Abstract
Renal interstitial cell tumors are benign tumors of renomedullary origin; however, malignant features have not been reported in dogs, to our knowledge. A 17-y-old spayed female Maltese dog was presented to a local animal hospital with a mass in the right abdomen. Clinicopathologic findings prior to surgery revealed renal insufficiency and anemia. Imaging revealed that the right kidney was enlarged by an amorphous mass with opaque areas, indicative of mineralization. Upon histologic examination, the mass was comprised of malignant mesenchymal cells that produced mucinous matrix. The tumor cells were positive for vimentin and COX-2, but negative for pancytokeratin; the matrix stained positively with alcian blue. Therefore, the mass was diagnosed as a renal interstitial cell tumor, with malignant features. COX-2 may be useful in the diagnosis of canine renal interstitial cell tumors, similar to its diagnostic role in humans.
In dogs, primary renal tumors are relatively uncommon, comprising 0.3–1.5% of all reported canine tumors.1,2,11 Nearly 70% of canine renal tumors are derived from epithelial cells, 25% are derived from mesenchymal cells, and 5% are nephroblastomas. 10 Notably, renal interstitial cell tumors have been reported only rarely. 10
Renal interstitial cell tumors are believed to arise from renal interstitial cells that contain prostaglandin and arachidonic acid. 10 Renal interstitial cells exhibit distinctive ultrastructural features such as lipid-rich cytoplasmic vesicles; this neutral antihypertensive lipid can lower arterial blood pressure.5,10 Most renomedullary interstitial cells express high levels of cyclooxygenase-2 (COX-2), which is the inducible isoform of prostaglandin-endoperoxide synthase 2.5,13 In humans, renal interstitial cell tumors are known to be COX-2 positive. 10
Moreover, in humans, these mesenchymal tumors are known as renomedullary interstitial cell tumors, which are benign neoplasms diagnosed only rarely prior to death; they were previously described as medullary fibromas or hamartomas.12,13 The tumors are commonly identified at autopsy, in up to 50% of autopsy cases; most are asymptomatic and < 5 mm diameter, hence the antemortem diagnostic rate is low. 12
In previously reported cases of canine renal interstitial cell tumors, only benign features were observed as in humans; moreover, there were no data regarding clinicopathologic or imaging findings (e.g., radiography, ultrasonography, or computed tomography). 4 We describe herein a renal interstitial cell tumor with malignant features in a dog.
A 17-y-old spayed female Maltese dog weighing 3.9 kg was presented because of anorexia and an enlarged right side of her abdomen. Blood was collected for a complete blood count and chemistry panel. Creatinine and urea concentrations were increased as were the activities of amylase and lipase; moderate anemia and leukocytosis were present (Table 1).
Clinicopathologic findings prior to surgery in a dog with a renal interstitial cell tumor.
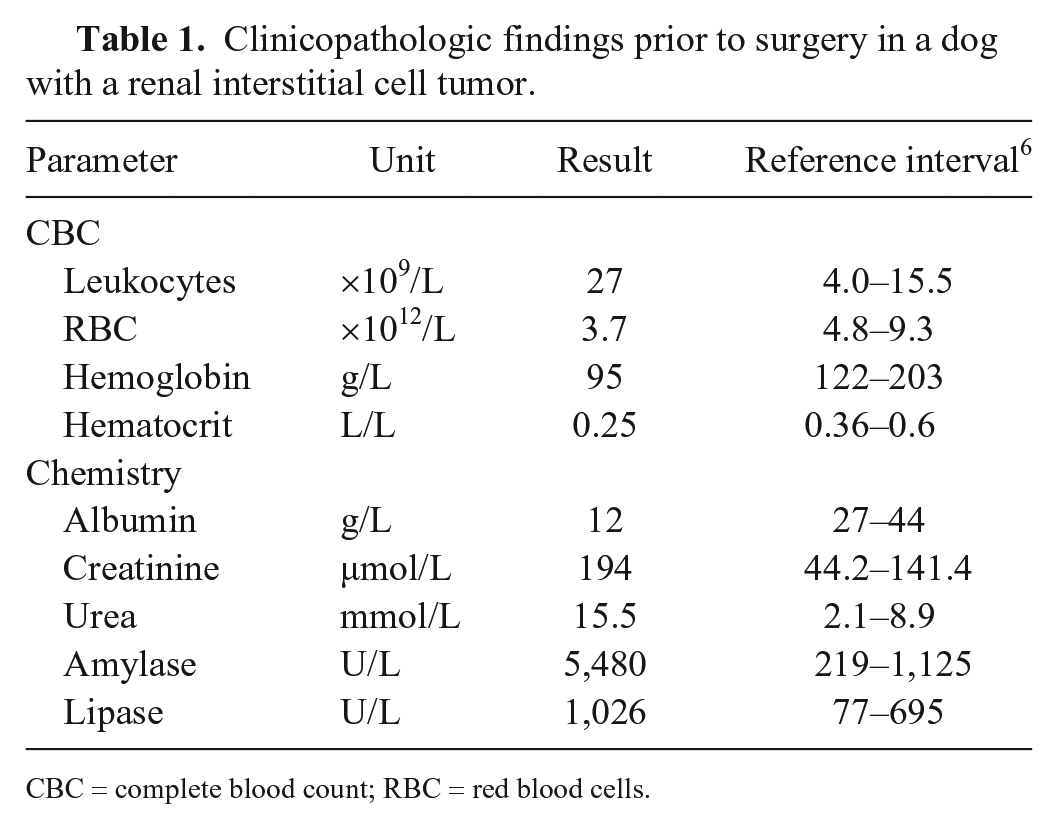
CBC = complete blood count; RBC = red blood cells.
Radiographic findings included an ovoid mass with soft tissue opacity in the right side of the abdominal cavity, as well as deviation of the pylorus and intestines in the opposite direction. A large amorphous structure with mineralization opacity was identified in the right cranio–mid-abdominal cavity (2.5 cm3) and was located in the mass. The right kidney was enlarged on ultrasonographic examination, and normal architecture was lost. A heterogeneously hyperechoic lesion was identified in the right renal mass, caudal to the anechoic lesion. Computed tomography revealed an irregularly enlarged caudal pole of the right kidney (71 × 81 × 54 mm; Fig. 1). A 2.5-cm3 stellate-shaped area of mineralization was identified in the right renal pelvis and diverticulum; the mass displaced other abdominal organs to the left. No other abnormalities or metastases of the tumor were found in other organs by imaging. The mass was removed surgically under general anesthesia. The excised mass was fixed in 10% neutral-buffered formalin, and was sent to the Department of Veterinary Pathology, College of Veterinary Medicine, Konkuk University, Republic of Korea. The dog died one week after surgery, and an autopsy was not performed at the referral hospital.
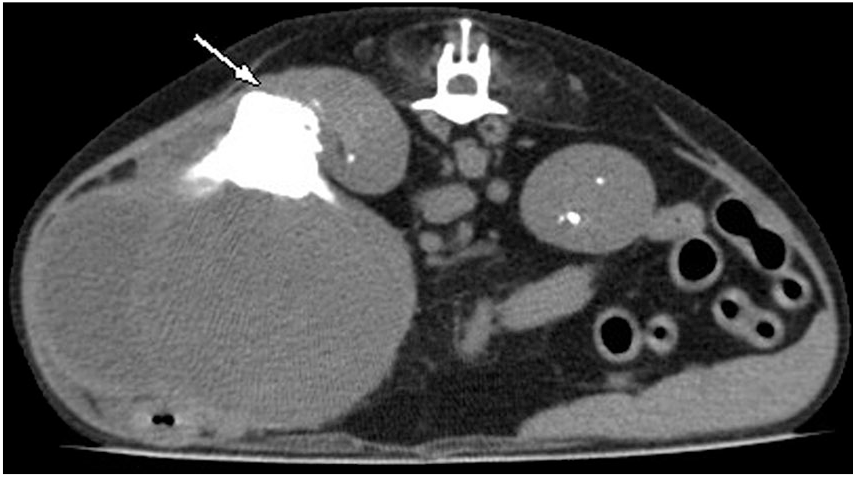
Computed tomography of renal interstitial cell tumor in a dog. The preoperative image showed an irregularly enlarged mass. The cranial region of the right kidney maintained a normal shape, but its position was changed in the cranial direction by the mass. The mineralized region (arrow) was found between normal kidney and the mass.
On gross examination, the right kidney contained one irregular, large, red-to-ivory mass (70 × 78 × 55 mm) on the elongated caudal pole; the cranial pole was of normal shape (Fig. 2). The center of the tumor was mineralized extensively, as noted in radiographic analysis and computed tomography; thus, the tumor was sectioned after demineralization. Cross-section of the cranial pole of the right kidney revealed tissue similar to that of normal kidney macroscopically.
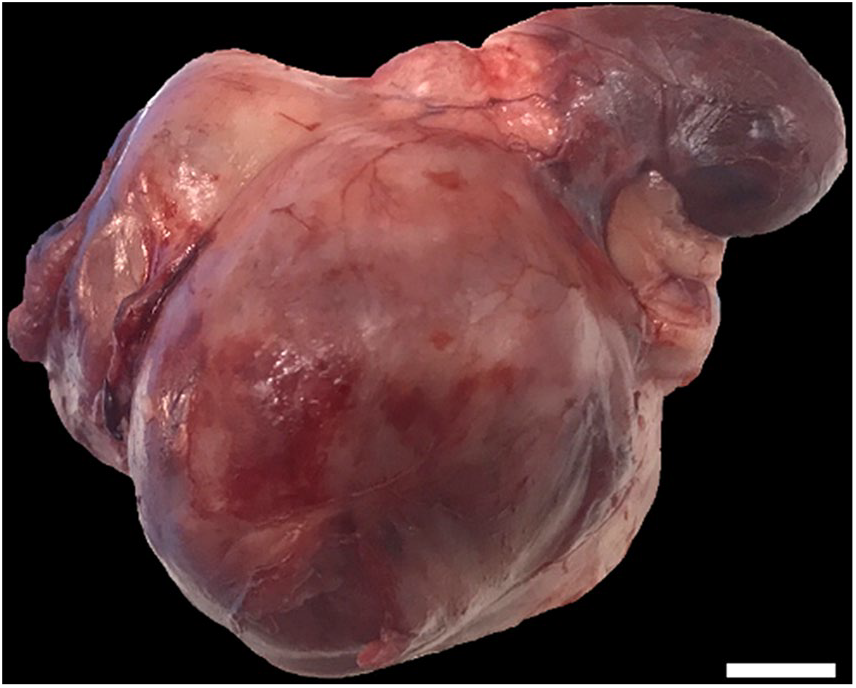
Renal interstitial cell tumor in a dog. The mass replaced ~30% of the right kidney; recognizable kidney tissue remained. Bar = 1 cm.
The mass was processed routinely, and sections were stained with hematoxylin and eosin for histologic examination. Microscopically, there was multifocal necrosis within the mass. The right adrenal gland was not clearly identified on the image and gross findings, but was confirmed near the mass on histologic examination. The tumor was nonencapsulated and comprised interlacing bundles of uniform spindle cells with basophilic nuclei and slight-to-moderate amounts of eosinophilic cytoplasm. The neoplastic cells invaded perinephric fat (Supplementary Fig. 1) and connective tissue adjacent to the adrenal gland (Supplementary Fig. 2). The neoplastic cells had intracytoplasmic vacuoles (Fig. 3) and mild-to-moderate pleomorphic nuclei with single-to-multiple prominent nucleoli; moreover, the mitotic count was high (53 mitoses in 2.37 mm2; Fig. 4). One bizarre mitosis per 2.37 mm2 was observed. The mineralized portion found between normal kidney and tumor was identified as extensive osseous metaplasia (Supplementary Fig. 3). Dilated tubules were observed in the cranial pole of the kidney, as well as mild lymphoplasmacytic interstitial nephritis (Supplementary Fig. 4).
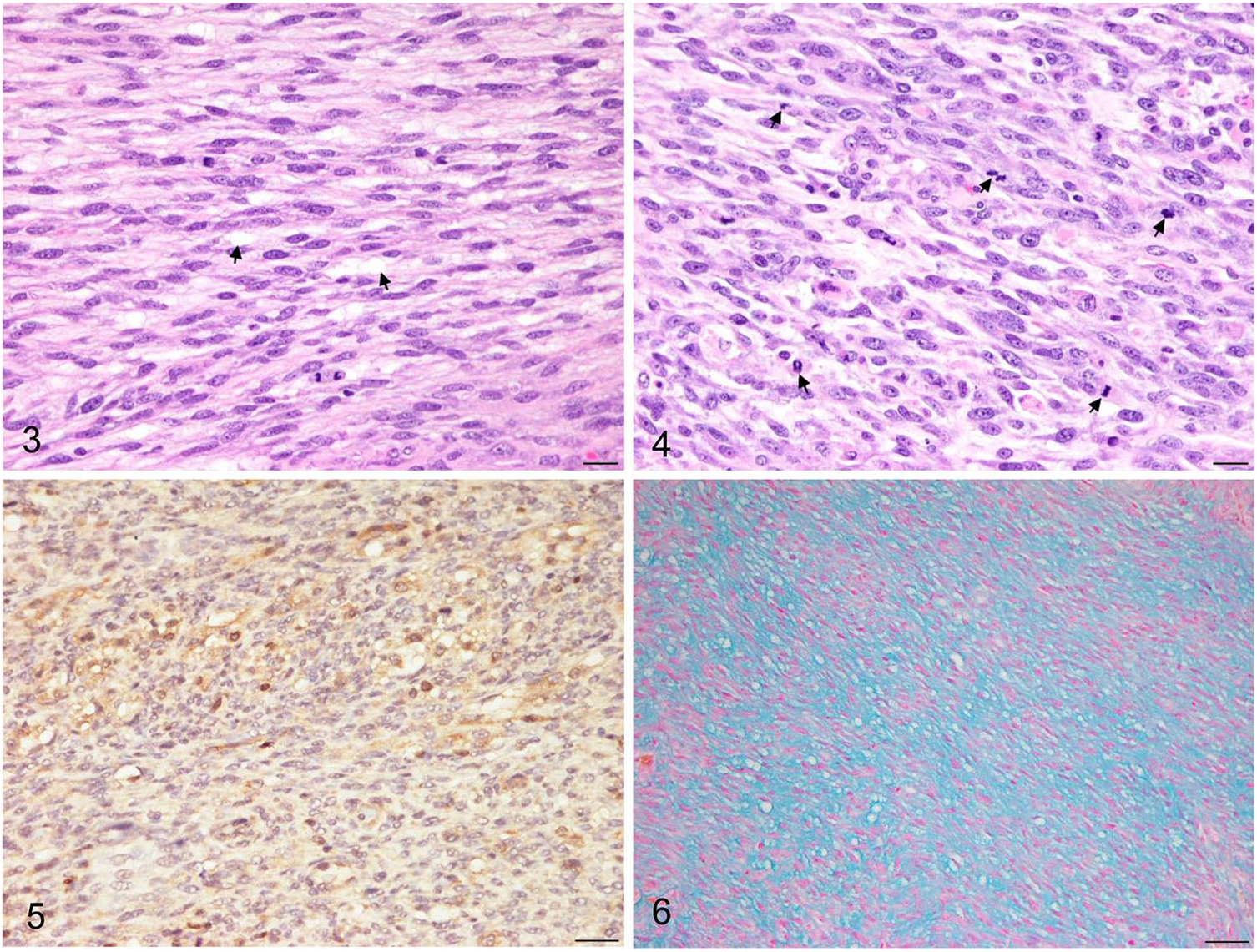
Histologic features of renal interstitial cell tumor in a dog. Some neoplastic cells contain intracytoplasmic vacuoles (arrows). H&E. Bar = 20 µm.
An autopsy was not performed on the dog, but there was no suspicion of metastasis, such as alterations in size in the lung or lymph nodes, on computed tomography. Therefore, a differential diagnosis was made based on the primary tumor in the kidney. Immunohistochemical staining using primary antibodies against vimentin (M0725; Dako, Glostrup, Denmark) and cytokeratin AE1/AE3 (M3515; Dako) was performed to rule out epithelium-derived tumors. 11 In addition, antibodies against CD31 (M0823; Dako) were used to rule out primary tumors of renal vascular origin. To distinguish between renal fibrous tissue–derived tumors and renal interstitial cell tumors, immunohistochemical staining for COX-2 (PG46; Oxford Biomedical Research, Rochester Hills, MI), alcian blue staining, and oil red O staining were performed. Deparaffinized slides were rehydrated in phosphate-buffered saline (PBS) and incubated in 3% hydrogen peroxide for 20 min. Heat-induced antigen retrieval using a microwave oven was performed in Tris–EDTA (pH 9.0) for detection of vimentin and CD31, and in citric acid (pH 6.0) for detection of cytokeratin AE1/AE3 and COX–2. Blocking was performed with 5% normal goat serum for 30 min at room temperature, and the slides were incubated with each of the primary antibodies as follows: 2 h for the antibody against vimentin, and 3 h for the antibodies against cytokeratin AE1/AE3, COX-2, and CD31; all incubations were performed at room temperature. The slides were washed 3 times in PBS and incubated with secondary antibody (K5007; Dako) for 40 min. All slides were counterstained with Gill hematoxylin. Positive control staining was performed using tissues from other dogs: canine normal skin tissue was used for cytokeratin AE1/AE3, vimentin, and CD31; canine colorectal cancer was used for COX–2. Isotype-matched immunoglobulins were used as negative controls.
The tumor cells were positive for vimentin and negative for cytokeratin AE1/AE3; thus, the tumor was confirmed to be of mesenchymal origin. Mesenchymal tumors originating from the kidney are derived from vascular tissue or fibrous tissue. 9 Vascular endothelial-derived tumors (e.g., solid hemangiosarcoma) were ruled out because of negative CD31 expression. The cytoplasm of several neoplastic cells was positive for COX-2 (Fig. 5), and the myxoid matrix was positive with alcian blue stain (Fig. 6). Oil red O staining showed a positive reaction in the cytoplasmic vacuoles of some cells. COX-2–positive reaction and cytoplasmic vacuoles are known to be present in the cytoplasm in human renomedullary interstitial cell tumors, 5 and alcian blue stain is regarded as a negative sign for fibroma 10 ; the mass was therefore diagnosed as a renal interstitial cell tumor.
The clinicopathologic findings (i.e., increased creatinine, urea, amylase, and lipase) indicated that the tumor might have caused renal insufficiency and anemia. Mild lymphoplasmacytic interstitial nephritis and tubule dilation were observed in the portions that appeared to be normal kidney on imaging and gross examination. Renal tubule dilation can arise from several conditions (nephropathy, obstruction, etc.). 8 In our case, the kidney tumor was large, and mineralization was present in the right renal pelvis and diverticulum, perhaps partially blocking the tubule pathway.
Enlargement of the caudal pole of the right kidney with extensive osseous metaplasia, approximately one-third the size of the kidney, was confirmed on radiographic, computed tomographic, and histologic examinations. This differed from the findings of a prior report, in which only small mineralized foci were observed. 4 In previous reports, 4 mineralized foci were observed only on radiographic analysis; these gradually increased in size and may have developed osseous metaplasia, as in our case.
Regarding histopathology features, renal interstitial cell tumors have been reported to consist of interstitial cells with lipid-rich cytoplasmic vesicles, high prostaglandin content, and positive extracellular alcian blue staining plus a positive lipid reaction in cytoplasmic droplets. 10 Renal interstitial cell tumors express cytoplasmic COX-2, whereas fibrosarcomas do not.5,7 Importantly, renal interstitial tumors occur in the cortex and medulla of older dogs (10–18 y old); the tumors are often bilateral and multiple. 4 In our case, although no mass was found in the opposite kidney by imaging, various features including imaging findings (location in kidney without metastatic lesions), age (17 y), and microscopic features (i.e., alcian blue–positive matrix, vimentin- and COX-2–positive reactions in the cytoplasm) met the criteria for a diagnosis of renal interstitial cell tumor.
In histologic examination, the tumor showed variable degrees of cell proliferation. There were regions with relatively few mitotic figures (e.g., 38 mitoses/2.37 mm2, minimal nuclear pleomorphism, and few nucleoli). Other regions had numerous mitotic figures (80 mitoses/2.37 mm2) and increased nuclear pleomorphism with many nucleoli per nucleus. In regions with low mitotic activity, relatively high numbers of cytoplasmic vacuoles were observed, whereas there were fewer cytoplasmic vacuoles in regions with high mitotic activity. We concluded that this aspect was consistent with the reduced degree of differentiation in the presence of increased anaplasia. 3
Although metastases were not found by computed tomography, an autopsy was not performed, and there may have been undetected metastases. The tumor showed malignant features, such as invasiveness, pleomorphic nuclei with multiple prominent nucleoli, marked mitotic activity, and bizarre mitoses. These histopathologic malignant features differ from those of the typical benign renomedullary interstitial cell tumors found in humans and dogs.4,10,12,13 In addition, no mitoses were observed in previous reports of canine interstitial cell tumors. Our case demonstrates that a renal interstitial cell tumor in a dog can be aggressive and can have malignant histologic features, and that COX-2 can be useful in the diagnosis of this tumor.
Supplemental Material
Supplementary_material – Supplemental material for Renal interstitial cell tumor in a dog: clinicopathologic, imaging, and histologic features
Supplemental material, Supplementary_material for Renal interstitial cell tumor in a dog: clinicopathologic, imaging, and histologic features by Seung-Hee Cho, Byung-Joon Seung, Soo-Hyeon Kim, Ha-Young Lim, Gyu-Seok Lee, Mi-Suk Chae and Jung-Hyang Sur in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF), funded by the Korean government (MSIT; grant 2016M3A9B6903437).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
