Abstract
Proliferative gill disease (PGD), caused by the myxozoan Henneguya ictaluri, has been the most notorious parasitic gill disease in the US catfish aquaculture industry. In 2019, an unusual gill disease caused by massive burdens of another myxozoan, Henneguya exilis, was described in channel (Ictalurus punctatus) × blue (Ictalurus furcatus) hybrid catfish. Targeted metagenomic sequencing and in situ hybridization (ISH) were used to differentiate these conditions by comparing myxozoan communities involved in lesion development and disease pathogenesis between massive H. exilis infections and PGD cases. Thirty ethanol-fixed gill holobranchs from 7 cases of massive H. exilis infection in hybrid catfish were subjected to targeted amplicon sequencing of the 18S rRNA gene and compared to a targeted metagenomic data set previously generated from clinical PGD case submissions. Furthermore, serial sections of 14 formalin-fixed gill holobranchs (2 per case) were analyzed by RNAscope duplex chromogenic ISH assays targeting 8 different myxozoan species. Targeted metagenomic and ISH data were concordant, indicating myxozoan community compositions significantly differ between PGD and massive branchial henneguyosis. Although PGD cases often consist of mixed species infections, massive branchial henneguyosis consisted of nearly pure H. exilis infections. Still, H. ictaluri was identified by ISH in association with infrequent PGD lesions, suggesting coinfections occur, and some cases of massive branchial henneguyosis may contain concurrent PGD lesions contributing to morbidity. These findings establish a case definition for a putative emerging, myxozoan-induced gill disease of farm-raised catfish with a proposed condition name of massive branchial henneguyosis of catfish (MBHC).
Proliferative gill disease (PGD), caused by the myxozoan Henneguya ictaluri, has significant, negative impacts on commercial catfish production, the largest sector of food fish aquaculture in the United States. The life cycle of H. ictaluri involves the development and release of the myxospore stage in the gills of channel catfish, Ictalurus punctatus, and the actinospore stage in the ubiquitous benthic oligochaete Dero digitata.2,17,18 Continuous exposure of catfish to the actinospore stage of H. ictaluri results in clinical PGD characterized by severe respiratory distress, high fish mortality, and substantial economic losses. 27 Pathognomonic lesions of PGD include swollen, red and white mottled, frequently truncated, gill filaments grossly and epithelial hyperplasia, lamellar fusion, intense inflammatory cell infiltrates associated with cartilage lysis and chondrocyte proliferation, and presporogonic stages of H. ictaluri microscopically.22,27 Although several other Henneguya spp. infect the gills of catfish, most do not cause overt disease. However, their presence in gill tissues during PGD outbreaks may induce further pathologic changes, including additional PGD-like lesions, and potentially exacerbate overall disease severity.22,23
In contrast to channel catfish, channel catfish × blue catfish (Ictalurus furcatus) hybrids demonstrate decreased transmission rates and arrested development of H. ictaluri.6,19,22 Although the specific mechanisms responsible for this differential host susceptibility to the parasite are unknown, the abundance of H. ictaluri is suppressed in hybrid catfish gill tissues and hybrid monoculture systems.6,8,21 -23 In addition, monoculture of hybrid catfish results in overall decreased myxozoan diversity and, potentially, enhanced selection of H. exilis within the pond environment. 21
H. exilis was first morphologically described in channel catfish by Kudo in 1929 followed by its molecular characterization 70 years later.13,14 H. exilis shares the same indirect life cycle as H. ictaluri involving the definitive oligochaete host, D. digitata, and intermediate fish host, the channel catfish. 14 Massive H. exilis infections have been associated with histopathologic changes during disease outbreaks in channel catfish and were historically considered to be one of three different forms of PGD prior to molecular characterization of catfish myxozoans and identification of H. ictaluri as the causative agent for PGD. 4 More recently, gross and microscopic lesions associated with outbreaks of respiratory distress and massive interlamellar burdens of Henneguya spp. in channel catfish × blue catfish hybrids were exclusively H. exilis, which was confirmed by laser capture microdissection and molecular sequencing. 20 The clinical and pathologic features associated with these outbreaks differed substantially from those of PGD.20,27
The objective of this study was to investigate whether massive H. exilis infections in the gills of catfish represented either another form of PGD or a distinct respiratory condition caused by H. exilis in ictalurid catfish. Targeted metagenomic sequencing and in situ hybridization (ISH) techniques were employed to compare myxozoan community compositions between cases of PGD and massive H. exilis infections and myxozoan distribution and contribution to lesion formation within infected gills.
Materials and Methods
Fish Collection and Histopathology
In the spring of 2018 and 2019, 7 catfish case submissions to the Mississippi State University Aquatic Research and Diagnostic Laboratory in Stoneville, MS, were diagnosed with massive H. exilis infection in accordance with previously reported gross and wet mount lesions. 20 During necropsies, right holobranchs were fixed in 10% neutral-buffered formalin for histological assessment and ISH, whereas left holobranchs were fixed in 70% ethanol for laser capture microdissection and targeted metagenomic analysis. 20 Thirty gill samples from 7 cases (2018, n = 2; 2019, n = 5) were included in the targeted metagenomic analysis. The fish were confirmed as hybrid catfish by duplex-polymerase chain reaction (PCR) using previously established protocols. 25 Formalin-fixed tissues were processed by standard methods, embedded in paraffin, sectioned at 4 µm onto charged slides, and either left unstained for ISH or stained with hematoxylin and eosin for light microscopic examination.
Targeted Metagenomic Sequencing and Analysis
Targeted metagenomic sequencing and analysis investigating myxozoan communities were performed as described previously. 23 Extracted DNA samples from ethanol-fixed gills were submitted to the Georgia Genomics and Bioinformatics Core, University of Georgia, Athens, GA, USA for library preparation and next generation sequencing (Illumina MiSeq, San Diego, California), generating paired, 300 bp reads. General myxozoan primers were used to amplify an approximately 432 bp segment of the myxozoan 18S rRNA gene containing a diagnostic variable region used for species identification of myxobolids.7,12 Primer sequences were as follows: H9, forward primer, 5’-TTACCTGGTCCGGACATCAA-3’ 9 and 1862R, reverse primer, 5’-ATTGTAGCGCGCGTGCAG-3’. 23 Paired end reads were merged, trimmed, and filtered by quality (maximum expected error rate of 1.0) and sequence length (370-490 bp) using USEARCH v10.0.240. 5 Unique reads were counted and binned into operational taxonomic units (OTUs), which were identified through a BLASTn search of the National Center for Biotechnology Information (NCBI) nonredundant nucleotide database. OTU counts were converted to percentages to assess relative abundances of myxozoan taxa. Using targeted metagenomic data derived from 2018 and 2019 channel and hybrid catfish PGD cases, myxozoan community composition was compared between PGD and massive H. exilis cases using permutational multivariate analysis of variance (PERMANOVA) analysis of dissimilarity (ADONIS) with the vegan package.16,23 Nonmetric multidimensional scaling plots were generated with the vegan package to visualize community differences in beta diversity.
In Situ Hybridization
Two blocks containing formalin-fixed gill tissue from each case were arbitrarily chosen. Each block was serially sectioned 4 times at a thickness of 4 µm and placed onto charged slides to create 4, serial unstained slides. The ISH was performed using previously developed probes and protocols. 22 Henneguya species–specific probes for 5 known and 3 suspect taxa were allocated into duplex assays as follows: (1) H. ictaluri and H. exilis, (2) Henneguya mississippiensis and Raabeia-type TGR-2014, (3) Henneguya postexilis and Myxozoan 12, (4) Myxozoan 7 and Myxozoan 11 (Advanced Cell Diagnostics Inc., Hayward, California). 22 The probes were diluted by half using equal parts of stock probe solution and RNAscope probe diluent (Catalog No. 300041, Advanced Cell Diagnostics Inc.). The assays were performed using the RNAscope duplex chromogen kit (RNAscope 2.5 HD Duplex Detection Kit User Manual, Part 2, Catalog No. 322500-USM, Advanced Cell Diagnostics Inc.) according to the manufacturer’s instructions with a 25-minute target retrieval step. Slides were counter-stained with hematoxylin and Tris-buffered saline then manually coverslipped. Duplex assay positive control slides included channel catfish gill tissue experimentally infected with H. ictaluri and hybrid catfish with H. exilis infection previously confirmed morphologically by myxospore morphometrics and molecularly by laser capture microdissection.20,23 Channel catfish adipose fins infected with Henneguya adiposa served as negative controls.
The data analyzed in this study are available as Supplemental Materials (Supplemental Table S1).
Results
Targeted Metagenomic Analysis
The sequencing run generated 1 931 376 paired reads with 1 483 440 reads remaining after merging and filtering for quality and sequence length (76.8%). Nearly all merged, filtered reads were related to myxozoan taxa (1 483 281 reads, 99.9%). A total of 11 myxozoan OTUs were identified in the samples, with all OTUs previously detected in PGD cases (Supplemental Table S1). 23 Myxozoan communities in massive H. exilis infections were dominated by H. exilis, with an average relative abundance of approximately 98.5% per gill sample (Fig. 1). In contrast, H. ictaluri was present in these samples at an average relative abundance of 1.3%, whereas all other taxa averaged <1% relative abundance (Fig. 1). When compared to PGD data from the same years, there was no overlap in myxozoan community composition between the 2 disease manifestations (P < .001, Fig. 2a, b), 23 indicating the massive H. exilis infections represent a distinct, myxozoan-induced gill disease in catfish separate from PGD.
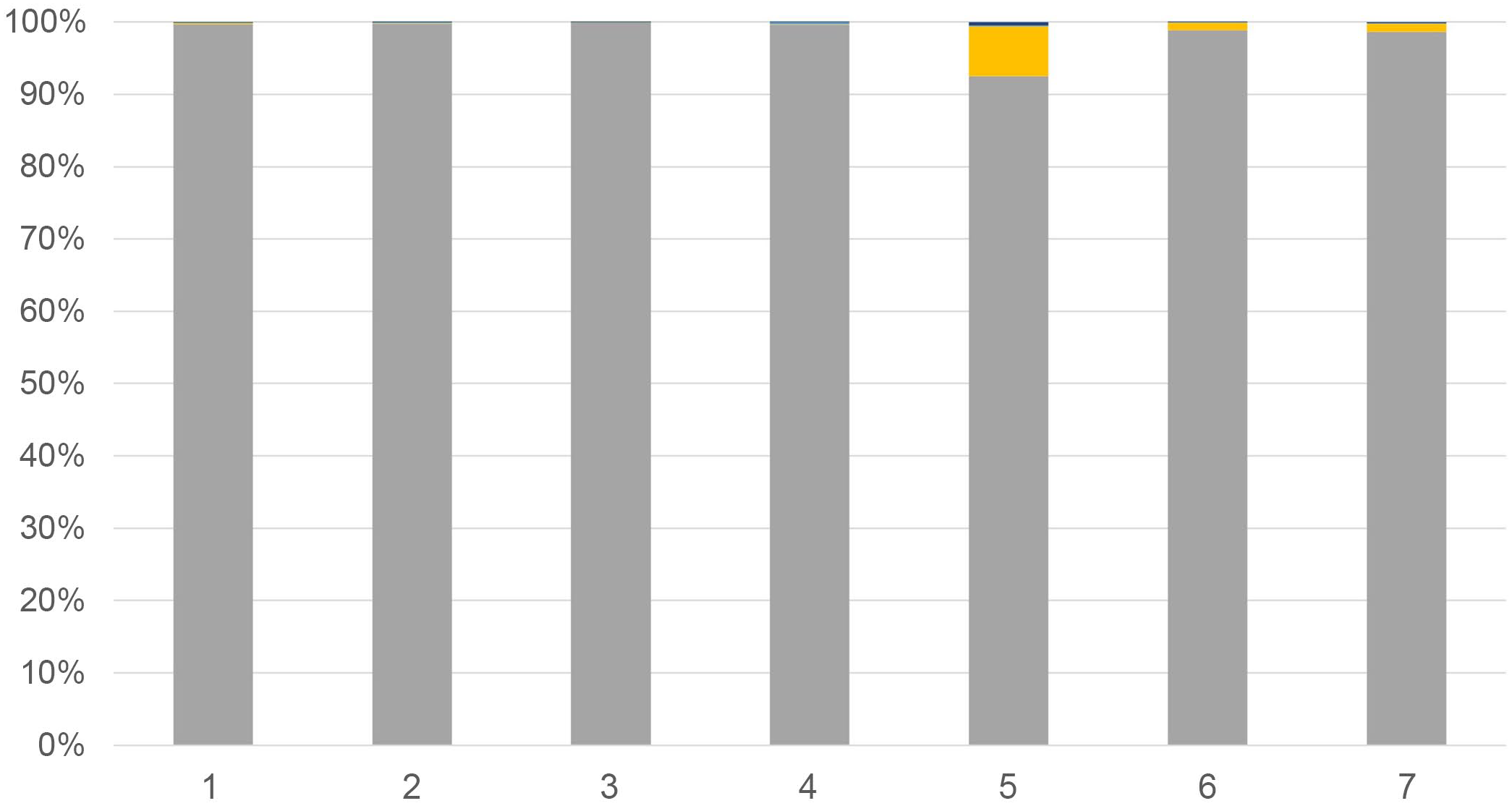
Relative abundances of myxozoan taxa in cases of massive branchial henneguyosis of catfish. The y-axis represents myxozoan relative abundance as stacked percentages whereas the x-axis lists case numbers. Each case represents a nearly pure infection of Henneguya exilis with few other myxozoans detected. Taxa are color-coded as follows: dark blue, all unclassified taxa; green, Raabeia-type; light blue, Henneguya mississippiensis; blue, Henneguya adiposa; gray, Henneguya exilis; yellow, Henneguya ictaluri. With the exception of H. exilis, and occasionally H. ictaluri, these myxozoans made up such a low relative abundance of the myxozoans identified that they are not visible in the graphs but were rarely detected.
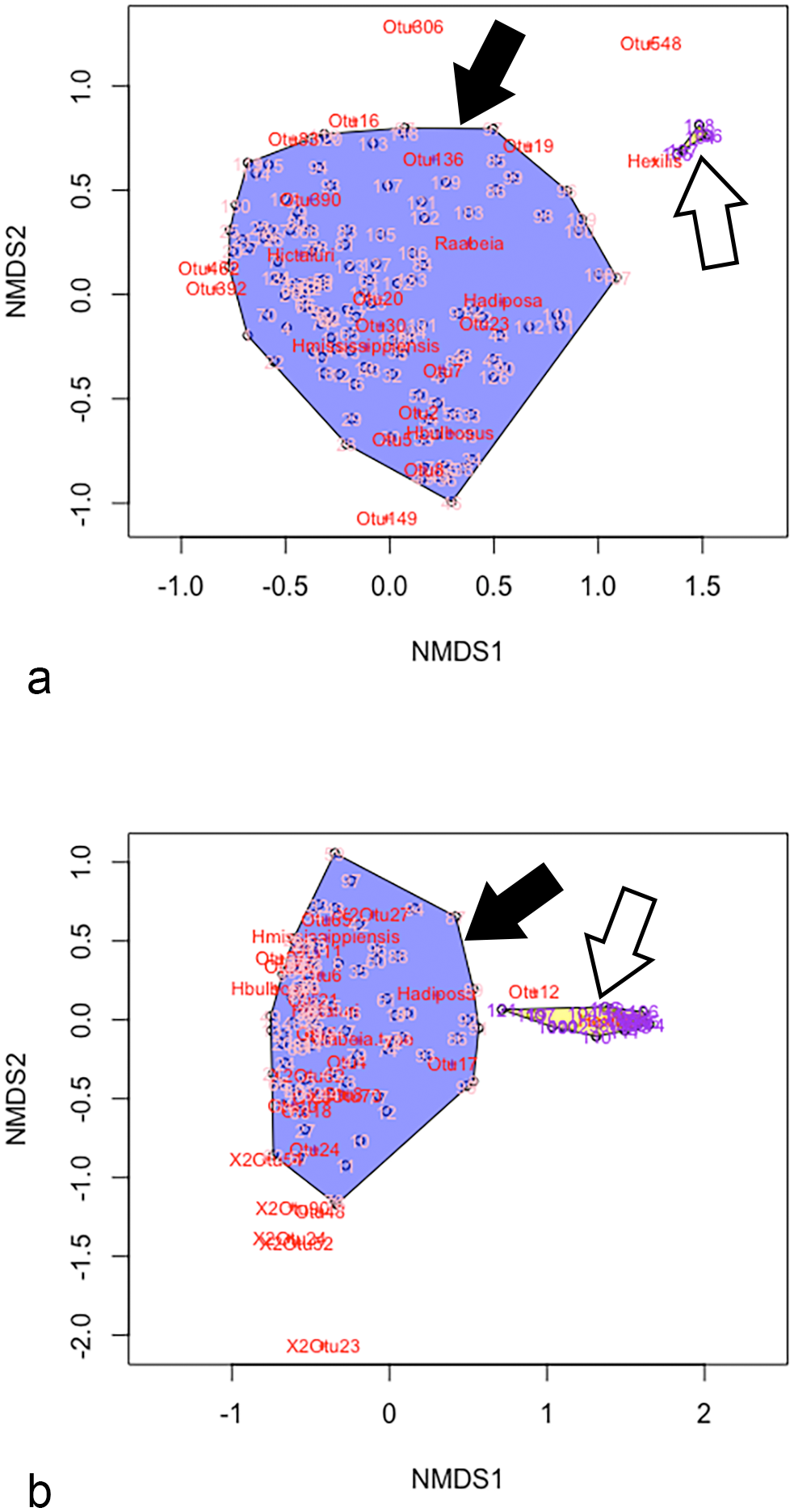
Nonmetric multidimensional scaling (NMDS) plots comparing myxozoan community composition and beta diversity between proliferative gill disease (PGD) cases (solid arrows) and cases of massive branchial henneguyosis of catfish (MBHC) infections (open arrows) in (a) 2018 and (b) 2019. Pink numbers within blue regions represent PGD communities, whereas purple numbers in yellow regions represent MBHC communities. Note there is no overlap in myxozoan community composition between the 2 diseases with PGD gills demonstrating considerable variability in community structure, whereas MBHC cases cluster in tight association with Henneguya exilis. NMDS stress values: 0.1452, 2018; 0.1299, 2019. OTU, operational taxonomic unit.
Histopathology and In Situ Hybridization
Pathology associated with cases from this work has been previously described. 20 Briefly, interlamellar spaces of segments or whole filaments were occluded by 1 or more myxozoan plasmodia embedded within the lamellar epithelium containing myxospores consistent with Henneguya sp. (Fig. 3a, c). Occasionally, these plasmodia ruptured internally, releasing myxospores into the filament stroma and inciting granulomatous inflammation (Fig. 3c). Photomicrographs of PGD are provided with Supplemental Figure S1 for comparison.
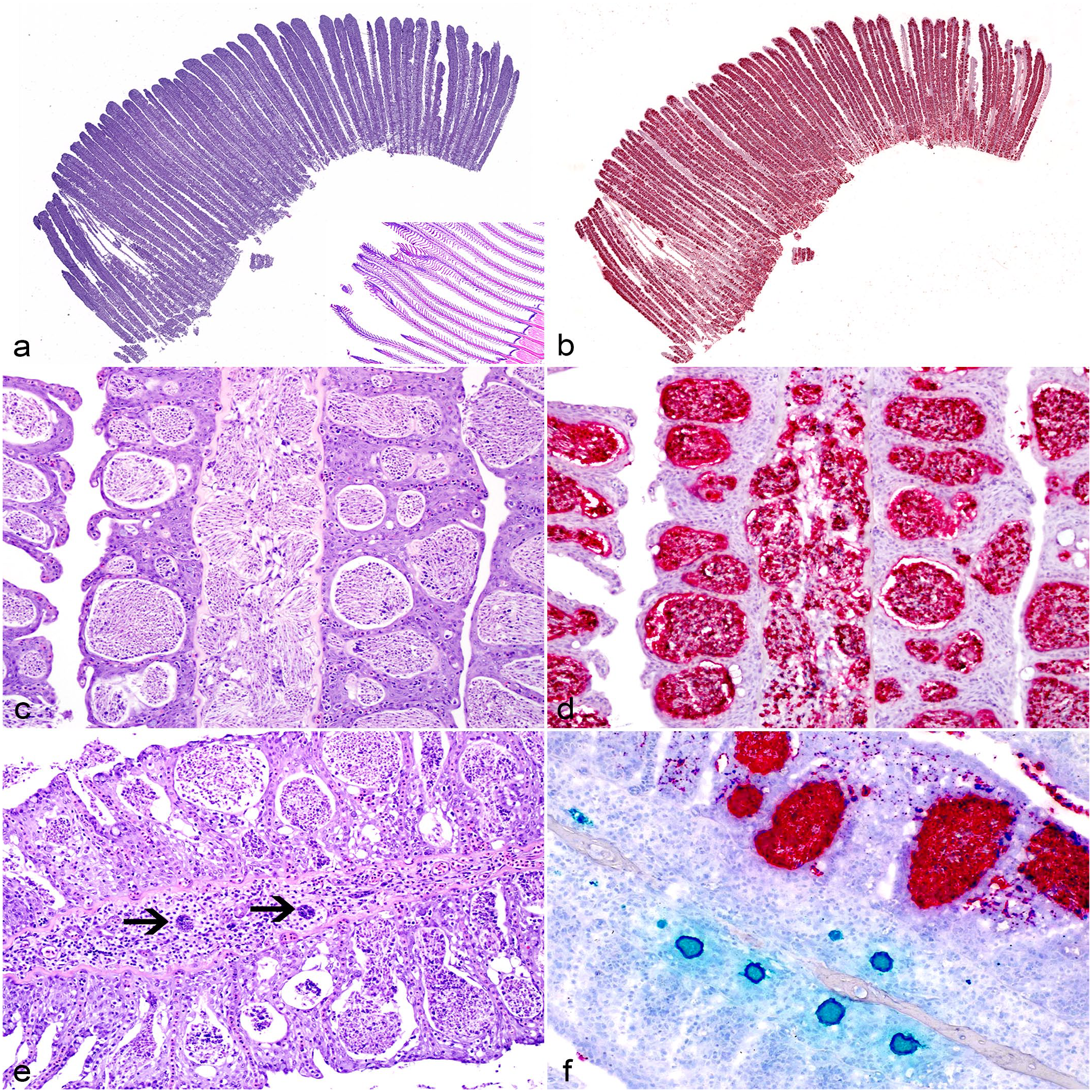
Histopathologic lesions associated with massive branchial henneguyosis of catfish in the gills of hybrid catfish. (a) Subgross image of a gill arch with diffuse thickening of filaments due to numerous interlamellar plasmodia. Inset: unaffected catfish gill tissue. Hematoxylin and eosin (HE). (b) Serial section of the gill arch in (a). Nearly all plasmodia are identified as Henneguya exilis by in situ hybridization (ISH) (red chromogen). Duplex H. ictaluri/H. exilis ISH. (c) Typical histologic lesions associated with MBHC with interlamellar spaces filled by epithelial hyperplasia, embedded myxozoan plasmodia, and inward rupture of plasmodia releasing myxospores into the filament stroma. HE. (d) Serial section of the gill in (c). All plasmodia and free myxospores are identified as H. exilis by ISH (red chromogen). Duplex H. ictaluri/H. exilis ISH. (e) Presporogonic life stages of a myxozoan (arrows) surrounded by granulomatous inflammation expanding the filament and plasmodia filling interlamellar spaces. HE. (f) Serial section of the gill in (e). Presporogonic life stages are identified as H. ictaluri by ISH (green chromogen) in association with proliferative gill disease lesions while interlamellar plasmodia are identified as H. exilis by ISH (red chromogen). Duplex H. ictaluri/H. exilis ISH.
Interlamellar plasmodia and free myxospores within the fibrous stroma and vasculature of the gill filament demonstrated consistent positivity with only the H. exilis-specific probe (Fig. 3b, d). Rare presporogonic stages in association with granulomatous inflammation and filament cartilage lysis only reacted with H. ictaluri-specific probes (Fig. 3e, f). Other probes showed rare, scattered hybridization throughout the sections, containing either few, scattered presporogonic stages (Raabeia-type TGR 2014) or single plasmodia (H. mississippiensis, H. postexilis, Myxo 11) (Table 1).
Presence of developing myxozoan life stages in the gills of hybrid catfish with natural massive branchial henneguyosis determined by in situ hybridization.
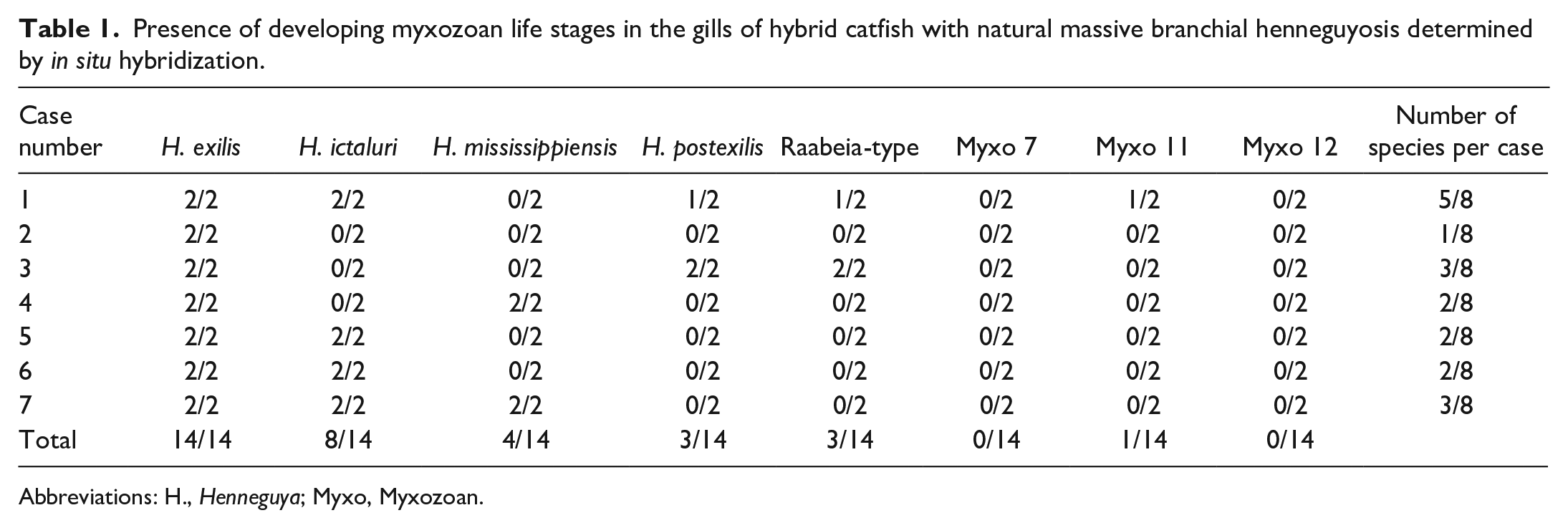
Abbreviations: H., Henneguya; Myxo, Myxozoan.
Discussion
Findings from this study indicate massive infections of H. exilis represent a myxozoan-induced gill disease distinct from PGD (Table 2), known hereafter as massive branchial henneguyosis of catfish (MBHC). Targeted metagenomic analysis of gill-infecting myxozoans revealed these cases in hybrid catfish resulted from nearly pure infections of H. exilis (>98.5%), a finding confirmed by only rare detection of other myxozoans, including H. ictaluri, by in situ hybridizaton. This is consistent with previous molecular characterization of lesions by laser capture microdissection. 20 The findings contrast with cases of PGD, where targeted metagenomic analysis and in situ hybridizaton studies have shown mixed infections.22,23 Although H. ictaluri is typically the dominant organism present and usually associated with characteristic PGD lesions, it is often accompanied by mixed infections with many other myxozoans and exhibits lower relative abundances compared to H. exilis in MBHC cases.22,23 Cases of massive H. exilis infection also had fewer discrete OTUs present than PGD cases, further demonstrating less diversity and species evenness in the myxozoan community compared to PGD. 23 All known and potential unknown taxa detected had direct matches to sequences previously identified in association with PGD, providing additional validation for both data sets. 23
Case definitions for massive branchial henneguyosis of catfish and proliferative gill disease in catfish aquaculture.

Outbreaks of MBHC result in high morbidity, but low mortality, whereas PGD often results in high morbidity and mortality. These differences are likely related to the underlying disease pathogenesis and pathology responsible for respiratory compromise, wherein respiratory distress in MBHC results from a transient mechanical obstruction, whereas PGD is a more injurious process, resulting in tissue destruction and filament collapse.20,27 It is hypothesized that massive plasmodial development by H. exilis interferes with oxygen exchange by physical impairment of water flow through obstructed gill interlamellar channels but causes minimal tissue damage and tissue function returns rapidly following plasmodial rupture and myxospore release. 20 In contrast, presporogonic stages of H. ictaluri incite profound destructive inflammatory, proliferative, and lytic changes, often with residual filament loss. 27 Proliferative gill disease outbreaks can persist in ponds for several weeks to months, often increasing in intensity over time. 26 Although fish may recover, the extensive tissue damage in PGD cases and persistent exposure to infective actinospores during disease progression often becomes insurmountable, resulting in death of the fish host.2,26,27 Despite differences in disease pathogenesis and pathology, similar clinicopathologic outcomes likely result from respiratory compromise in both diseases. Proliferative gill disease results in decreased oxygen partial pressure (pO2) and increased carbon dioxide partial pressure (pCO2) in the blood of affected fish. 1 Whether this occurs in MBHC affected fish remains to be determined.
The diseases both occur in mid to late spring and early summer yet involve different stages of myxozoan development, with MBHC involving numerous plasmodia with mature myxospores and PGD consisting largely of presporogonic stages associated with the destructive branchitis.20,27 This implies infection with H. exilis occurs months before clinical presentation with MBHC, whereas clinical PGD occurs within weeks of infection with H. ictaluri.6,19,20 Given that H. exilis and H. ictaluri share the same life cycle, it would be assumed that similar factors predispose to infection and disease outbreaks, although differences in the developmental progression and seasonal timeline of these diseases suggest otherwise.14,17,20 These differences could be related to either differing myxozoan virulence between H. exilis and H. ictaluri, fish host susceptibility, seasonal changes in fish immune status, or a combination of factors, which require further investigation.
Research has demonstrated differences in fish host species susceptibility to myxozoan infection and sporogenesis influence the myxozoan communities and diversity present in host tissues and environments.21,23 Differences in susceptibility to particular myxozoans exist between fish host species as well as between different strains of the same fish species.6,10,11,19,22 Massive H. exilis infections in hybrid catfish potentially reflect either increased susceptibility to infection and development of H. exilis (with hybrid catfish providing positive selective pressure for H. exilis proliferation), decreased susceptibility to other Henneguya spp. (with hybrid catfish providing negative selective pressure for other Henneguya sp.), or a combination of both. The latter is supported by the demonstration of myxozoan communities in ponds dedicated to hybrid catfish monoculture having decreased H. ictaluri abundance, Shannon diversity, and species evenness with a distinct peak in H. exilis relative abundance during each spring.8,21 This suggests that H. exilis exhibits some competitive advantage over other myxozoans in hybrid catfish systems.20,21 Progressive amplification and accumulation of H. exilis within continuous hybrid catfish monoculture ponds over multiple production cycles could potentially lead to myxozoan numbers associated with debilitating parasite burdens and disease.
Few myxozoans cause disease through heavy parasite burdens alone. Similar to these H. exilis infections, Myxobolus koi in koi carp, Cyprinus carpio, and Henneguya pseudoplatystoma in pinado, a hybrid South American catfish bred from crossing Pseudoplatystoma corruscans and Pseudoplatystoma fasciatum, form large plasmodia that obscure gill filaments and reduce respiratory capacity and surface area.3,15 These outbreaks were associated with pond or aquaculture settings which presumably contained a suitable definitive host in close proximity to large fish host biomass, and conditions allowing for completion and amplification of parasite replication. This suggests that other myxozoans with similar tissue tropisms may produce heavy parasite burdens and disease potential if introduced to intensive aquaculture or pond systems.20,24
Targeted metagenomic sequencing and ISH techniques clearly demonstrated the etiology and features of a distinct, myxozoan-induced gill disease of hybrid catfish caused by H. exilis. Although MBHC and PGD both induce respiratory distress, massive H. exilis infections are differentiated clinically from PGD based on levels of morbidity and mortality. Microscopic distinctions include the location and severity of tissue changes, as well as the myxozoan life stages present and their specific tissue tropisms. Molecular evidence indicates near pure infections of H. exilis occur in cases of MBHC, whereas cases of PGD involve a diverse community of myxozoans, often dominated by H. ictaluri.20,22,23 Although epidemiologic factors leading to outbreaks of H. exilis are unknown, similar targeted metagenomic analysis of controlled pond water environments dedicated to hybrid catfish monoculture revealed distinct peaks in relative abundance of H. exilis, suggesting increased host susceptibility and selection for H. exilis in hybrid catfish ponds. 21 Research into optimizing conditions for life cycle maintenance and propagation of catfish infecting Henneguya spp. within the laboratory setting is required to allow for the experimental challenges necessary to fulfill all of Koch’s postulates for this disease entity. Regardless, the pathologic and molecular evidence provided herein offers strong support for a myxozoan-induced gill disease of ictalurid catfish distinct from PGD and clearly associated with massive H. exilis infection. Finally, the repeated and continued diagnosis of MBHC in hybrid catfish since 2018 and increased commercial production of hybrid catfish suggest that this condition may represent an emerging disease of hybrid catfish monoculture. Further investigations are needed to determine the true prevalence of the disease in the catfish aquaculture industry and epidemiologic factors associated with outbreaks.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241259181 – Supplemental material for Massive branchial henneguyosis of catfish: A distinct, myxozoan-induced gill disease caused by severe interlamellar Henneguya exilis infection in catfish aquaculture
Supplemental material, sj-pdf-1-vet-10.1177_03009858241259181 for Massive branchial henneguyosis of catfish: A distinct, myxozoan-induced gill disease caused by severe interlamellar Henneguya exilis infection in catfish aquaculture by Justin M. Stilwell, Matt J. Griffin, John H. Leary, Lester H. Khoo and Alvin C. Camus in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Dr Larry Hanson and Dr Graham Rosser for providing additional samples for this work.
Author Contributions
LHK made original diagnoses and collected sample material. JMS and JHL designed and performed the experiment. MJG and ACC contributed to experimental design. JMS performed statistical analysis of targeted metagenomic sequence data. The manuscript was written by JMS with contributions from all coauthors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by the United States Department of Agriculture Agricultural Research Service (Project No. 58-6402-2729), the United States Department of Agriculture Catfish Health Research Initiative (CRIS 6402-31320-002-02), the National Center for Veterinary Parasitology, the Mississippi State University College of Veterinary Medicine, the Mississippi Agricultural and Forestry Experiment Station (MAFES), and the University of Georgia College of Veterinary Medicine.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
