Abstract
Visceral toxicosis of catfish (VTC) is a syndrome characterized by sudden mortality in apparently healthy market- and brooder-sized catfish (Ictalurus punctatus). This paper reports the design of a catfish neutralization assay to detect botulinum in catfish with VTC and verification by Endopep mass spectrometry (Endopep-MS). Sera from 6 affected catfish were incubated with botulinum antitoxin serotypes A, B, C, D, E, or F. For each serum sample, 3 experimental fingerlings were injected intracoelomically with each serotype-serum mixture and placed separately in an aquarium. Three fish were injected with VTC-affected serum only, and 3 fish were injected with unaffected serum only and also placed in separate aquaria. Signs of morbidity and mortality were seen in fish injected with sera combined with serotype A, B, C, or D, as well as in positive controls. No morbidity or mortality was seen in fish injected with sera combined with antitoxin serotypes E or F or negative control serum. Sera from affected and unaffected catfish were sent to Centers for Disease Control and Prevention for detection and differentiation of botulinum neurotoxin. Aliquots of 0.5 ml of sera were incubated with magnetic beads coated with antibodies to botulinum, and the beads were subjected to the Endopep-MS reaction. Sera from affected catfish tested positive for botulinum E. Sera from 34 unaffected catfish tested negative for botulinum. Although there was not enough botulinum present in affected samples to obtain exact quantification, the estimated quantity of botulinum E in these sera samples was between 0.01 and 0.5 mouse LD50/ml.
Introduction
Visceral toxicosis of catfish (VTC) was first recognized in the spring of 1999 in the Mississippi Delta in channel catfish (Ictalurus punctatus). 24 This syndrome is characterized by the sudden death of otherwise healthy market-sized fish when pond water temperatures are approximately 18 to 22°C. Because diagnostic submissions were negative for bacterial, parasitic, fungal, and viral pathogens, the etiology of this disease was considered to be a toxin. Clinical signs observed included erratic swimming and progressive muscular weakness leading to paralysis, lethargy, and death. Previously reported lesions of VTC include exophthalmia, intussusception of the intestines, ascites, congested spleen, and blanching of the intestinal tract. 24 One potential cause of these neurological signs is botulism. Intoxication with botulinum toxin has been reported in previous fish die offs with similar signs. 3,7,8,14 Seven serotypes of botulinum neurotoxin (BoNT) have been identified. 22 In North America, ingestion of botulinum serotype E formed in decomposing carcasses in contact with Clostridium botulinum spores on pond sediments is most commonly associated with botulism in fishes. 8,10
Several assays were used to detect botulinum in catfish with visceral toxicosis. These tests included a mouse bioassay of affected serum; an enzyme-linked immunosorbent assay of affected catfish serum for botulinum; anaerobic cultures of intestinal contents for C. botulinum; and polymerase chain reaction (PCR) of the liver, kidney, spleen, and intestine for C. botulinum. All these conventional tests were negative.
Botulism can be difficult to diagnose because some animal species are extremely susceptible to the toxin 19,21 and C. botulinum is not always detectable in intoxicated animals by culture or PCR. 5,10,21 If botulism was the possible cause of VTC, the conventional assays may lack the sensitivity for its detection. This manuscript reports the design of a catfish neutralization assay based on the mouse neutralization bioassay 4 with sufficient sensitivity to detect botulism in catfish with visceral toxicosis and the confirmation of this method by the Endopep mass spectrometry (Endopep-MS) assay.
Materials and methods
Catfish suspected of having VTC were selected for inclusion in this study based on postmortem findings and culture results. Necropsy of affected fish revealed well-fleshed, primarily 1- to 2-pound, market-sized fish with no external signs of disease. Internal examination of the fish revealed chylous or clear fluid (ascites) in the coelomic cavity, intussusception of the intestinal tract, reticulated pattern to the surface of the liver, congested spleen, and eversion of the stomach into the oral cavity. These lesions were consistent with previously reported VTC lesions. 24 Wet mounts of gill clips showed an occasional superficial parasite but not numerous enough to cause mortality. Cultures from these fish for bacteria were negative, and virus isolation on channel catfish ovary cells showed no cytopathic effects.
Blood was taken from 11 affected catfish and 34 healthy catfish not showing signs of VTC to serve as negative controls. Serum was collected in a routine manner and refrigerated at 4°C. Bloodletting yielded less than 5 ml of serum per fish.
For each assay, fingerling catfish weighing between 5 and 10 g were maintained in 20-gallon aquaria supplied with deep well water with a single pass rate of approximately 500 ml/minute. The temperature of the water was between 23 and 25°C, and water quality chemistries were within normal aquaculture conditions for catfish. 23
Bioassay method
VTC assay. Bioassays were conducted within 8 hours after the sera were collected. Serum (0.1 ml) from affected fish was injected intracoelomically into each of 3 experimental channel catfish fingerlings. Fish were checked 3 times a day for development of clinical signs and lesions previously reported in VTC. 24 Death of experimental fish within 96 hours was interpreted as diagnostic of VTC in the affected catfish used as a serum source. As a control, other experimental channel catfish fingerlings were concurrently injected with 0.1 ml of serum from clinically healthy catfish. Four days post injection, any surviving fish were euthanized with tricaine methanesulfonate. a Sera that caused VTC-like signs and lesions were used for neutralization assays.
Preliminary neutralization assay. Bioassays were conducted within 21 days after the sera were collected. Three treatment groups were used to determine if botulinum serotype E antitoxin b neutralized the VTC toxin. Group 1: VTC serum (0.3 ml) was mixed with 0.1ml botulinum antitoxin E b (diluted with sterile glycerol c and saline according to the manufacturer's directions). 4 Group 2 (positive control): VTC serum (0.3 ml) was mixed with 0.1 ml of the glycerol c and saline carrier. Group 3 (negative control): Three-tenths (0.3) ml unaffected (control) catfish serum was mixed with 0.1 ml of carrier. After 30 minutes of incubation at room temperature (∼22°C), each mixture was drawn into a separate 1-ml syringe. Fingerlings ranging in weight from 5 to 10 g were each injected intracoelomically with 0.125 ml/mixture. The number of fingerlings injected per mixture ranged from 2 to 5 depending upon the quantity of VTC sera available. Fish were grouped according to treatment in 1 of 3 80-liter aerated aquaria with free-flowing water. During the experiment, fish were fasted and observed 3 times a day for unusual signs of swimming, lethargy, or death. Fish that died were necropsied. Four days post injection, any surviving fish were euthanized with tricaine methane-sulfonate a and necropsied. This preliminary study tested sera from 4 different VTC-affected catfish.
Catfish neutralization assay. To test whether type E botulinum antitoxin b was type specific for the VTC toxin, neutralization assays were conducted using monospecific botulinum antitoxin serotypes A, B, C, D, E, and F b as they were received from Centers for Disease Control and Prevention (CDC) in Atlanta, Georgia. Monospecific antitoxin for BoNT C, D, and F d were not available initially for testing the VTC sera when these outbreaks occurred. The specificity of type E neutralization was tested for each serum and was assessed against the other antitoxins when they were received. There was enough serum remaining from fish #4 to repeat the assay when the C and D monospecific antitoxins b became available.
Bioassays were conducted within 21 days after the sera were collected. The design of the study was similar to the preliminary study but included VTC sera incubated with the monospecific serotypes of antitoxin in the saline/glycerol c carrier, as well as the positive and negative control groups (a maximum of 8 treatment groups). Fish were observed for morbidity and mortality as described above. To confirm results of the catfish neutralization assay, 6 sera samples from VTC-positive fish and 34 negative control catfish sera samples were packed on ice and sent overnight to CDC for analysis of BoNT by Endopep-MS.
Endopep MS assay
Materials. The BoNT E complex d was provided at 1 μg/mL total protein in 50 mM sodium acetate, 2 mg/mL gelatin, 3 mg/mL bovine serum albumin, pH 4.2. The polyclonal rabbit-specific IgGs d were provided in 150 mM potassium phosphate, pH 7.4 at 8.26 mg/mL. Dynabeads Protein G e were provided at 1.3 g/cm3 in phosphate-buffered saline (PBS), pH 7.4, containing 0.1% Tween-20 f and 0.02% sodium azide. The sequence of the synthesized peptide substrate was IIGNLRHMALDMGNEIDTQNRQIDRIMEKAD. g
IgG binding to protein G beads. The IgG was immobilized to the Dynabeads Protein G e as described in the manufacturer's protocol using 50 μg of IgG for BoNT E b diluted into 500 μl of PBS for every 100 μl of Dynabeads Protein G. e The IgG-coated Dynabeads d e also were cross-linked using the manufacturer's protocol. Cross-linked IgG-coated Dynabeads d e were stored in PBS-Tween buffer f (PBS with 0.05% Tween-20 f )at 4°C for up to 12 weeks.
Botulinum neurotoxin isolation from serum. Botulinum neurotoxin was handled within a class 2 biosafety cabinet equipped with HEPA filters. The volume of each sample tested, including controls, was 500 μl of serum. As a positive control, 2 mouse LD50 of BoNT E d was spiked into 500 μl of normal catfish serum. A negative control consisted of 500 μl of normal catfish serum. Twenty-five μlof 1% Tween f was added to each 500-μl serum aliquot, including the controls. Twenty μl of antibody-coated beads d e were added to each serum sample, and the mixture was agitated for 1 hour at room temperature. The beads were then washed twice with 1 ml each of PBS-Tween buffer f followed by 1 wash with 200 μl each of water. The water was then removed from the beads.
Number of experimental catfish dead after intracoelomic injection of visceral toxicosis of catfish (VTC) sera, VTC sera neutralized with antitoxin E, or control serum

Endopep mass spectrometry reaction. The reaction was performed as previously described 15,16 with a few modifications. In all cases, the final reaction volume was 20 μL; the final concentration of the reaction buffer was 0.05 M Hepes (pH 7.3), f 25 mM dithiothreitol, f 200 mM ZnCl2, f and 1 mg/ml bovine serum albumin/ and the final concentration of the peptide substrate g was 50 pmol μl. All samples then were incubated at 37°C overnight without agitation.
Mass spectrometry detection. After incubation, 2 μl of each 20-μl reaction supernatant was combined with 18 μl of matrix-assisted laser desorption/ionization (MALDI) matrix solution, h which is alpha-cyano-4-hydroxy cinnamic acid at 5 mg/ml in 50% acetonitrile, 0.1% TFA, and 1 mM ammonium citrate. Five-tenths μl of this mixture was pipetted onto each spot of a 192-spot MALDI plate. h Mass spectra of each spot were obtained by scanning from 700 to 4200 m/z in MS-positive ion reflector mode on an Applied Biosystems 4700 Proteomics Analyzer. h The instrument uses a nitrogen laser at 337 nm, and each spectrum is an average of 2,400 laser shots.
Results
The data from the preliminary study that assessed the specificity of botulinum serotype E antitoxin for the VTC toxin against negative and positive controls are presented in Table 1 All 3 fish in the positive control group (treatment group 2) died within 24 hours and had lesions compatible with VTC. All fish from treatment groups 1 and 3 (VTC sera neutralized with botulinum antitoxin type E and control serum, respectively) survived and showed no clinical signs nor had lesions compatible with VTC. This preliminary study was repeated with sera from 3 other VTC-positive fish with similar outcomes (Table 1).
The data from the studies to assess the specificity of botulinum antitoxin type E (vs. the other serotypes of botulinum antitoxin) are presented in Table 2. Fish injected with sera incubated with botulinum antitoxin serotypes A, B, C, or D and fish injected with only positive control serum showed signs of progressive muscular weakness manifested as erratic swimming and died within 48 hours. Fish injected with VTC sera combined with botulinum antitoxin serotypes E or F and fish injected with only control (unaffected) serum showed neither clinical signs nor died.
Figure 1A depicts the mass spectrum of 2 mouse LD50 of BoNT E concentrated from 500 μl of catfish serum (4 mouse LD50/ml) using Ab-coated magnetic beads followed by a reaction in buffer with peptide substrate. The intact substrate peptide IIGNLRHMALDMGNEIDTQNRQIDRIMEKADSNKT is present at 4,043.32 m/z. The peak at m/z 2,923.52 corresponds to the peptide with the sequence IIGNLRHMALDMGNEIDTQNRQIDR. This pep- tide is the N-terminal cleavage product of the substrate when cleaved by only BoNT E, so the presence of this peak is evidence for the presence of BoNT E in this sample. A negative control of 500 μl of catfish serum without BoNT E did not yield a peak at m/z 2,923.5 (Fig. 1B), and the dominant peak in this spectrum is the intact substrate peptide at 4,043.30 m/z. In total, 40 serum samples (including 34 negative control samples) were tested for the presence of BoNT E, and 6 of those samples were positive for BoNT E by Endopep-MS: 36–1, 35–2, 303, 2–4, 21–5, and 18–6. The spectra from these 6 samples all contained N-terminal cleavage products at 2,923.5 m/z, demonstrating the presence of BoNT E in these samples. Figure 1C is the mass spectrum of the Endopep-MS reaction of sample 30–3. The peak at 2,923.47 m/z proves the presence of BoNT E in this sample. Although there was not enough BoNT present in these samples to quantify the toxin concentrations, these sera samples were estimated to contain between 0.01 and 0.5 mouse LD50/ml of BoNT E.
Number of experimental catfish dead after intracoelomic injection of visceral toxicosis of catfish (VTC) sera, VTC sera incubated with monospecific antitoxins, or control (negative) serum

N/A = not applicable. Monospecific antitoxin was not available at the time of testing. Three fish were bioassayed treatment with the exception of sample 2–7, in which there were 2 fish/treatment. Sample 30–3† was reassayed when all monospecific antitoxins were available.
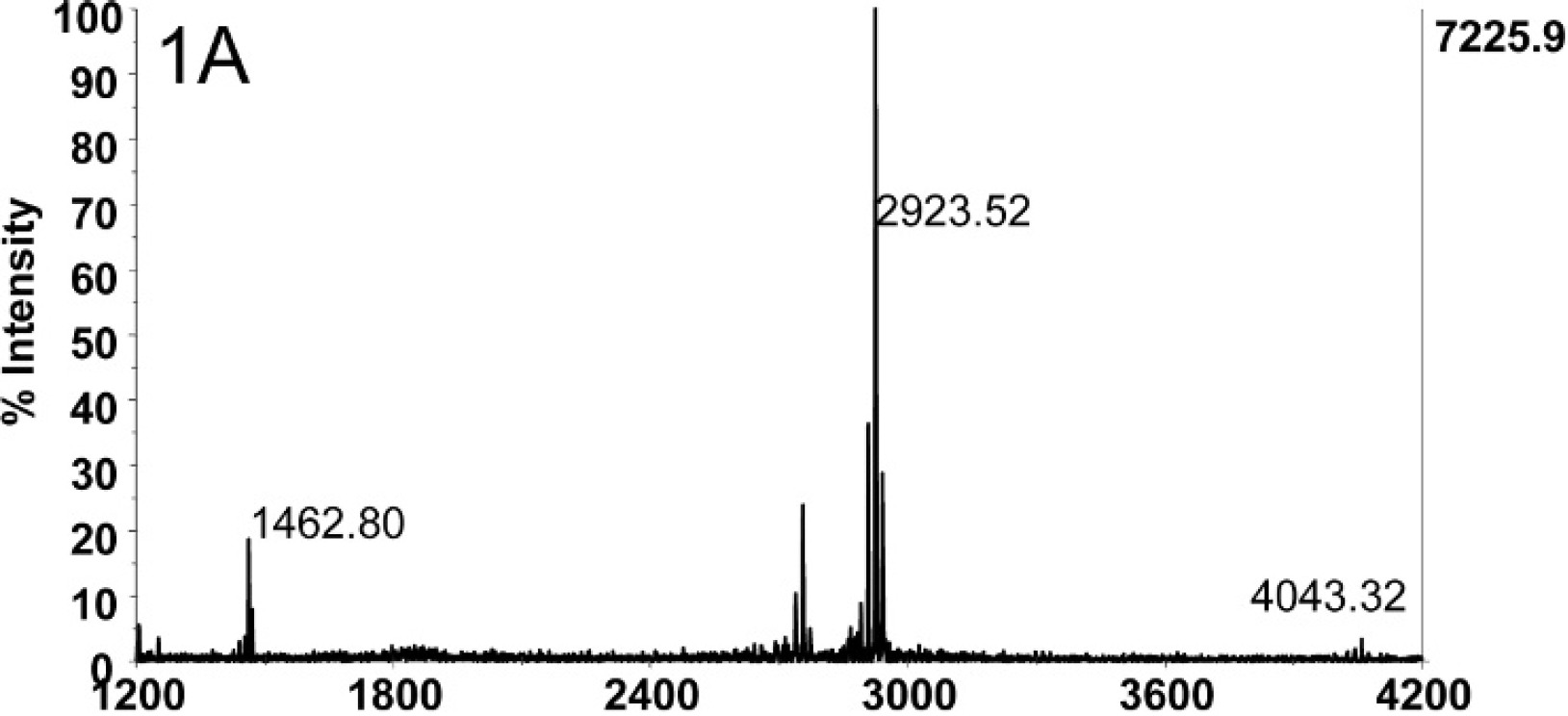
Mass spectrum of 2 mouse LD50 of BoNT E spiked in 500 μl of catfish serum. The intact substrate is present at 4,043.32 m/z. The peak at 2,923.52 m/z corresponds to the product of the substrate that was cleaved by BoNT E.
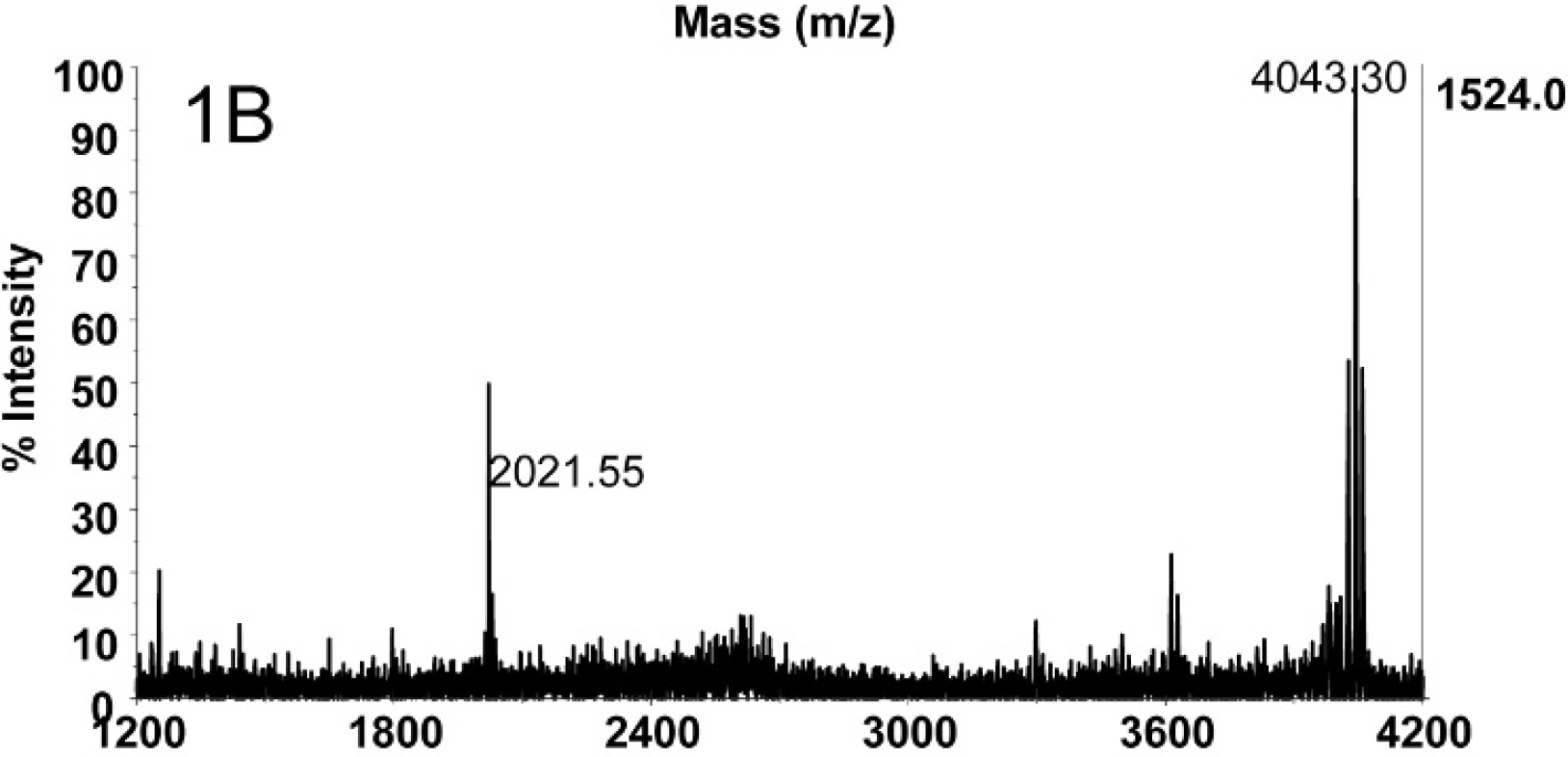
The negative control of catfish serum without BoNT E. There is no peak at m/z 2,923.52.
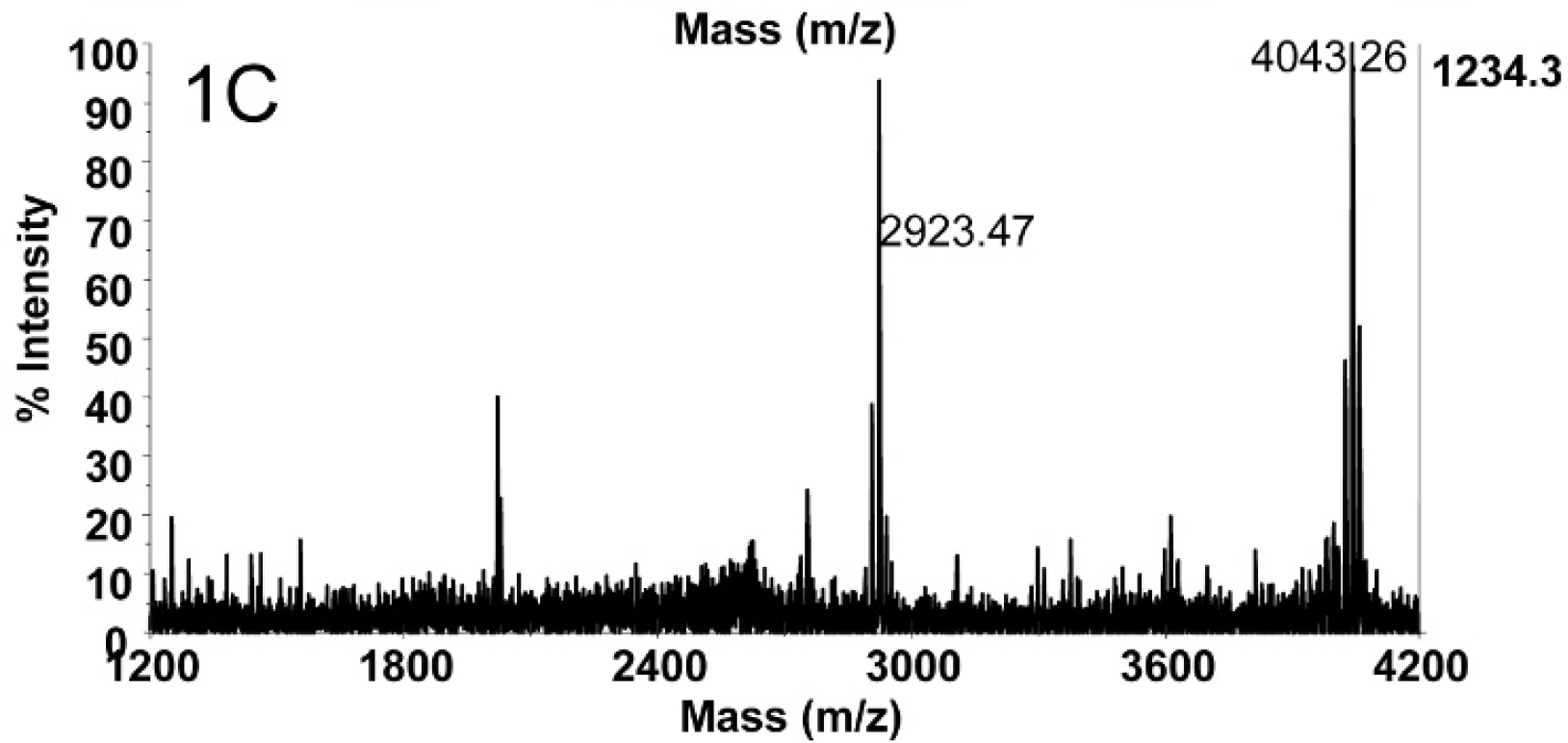
Mass spectrum of the Endopep-MS reaction of sample 30–3. The peak at m/z 2,923.47 demonstrates the presence of BoNT E.
Discussion
The mouse neutralization assay is considered the definitive test for diagnosis of botulism. 4,5 However, this assay did not detect BoNT when previously tested with the VTC catfish sera. By definition, the toxin activity required for lethality in a mouse is 1 mouse LD50 BoNT E, whereas the endopep MS assay estimated the VTC sera contained between 0.01 and 0.5 mouse LD50/ml BoNT E. Although the botulinum neutralization assays have never been performed with the same sera concurrently in mice and catfish, the estimated quantity of BoNT E in the serum of affected catfish was below the mouse bioassay's detection threshold. In the investigation of botulism outbreaks, toxin detection in substantial amounts in blood is considered to be diagnostic. 12 Whether the small amount of BoNT present in VTC catfish sera is substantial enough to be solely responsible for the syndrome will be determined in future botulinum LD50 assays.
The bioassays' results demonstrate different sensitivities to BoNT E in catfish and mice. One-tenth ml of serum from affected catfish injected intracoelomically in 5- to 10-g experimental fingerlings produced mortality, but 0.4 ml of the same sera injected intraperitoneally in experimental 20-g mice (used for the mouse bioassay) did not cause mortality, which suggests that the catfish is more sensitive than the mouse to BoNT E. Reports that domestic animals are more sensitive to BoNT than the mouse are well documented. 13,19,21 Previous experiments with BoNT in salmonids demonstrate that salmonids are twice as sensitive as the mouse when the toxin is given by the intraperitoneal route. 8 Titration studies are needed to determine the LD50 of BoNT E for the catfish.
Guidelines for the mouse neutralization assay state that results from multiple specimens should be consistent. 5 The catfish neutralization assay described in this report gave consistent neutralization with antitoxin BoNT E with many different catfish sera. In addition, the negative controls were consistently negative, indicating that the catfish neutralization and the endopep MS assays are highly specific.
No bioassay catfish died when injected with the sera incubated with antitoxin E, but some fish did not die when the VTC sera were incubated with antitoxin BoNT A or B or with VTC sera alone. This finding is also encountered in mouse bioassays and is attributed to such factors as biological variation of mice 2 and loss of potency of toxin. 5,9 Because the limit of detection of the catfish neutralization is not known, the quantity estimated in these sera could be approaching the lower limit of the assay. It should be considered that only free BoNT circulating in blood can be detected after injection into bioassay animals. 26 To produce its toxic effect, BoNT must bind to its substrate at the neuromuscular junction, and the bound BoNT is not free to circulate in blood. 13,19,21 Therefore, the quantity of free BoNT in the sera used for the catfish bioassay and Endopep-MS assay was likely less than the original blood concentration that killed the affected catfish.
The mouse neutralization assay generally distinguishes between the specific serotypes of botulinum. 4,18 However, cross-reactivity between botulinum antitoxin serotypes E and F has been reported. 11,13,18 The data from the catfish neutralization assays indicated that botulinum antitoxin serotypes E and F neutralized the toxic sera in experimental fish, so it was unclear from the neutralization trials which toxin was present in the VTC sera. Endopep-MS determined that serotype E, not F, was implicated as a cause of VTC.
The Endopep-MS method is an activity-based, mass-spectrometric assay that detects the BoNT. This method has proved successful for detecting all 7 BoNT toxin types 1,2 and involves incubation of BoNT with a peptide substrate that mimics the toxin's natural target. Each BoNT cleaves the peptide substrate in a specific, toxin-dependent location that is unique for each of the BoNT toxin types. 1,2,20 The reaction mixture is then assayed by mass spectrometry, which detects any peptides and determines their masses. Detection of the peptide cleavage products corresponding to their specific toxin-dependent location indicates the presence of a particular BoNT toxin type. If the peptide substrate either remains intact or is cleaved in a location other than the toxin-specific site, then that BoNT toxin type is not present. This method can detect BoNT at levels comparable with or lower than levels detected with mouse bioassays. 1,2,15–17
The use of fish bioassays to establish LD50s and sensitivities for botulinum has been described, 6,25,26 but no fish neutralization bioassays have been reported. This manuscript reports a catfish neutralization bioassay confirmed by Endopep-MS to link botulinum in the syndrome of VTC. Because of their high sensitivity and specificity, these techniques have the potential for wide application in the field of botulinum diagnostics and research.
Footnotes
a.
Western Chemical, Inc., Ferndale, WA.
b.
Centers for Disease Control and Prevention, Atlanta, GA.
c.
Fisher Scientific, Fairlawn, NJ.
d.
Metabiologics, Madison, WI.
e.
Dynal, Lake Success, NY.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Los Alamos National Laboratory, Los Alamos, NM.
h.
Applied Biosystems, Framingham, MA.
