Abstract
An 11-year-old cat with a retained testis was presented with a chronic history of dysuria and bladder atony. Medical therapy failed to alleviate the clinical signs. Contrast radiography demonstrated a diffusely narrowed urethra. During a celiotomy and prepubic urethrostomy, a retained testis, stenosed urethra, and irregularly enlarged prostate were observed. Histopathologic diagnosis was retained testis with a well-differentiated interstitial cell tumor, a poorly differentiated interstitial cell tumor, and marked squamous metaplasia of the prostatic epithelium with suppurative prostatitis. Neoplastic interstitial cells were immunoreactive for Melan A, consistent with reports of Melan A expression in steroid hormone-producing tissue. This is the first report of prostatic squamous metaplasia associated with testicular neoplasia in a felid.
Testicular tumors are uncommon in domestic cats. Interstitial cell tumors, testicular teratomas, Sertoli (sustentacular) cell tumors, and seminomas have been described in this species, and reported cases have recently been summarized. 15 In addition, there have been reports of interstitial cell and Sertoli cell tumors in ectopic testes or extratesticular locations. 6, 22 Cryptorchidism is an important risk factor for the development of testicular tumors in dogs, but the risk of testicular tumors in retained testes in cats is not known. Masculinization because of androgen-producing tumors has been well characterized, 15 but feminization, bone-marrow aplasia, or other changes because of estrogen production by testicular tumors have not been reported in cats. Squamous metaplasia of the prostatic and urethral epithelium has also been described in dogs with estrogen-secreting testicular tumors 12 but not in cats. This report describes interstitial cell neoplasia in a retained testis with associated prostatic epithelial squamous metaplasia and suppurative prostatitis in a cat.
A 5.6-kg, 10-year-old male domestic shorthaired cat was presented for investigation of stranguria and pollakiuria. A large, flaccid bladder was present on physical examination, but it was difficult to express. Passage of a urinary catheter demonstrated urethral patency. The urine specific gravity was 1.050 and urine pH was 6.0, with significant hematuria and proteinuria. Treatment consisted of a 2-week course of clavulanic acid potentiated amoxicillin (Clavamox, GlaxoSmithKline, USA) and phenoxybenzamine. The cat was presented to the University of Tennessee College of Veterinary Medicine, (UTCVM) 3 weeks later for recurrence of clinical signs.
Complete blood cell count and plasma biochemistry were within the reference range. Urinalysis did not differ from previous findings, and bacteriologic culture of urine was negative. Thoracic radiographs were unremarkable, abdominal radiographs showed bilateral irregular renal silhouettes consistent with chronic renal disease. A human gonadotropin (hCG) stimulation test (250 µg intramuscular [IM]) was performed. Baseline, 2-hour, and 4-hour concentrations of testosterone were 0.10 ng/ml, 0.18 ng/ml, and 0.11 ng/ml, respectively. Urethral spasm and bladder atony secondary to feline idiopathic cystitis (FIC) or a previous partially obstructive urolith was presumptively diagnosed. The cat was observed to void urine normally before discharge.
Approximately 1 year later, the cat was returned to UTCVM with a recurrence of clinical signs. The cat's weight had decreased to 3.7 kg. A distended bladder was palpated, but it could not be manually expressed. Clinicopathologic data were normal, with the exception of hematuria. A diffusely thickened bladder wall and proximal urethra were evident on abdominal ultrasound. A retrograde cystourethrogram showed essentially no change from the previous study, and there was narrowing, with smooth margination of the entire urethra; there was no evidence of a focal stricture or intraluminal filling defect. The condition was refractory to medical treatment. Therefore, a perineal urethrostomy was performed. The urethra was stenotic for at least 1-cm cranial to the bulbourethral glands and so the surgery was converted to an antepubic urethrostomy. During the exploratory celiotomy, a retained left testis was removed, and there was mineralization of both vasa deferentia. A 1.3-cm-diameter, multilobulated, smooth, white-to-tan mass that surrounded the pelvic urethra was observed (Fig. 1). The periurethral mass and stenotic distal penile urethra were resected. The cat was managed, after surgery, with an indwelling urinary catheter for 4 days but was unable to urinate voluntarily after the catheter was removed. A repeated exploratory celiotomy revealed a severely stenosed urethra. At the owners' request the cat was euthanatized. At this point, the owners reported that the cat had been neutered years before the onset of clinical signs and that a unilateral gonadectomy had been performed because only 1 descended testis was found.
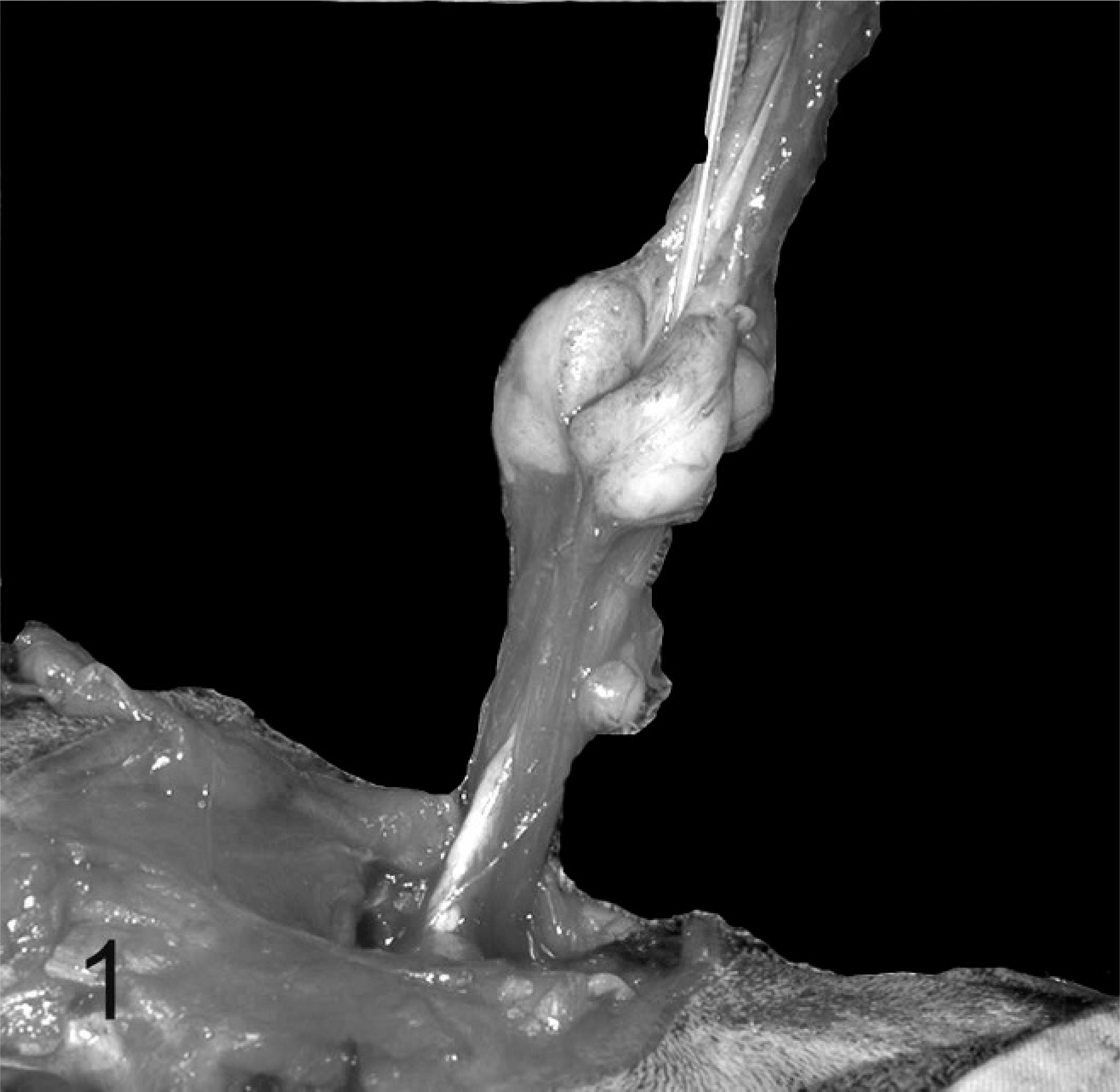
Prostate and urethra; cat. Intraoperative dissection of the urethra (with catheter in place) and prostate. Note the multilobulated enlargement of the prostate.
Histopathologic examination of the left testis revealed an interstitial cell tumor that involved approximately two thirds of the cross section of the testis (Fig. 2). It was composed of a well-demarcated, compressive proliferation of large polyhedral cells arranged in small packets or lobules. These cells contained round, uniform, nuclei and large amounts of eosinophilic cytoplasm. Many of the cells contained lipid vacuoles and small amounts of yellow cytoplasmic pigment. Cell borders were indistinct. There was atrophy of seminiferous tubules adjacent to the neoplasm, with intramural deposition of fibrous tissue. There was no spermatogenesis in the few remaining seminiferous tubules lined by Sertoli cells. A second neoplastic focus was composed of an unencapsulated, infiltrative, randomly arranged proliferation of polygonal to pyriform cells (Fig. 3). These cells had distinct borders, variably abundant, vacuolated, eosinophilic cytoplasm, and ovoid nuclei. Cellular pleomorphism was marked and cell polarity was lacking. Nuclei were hyperchromatic in smaller cells but had finely stippled chromatin and variably prominent nucleoli in larger cells. Round intranuclear cytoplasmic invaginations were occasionally present. Mitotic figures averaged 1 per high-power (400×) field. The second mass was also an interstitial-cell tumor. The mass associated with the stenotic urethra was histologically confirmed as prostate. Prostatic glandular acini were frequently distended and were lined by multiple layers of squamous epithelial cells (Fig. 4). There was marked accumulation of neutrophils, necrotic cells, cellular debris, and occasional acicular (cholesterol) clefts within acini. The interstitial connective tissue was infiltrated with neutrophils, macrophages, plasma cells, and lymphocytes.
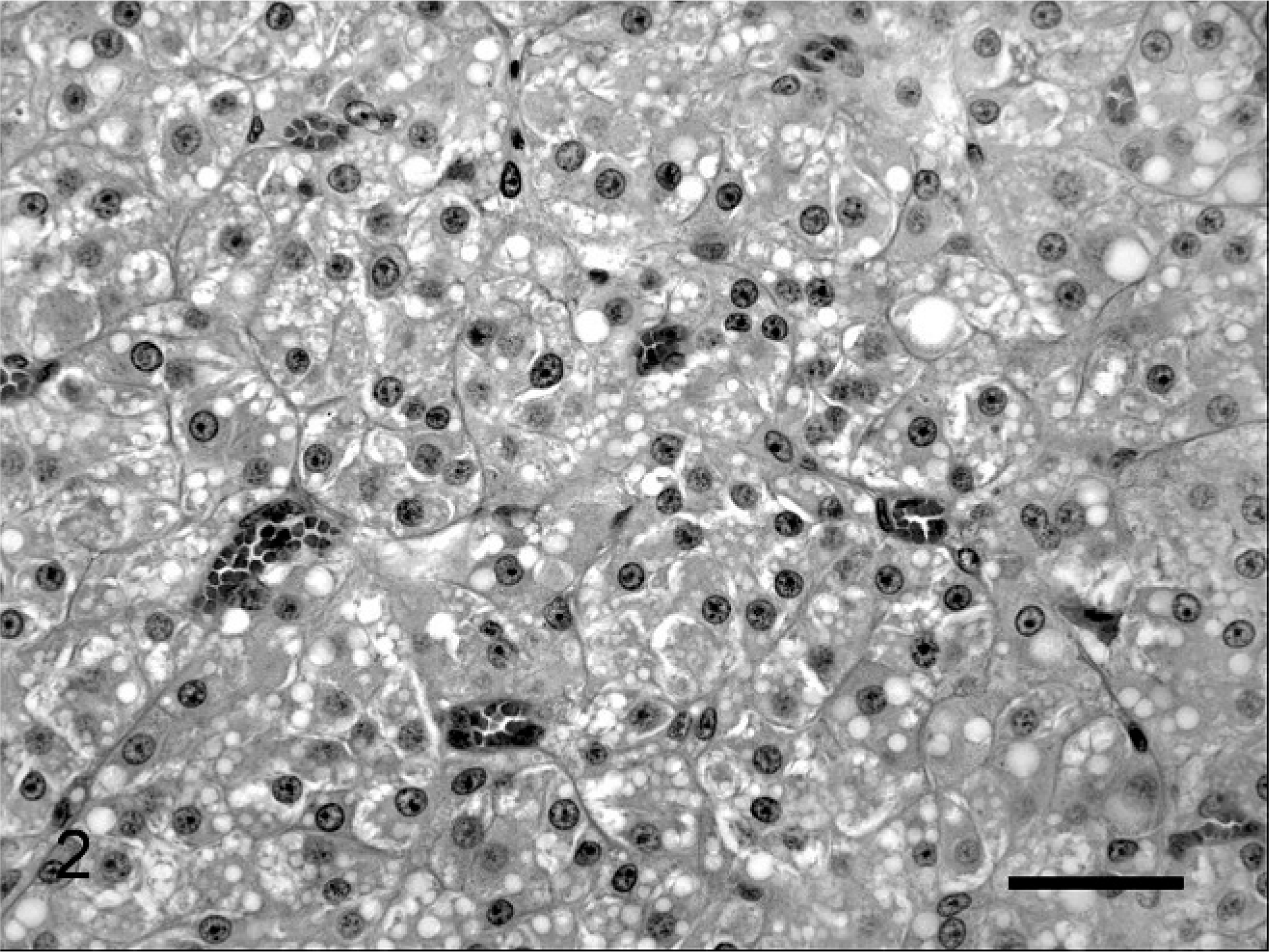
Interstitial cell tumor, testis; cat. Lobules of neoplastic polygonal cells with abundant vacuolated cytoplasm are arranged in packets within a fine stroma. HE. Bar = 50 µm.

Interstitial cell tumor, testis; cat. Pleomorphic neoplastic interstitial cells have intranuclear cytoplasmic invaginations (∗). HE. Bar = 50 µm.
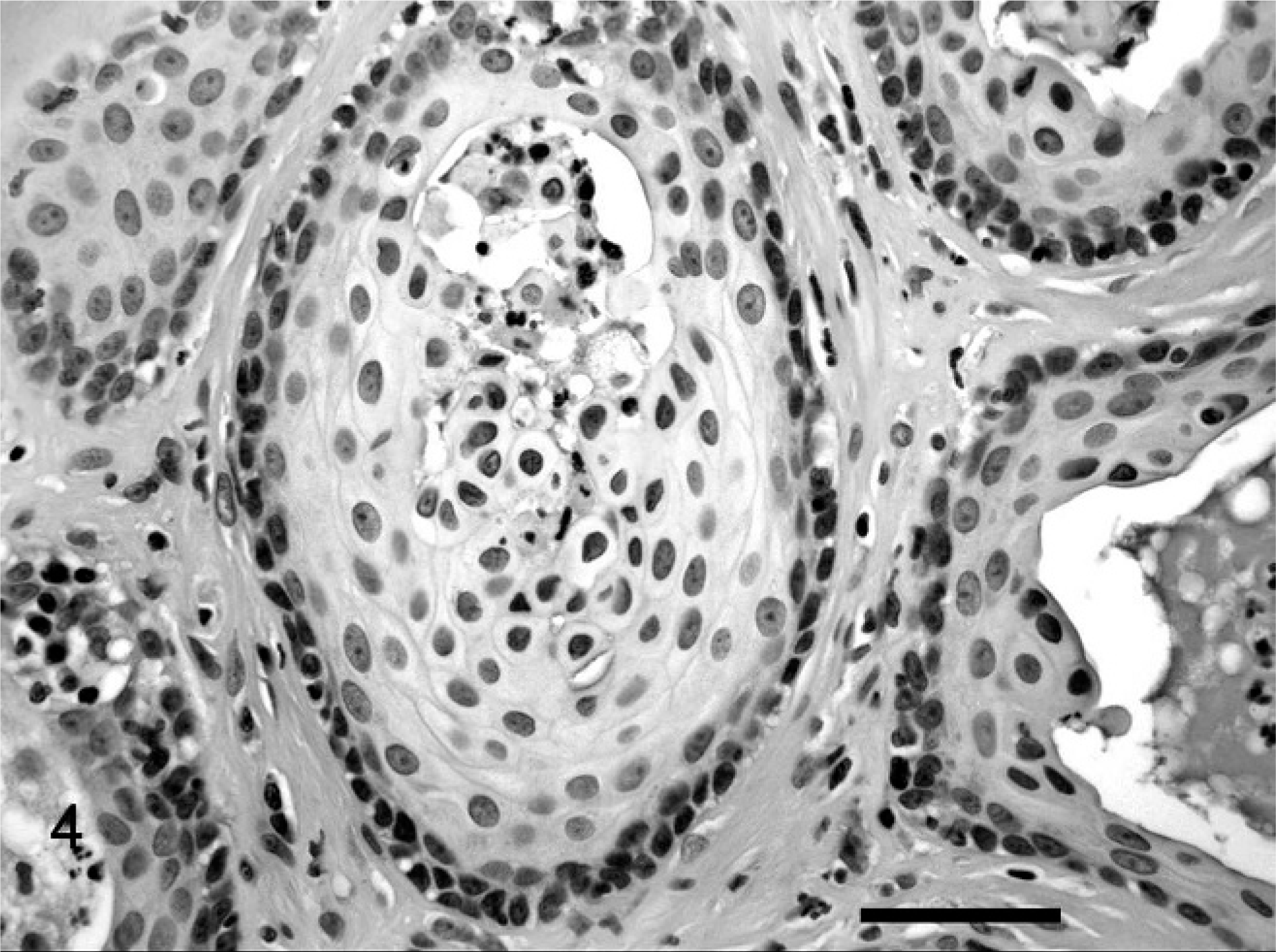
Prostate; cat. Distended acini lined by keratinized stratified squamous epithelium contain neutrophils and sloughed keratinocytes. HE. Bar = 50 µm.
Sections of testis and prostate were obtained from a 3.6-kg adult male cat, with no gross or histologic evidence of urinary-tract or reproductive-tract lesions to serve as controls. Unstained sections of prostate and testis from both cats were rehydrated. Peroxidase blocking with 3% hydrogen peroxide and a serum-free nonspecific protein blocking were applied for 5 minutes each. 17 Commercial antibodies were used to assess immunoreactivity for cyclo-oxygenase 2 (COX2) (rabbit polyclonal, 1 : 1000, Cayman Chemical, Ann Arbor, Michigan, 48108, USA), Melan A (mouse monoclonal, 1 : 50, Dako, Glostrup, DK-2600 Denmark), neuron specific enolase (NSE) (mouse monoclonal, 1 : 450, BioGenex, San Ramon, California, 94583, USA), and S 100 (rabbit polyclonal, 1 : 2000, Dako). Incubation times were 30 minutes for all primary antibodies. Positive staining was highlighted by 3,3′-diaminobenzidine (DAB) substrate precipitation, except for the Melan A, in which permanent red was used. Mixed feline tissue external controls were run, in addition to the feline normal testis and prostate. Negative controls were run by using the same technique with rabbit (COX2, S100) or mouse (Melan A, NSE) serum instead of the primary antibody. For differentiating S100a from S100, unstained paraffin-embedded sections of testis were processed at the immunohistochemistry laboratory of the University of Pennsylvania School of Veterinary Medicine, New Bolton Center, Kennett Square, Pennsylvania, by their standard protocols. For estrogen receptor alpha clones cc4-5 and 1D5 unstained paraffin-embedded sections of prostate were processed at the Veterinary Medical Teaching Hospital Laboratory at the University of California, Davis, California, by their standard protocols. In the testis from the intact male cat, interstitial cells consistently had immunoreactivity for S100 within the cytoplasm. The neoplastic interstitial cells and interstitial cells surrounding atrophic seminiferous tubules had cytoplasmic immunoreactivity for S100. Immunoreactivity for S100a was most pronounced in the pleomorphic neoplastic population. The neoplastic interstitial cells and the Sertoli cells in atrophic seminiferous tubules had less consistent S100a immunoreactivity.
In the testis from the intact male cat, interstitial cells and Sertoli cells had immunoreactivity for NSE in the cytoplasm. The well-differentiated and pleomorphic neoplastic interstitial cells demonstrated cytoplasmic immunoreactivity for NSE. In this section, the few remaining interstitial cells within seminiferous tubules had consistent immunoreactivity for NSE. Peripheral nerves in the sections were immunoreactive for NSE, S100, and S100a, and served as internal controls. In the testis from the intact male cat, interstitial-cell cytoplasm was immunoreactive for Melan A, as were rare granules within Sertoli-cell cytoplasm. In both interstitial-cell tumors, the cytoplasm of the neoplastic cells was immunoreactive for Melan A. The remaining Sertoli cells had occasional cytoplasmic foci with immunoreactivity for Melan A.
In the prostate, cytoplasmic immunoreactivity for estrogen alpha receptor clones 1D5 and cc4-5 was present in basal epithelial cells and in stromal cells. Immunoreactivity for cc4-5 was present in scattered clusters of squamous epithelial cells. Immunoreactivity was present for COX2 in the cytoplasm of stromal cells, basal epithelial cells, and in occasional squamous epithelial cells in the affected prostate but was not present in the prostate from the intact male cat.
The uniform diameter of the urethral lumen and smooth margination of the urethra on the first retrograde cystourethrogram made current urethral calculi or neoplasia unlikely. No prostatomegaly was noted on imaging studies. The enlarged prostate may have been positioned within the pelvic canal, hindering radiographic and ultrasonographic interpretation. In previous reports of feline prostatic disease, obstipation and dyschezia were the main clinical signs. 11, 23 However, the clinical signs in this case were limited to the urinary system.
Prostatic disease is uncommonly reported in the cat. Previous case reports were limited to bacterial prostatitis, 23 periprostatic cysts, 16 adenocarcinoma, 9 and carcinoma. 2, 11 Prostatic squamous metaplasia may be the result of estrogen-induced proliferation of prostatic acinar basal reserve cells, with squamous differentiation rather than androgen-driven differentiation into secretory epithelium. 8 This occurs in dogs with exogenous hyperestrogenism 7, 14 and in dogs with testicular neoplasia but has not been reported in cats. Prostatic squamous metaplasia and keratinized prostatic cysts, with prostatitis, are described in neutered ferrets associated with hyperestrogenism because of hyperplastic or neoplastic adrenocortical lesions. 3
An hCG stimulation test used to assess production of testosterone may be used to test for the presence of retained testes 13 in felids. In intact male cats, serum testosterone concentration should increase after hCG administration (250 µg IM), and post-hCG serum concentration should exceed 3.1 ng/ml. 10 In this case, the concentration of testosterone did not increase after administration of hCG, consistent with a neutered cat status. Dogs with interstitial-cell tumors or Sertoli cell tumors can have reduced concentrations of testosterone secondary to negative feedback of estrogen and inhibin on the gonadotropic hormones (follicular stimulating hormone and luteinizing hormone). 19 Unfortunately, no preoperative serum was available to assay for estrogen, inhibin, and testosterone concentrations at the time the testicular and prostatic changes were discovered.
Previous reports of hormonally functional feline testicular tumors described interstitial-cell tumors with androgenic effects; cats were suspected of having the tumor based on the development of typical male behavior and secondary sexual characteristics at least 5 years after gonadectomy. 15 A cat with both an interstitial-cell tumor and a Sertoli-cell tumor within a retained testis had male behavior and secondary sexual characteristics 7 years after a reported gonadectomy. Only the interstitial-cell tumor was determined to be hormonally productive in that case. 15
Immunohistochemistry has been used to characterize different types of testicular tumors in dogs. Interstitial-cell tumors are NSE-negative in dogs, 6, 18 but our results demonstrated immunoreactivity for NSE in both normal and neoplastic interstitial cells in the cat. Immunoreactivity for S100 in normal testicular tissue varies by species, 1 but, in the cat, immunoreactivity for S100 alpha is described in Sertoli cells and in epithelial cells of the transitional segment of the seminiferous tubules, whereas S100 beta immunoreactivity is in the interstitial cells. 4 In this case, S100 immunoreactivity was strongest in both normal and well-differentiated neoplastic interstitial cells, whereas S100 alpha immunoreactivity was present in interstitial cells and Sertoli cells, and was strongest in the pleomorphic neoplastic population. Steroid-producing (hormonally active) testicular interstitial-cell tumors in the dog 6, 20 and the cat 15 were reported as Melan A positive. A cross reaction to a protein expressed in steroid hormone producing cells is postulated 20 rather than the presence of the melanocytic-differentiating protein. The immunoreactivity was clearly present in both neoplastic populations, yet estrogen-secreting interstitial-cell tumors have not been reported in the cat. However, given the low levels of antemortem testosterone, and the putative estrogenic effects described in the prostate of this cat, we assumed that the neoplastic interstitial cells were not producing testosterone.
The presence of estrogen receptor alpha in prostatic squamous metaplasia was described in the mouse. 21 In addition prostatic epithelial and stromal expression of COX2 under estrogen-induced epithelial differentiation was demonstrated in neutered and intact dogs. 5 Although, in the case reported here, the expression is not strong, the presence of estrogen receptor alpha immunoreactivity in stromal and basal epithelial cells with occasional expression by differentiated squamous epithelial cells was consistent with the described interaction of estrogen on both stromal and epithelial cells when inducing squamous metaplasia. The COX2 expression was in similar populations and had no particular association with inflammation, again consistent with estrogenic stimulation of the receptors.
Unfortunately, it was not possible to pursue hormone assays, beyond the hCG stimulation test, on serum from this case. Nonetheless, the histologic and immunohistochemical results are consistent with the diagnosis of interstitial-cell neoplasia in the retained testis of a cat with estrogenic effects. This is the first report of prostatic squamous metaplasia with secondary prostatitis induced by interstitia-cell neoplasia in a cat.
Footnotes
Acknowledgements
We thank Dr. M. D. McCracken, Dr. L. E. Craig, and Dr. K. Newkirk in the Department of Patholobiology, and Dr. K. Tobias in the Department of Small Animal Clinical Science (surgery) for their involvement in this case; the UTCVM Diagnostic Imaging Department for its assistance in performance and interpretation of radiographic and ultrasonographic studies; and the UTCVM Immunohistochemistry Laboratory for its contribution to this case.
