Abstract
Myxobolus albi was diagnosed in the cartilage of captive lumpfish (Cyclopterus lumpus) from 2 public aquaria. Eleven fish were affected, with the most common clinical signs being exophthalmos and grossly visible 1- to 2-mm white to tan scleral nodules. Myxozoan cysts were identified in the cartilage of the skull, branchial arch, sclera, vertebrae, tongue, all fin insertions, and the pectoral girdle. Cysts resulted in expansile, deforming, space-occupying lesions, resulting in exophthalmos but often lacking significant tissue damage or inflammation. Once cysts ruptured, free spores elicited a mild to marked inflammatory response. Spores measured 7.5 to 9.0 µm × 3.0 to 6.0 µm and contained 2 pyriform polar capsules oriented at one pole as well as occasional 1-µm-diameter basophilic nuclei. Identification was based on spore morphology together with polymerase chain reaction and sequence comparison of 18S ribosomal DNA. Isolates had 99% similarity to M. albi.
Introduction
Myxozoans are metazoan parasites that mainly infect poikilothermic animals. Over 700 species of myxozoans have been described in fishes. 19 Myxobolus cerebralis, Henneguya ictaluri, and Tetracapsuloides bryosalmonae, the species associated with whirling disease of the salmon, proliferative gill disease of the catfish, and proliferative kidney disease of the salmon, respectively, have been responsible for high morbidity and mortality and great economic losses.7,14,33 Myxozoans have a complex life cycle that often alternates between vertebrate and invertebrate hosts with sporogony in each (i.e., alternating bisporogony).4,11
Lumpfish (Cyclopterus lumpus) are semipelagic fish found in the eastern and western North Atlantic. Their pelvic fins have been modified into a single ventral sucker, allowing them to adhere to coastal rock structures and floating objects, such as algae and debris. They are an important commercial fish collected and reared for their meat and roe.5,8 Due to their unique body shape and behavior, lumpfish are also a popular exhibit fish in public aquaria. The current report describes the clinical signs and pathology of Myxobolus albi infection in the cartilage of captive lumpfish at the New England Aquarium in Boston, Massachusetts (NEAq) and the John G. Shedd Aquarium in Chicago, Illinois (JGSAq).
Materials and methods
Animals
Lumpfish were wild-caught by hand using SCUBA in Eastport, Maine, during October from 2005 through 2008 and were exhibited at the 2 aquaria. Cases 1–5 occurred at NEAq, while cases 6–11 originated from JGSAq. All animals completed routine quarantine protocols including immersion formalin at NEAq and immersion nitrofurazone, praziquantel, and oral fenbendazole at JGSAq as well as any other antimicrobial or antiparasitic medications required based on the results of skin scrapes and gill biopsies at the time of arrival. Animals were housed in multitaxa exhibits with various fishes and invertebrates at temperatures of 7–10°C. The fish were fed a mixture of live or frozen zooplankton and frozen fish or squid depending on the size of the lumpfish. Weights of affected animals ranged from 0.5 kg to 3.8 kg including both juveniles and adults. Cases 1, 2, and 11 were euthanized using an overdose of tricaine methanesulfonate a at approximately 250 ppm.
Cytological and histological examination
A premortem fine-needle aspirate was conducted on case 2 in order to obtain cytological specimens from scleral nodules. Samples were expressed onto glass slides and stained with Romanowsky staining for microscopic examination. Tissues from enucleations and gross necropsies were fixed by immersion in 10% neutral buffered formalin and submitted to the Connecticut Veterinary Medical Diagnostic Laboratory, University of Connecticut (Storrs, CT; cases 1–5) and the Zoological Pathology Program, University of Illinois (Maywood, IL; cases 6–11) for histopathological evaluation. The tissues were fixed for a minimum of 48 hours, grossly evaluated, trimmed to fit plastic cassettes, processed routinely, embedded in paraffin, sectioned at 4 or 5 µm, and stained with hematoxylin and eosin and Ziehl–Neelsen acid-fast stains for light microscopic examination. The dimensions and shapes of myxozoan spores in histological sections of formalin-fixed tissues as well as the number, size, and location of polar capsules were compared to previously identified species. 25
Molecular analysis
Myxozoan identification was performed by polymerase chain reaction (PCR) and sequence comparison of 18S ribosomal DNA (rDNA) of the species in question and other known myxozoans. Formalin-fixed, paraffin-embedded tissue blocks from case 2 were submitted to the Fish Pathology Laboratory, University of California (Davis, CA). Portions of the paraffin-containing myxozoan cysts in scleral cartilage were cut from blocks, and genomic DNA was extracted using a silica membrane–based genomic DNA extraction kit b with deparaffinization according to the manufacturer’s instructions. Concentrations of genomic DNA samples were measured by a spectrophotometer, c and approximately 100 ng of genomic DNA was used for degenerate PCR. Combinations of degenerate primers (Myxgen4f and ACT1r, Myxgen4f and Myxgen2r; Table 1) targeting short fragments of the myxozoan 18S rDNA (<250 bp) were used for the reactions, as degradation of DNA was suspected due to formalin fixation.16,20 Polymerase chain reaction was conducted in 50-µl reaction volumes that included 200 µM of each dNTP, 1.5 mM of MgCl2, 40 pmol of each primer, 1 U of Taq DNA polymerase, d and 5 µl of 10× reaction buffer. The cycling conditions were as follows: initial denaturation step at 95°C for 5 min and then 40 cycles at 95°C for 30 sec, 50°C for 30 sec, and 72°C for 1 min, followed by a final extension step at 72°C for 5 min and then held at 4°C. The PCR products were separated on 2% agarose gels followed by ethidium bromide staining. The DNA bands were observed under ultraviolet transillumination, and amplicons of the expected size were excised and recovered using a gel extraction kit. e Extracted DNA was ligated into a cloning plasmid vector f for transformation of Escherichia coli DH5α competent cells. g The plasmid carrying the PCR product was extracted using a plasmid DNA extraction kit h according to the manufacturer’s instructions. The sequence of the inserted fragment was determined using M13 forward and reverse primers by fluorescently labeled dideoxynucleotide terminator sequencing using an automated DNA sequencer i from 3 clones with the suspected M. albi genomic DNA fragment. Sequence similarity searches were performed using the Basic Local Alignment Search Tool for Nucleotide sequences (BLASTn) program available at the National Center for Biotechnology Information (NCBI) website (http://blast.ncbi.nlm.nih.gov/Blast.cgi). 1 Pairwise comparisons of the parasite obtained in the current study with closely related species were performed using BioEdit software. j
Primers used for amplification of 18S ribosomal DNA from a myxozoan parasite of lumpfish (Cyclopterus lumpus).
Another primer set targeting 1.5 kb of M. albi 18S rDNA (538MyxFw and 540MyxRv; Table 1) was designed based on the reference sequence (EU420055). 30 Genomic DNA was extracted from frozen material of myxozoan cysts within the ocular cartilage from cases 9 and 10 using the silica membrane–based genomic DNA extraction kit, a as described previously, without deparaffinization. Polymerase chain reaction using this second primer set was performed using the same composition of PCR reagents but with a longer extension time: 95°C for 5 min and then 30 cycles at 95°C for 30 sec, 50°C for 30 sec, and 72°C for 2 min. The sequence of the amplified fragment (1.5 kb) was determined as described previously.
Results
Clinical findings
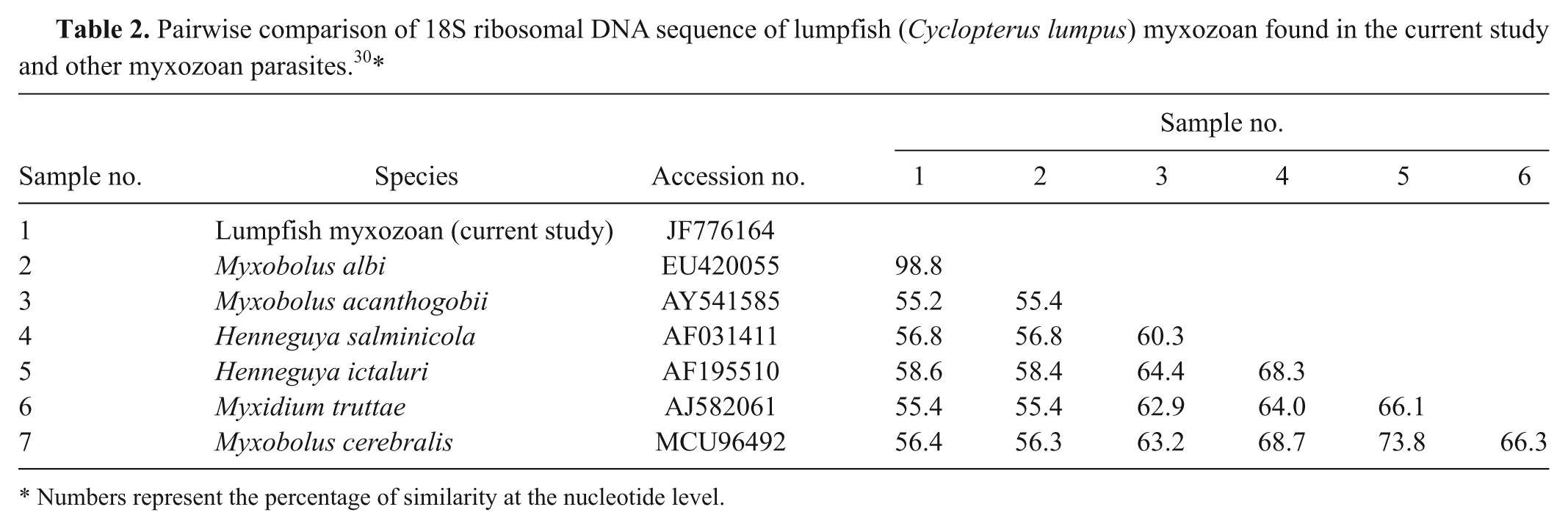
Mild to severe unilateral or bilateral exophthalmos was noted in all cases (Fig. 1). Case 1 was observed acutely spinning and then lying on its side with increased respiratory rate and effort. The fish was euthanized due to a poor prognosis. Bilateral exophthalmos was noted at gross necropsy. White or tan 1- to 2-mm-diameter nodules affected approximately 70–90% of the sclera in all cases. Hyphema was noted in case 2 (Fig. 1). Microscopic examination of scleral fine-needle aspirate from case 2 revealed multiple aggregates of 10- to 13-µm basophilic, circular to ovoid spores with 2 similarly sized pyriform polar capsules at one end. Occasional spores contained a darkly basophilic nucleus (Fig. 2). Case 3 had emphysema in the anterior and posterior chambers and a cranioventrally subluxated lens at initial examination of the right eye. Cases 5, 6, and 11 were initially examined due to unrelated causes, and exophthalmos was incidentally noted at that time. Cases 8 and 10 presented with unilateral exophthalmos, each undergoing enucleation of the affected eye.

Severe bilateral exophthalmos and hyphema of the left eye are observed in a lumpfish (Cyclopterus lumpus) infected with Myxobolus albi (case 2).
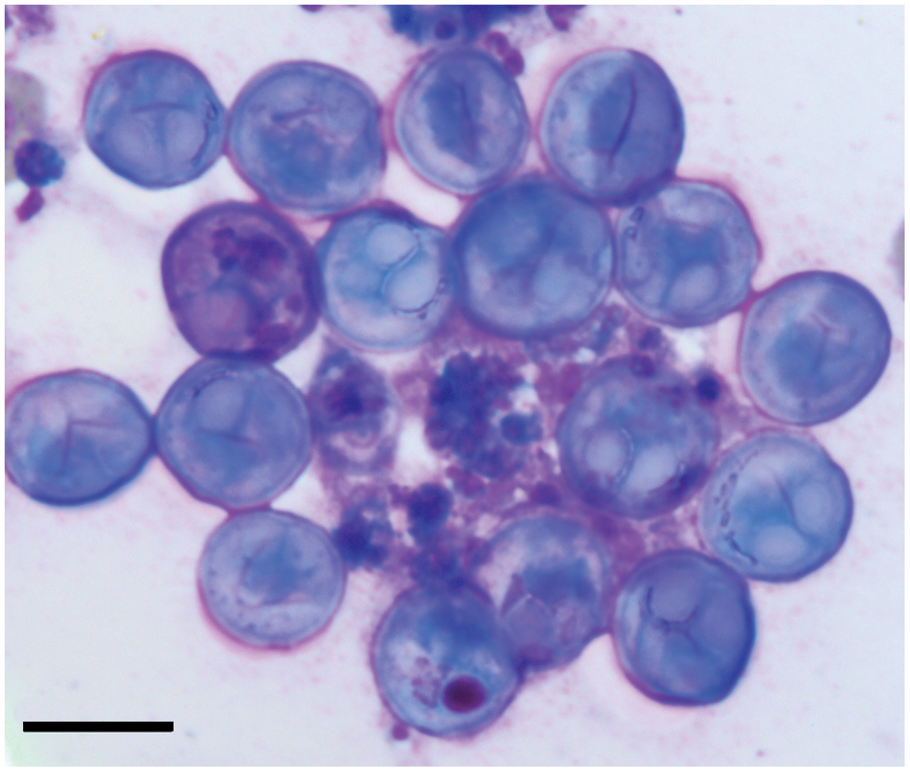
Lumpfish (Cyclopterus lumpus). Multiple, round myxozoan spores are present in this cytology of a fine-needle aspirate from the sclera of case 2. A pair of ovoid to pyriform polar capsules is discernable in several of these spores. Romanowsky stain. Bar = 10 µm.
All of the cases either died or were euthanized at NEAq or JGSAq between August 2006 and January 2011 due to the severity of the myxozoan infection (cases 2–4) or unrelated causes including potential drug reaction (case 5), bacterial septicemia (cases 6, 11), disseminated mycotic infection (cases 7, 8, 11), jejunitis secondary to foreign material impaction (case 9), and acute, severe postovariectomy blood loss (case 10).
Pathological findings
Gross findings included multifocal to coalescing myxozoan cysts, each composed of sporogonic plasmodia up to 2 mm in diameter. Cysts were identified in the cartilage of the skull, branchial arch, sclera, vertebrae, tongue, all fin insertions, and the pectoral girdle (Fig. 3A–C). Cysts most commonly resulted in expansile, deforming, space-occupying lesions lacking significant tissue damage and inflammation (Fig. 4A–C). Cysts contained numerous 7.5- to 9-µm-long × 3.0- to 6.0-µm-wide ovoid spores, each with a 1- to 3-µm-thick refractile wall, 2 pyriform polar capsules oriented at 1 pole, and occasionally a 1-µm-diameter basophilic nucleus. However, in some areas, large and coalescing cysts were associated with cartilage degeneration and necrosis, disruption of cortices of adjacent bone, and extension into surrounding tissue. Outside of cysts, free spores elicited a mild to marked granulomatous inflammatory response of the surrounding cartilage. In case 8, aggregates of myxozoan spores separated the retina from retinal pigmented epithelium and extended throughout the choroid, vitreous, posterior, and anterior chambers and into the ciliary body and iris. Drainage angles were obscured by myxozoan spores, histiocytes, erythrocytes, fibrin, and necrotic cellular debris. Myxozoan spores with similar morphology to those identified in the sclera or histiocytes with phagocytized fragments of spores were also scattered throughout parenchyma, interstitium, tubules, and glomeruli of the kidneys of cases 3 and 9, sometimes associated with inflammation or necrosis.
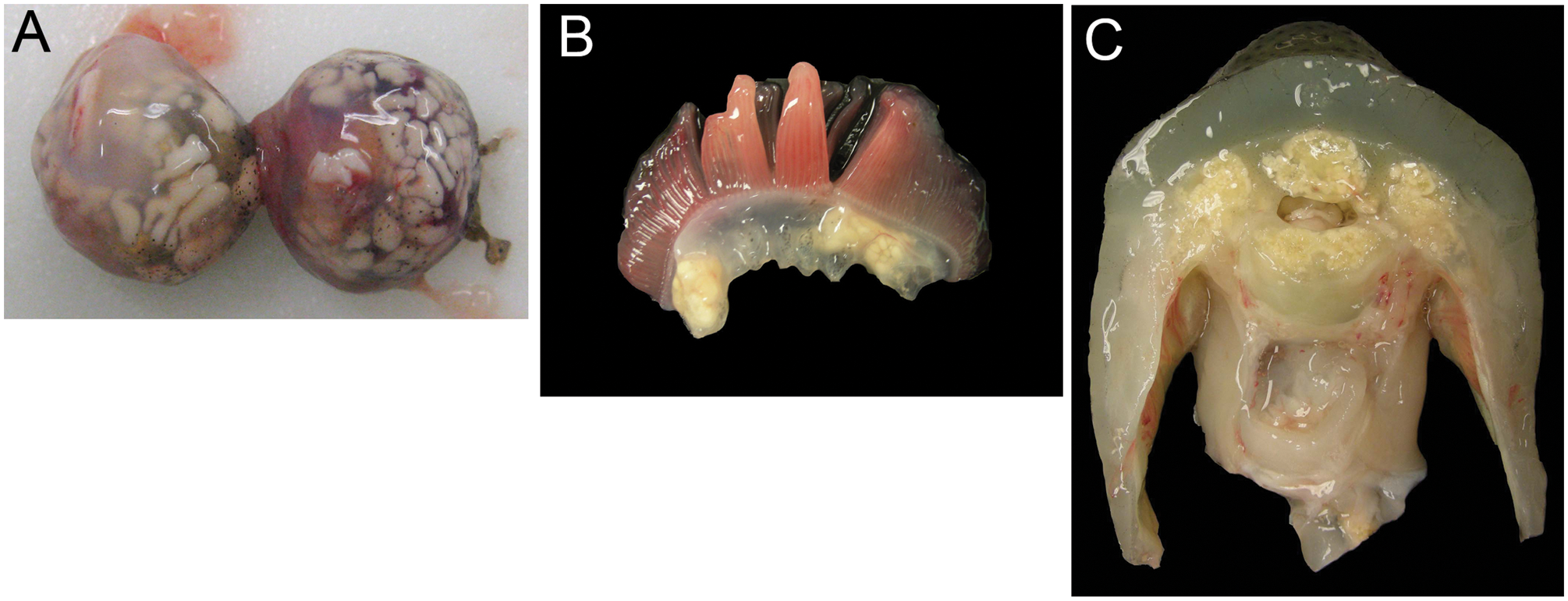
Lumpfish (Cyclopterus lumpus). Myxobolus albi cysts are dispersed throughout the sclera (
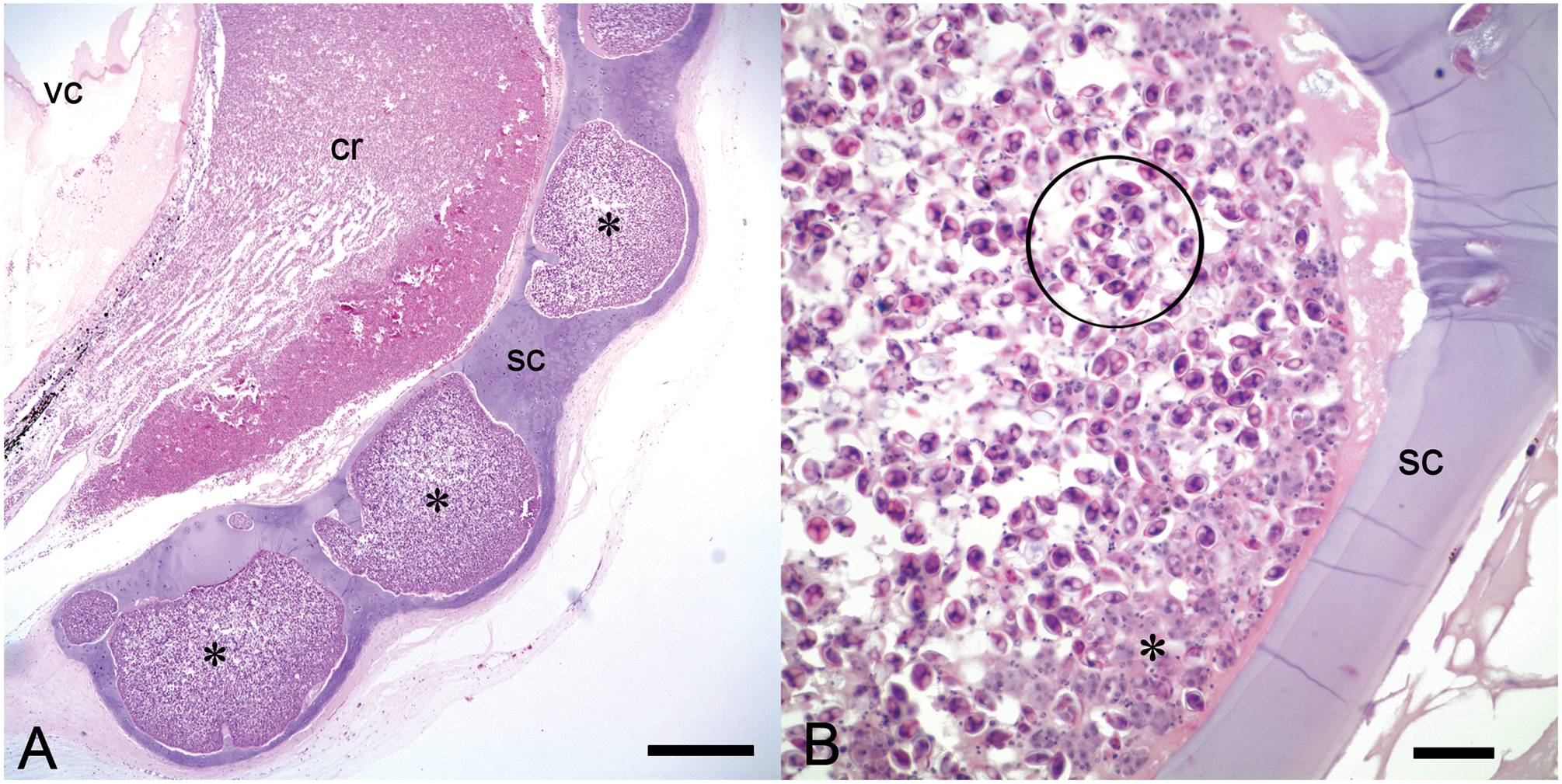
Lumpfish (Cyclopterus lumpus).
Other significant pathological findings included acute, severe myocardial necrosis (case 1), marked diffuse atrial endothelial hypertrophy (cases 3, 10), granulomatous nephritis without identification of myxozoan spores (case 2), and multiorgan changes due to fungal or bacterial septicemia (cases 6–8, 11). These findings were considered evidence of intercurrent disease processes unrelated to the myxozoan infection.
Molecular analysis
An 18S rDNA fragment (153 bp) was successfully amplified by the Myxgen4f and ACT1r primer set from a NEAq lumpfish (case 2) with unknown myxozoan parasites using formalin-fixed, paraffin-embedded material. The longer fragment (1,496 bp; JF776164) covering almost the entire 18S rDNA sequence was amplified from frozen tissue from cases 9 and 10 submitted by JGSAq using the 538MyxFw and 540MyxRv primer set. Both amplified DNA sequences had 99% similarity to M. albi 18S rDNA at the nucleotide level (EU420055). Pairwise comparisons with other myxozoan parasites shown in Table 2 indicate that the sequence is distinct from other myxozoan species. A summary of clinical, pathological, and molecular findings is provided in Table 3.
Numbers represent the percentage of similarity at the nucleotide level.
Clinical, pathological, and molecular findings for 11 lumpfish (Cyclopterus lumpus) infected with Myxobolus albi.*

NEAq = New England Aquarium (Boston, MA); OS = oculus sinister (left eye); FNA = fine-needle aspirate; OD = oculus dexter (right eye); JGSAq = John G. Shedd Aquarium (Chicago, IL); rDNA = ribosomal DNA.
Discussion
Myxobolus albi was recently described as a new myxozoan species in the gill arch of the common goby (Pomatoschistus microps) in Scotland. 30 Several myxozoan species are known to infect cartilage and have been associated with some of the most economically significant diseases of fish including proliferative gill disease of the channel catfish and whirling disease of salmonids.14,33 Once mostly a disease of farmed fish, whirling disease has recently been found in wild stock, causing concerns of population decline.14,27
While several species of myxozoans cause clinical illness,22,25 the majority of infections cause chronic subclinical diseases with little to no clinical signs or host immune response. 15 In the current cases, all lumpfish exhibited unilateral or bilateral exophthalmos. Despite extensive cartilaginous expansion and deformation, there was often little inflammation unless there was rupture of the plasmodia and release of spores into the surrounding tissue, allowing interaction of antigenic spores with host macrophages and lymphocytes and thus promoting the host inflammatory response. This release of spores and degeneration of ocular tissues and systemic cartilage are likely important to the natural life cycle of the myxozoan, allowing the release of spores into the environment. Case 1 exhibited increased respiratory effort and acute spinning behavior prior to euthanasia, the latter likely being a nonspecific sign of illness, as no central nervous system lesions were observed in infections by this myxozoan. Case 3 was 1 of only 3 cases in which myxozoan infection was considered significant in the death of a lumpfish. During gross necropsy, cysts were noted within the sclera and other cartilage as in all other cases. Renal tubular necrosis was associated with myxospores having similar morphology to those identified in cartilage, which were observed in glomeruli and renal tubules. Several reports have suggested that such a severe reaction may be due to infection of an inappropriate tissue or host.23,25,26 However, the kidney was not tested by PCR to confirm the identity of these myxospores. Atrial endothelial hypertrophy was attributed to systemically circulating myxozoan antigen. Renal mineralization was also observed, but this could have been the result of a concurrent disease process.
The method of transmission of M. albi in lumpfish is not fully understood. It is widely demonstrated that an invertebrate host is usually needed to complete the myxozoan life cycle.4,11,34 However, direct fish-to-fish transmission has been shown in some circumstances.9,24,31 Lumpfish were wild-caught by NEAq staff and subjected to quarantine prior to mixing with previously collected animals or being transported to other institutions. It seems most plausible that animals arrived infected, although transmission directly between fish and acquisition of infection via an unrecognized invertebrate host within the exhibit systems are hypothetical alternatives. It is notable that 2 adult lumpfish born at NEAq have no grossly visible scleral nodules at the time of publication and that all cases to date have been diagnosed in lumpfish weighing over 400 g, despite the presence of smaller specimens in the collection. It is possible that animals under 400 g are too small to have grossly visible scleral cysts or that there is a latent subclinical period, with clinical signs developing after a certain age or body size or after a period of acute or chronic stress. 29
To date, there are no consistently effective medical treatments for myxozoan infections in fish. Several methods of disinfection including ultraviolet exposure, chlorine bleach, and hydrogen peroxide have been previously used to kill environmental infective stages.18,32 The coccidiostat salinomycin with or without amprolium has successfully reduced the severity of branchial and renal myxozoan infection in sharpsnout sea bream (Diplodus puntazzo) and tapir fish (or elephantnose fish, Gnathonemus petersii).3,10,21 Fumagillin also has some efficacy against myxozoa but has a high incidence of toxic side effects.3,17,21 Multiple individualized treatments including, but not limited to, systemic acetazolamide, intraocular gentamicin, and systemic and topical broad-spectrum antibiotics were unsuccessfully attempted in the current cases to correct the exophthalmos but were not specifically aimed at eliminating the myxozoan infection. Further investigations into therapeutic agents for the treatment of myxozoan infections are warranted. Clinical and pharmacokinetic trials using salinomycin or other coccidiostats would be beneficial in determining the effectiveness of these drugs on myxospores in specific tissues and organs (e.g., therapeutic drug levels in the cartilage and scleral ossicle).
The pairwise comparisons of the amplified 18S rDNA sequences from infected lumpfish at JGSAq revealed significantly high similarity at the nucleotide level to that of M. albi (99%) and lower similarity to those of other representative myxozoans (<60%) as shown in Table 2, suggesting that the myxozoan found in the cartilage of these lumpfish is most likely one of the isolates in the species M. albi. Prior to molecular testing, the parasites were presumptively identified as Myxobolus aeglefini Auerbach, 1906 based on myxospore morphology and previous reports of M. aeglefini in the cartilage and bone of the head of lumpfish and in the cornea of the blue whiting (Micromesistius poutassou). 25 Spores from M. aeglefini and M. albi share several morphological features including similar size, shape, and location and number of polar capsules.25,28,30 Multiple recent studies of other myxozoan species have reported that re-evaluations of isolates based on 18S rDNA sequence comparisons often confirm the presumptive identification of isolates but occasionally determine that myxozoans previously identified as different species are the same.19,28,35 Currently, DNA sequences from M. aeglefini are not available; therefore, it is possible that previously described parasites in the cartilage of lumpfish 25 were misidentified as M. aeglefini based on myxospore morphology and were actually the yet unknown M. albi or that the 2 species are actually the same.
The current study and other reports regarding the use of molecular techniques to further describe myxozoan species or their phylogenetic relationships suggest the need for a comprehensive database linking genetic information and morphological features of the organisms.2,6,12,13 Historically, myxozoans have been grouped based on spore morphology, host, or site fidelity and geographic location. In the present study, the presence of M. albi was demonstrated in a different host, distant geographic location, and different environment (marine vs. estuarine) from that in which it was originally described, a link made possible by comparison of 18S rDNA.
Footnotes
Acknowledgements
The authors acknowledge Stephen Atkinson at Oregon State University for providing the primer sequences, Dr. Ronald P. Hedrick for guiding the molecular procedures, Pilar J. Nelson for ![]() , and Alexandra Young for assistance with graphics and digital artistry. The authors also appreciate the husbandry and veterinary staff of the New England Aquarium and John G. Shedd Aquarium for their care of the affected animals and assistance with necessary medical treatments.
, and Alexandra Young for assistance with graphics and digital artistry. The authors also appreciate the husbandry and veterinary staff of the New England Aquarium and John G. Shedd Aquarium for their care of the affected animals and assistance with necessary medical treatments.
a.
Finquel®, Argent Chemical Laboratories, Redmond, WA.
b.
QIAamp® DNA Mini Kit, Qiagen Inc., Valencia, CA.
c.
BioPhotometer plus, Eppendorf North America, Hauppauge, NY.
d.
Platinum Taq DNA Polymerase, Invitrogen Corp., Carlsbad, CA.
e.
QIAEX II® Gel Extraction Kit, Qiagen Inc., Valencia, CA.
f.
pGEM®-T Easy Vector System, Promega BioSciences, San Luis Obispo, CA.
g.
Escherichia coli DH5α Competent cells, Invitrogen Corp., Carlsbad, CA.
h.
QIAprep® Spin Miniprep Kit, Qiagen Inc., Valencia, CA.
i.
ABI PRISM® 3730 DNA Analyzer, Applied Biosystems, Carlsbad, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) declared that they received no financial support for their research and/or authorship of this article.