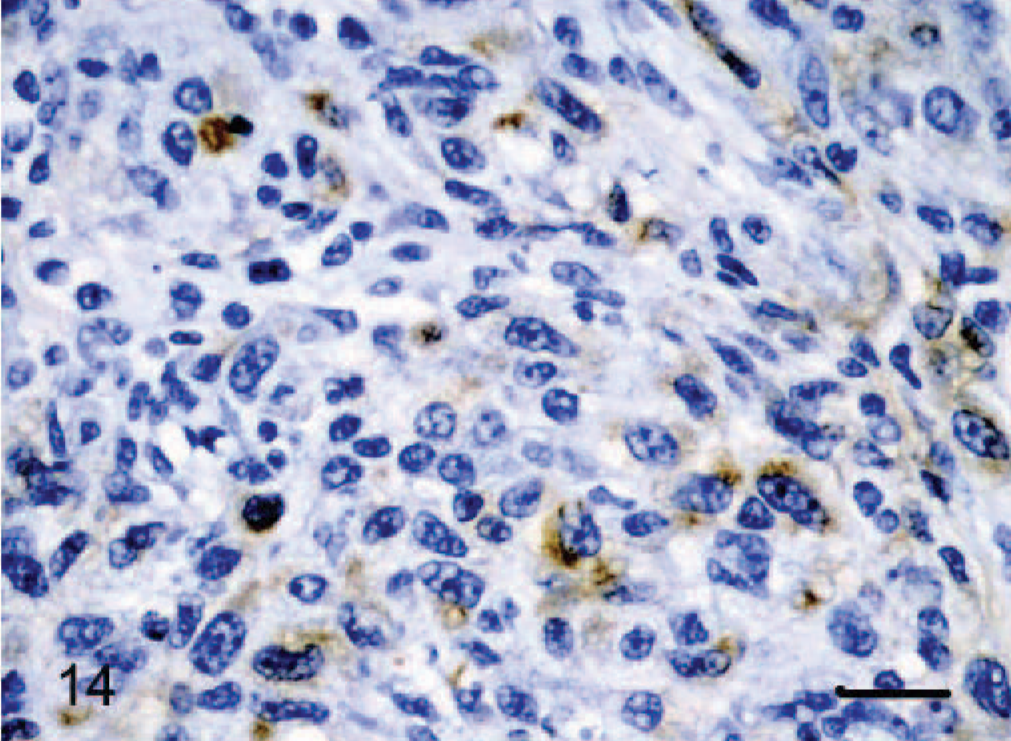
Dermis-subcutis; dog No. 8. Epithelioid hemangiosarcoma: tumors with the most malignant histologic features show weak positive cytoplasmic staining for vWF immunohistochemistry. Streptavidin–biotin immunoperoxidase method, Mayer's hematoxylin counterstain. Bar = 24 μm.
CD31 staining was positive in 9/9 tumors stained and, in contrast to vWF factor, showed linear cell membrane staining rather than cytoplasmic. Only 2 of the cases designated as epithelioid hemangiomas (horse Nos. 1 and 3) had tissue available for CD31 staining. Horse No. 1 showed strongly positive cell membrane staining and horse No. 3 showed weakly positive cell membrane staining. Of the epithelioid hemangiosarcomas, tissue was available for dog Nos. 4, 6, 8, and 9–12 for staining with CD31. Dog Nos. 4, 6, 8, 9, and 12 had strongly positive cell membrane staining (Fig. 15). Dog Nos. 10 and 11 had weakly positive cell membrane staining.
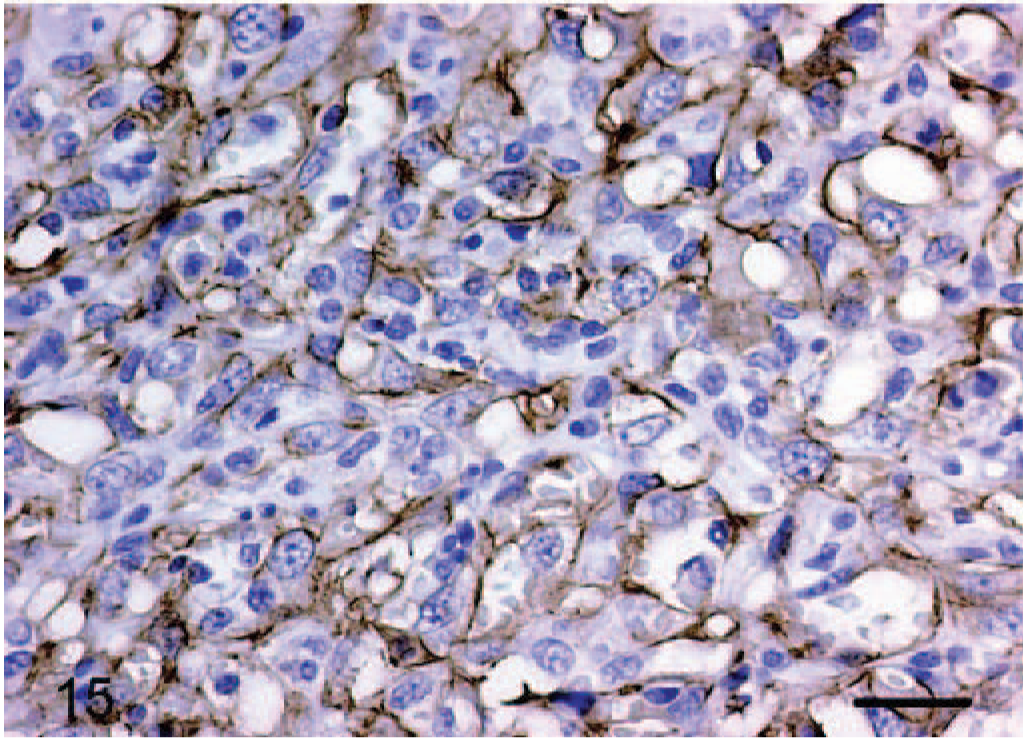
Dermis; dog No. 12. Epithelioid hemangiosarcoma: endothelial cells have diffuse strong positive cell membrane staining for CD31 (PECAM) antigen. Streptavidin–biotin immunoperoxidase method, Mayer's hematoxylin counterstain. Bar = 24 μm.
Cytokeratin staining was negative in all cases. There was strong positive staining of adnexal structures, follicles, and in addition, when present, overlying epidermis acting as an internal positive control (Fig. 16).
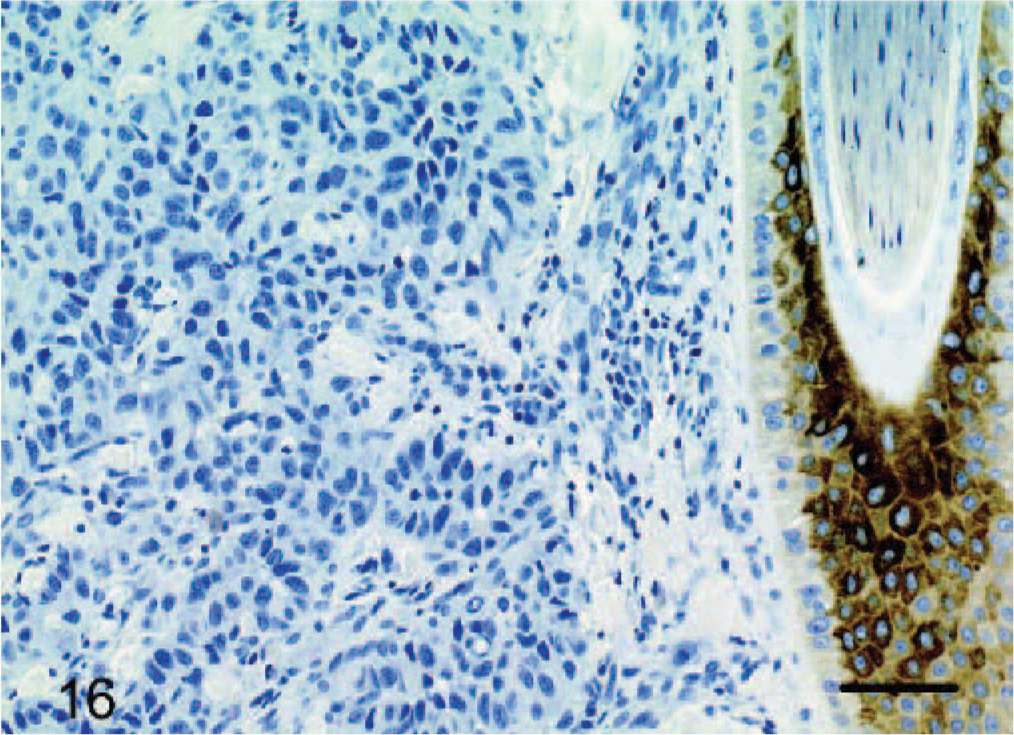
Dermis; dog No. 10. Epithelioid hemangiosarcoma: endothelial cells do not stain for cytokeratin, whereas the adjacent follicular wall has strong cytoplasmic staining. Streptavidin–biotin immunoperoxidase method, Mayer's hematoxylin counterstain. Bar = 48 μm.
Discussion
In this article, we describe a series of novel epithelioid endothelial tumors in animals that are but briefly mentioned in the veterinary literature and indeed seem to be largely unrecognized as judged by the paucity of reports. These 12 tumors share many features with a well described family of epithelioid vascular tumors in humans (see below) that historically have been confused with epithelial and histiocytic neoplasms. Subtle vascular differentiation is often the only feature indicating their true nature. 20 Vascular channels are instead small and recapitulate channels seen in early development. Within bundles of mesenchymal cells in these tumors, small intracytoplasmic vacuoles, representing primitive attempts at angiogenesis, are prominent. 20 Various adjunct diagnostic procedures were performed to establish that these tumors in humans were indeed vascular endothelial in origin, including varying degrees of positivity to vWF and CD31, and reticulin stains delineating basal lamina around small capillary-like vascular channels. 20 In the original description by Weiss and Enzinger, 20 electron microscopy showed features consistent with endothelial cells, including numerous tubular structures consistent with Weibel Palade bodies, pinocytotic vesicles, and prominent basal lamina. To our knowledge, no molecular basis for the unusual epithelioid morphology has been elucidated.
On the basis of a combination of microscopic features and behavior, endothelial vascular tumors in humans are subdivided into 3 major groups, which are designated hemangiomas, hemangioendotheliomas, and angiosarcomas. 11 In brief, hemangiomas are the most benign of endothelial tumors that in some cases regress spontaneously; hemangioendotheliomas are an intermediate group that shows local recurrence and some metastatic potential, whereas angiosarcomas reveal the greatest pleomorphism and highly malignant behavior with widespread dissemination. Although these 3 major groups are identified, it is probable that a continuum exists with some cases falling between categories. 17 Of the 12 cases reported here, all 8 canines and 1 equine case were diagnosed as hemangiosarcomas whereas 2 of the equine and the single bovine case were identified as hemangiomas. Interestingly, of the 8 canines, six were Golden Retrievers. This breed seems to be at increased risk for hemangiosarcomas, with the Golden Retriever Club of America national health survey 1998–1999 reporting this as the most common confirmed cause of death, with 18.5% of deaths attributable to hemangiosarcoma. It is not surprising that they would be highly represented in the epithelioid variant group of hemangiosarcomas.
The tripartite classification of endothelial tumors in humans has been applied to the epithelioid variants. 11, 19 Epithelioid hemangiomas in humans are benign hemangiomas of well formed sometimes thick-walled vessels. 11 Endothelial cells are large and cuboidal with abundant eosinophilic cytoplasm, large vesicular nuclei, and characteristic prominent cytoplasmic vacuoles that occasionally contain a single erythrocyte. There is no cellular atypia, and there are inconspicuous nucleoli and no or scarce mitoses. Inflammatory lymphoid infiltrates and abundant scattered eosinophils are additional features of these tumors in humans, with synonyms for epithelioid hemangiomas including “angiolymphoid hyperplasia with eosinophilia” and “atypical pyogenic granuloma” 11, 19 ; historically, these masses were thought to be reactive rather than benign tumors. There is often strong expression of vWF with the majority of cells staining positively. 11, 19 The 3 epithelioid hemangiomas we have described in 2 horses and a cow share clinical behavioral and pathologic features described for these tumors in humans. In these cases, the prominent architectural feature (animal Nos. 2 and 3) was the formation of thick-walled blood vessels lined by plump endothelium whereas the third case (horse No. 1) was uniformly glandlike with prominent endothelial cytoplasmic vacuoles. Cellular and nuclear atypia and mitoses were minimal in all 3 cases. Prominent inflammatory lymphoid infiltrate, described in human tumors, was present in only 1 case (horse No. 3) and rather than being predominantly peripheral, infiltrates dissected throughout the mass. Considering the young age of cow No. 2 (7 months) and the described distribution of the growth, a hamartoma was initially considered, however as this mass was composed of aberrant, malformed vessels rather than an exuberant proliferation of normally formed vessels, the diagnosis of hemangioma was favored. All 3 tumors had strong positive cytoplasmic staining in the majority of tumor cells for vWF although there was some variation in the proportion of the cells.
Epithelioid hemangioendothelioma in humans represents a tumor of low-grade malignancy positioned between an epithelioid hemangioma and epithelioid angiosarcoma. The term hemangioendothelioma has been employed sporadically in veterinary pathology 1, 5, 12, 13 and where used seems to describe a slightly more aggressive hemangioma. Two of the described veterinary hemangioendotheliomas additionally showed epithelioid morphology. 5, 13 Three tumors from this series (dog Nos. 6, 10, and 11) have cytologic features suggestive of low-grade or borderline malignancy. As the diagnosis of hemangioendothelioma has prognostic implications in humans, it was salient to consider if these 3 tumors (dog Nos. 6, 10, and 11) might qualify. Epithelioid hemangioendotheliomas in humans tend to form poorly marginated and infiltrative masses with multicellular or unicellular trabecular patterns with little or no vasoformative growth. Generally, neoplastic cells are epithelioid, although they infrequently blend into spindled areas. There is abundant pale eosinophilic cytoplasm showing similar striking cytoplasmic vacuolation seen in epithelioid hemangiomas and epithelioid angiosarcomas. Cytologic atypia is minimal, and the mitotic rate is low. 11, 19 A prominent myxohyaline matrix is described as an essential feature for the diagnosis of these tumors. Intravascular and intralymphatic growth is also commonly present with tumor cells showing a propensity to spread along pre-existing blood and lymphatic vessels. 17 Generally immunohistochemistry is positive for at least 1 endothelial marker (vWF, CD34, or CD31), and some are occasionally positive for keratin. The tumors in dog Nos. 6, 10, and 11, show some, but not all, features described in humans for this subset of tumors. Notably, no tumors showed a myxohyaline stroma, and none had the consistent architectural feature of cords of single cells. These 2 features are considered essential defining histologic features of this tumor subset in humans, 17 and in their absence, we were disinclined to use this classification. Thus, although there are features of intermediate-grade malignancy in these 3 tumors, namely reduced invasiveness, a lower mitotic rate, and fewer features of nuclear and cytologic atypia, these tumors do not fit the histologic description of epithelioid hemangioendotheliomas in humans. Consequently, we prefer to view them as low-grade hemangiosarcomas although these 3 cases could be seen as epithelioid hemangioendothelioma-like to convey their partial intermediate features. Whereas just a few vascular tumors in animals have been published as hemangioendotheliomas, the term seems often to have been used to convey a vascular endothelial tumor but nothing more. 5, 12, 13, 16, 18
Human epithelioid angiosarcomas generally are diffusely infiltrative, forming multilayered to solid endothelial proliferations as well as vasoformative structures. Early lesions are described as a proliferation of small, anastomosing vascular channels lined by a single layer of plump hyperchromatic endothelial cells that occasionally form tufts or micropapillations. These channels tend to dissect among dermal collagen and deep to the subcutis. More advanced lesions are highly cellular and form sheets or nodules. Cells tend to be atypical with large hyperchromatic nuclei and frequent atypical mitoses. As atypia increases, cells become closely packed and spindled with progressive loss of vascular channels. Tumors may or may not stain for vWF, are commonly positive for CD31 and occasionally positive for CD34, and up to 50% are keratin positive. 11, 15, 19
Tumors described from animal Nos. 4, 5, 7–9, and 12 share many of the features described for human epithelioid angiosarcomas. Growth patterns varied from tubular forms, akin to the early human epithelioid angiosarcomas, to glandlike to solid, similar to advanced human epithelioid angiosarcomas. Cellular and nuclear atypia was moderate to high, and the mitotic rate ranged between 4 and 43 in 10-high power fields. There was variable staining with vWF that reduced in intensity as nuclear atypia and mitotic rate increased. CD31 was positive with varying intensity in dog Nos. 4, 6, and 8–12 (tissue not available for animal Nos. 5 and 7). No tumor cells stained positive for cytokeratin in contrast to approximately 50% of human epithelioid angiosarcomas showing at least some positive staining. The sample size examined here, however, is small, a single cocktail of antikeratin antibodies was used (AE1/AE3), and it is to be expected that not all features would be present in the tumors examined. Potential positivity of epithelioid hemangiosarcomas in animals for cytokeratin should be kept in mind, however, particularly considering that carcinomas are the main differential diagnosis for this group of neoplasms. Cytokeratin expression in neoplastic endothelial cells may appear aberrant but endothelia of lower vertebrates, including trout and Xenopus frog, normally coexpress keratin and vimentin. 10, 14
We assigned the tumors as hemangiomas (n = 3) or hemangiosarcomas (n = 9), with dog Nos. 6, 10, and 11 of the second group having histologic features that are borderline/low-grade in malignancy. We were interested to learn how this classification related to the post-excisional behavior. A comprehensive questionnaire was sent to all hospitals that had submitted cases. Clinical follow-up was only attainable for 6 cases (animal Nos. 1, 3, 7, 9, 10, and 12), which ranged from 20 days to 15 years post-excision (Table 1). Two of the 6 cases for which follow-up was available (horse Nos. 1 and 3) were diagnosed as epithelioid hemangiomas. No tumor recurrence was noted in horse No. 1, and the animal was euthanized 15 years later because of an unrelated disease process (colic). Horse No. 3 did recur at the surgical site 1 month after the biopsy was reported. However, the original submission was a punch biopsy of a larger mass that was not fully excised. It is reasonable to assume that recurrence of this tumor is attributable to continued growth of the remaining tumor rather than regrowth; however, malignant behavior resulting in regrowth of the tumor cannot be excluded.
Of the epithelioid hemangiosarcomas, follow-up information was available for animal Nos. 7, 9, 10, and 12. In horse No. 7 the tumor did not recur. There was local recurrence of the tumor 6 months after excision in dog No. 9, and in dog No. 10 there was lymph node metastasis at the time of diagnosis. Both animals (dog Nos. 9 and 10) were euthanized directly because of the tumor. In dog No. 12, there was a recurrence at multiple dermal sites distant to the original excision sites. Although we interpreted this as metastatic spread, we recognize that de novo occurrence may have been the case. Dog Nos. 6, 10, and 11 were thought to be of lower grade based on fewer features of malignancy; however, at least dog No. 10 had the propensity for more aggressive behavior, regionally metastasizing to the local lymph node. This is compatible with the behavior of epithelioid hemangioendotheliomas in humans. Whereas these tend to be indolent and associated with long-term survival, rarely these tumors will recur locally and occasionally will metastasize to local lymph nodes and distant sites including the lung, liver, and bone. 17 However, as there is follow-up information for only 4 of the 9 hemangiosarcomas, no clear association between morphologically high- and low-grade tumors and clinical outcome can be made. Whereas it appears that the behavior of most of the 12 cases was favorable to long-term survival, a larger series will be needed to firmly establish the frequency of regional or widespread malignant behavior of this group of neoplasms.
Epithelioid hemangiomas and hemangiosarcomas in animals represent a previously poorly described variant of vascular tumors in animals that resemble tumors of epithelial origin. Their importance lies in the ambiguity of their features and the diagnostic challenge they pose. There is no evidence to suggest these tumors will act in a manner different to conventional cutaneous hemangiomas and hemangiosarcomas although this will more securely be determined by the study of a larger group.
Footnotes
Acknowledgements
We thank Drs Peter Rowland, Cameron Knight, Taryn Donovan, and Melissa Czajkowski for case material related to animal Nos. 10, 9, 11, and 12, respectively, and Patricia Fischer for technical assistance with the images and CD31 immunohistochemical staining. The authors are grateful to the technical assistance of Joy Cramer and Crystal Freitas of the immunohistochemistry laboratory at Cornell University.
