Abstract
Tumors in boars are uncommon, and testicular tumors even rarer. This study describes the pathological and immunohistochemical characteristics of a case series of testicular tumors in commercial boars with fertility problems. Tumors were detected in 19 of 333 animals (19/333, 5.9%). Macroscopically, tumors were observed in 13 (13/19, 68%) boars, while 6 cases (6/19, 32%) were only detected by microscopic examination. Testicular enlargement was observed in 1 boar, while in the others, tumors were only observed after removal of the scrotal skin or after sectioning of the testis. Histologically, tumors were classified as seminomas (16/19, 84%), mixed germ cell-stromal tumors (2/19, 11%), and B-cell lymphoma (1/19, 5%). Seminomas had 3 different growth patterns: intratubular (6/16, 38%), diffuse (4/16, 25%), and intratubular/diffuse (6/16, 38%). All tumors that were not evident on macroscopic examination were intratubular seminomas. Intratesticular metastases were observed in 2 cases and extratesticular metastases, located in the pampiniform plexus, were observed in 1 case. In 1 seminoma, the rete testis was also involved. By immunohistochemistry, all intratubular seminomas were negative for c-kit, cytokeratin, and vimentin. In diffuse seminomas, c-kit and cytokeratin were also negative, while vimentin showed granular or perinuclear cytoplasmic labeling in some areas. PAX-5 and CD-3 antibodies classified the lymphoma as a B-cell lymphoma. This study suggests that testicular tumors in boars may be more common than previously reported, especially when microscopic examination is performed. It also shows that testicular tumors in pigs are predominantly seminomas.
Keywords
Tumors in pigs are uncommon because they are typically slaughtered at 6–8 months of age, long before cancer usually develops. Although tumors have been described in young animals, they are typically sporadic and frequently congenital or hereditary.18,31,33 The importance of age is demonstrated by the increasing number of descriptions of tumors in pot-belly pigs, which live longer as pets.21,36 Various tumors have been reported in swine, with lymphoma and melanoma being the most frequent.3,9,16,20,22,29,34 Some authors, however, have found that nephroblastoma is more frequent than melanoma or lymphoma. 33
Testicular tumors are reported mainly in humans and dogs.2,7,40 In dogs, the majority are germ cell tumors, especially seminomas, and sex-cord stromal tumors, such as sustentacular (also referred to as Sertoli) or interstitial (also referred to as Leydig) cell tumors. 2 Less frequently, mixed tumors composed of germ cells and sex-cord stromal cells are reported.4,19 Other germ cell tumors, such as embryonal carcinoma, yolk sac carcinoma, or teratoma are more rarely reported in a variety of species. 2 Epithelial tumors of the testes, which typically arise from the rete testis, are extremely rare in humans and other animals.2,7
In boars, reproductive tumors are infrequently reported.6,9,17 Most references come from adult pet pigs,21,31,38 while references in commercial boars are very uncommon. 6 Although scrotal tumors appear to be relatively common in commercial pigs,9,33 testicular tumors are very rare, and individual cases have been reported sporadically.2,38 Among testicular neoplasms in pigs, germ cell tumors and sex-cord stromal tumors are the most commonly reported.6,9,17 Scrotal tumors are typically cutaneous capillary hemangiomas and have occasionally been associated with testicular hemangiomas. 33
Immunohistochemistry (IHC) has been used widely to classify testicular tumors in humans and dogs and less frequently in other animal species.7,14,23,24,32 For instance, seminomas can be classified as classical or spermatocytic based on c-kit and placental alkaline phosphatase (PLAP) labeling. However, differences between species have been demonstrated. C-kit has been shown to be a sensitive marker of classical seminoma in humans; however, in dogs, strong c-kit labeling has been demonstrated in spermatocytic seminomas. 23 Additionally, variable germ cell immunoreactivity with vimentin has been demonstrated in normal canine testes; some reports 24 describe cytoplasmic labeling, while others 23 did not observe any labeling. In porcine tumors, IHC has been used sporadically to identify specific cells.6,13,17,26
The objective of this study was to describe the pathological and immunohistochemical characteristics of testicular tumors detected in commercial boars rejected due to fertility problems.
Materials and Methods
Cases
The study population included 333 male pigs kept for breeding in 2 insemination centers and eliminated due to impaired fertility or genetic reasons. Fertility problems included an abnormal spermiogram, low conception rates, or prolificacy. Semen quality was evaluated microscopically, and pigs were classified into oligospermic, azoospermic, and teratozoospermic groups. Pigs that had a spermatozoa concentration less than 150 × 106/mL were considered oligospermic, and pigs that had no spermatozoa were considered azoospermic. Teratozoospermia was the cause of rejection when more than 30% abnormal spermatozoa were observed. The animals were slaughtered at one abattoir, and the scrotal skin with the testes and spermatic cord were collected and transported to the Department of Pathology of the Faculty of Veterinary Medicine of the University of Zaragoza. A detailed macroscopic and microscopic evaluation of these samples was carried out to detect the cause of rejection. The study sample included all the boars that had testicular tumors.
Pathological Study
Gross evaluation of the specimens included an initial external exam for evidence of scrotal lesions or lesions in the spermatic cord. Subsequently, the scrotal skin was removed and the scrotal cavity and epididymis were evaluated. The external surface of the testes was evaluated to detect alterations in morphology, such as abnormal testicular size or the presence of nodules. Subsequently, several transverse and longitudinal incisions were made through both testes. If no lesions were observed, a transverse sample of approximately 4 × 2 cm was taken from the central testicular parenchyma of a single testis for histopathology. The head, tail, and body of the epididymis were sampled. Samples were taken from the spermatic cord when it was enlarged. Tumor samples were obtained based on size and appearance of the lesions. In cases with multiple homogeneous tumors, samples were taken from several lesions. In variegated tumors, samples were obtained from areas with different appearances. For all lesions, samples always included the periphery of the lesion. The samples were fixed in 10% buffered formalin for 48 hours, embedded in paraffin, and sectioned at 4 µm. Sections were stained with hematoxylin and eosin. The mitotic count was determined by counting mitotic figures in an area of 2.37 mm2, using consecutive fields. All seminomas were evaluated for the presence of multinucleated cells, although they were not counted.
Immunohistochemistry
IHC for c-kit, vimentin, cytokeratin, and inhibin-α was performed on all tumors. IHC for CD3 and PAX-5 was performed in the lymphoma case. IHC was performed using the Dako Envision system on a semiautomatic stainer (Dako), following the manufacturer’s instructions. Details of the antibodies and protocol are available in Table 1. This method is based on a horseradish peroxidase-labeled polymer that is conjugated with mouse or rabbit secondary antibodies. All antibodies were from Dako and antigen unmasking was performed on a Dako PT Link module; it was carried out at 96º C for 20 minutes. A normal porcine testis was used as a positive control for c-kit, vimentin, cytokeratin, and inhibin-α, and residual normal testicular tissue in tumor cases was used as an internal control. For the CD3 and PAX-5 antibodies, a lymph node from a pig was used for positive control. The negative control was the sample without the primary antibody, which was replaced by buffer. Cross-reactivity of the antibodies was previously tested in appropriate tissues using canine and porcine samples, such as intestine, skin, and testis (c-kit, vimentin, cytokeratin, and inhibin-α) and lymph nodes (CD3 and PAX-5). A canine mastocytoma was also used to determine c-kit cross-reactivity and then used to validate c-kit in pigs. Labeling of mast cells using the c-kit antibody served as an internal control for this antibody and allowed for the estimation of the presence of mast cells in the tumors, although a detailed study was not performed. An antibody against PLAP was also used; however, it did not show reactivity in porcine tissues.
Antibodies and procedures for immunohistochemistry.
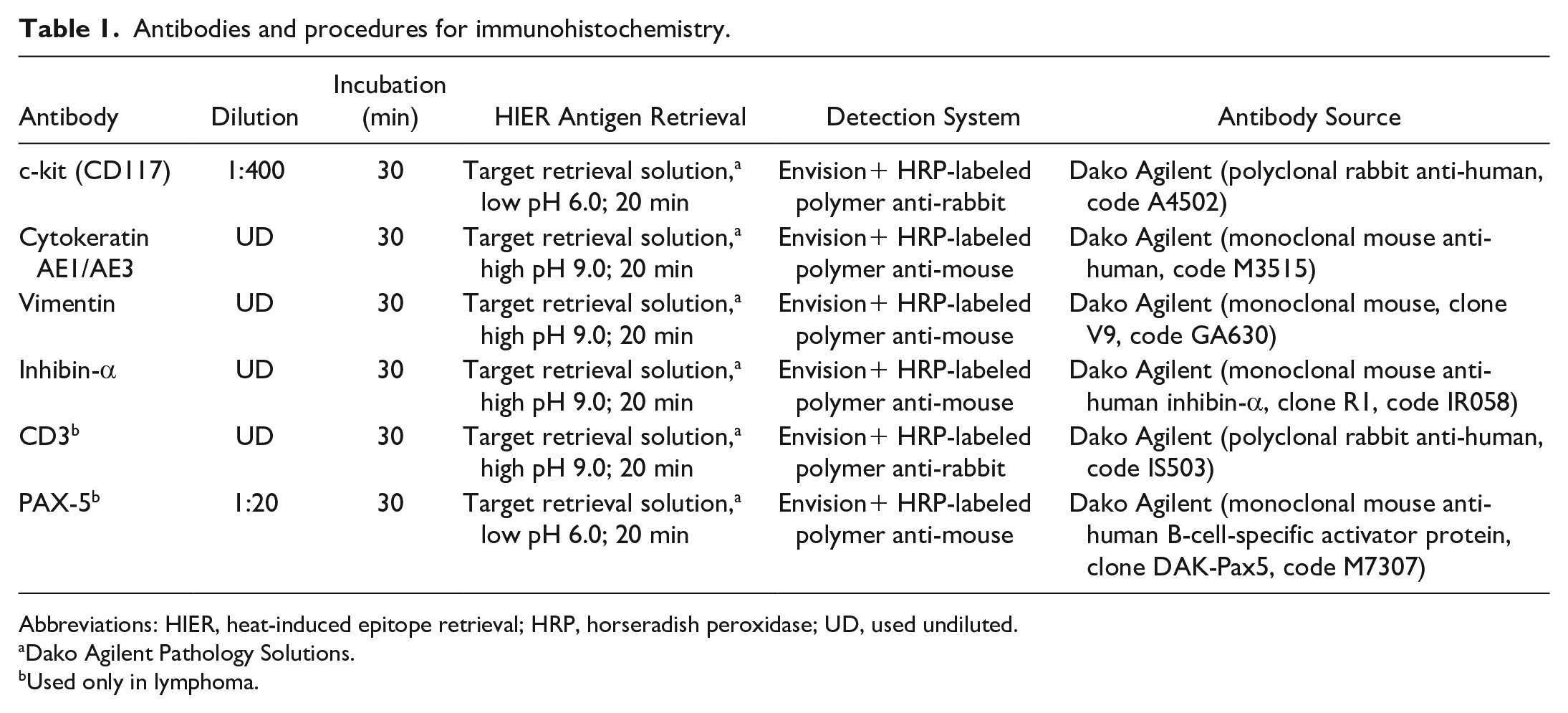
Abbreviations: HIER, heat-induced epitope retrieval; HRP, horseradish peroxidase; UD, used undiluted.
Dako Agilent Pathology Solutions.
Used only in lymphoma.
Results
Cases
A total of 19 of the 333 boars (19/333, 5.9%) studied had testicular tumors (Table 2). Fourteen cases (14/19, 74%; cases 1–3, 5–8, 10–12, and 15–18) were found in the Pietrain breed, 2 cases (2/19, 11%; cases 9 and 13) in the Landrace breed, 1 case (1/19, 5%; case 4) in the large white breed, and 2 cases were from an unknown breed (2/19, 11%; cases 14 and 19). The age of the animals ranged between 10 months and 7 years, with an average of 3.4 years. In 6 cases, the age was unknown. Six boars (6/19, 32%; cases 4–7, 9, and 15) were eliminated for teratozoospermia, 7 (7/19, 37%; cases 1–3, 10, 12, 13, and 17) for oligospermia, and 1 (1/19, 5%; case 18) for azoospermia. One oligospermic boar (1/19, 5%; case 12) also presented with severe arthritis. Five oligospermic boars (5/19, 26%; cases 2, 3, 10, 13, and 17) were also eliminated by genetic selection. Five pigs (5/19, 26%; cases 8, 11, 14, 16, and 19) were eliminated for unknown reasons. Tumors were observed macroscopically in 13 animals (13/19, 68%; cases 1–3, 5–7, 9–11, 13–15, and 17) and only microscopically in 6 (6/19, 32%; cases 4, 8, 12, 16, 18, and 19).
Summary of macroscopic and microscopic findings and epidemiologic data of 19 breeding boars with testicular tumors.
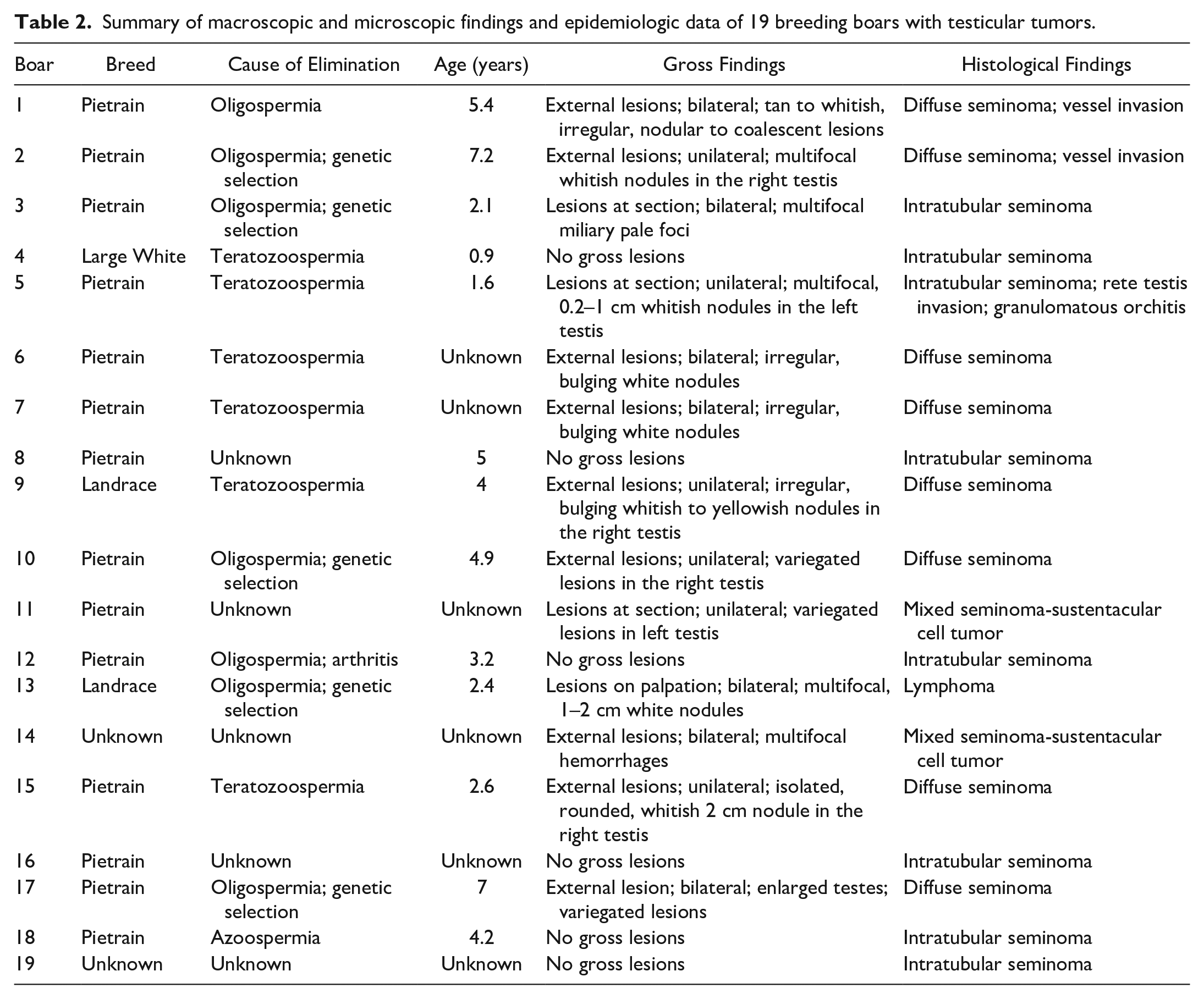
Gross Findings
Bilateral enlargement of the testes was observed in 1 pig (1/19, 5%; case 17), while in the remaining 12 cases with grossly evident tumors (12/19, 63%), the lesions were only detected after the removal of the scrotal skin, with 8 tumors (8/19, 42%; cases 1, 2, 6, 7, 9, 10, 14, and 15) protruding into the testicular capsule and others detected only by palpation (1/19, 5%; case 13) or after sectioning (3/19, 16%; cases 3, 5, and 11). The tumors observed in the testicular capsule were rounded, slightly to moderately prominent nodules of various sizes (Fig. 1a, Supplemental Figure S1a). Nodules were also observed in the right testis of the pig with testicular enlargement. Bilateral tumors were observed in 7 pigs (7/19, 37%; cases 1, 3, 6, 7, 13, 14, and 17), although typically one testis had more extensive lesions.
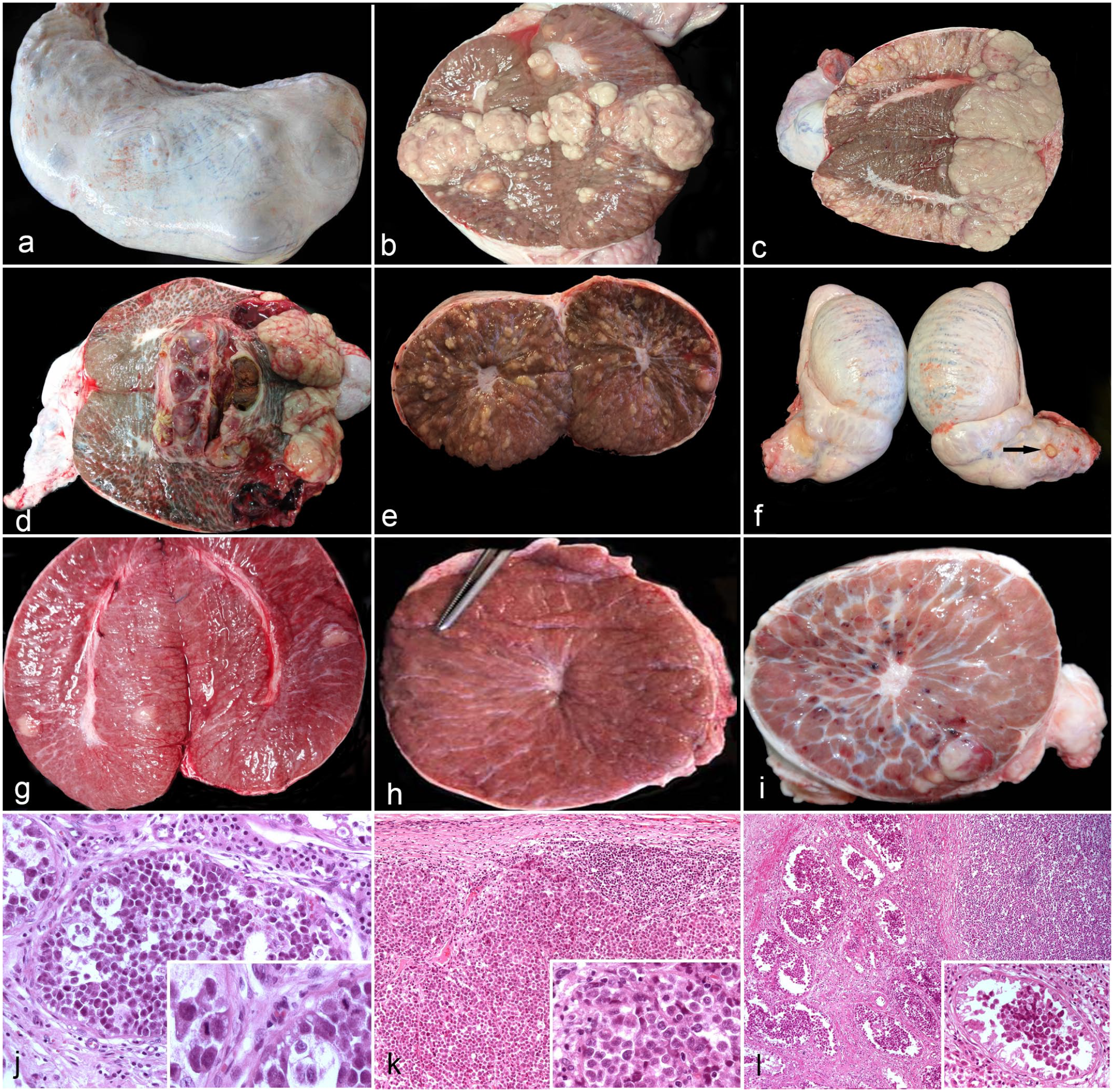
Testicular tumors in commercial boars. Gross and microscopic characteristics. (a) Nodules of seminoma on the testicular surface. Case 7. (b) Seminoma showing bulging tan to whitish masses of various sizes. Case 9. (c) Seminoma manifesting as bulging tan masses involving most of the testicular parenchyma. Case 1. (d) Seminoma with bulging tan masses containing necrotic and hemorrhagic cystic areas. Case 10. (e) Seminoma comprising tan to yellow, slightly elevated and irregular nodules, and is less than 1 cm in diameter, affecting the entire left testis. Case 5. (f) Spermatic cord of the pig with the tumor represented in (e) with a small nodular lesion on its surface (arrow) that microscopically corresponded to a metastasis. Case 5. (g) Lymphoma. Scattered gray nodules approximately 1–2 cm in diameter are observed. Case 13. (h) Seminoma comprising white, pinhead-sized lesions. Case 3. (i) Seminoma with tan nodules and small scattered hemorrhages throughout the testicular parenchyma suggestive of a vascular tumor. Case 14. (j) Intratubular seminoma. A tubule is filled with round cells with small to moderate amounts of eosinophilic cytoplasm. Inset: cells have round, oval, or indented nuclei. Two mitotic figures are observed near the basement membrane of 2 adjacent seminiferous tubules. Hematoxylin and eosin. (HE). (k) Diffuse seminoma. Numerous cells with a “starry sky” appearance delimited by a thin fibrous capsule replace the parenchyma. Inset: higher magnification of the large, round neoplastic cells. HE. (l) Diffuse/intratubular seminoma showing both appearances described in figures (j) (left side of the image) and (k) (right side of the image). Inset: tubule with intratubular seminoma with neoplastic cells in the lumen. HE.
On sectioning, most tumors were tan to whitish, bulging, soft to firm, variably sized, nodular to coalescent masses, occupying approximately 20 to 80% of the testis (Fig. 1b–d). Lesions were more frequently located near the surface, although more extensive tumors invaded central areas. In 4 cases (4/19, 21%; cases 10, 11, 14, and 17), necrotic, hemorrhagic, or cystic areas were also observed (Fig. 1d). In case 5, the macroscopic features were slightly different, with tan to whitish, slightly raised and irregular nodules, less than 1 cm in diameter, involving the entire left testis (Fig. 1e). In addition, this pig had a nodular lesion on the surface of the spermatic cord that was microscopically identified as a metastasis (Fig. 1f). In case 13, scattered, soft, gray nodules of approximately 1–2 cm in diameter were observed in both testes (Fig. 1g). In case 3, firm, white, pinhead-sized lesions (Fig. 1h) corresponded with an intratubular seminoma on microscopic examination. In case 14, whitish nodules mixed with small hemorrhages, suggestive of a vascular tumor, were observed scattered through the testicular parenchyma (Fig. 1i).
Other lesions were observed in the testes with tumors; some of them were considered incidental. Cysts near the head of the epididymis were observed in 4 pigs (4/19, 21%; cases 1, 3, 4, and 7). The cysts were located in the serosal layer at the intersection of the testis and epididymis and were covered by a thin capsule. Three were filled with clear fluid and one with hemorrhagic fluid (Supplemental Figure S1b). Enlarged and edematous spermatic cords with dilated blood vessels were frequently observed (7/19, 37%; cases 1, 2, 5, 7, 8, 10, and 14) (Supplemental Figure S1a). Pale areas of variable extension were observed (5/19; 26%; cases 2, 5, 7, 10, and 14) on the cut surface of some testes. In 4 pigs (4/19, 21%; cases 9, 10, 11, and 18), a variable number of reddish to blackish strawberry-like polypoid nodules, approximately 1 to 2 cm in diameter, were observed on the scrotal skin (Supplemental Figure S1c).
Histological Findings
Microscopically, 16 (16/19, 84%) tumors were seminomas, 2 (2/19, 11%; cases 11 and 14) were mixed germ cell-stromal tumors, and 1 (1/19, 5%; case 13) was lymphoma. Seminomas had 3 different growth patterns, intratubular (6/16, 38%), diffuse (4/16, 25%), and intratubular/diffuse (6/16, 38%) (Fig. 1j–l). All tumors that were only detected by microscopic examination were intratubular seminomas (Fig. 1j). In these testes, between 5 and 30% of the tubules were occupied by large, round cells with scant eosinophilic cytoplasm and round, oval, or indented vesicular nuclei with coarse chromatin and 1 or 2 prominent nucleoli. Some cells had clear cytoplasm. The mitotic count varied from 0 to 30 / 2.37 mm2 (Fig. 1j). Some tubules had tumor cells mixed with areas of spermatogenesis, while others were totally occupied by tumor cells, and in others, tumor cells were located in the center of the tubule (Fig. 1l). Occasionally, sustentacular cells were absent in tubules occupied by tumor cells, as demonstrated by immunohistochemistry for vimentin.
In diffuse seminomas, the testicular parenchyma was replaced by neoplastic cells, frequently with a nodular pattern, in which nodules of variable size were delimited by a fibrotic layer of variable thickness. The cells were rounded or polygonal and of 3 different sizes, with large, intermediate, and small cells (Fig. 2a). In general, intermediate cells predominated, although in some areas small cells were abundant. The cells were normally grouped together and separated by thin fibrovascular connective tissue. Mitotic figures were more frequent than in the intratubular pattern, and the number of mitotic figures varied within different areas of the same tumor; the mitotic count varied from 20 to 60 / 2.37 mm2. Multinucleate cells were more frequent in diffuse seminomas (Fig. 2a). In 2 tumors with a diffuse pattern, clear cells with abundant vacuolated cytoplasm were observed in some areas (Fig. 2b). A lymphocytic infiltrate was observed in 3/19 (18%) tumors, mainly near the fibrous septa. In 2 diffuse seminomas (cases 1 and 7), the infiltration was moderate to strong, while in a mixed tumor (case 14), it was mild (Fig. 2c). Necrosis, hemorrhages, and calcification were observed mainly in tumors with a diffuse pattern. Marked fibrosis was present in some areas of these tumors. In 2 cases (cases 1 and 2) of diffuse seminoma, invasion of the lymphatics of the testicular capsule by tumor cells was observed. The cells formed aggregates mixed with fibrin and blood inside the vessels. In the 2 tumors (cases 11 and 14) classified as mixed, seminoma predominated. In these tumors, tubules with proliferation of both sustentacular cells and seminoma cells were observed. These tubules often had sclerotic membranes and fewer spermatic cells, as demonstrated by labeling for vimentin (Fig. 2d).
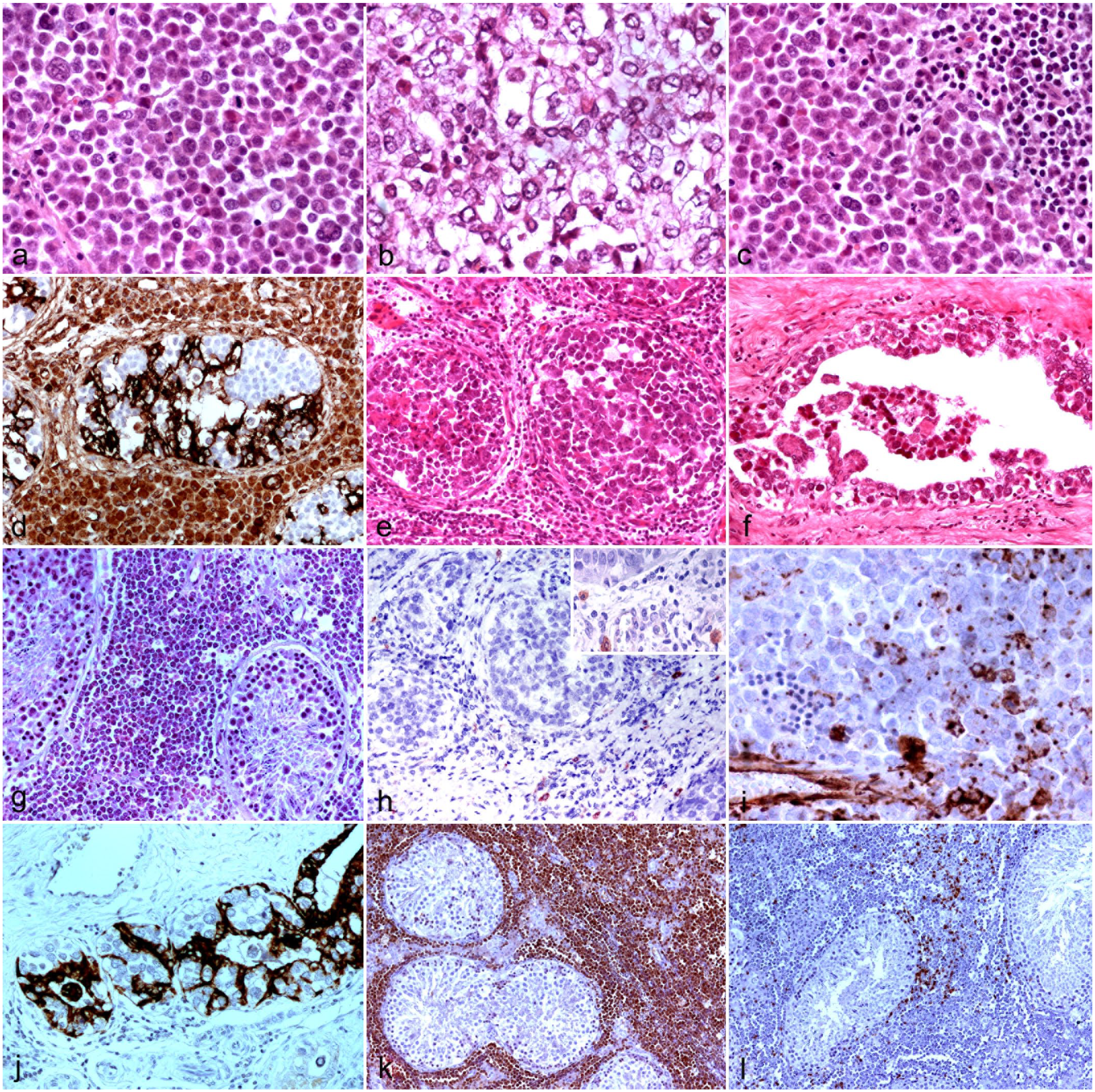
Testicular tumors in commercial boars. Microscopic and immunohistochemical findings. (a) Diffuse seminoma with round cells of 3 different sizes (large, intermediate, and small). Multinucleated cells are also observed. Hematoxylin and eosin (HE). (b) Diffuse seminoma with clear cells containing vacuolated cytoplasm. HE. (c) Diffuse seminoma with a lymphocytic infiltrate. HE. (d) Mixed germ cell-stromal tumor with intratubular proliferation of atypical sustentacular cells surrounding groups of seminoma cells. A slightly sclerotic tubular membrane is also observed. Immunohistochemistry (IHC) for vimentin. (e) Intratubular seminoma with tumor cells mixed with granulomatous inflammation and necrosis. HE. (f) Intratubular seminoma in (e) with invasion of the rete testis. HE. (g) Lymphoma with lymphoid cells infiltrating the interstitium. HE. (h) Intratubular seminoma with c-kit negative tumor cells and c-kit positive mast cells in the interstitium, more evident in the lower right corner. Inset: higher magnification of the c-kit positive mast cells. IHC for c-kit. (i) Diffuse seminoma with some cells showing complete or partial, granular or perinuclear cytoplasmic immunoreactivity for vimentin. IHC for vimentin. (j) Seminoma with invasion of the rete testis, showing irregular expression of cytokeratin in the epithelial cells but not in the seminoma cells that appear grouped. IHC for pan-cytokeratin AE1/AE3. (k) Testicular lymphoma with numerous PAX-5-positive neoplastic cells infiltrating the interstitium. IHC for PAX-5. (l) Testicular lymphoma with few CD3-positive lymphocytes scattered among the neoplastic B cells. IHC for CD3.
In the 4 cases with a different macroscopic presentation, various lesions were observed. Multifocal, intratubular, granulomatous, and necrotic inflammation and intratubular seminoma (Fig. 2e) were observed in the pig with irregular, slightly raised, tan to yellow nodules (case 10; Fig. 1e). In this pig, tumor cells invaded the rete testis (Fig. 2f) and the surface of the spermatic cord. The epithelium of the rete testis was irregularly invaded by round to polygonal cells, which were similar to the cells of the seminoma. The invading cells were irregularly distributed and frequently detached into the lumen of the rete testis (Fig. 2f). In the pig with bilateral, scattered, gray, and soft lesions (case 13; Fig. 1g), neoplastic lymphoid cells infiltrated the interstitium (Fig. 2g). In the pig with pinhead-sized lesions (case 3; Fig. 1h), an intratubular seminoma was observed on microscopic examination. In the pig with gross hemorrhages (case 14; Fig. 1i), a seminoma with central hemorrhages was observed.
Microscopically, epididymal cysts were lined by a single layer of ciliated, cuboidal, or columnar cells surrounded by various amounts of fibrous tissue (Supplemental Figure S1d). In the enlarged and edematous spermatic cords, the pampiniform plexus had tortuous and dilated, occasionally calcified, veins accompanied by multifocal mononuclear infiltrates in the interstitium. The affected veins were small to medium-sized. Fibrin emboli were observed in some vessels (Supplemental Figure S1e). The pale areas noted grossly were composed of tubules with partial or total hypospermatogenesis, the latter corresponding to sustentacular cell-only tubules. The scrotal lesions were capillary hemangiomas (Supplemental Figure S1f).
Immunohistochemical Findings
In normal testes, the cytoplasm of sustentacular and interstitial cells, but not germ cells, was strongly positive for vimentin. The sustentacular cells had a pyramidal shape with the base adjacent to the basement membrane of the tubule and were regularly distributed (Supplemental Figures S2a-S2b). Immunoreactivity for vimentin was also observed in the epithelium of the rete testis, the capillary walls, and the fibroblasts of the interstitium. Cytokeratin was only expressed in the epithelium of the rete testis (Supplemental Figure S2c). Expression of c-kit was observed in the cytoplasm of primordial germ cells, located in the basement membrane of the tubules, between the sustentacular cells, and in the cytoplasm of mast cells located in the interstitium between the interstitial cells (Supplemental Figure S2d). Immunoreactivity for inhibin-α was only observed in the cytoplasm of interstitial cells (Supplemental Figures S2e-S2f). Occasional germinal and sustentacular cells were also labeled. IHC for PALP did not show reactivity in the present study.
In the tumors, all intratubular seminomas were negative for c-kit, cytokeratin, vimentin, and inhibin-α. In some areas, interstitial cells located between the tubules with intratubular seminomas were smaller and less immunoreactive for inhibin-α. All diffuse seminomas were negative for c-kit, cytokeratin, and inhibin-α, although IHC for c-kit highlighted a moderate increase in mast cells in the interstitium of 2 tumors (Fig. 2h). Interstitial cells located between the seminoma cells labeled for inhibin-α, although the intensity was milder than in normal testes. In some areas of diffuse seminomas, there was granular cytoplasmic or perinuclear immunolabeling for vimentin that was complete or partial (Fig. 2i). In the pig with the metastatic seminoma and invasion of the rete testis (case 5), the epithelial cells of the rete testis, but not the seminoma cells, were positive for cytokeratin and vimentin (Fig. 2j). In the mixed germ cell-stromal tumors, IHC for vimentin showed proliferation of sustentacular cells with altered morphology (Fig. 2d). In the lymphoma, most of the cells were positive for PAX-5 (Fig. 2k) and some were positive for CD-3 (Fig. 2l), and consequently, it was classified as B-cell lymphoma.
Discussion
This study suggests that testicular tumors in pigs may be more prevalent in older boars or may require microscopic examination of the testes to identify them, as previously suggested.6,9,17,25,33,37 However, this measure of prevalence could be biased since these boars were selected from pigs with fertility problems.
This study has also shown that testicular tumors may be present in young boars, under 2.5 years, and this finding is in contrast to most reports.25,33,37 In one study, 37 3 of 30 castrated males older than 5 years of age had intratubular seminomas identified on microscopic examination. In that study, a 3.5-year-old boar had a macroscopic seminoma composed of grayish-yellow, glass pinhead-shaped foci. 37 This description is similar to case 3 (Fig. 1h) of the present study. Similarly, in another study, microscopic tumors were found in 4 of 80 castrated boars between 8 and 60 months of age, although their age distribution was not reported. 25 Microscopic intratubular seminomas were also observed in a 9.5-year-old Pietrain who had a diffuse seminoma in an enlarged testis 6 and in pigs with testicular hemangiomas. 33 These authors studied the testis of 12 boars with scrotal hemangiomas and detected gross testicular hemangiomas in 3 of them; in 2 of them, microscopic study identified 1 intratubular seminoma and 1 sustentacular cell tumor, respectively. 33 Interestingly, these 2 animals were 5-years-old, while hemangiomas were even found in boars less than 1 year. In the present study, 6/19 (32%) tumors were only detected microscopically, although the prevalence may have increased if multiple sections of both testes were collected in each case. Sporadically, testicular tumors have been reported in very young pigs; one study reported a metastatic, malignant sustentacular cell tumor mixed with an interstitial cell tumor in an enlarged testis of a 6-month-old boar. 17 In the present study, tumors were found in 4 boars that were considered young out of the 13 (31%) animals of known age. However, the boar under 1 year of age had a microscopic tumor.
Testicular tumors are predominantly classified as germ cell tumors, most commonly seminomas, and stromal tumors, such as sustentacular or interstitial cell tumors, in all species.2,11 Seminoma is divided into intratubular and diffuse forms, and some tumors present both patterns. Mixed germ cell sex-cord stromal tumors and other types have also been described, although less frequently. Mixed tumors are composed of germ cells and sex-cord stromal cells, although 1 cell type tends to predominate.4,19 However, there is some controversy regarding mixed tumors, especially in human pathology.2,19 Some authors believe the germ cells are not neoplastic and are just entrapped between neoplastic sustentacular cells, while others report that these germ cells have microscopic and immunohistochemical characteristics of seminoma, and the tumors can therefore be classified as mixed tumors. 19 In addition, some authors diagnose mixed tumors when seminoma and sustentacular cell tumors about each other, but these tumors should be classified as collision tumors. 2 True mixed tumors are those in which tubules with neoplastic germ cells are intimately admixed with tubules with neoplastic sustentacular cells. In the present study, this appearance was observed in 2 cases.
Unexpectedly, seminoma was the predominant tumor in the present study, and no pure sex-cord stromal tumors were detected. Regarding tumors only detected microscopically, our results agree with other authors,33,37 and with what has been described in dogs; 11 these tumors are predominantly seminomas. This only differs from those reported by Pereira et al, 25 who found that 3 of 4 microscopic tumors were interstitial cell tumors, and only 1 was a seminoma. Intratubular proliferation of germ cells seems to be the precursor of seminoma, and this may explain the high prevalence of seminomas found in this study. Microscopic intratubular seminomas could evolve into macroscopic seminomas if affected boars lived longer. A variegated appearance is more typically associated with interstitial cell tumors in dogs. 2 In the present study, 3 tumors showed this appearance but microscopically corresponded with seminoma. In humans, spermatocytic seminomas may show a more varied appearance. 15
Seminomas in humans have been classified into classical and spermatocytic, which have different microscopic and immunohistochemical patterns. 15 Classical seminomas are composed of homogeneous cells that express c-kit because they are derived from undifferentiated germ cells. In contrast, spermatocytic seminomas are pleomorphic with small, medium, and large cells and are negative for c-kit because the cells are derived from more differentiated germ cells. Classical and spermatocytic seminomas in humans have different behaviors, with the classical seminoma being more aggressive and the spermatocytic seminoma being locally extensive and affecting older people. In dogs, most seminomas resemble spermatocytic seminomas, although their prevalence varies according to different authors. 2 In this study, the microscopic and immunohistochemical characteristics suggested the spermatocytic form. Furthermore, all but 2 were considered benign, as no invasion of lymphatic vessels or adjacent tissues was observed. The infiltration of tumor cells into vessels or in tissues adjacent to the testes differentiates malignant from benign seminomas since their cellular morphology suggests malignancy in both cases. 2 Lymphocytic infiltrates in seminomas appear to be common; however, there are differences between humans and dogs. While in dogs, the infiltrates are mainly found in spermatocytic seminomas, in humans, they occur in classical seminomas. 2 In the present study, lymphocytic infiltrates were observed in 3 tumors, suggesting that spermatocytic seminomas in pigs have less lymphoid reaction compared to dogs and are more similar to what is observed in human spermatocytic seminomas. 15
IHC to differentiate testicular tumors has been widely used in humans and dogs and has helped classify undifferentiated tumors. However, species differences have been observed in both normal testis and tumors.23,24,32 These differences are frequently attributed to the antibodies or techniques used. 23 Some studies on porcine tumors have also used immunohistochemistry, although with variable results and antibodies used.6,17,32,33 In the present study, IHC for PLAP, a marker commonly used for seminoma differentiation, did not show reactivity in porcine tissues.
One of the most frequent uses of immunohistochemistry has been to differentiate classical and spermatocytic seminomas in humans 7 and dogs.11,14 Surprisingly, a study 23 reported a spermatocytic seminoma in a dog with strong c-kit labeling of the germ cells. However, most authors report IHC results similar to those observed in humans.11,14 In the present study, none of the seminomas were positive for c-kit, suggesting that this tumor is spermatocytic in pigs, although the proportion of the 3 different cell types characteristic of this seminoma was variable.
Vimentin is the main intermediate filament in sustentacular cells and is considered a useful marker of sustentacular and interstitial cells in both normal testis and testicular tumors, with strong cytoplasmic labeling.24,27 Immunoreactivity of germ cells with vimentin is variable. In normal canine germ cells, some authors have described perinuclear or granular cytoplasmic labeling, 24 while others reported no labeling. 23 In the present study, IHC resulted in strong cytoplasmic vimentin labeling in porcine interstitial and sustentacular cells, while germ cells were negative. These results agree with the only study carried out in pigs that used this marker. 33 Interestingly, in 2 tumors with cells resembling interstitial cells and initially classified as mixed tumors, immunohistochemistry revealed focal and cytoplasmic perinuclear labeling for vimentin similar to that observed in germ cells in normal dogs, and the tumors were subsequently classified as diffuse seminomas. Moreover, in these tumors, inhibin-α only labeled interstitial cells between seminoma cells. Inhibin-α has been used as a marker of interstitial and sustentacular cells, although with variable results depending on the animal species.10,23,32 In the dog, controversial data have been reported, with expression only in interstitial cells10,32 and in both interstitial and sustentacular cells. 23 In the present study, inhibin-α was only expressed by testicular interstitial cells, similar to what has been reported in cattle and horses 23 and to what has been reported in dogs by some authors,10,32 but it differed from other reports 23 and from other species.10,23 The various results appear to depend on the age of the animals.10,23,32 It is unclear whether this age-dependent expression of inhibin-α occurs in swine, as only mature boars were included in the present study.
Cytokeratin is another marker used in seminomas. Its expression has been demonstrated in a proportion of human seminomas, which suggests differentiation toward other germ cell tumors. 5 In dogs, cytokeratins have been shown to label sustentacular cell tumors but not seminomas.14,24 In this study, no immunoreactivity for cytokeratin was observed in seminomas, suggesting that the cells were more differentiated. These findings are consistent with our results of the c-kit IHC. Nevertheless, additional markers such as PLAP or melan A, some of them non-reactive in the present study, might add valuable additional information on testicular tumors in pigs.
Invasion of the rete testis by testicular tumors has been described in humans and is mainly associated with germ cell tumors, especially seminomas, and less frequently with sex-cord stromal tumors. 8 Two types of spreading into the rete testis have been described, a pagetoid extension and invasion from direct contact with the tumor. 8 In the present study, invasion of the rete testis by tumor cells was observed in a seminoma. Nonetheless, invasion of the rete testis should be differentiated from other lesions in the rete testis such as hyperplasia or primary tumors. Primary tumors of the rete testis are rare in human and domestic animals, 28 and several criteria have been suggested to differentiate invasion from primary lesions. 8 IHC has proven to be very useful to differentiate between lesions. Cytokeratin and vimentin are expressed in primary rete tumors, whereas adenomatous rete hyperplasia is negative for vimentin. 28 In this case, immunohistochemistry with cytokeratin and vimentin revealed labeling of the rete epithelium as previously reported, 35 but not of the invasive seminoma cells. It remains uncertain whether rete dissemination in porcine seminomas is common since the rete testis was only studied in 1 case in this study due to the atypical gross presentation compared to the other tumors. It would be advisable to assess the rete testis in all testicular tumors.
Vascular tumors, especially hemangiomas, appear to be common in the reproductive system of pigs, especially in the ovary and scrotal skin.9,33 However, vascular tumors in the testes seem to be rare, with only 2 reports found in the literature.9,33 Fisher and Olander, 9 studying 15,782 necropsy and biopsy cases over 10 years in a diagnostic laboratory, found only 1 vascular tumor, a hemangiosarcoma, in the testis of a 5-year-old Yorkshire boar. Teankum et al 33 observed a close association of scrotal hemangiomas with the presence of testicular hemangiomas. The present study suggests that scrotal hemangiomas are common, but testicular vascular tumors are rare. Although scrotal hemangiomas were frequently observed, no testicular hemangiomas were found in any of the 333 testes studied. Hemangiomas were macroscopically suspected in the testes of only 1 boar; however, microscopic examination revealed a seminoma with intratumoral hemorrhages.
Lymphoma is one of the most common tumors described in pigs and typically has a multicentric distribution with a female predominance and is mostly of B-cell origin.3,20,34 Testicular lymphoma, whether primary or part of disseminated presentation, is rare in humans and domestic species and has not been reported in pigs. A case of testicular involvement has been reported in a 5-month-old crossbreed boar with B-cell acute lymphoblastic leukemia. 30 The authors reported a pale tan nodule, 5-mm in diameter, on the cut surface of a testis. In our case, the lymphoma was observed in a 2.3-year-old Landrace pig and grossly manifested as bilateral pale tan nodules. The lymphoma was considered primary testicular lymphoma, since no lesions in other organs were reported. Immunohistochemistry confirmed a B-cell lymphoma in the present study. A small to moderate number of T lymphocytes was also present in some areas of the tumor.
Melanomas are common skin tumors in pigs, especially dark breeds. Most are benign, 9 although malignant and disseminated lesions have also been reported. Melanoma metastases to the testes are unusual in humans, and sporadic cases have been described in domestic species. 1 In this study, the majority of pigs were of the Pietrain breed, but no melanomas were observed on the skin or in the testes.
The other reproductive lesions observed in boars with testicular tumors were relatively frequent findings during the macroscopic evaluation of the 333 cases studied, although the majority were considered incidental findings. 2 Epididymal cysts are typically found in the head of the epididymis, are considered an incidental congenital malformation in pigs, and are not related to fertility problems. They must be differentiated from other cystic lesions, such as inclusion cysts or spermatocele. The presence of spermatozoa in the latter helps with differentiation. In the present study, the ciliated epithelium suggested that the cysts were remnants of embryonic ducts. 2 Dilated vessels of the pampiniform plexus that were not clinically suspected or identified were frequent incidental findings in our study, and no relationship with tumors was established. Pale areas corresponding to tubules with partial or total hypospermatogenesis were occasionally observed in sections of the testes that contained neoplasms. This was expected because it was a common finding in all boars studied. An interesting finding of uncertain significance in this study was the apparent increase in mast cell numbers in some testes with seminomas. Mast cells have been associated with subfertility and some seminomas in humans. 39 In the present study, it is not clear whether the mast cells were related to the tumor or to the observed hypospermatogenesis. Further studies should be done to investigate the relationship between mast cells and seminomas. Scrotal hemangiomas are frequent in pigs and are typically capillary hemangiomas, as observed in the present study. 2
In conclusion, this study suggests that testicular tumors in boars may be more common than previously reported, especially when microscopic examination is performed in both young and older boars. In this study, testicular tumors in pigs were predominantly spermatocytic seminomas. We recommend taking additional testicular samples, including at least 3 sections from both testes and the rete testis, for a more accurate estimation of the incidence of testicular tumors and documentation of their behavior in pigs. Finally, additional studies using new antibodies that work in porcine tissues could further characterize testicular tumors and allow evaluation of the immune microenvironment of testes with seminomas.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241269836 – Supplemental material for Testicular tumors in commercial boars with infertility: A gross, histologic, and immunohistochemical study
Supplemental material, sj-pdf-1-vet-10.1177_03009858241269836 for Testicular tumors in commercial boars with infertility: A gross, histologic, and immunohistochemical study by Raquel Ausejo, María Victoria Falceto, Olga Mitjana, Ana María Garrido, Juan José Badiola and Bernardino Moreno in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff at the insemination centers for providing information on the clinical cases, the slaughterhouse staff for allowing sample collection and the Magapor workers for taking the samples from the slaughterhouse to the University. We also thank the histology technicians at the Research Center for TSE and Emerging Transmissible Diseases from the Veterinary Faculty at Zaragoza University.
Authors’ Contributions
RA, MVF, and BM designed the study and protocols; RA, OM, MVF, AMG, and BM collected the samples and performed macroscopic evaluations; RA and BM performed immunohistochemical study; RA, JJB, and BM performed histologic evaluations; the manuscript was written by BM and RA with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Magapor, the University of Zaragoza through grants for doctoral studies (III convocatoria de ayudas para el Desarrollo del programa de doctorados industriales/empresariales published in the Boletín Oficial del Estado, n°1, 2 January 2018), the “Research Center for TSE and Emerging Transmissible Diseases,” and the Government of Aragón through the Research Group “Enfermedades priónicas, vectoriales y zoonosis emergentes (Ref. A05_20R).”
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
