Abstract
Fixation and demineralization protocols for bone marrow (BM) across diagnostic laboratories are not standardized. How different protocols affect histomorphology and DNA amplification is incompletely understood. In this study, 2 fixatives and 3 demineralization methods were tested on canine BM samples. Twenty replicate sternal samples obtained within 24 hours of death were fixed overnight in either acetic acid-zinc-formalin (AZF) or 10% neutral-buffered formalin (NBF) and demineralized with formic acid for 12 hours. Another 53 samples were fixed in AZF and demineralized with hydrochloric acid for 1-hour, formic acid for 12 hours, or ethylenediamine tetraacetic acid (EDTA) for 24 hours. Histologic sections were scored by 4 raters as of insufficient, marginal, good, or excellent quality. In addition, DNA samples extracted from sections treated with the different fixation and demineralization methods were amplified with 3 sets of primers to conserved regions of T cell receptor gamma and immunoglobulin heavy chain genes. Amplification efficiency was graded based on review of capillary electrophoretograms. There was no significant difference in the histomorphology scores of sections fixed in AZF or NBF. However, EDTA-based demineralization yielded higher histomorphology scores than demineralization with hydrochloric or formic acid, whereas formic acid resulted in higher scores than hydrochloric acid. Demineralization with EDTA yielded DNA amplification in 29 of 36 (81%) samples, whereas demineralization with either acid yielded amplification in only 2 of 72 (3%) samples. Although slightly more time-consuming and labor-intensive, tissue demineralization with EDTA results in superior morphology and is critical for polymerase chain reaction (PCR) amplification with the DNA extraction method described in this article.
Keywords
In clinical practice, bone marrow (BM) aspirates and core biopsies are typically obtained to investigate unexplained leukocytosis, leukopenia, anemia, thrombocytopenia, circulating atypical or neoplastic cells, and to diagnose certain infections. Marrow tissue is also collected as part of complete autopsies. Processing of trabecular marrow, comprised of bone and hematopoietic tissue, requires fixation followed by demineralization, paraffin embedding, sectioning, and staining. Assays in addition to histopathology, such as polymerase chain reaction (PCR) for clonally rearranged lymphocyte antigen receptor genes (PARR) and immunohistochemistry (IHC), are useful to investigate suspected neoplastic and infectious conditions. Formalin-based fixation and acidic demineralization are considered to reduce epitope integrity and accessibility for IHC, and DNA quality for PARR, but the effect of specific protocols on either assay is unclear.7,16,22
Fixatives and demineralizing agents are mostly chosen according to individual laboratory-developed protocols with 10% neutral-buffered formalin (NBF) being the most widely used fixative. The penetration rate, ambient temperature, and time of exposure influence fixation. 12 In general, NBF penetrates tissue at approximately 1 mm per hour; therefore, 24 hours of fixation is considered adequate for tissues <1 cm3, such as marrow core biopsies. 12 Formaldehyde-based fixatives preserve tissue by forming crosslinks within and between proteins, glycoproteins, nucleic acids, and polysaccharides.11,13 Crosslinking can interfere with antigen recognition by antibodies and be detrimental to the availability and quality of DNA and RNA.1,14,22 Therefore, a fixative that optimally preserves cell morphology and retains integrity of proteins and nucleic acids is desirable. Alternate fixatives such as acetic acid-zinc-formalin (AZF) are suggested to yield superior quality marrow sections but are not in common use.1,17,20 Tissue penetration of AZF is similar to that of NBF, but AZF purportedly allows for higher DNA yield and better preservation of epitopes.1,10,14,24 The fixative B5, consisting of formaldehyde and mercuric chloride, yields high-quality hematopoietic tissue sections, but mercuric chloride is an environmentally persistent toxin prohibited for use in many countries, including Canada. 1
Samples containing bone must be demineralized after fixation through removal of insoluble calcium and phosphorus salts prior to sectioning. The mineral salts form hydroxyapatite, which combined with an organic protein matrix, imparts hardness to bone and hence resistance to sectioning. 8 Therefore, inevitably, intertrabecular hematopoietic tissue is also exposed to demineralizing solutions, which may further impact tissue morphology and macromolecular integrity.2,5 Demineralization is the removal of various minerals from bone, most of which is calcium. Therefore, the terms demineralization and decalcification are used interchangeably in this article. Commonly used decalcifying agents are inorganic acids like hydrochloric acid (HCl), organic acids such as formic acid (FA), and neutral calcium-chelating substances such as ethylenediamine tetraacetic acid (EDTA). 4 Acids induce ion transposition and subsequent formation of calcium salts in the decalcifying solution. 23 FA, a weaker acid, is reported to have a less detrimental effect on nucleic acid recovery than HCl. 2 Demineralization with EDTA is considered superior to acid-based approaches for tissue preservation, nucleic acid integrity, and IHC.5,21 However, EDTA is not commonly used since submersion for up to 24 hours is required, while acid-based demineralization typically requires 10 to 60 minutes.5,21 As with all tissues, sample volume is an important factor for fixation and demineralization. Trabecular core biopsies with a typical thickness of 2 to 4 mm are more quickly fixed and decalcified than larger BM samples obtained at autopsy. 12
In vivo acquisition of marrow core biopsies in veterinary patients requires sterile technique, special needles, and substantial technical skill. Hence, optimal processing methods are important to maximize the potential utility of such precious samples. In this study, it was hypothesized that AZF fixation provides superior morphology scores and PCR amplification relative to NBF and that demineralization with EDTA yields superior results compared to acid-based demineralization. The specific objectives were to compare the effects of different fixation and demineralization procedures on (1) histomorphology and (2) amplification of DNA extracted from canine marrow.
Materials and Methods
Cases
Samples from 73 dogs that were euthanized (n = 42) or died (n = 21), and were submitted for postmortem examination, were available. Inclusion criteria were weight >5 kg and initiation of autopsy <24 hours after death. The dogs had a wide range of ages and weights (Table 1), and a variety of lesions, such as bronchopneumonia, dilated cardiomyopathy, hemangiosarcoma, peripancreatic fat necrosis, cerebellar herniation, intestinal adenocarcinoma, osteosarcoma, metastatic melanoma, sepsis, encephalomyelitis, histiocytic sarcoma, lymphoma, hepatocellular neoplasia, and others. Hemangiosarcoma was the most common diagnosis (n = 9). None of the dogs were euthanized due to primary hematopoietic disease. The procedures were exempt from Animal Use Protocols by the Animal Care Committee of the University of Guelph since sampling of marrow is part of standard autopsy protocol.
Animal and study parameters.
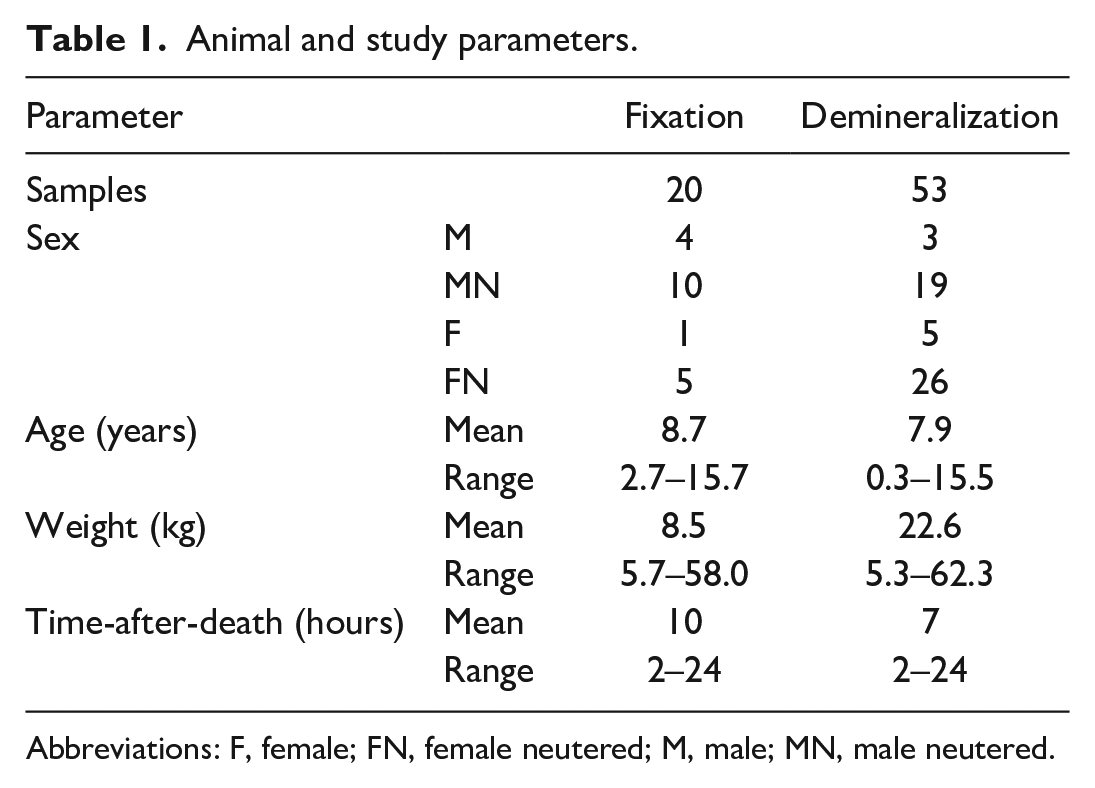
Abbreviations: F, female; FN, female neutered; M, male; MN, male neutered.
Specimens
All samples were collected from the sternum. 9 The sternum was isolated by dissection with a scalpel to remove adherent soft tissue and placed in a thin-slot miter box (37-240 Zona Tools) for immobilization. Replicate adjoining 2 to 4 mm transverse slices were cut with a general-purpose razor saw (3550, Zona Tools; Supplemental Fig. S1). The time-after-death (TAD) was recorded as the interval between death and initiation of postmortem examination, and the cause of death was noted.
Fixation
The AZF solution was prepared fresh monthly (Supplemental Table S1) from individual components, 17 and 10% NBF was obtained from a commercial source (Leica Biosystems, Concord, ON, Canada). Adjoining sawed slices of sternum were submerged in 60 ml of AZF or NBF and placed on a shaker for 14 to 20 hours at room temperature. After fixation, the cortical bone was manually removed with a scalpel blade. The remaining BM was rinsed with tap water for 30 seconds and placed in FA demineralization solution (Cal-Ex II Fixative/Decalcifier, Fisher Scientific, Ottawa, ON, Canada; 10.6% FA, 7.4% formaldehyde, 1% methyl alcohol) on a shaker for 12 hours. Samples were then rinsed with tap water for 30 seconds, re-placed into the original fixative, and routinely processed by dehydration, embedding in paraffin, sectioning at 2 to 3 µm, and staining with hematoxylin and eosin (H&E; Animal Health Laboratory, University of Guelph, Guelph, ON, Canada).
Demineralization
Three replicate sternal slices were placed in separate containers of AZF for 14 to 20 hours on a shaker at room temperature. As above, after fixation, the cortical bone was removed with a scalpel blade, and the remaining slices were rinsed with tap water for 30 seconds. Demineralization solution containing 14% EDTA was prepared fresh each month from stock reagents (Supplemental Table S2). Slices were then placed in 60 ml of either HCl demineralization solution (SurgiPath Decalcifier II, Leica; 7% HCl) for 55 to 60 minutes, FA demineralization solution for 12 hours, or EDTA solution for 24 hours on a shaker. 3 The EDTA solution was replenished at 12 hours. The optimal time for demineralization was estimated from a preliminary study of 8 samples based on the ability to section sternal marrow slices free of cortical bone with a scalpel blade without the use of force. After demineralization, samples were processed as above.
Histomorphology Scoring
Sections were microscopically evaluated by 4 assessors who were blinded to the fixation and demineralization protocol. Scores were 1 = insufficient; 2 = marginal; 3 = good, and 4 = excellent quality for interpretation. Features for fixative scores considered were preservation of tissue architecture, integrity of cell membranes, and nuclear and cytoplasmic staining. Samples decalcified by different methods were scored using the same scale and included assessment for the presence of fragmented or folded bone, insufficiently decalcified bone (brown to black color), homogeneously eosinophilic bone matrix, and osteocyte preservation.
DNA Amplification
Replicate slices of sternum from 6 dogs were fixed overnight in either AZF or NBF. Samples were then demineralized in either HCl, FA or EDTA for 1, 12, or 24 hours, respectively, for a total of 36 samples (2 fixatives × 3 decalcifiers × 6 dogs). Tissues were then routinely processed for histology. One approximately 3 mm2 and 10-µm thick scroll was cut from each paraffin block into a microtube. Next, DNA was isolated (QIAamp DNA FFPE Advanced Kit, Qiagen, Hilden, Germany), and the concentration and quality were measured by spectrophotometry (NanoDrop, Thermo Fisher, Waltham, Massachusetts). Subsequently, approximately 50 ng of DNA was subjected to PCR with primers targeting conserved regions of the T cell receptor gamma (TRG) and the immunoglobulin (Ig) heavy chain (IgH framework 2, FR2, and IgH framework 3, FR3) genes to generate amplicons of 75 to 170, 150 to 210, and 210 to 300 base pairs, respectively, as described previously. 15 A total of 216 amplifications were performed (108 in duplicate) with NEB Q5 high-fidelity polymerase according to the manufacturer’s instructions, and amplification efficiency was scored from 0 to 3 based on review of capillary electrophoretograms (Qiagen; high-resolution cartridge, OL 500; eGene HDA-GT12 analyzer, eGene Inc, Irvine, California; with BioCalculator software) as 0 = no amplification; 1 = weak amplification (discernible result above baseline but less than 1 relative fluorescence unit [RFU]); 2 = moderate amplification (between 1 and 2 RFU); or 3 = strong amplification (>2 RFU).
Statistical Analysis
Data were analyzed with Statistical Analysis Software (SAS 9.4, Cary, North Carolina), and differences were considered significant at P ≤ .05. A Wilcoxon signed-rank test was used to account for pairing of the mean scores of AZF and NBF to analyze the difference between the fixatives. Friedman’s test was performed to compare the mean score of fixation and demineralization methods. Spearman’s rank correlation test was used to evaluate the relationship between the TAD and the average section score for each fixation and demineralization procedure. This analysis was based on the average scores from the 4 raters and performed first by treatment to evaluate if there was a relationship between the treatments, and then by TAD relative to the mean score. Graphs were prepared with Prism (version 10.0.2, GraphPad).
For the DNA amplification study, in addition to descriptive contingency tables, a Pearson chi-square test was applied to evaluate if there was a general relationship between the variables (primers, fixatives, and decalcifiers) and the scores. Initial analysis started with univariable modeling of scores to evaluate 1 explanatory factor at a time with Friedman’s test. As the interactions could not be evaluated, the data were transformed to binomial (amplification scores 1, 2, and 3 vs a score of 0) using a binomial random effect logistic regression by a 3-factor factorial randomized block to model the probability of DNA amplification. Approximate 95% confidence intervals (CI) of parameter estimates were generated. The raw data from this study are available by contacting the corresponding author.
Results
Histomorphology of Sections Prepared With Different Fixatives
The mean scores for 20 replicate samples of sternal BM fixed with AZF and NBF were not significantly different from each other and were significantly correlated (Figs. 1, 2). The mean scores for AZF and NBF were 2.925 (standard deviation [SD] = 0.659) and 2.850 (SD = 0.603), respectively. Scores of sections fixed with either AZF or NBF were not significantly correlated with the TAD (Fig. 3). Spearman’s correlation coefficients of TAD with fixative were −0.236 (P = .315) for AZF and −0.133 (P = .575) for NBF. The mean histomorphology scores did not differ significantly between samples from dogs that were euthanized or that died (P = .381).
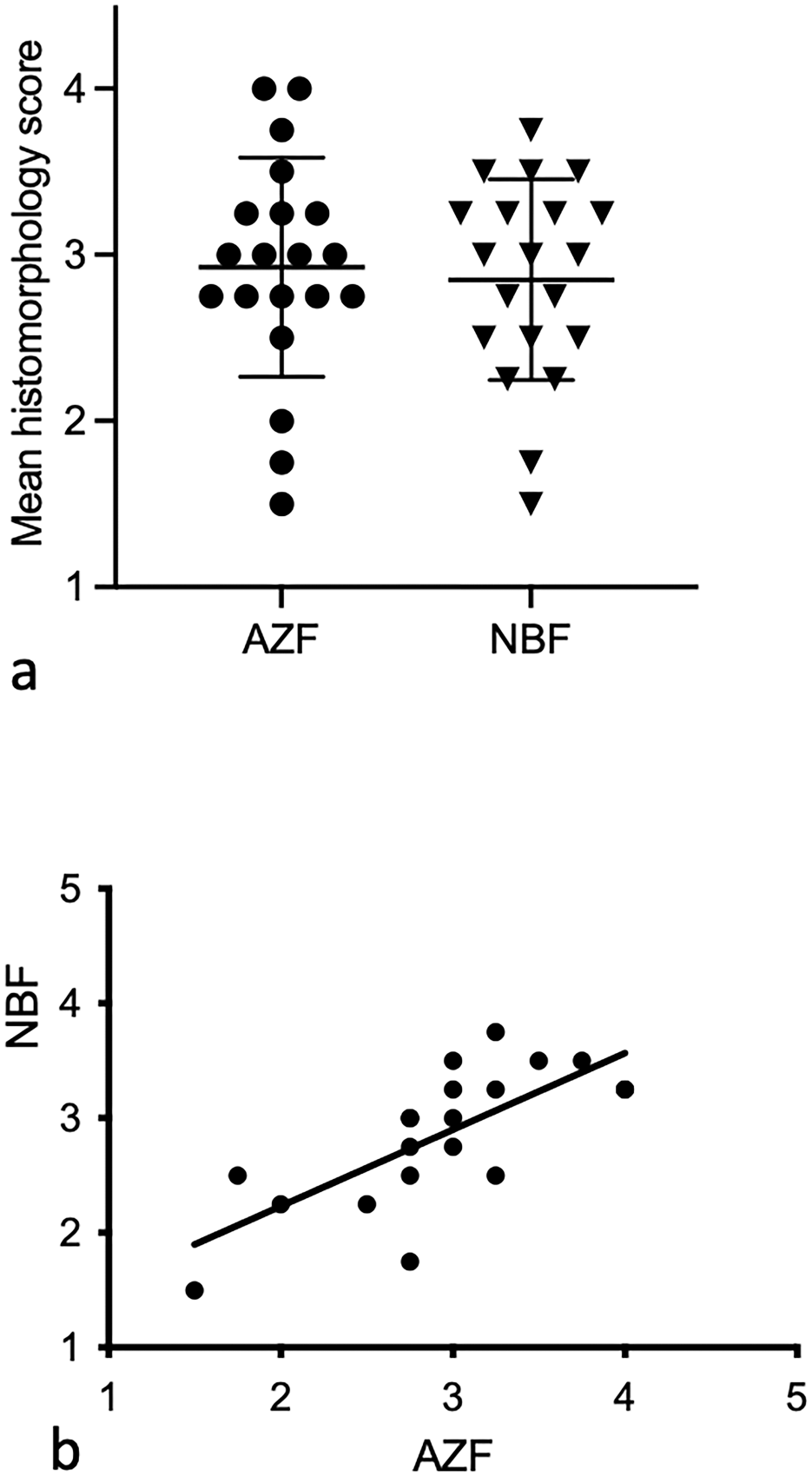
The mean ± standard deviation of histomorphology scores for 20 bone marrow samples fixed with acid-zinc-formalin (AZF) and neutral-buffered formalin (NBF) were (a) not significantly different (P = .566) from each other and (b) significantly correlated with each other (P < .001, r = 0.730).
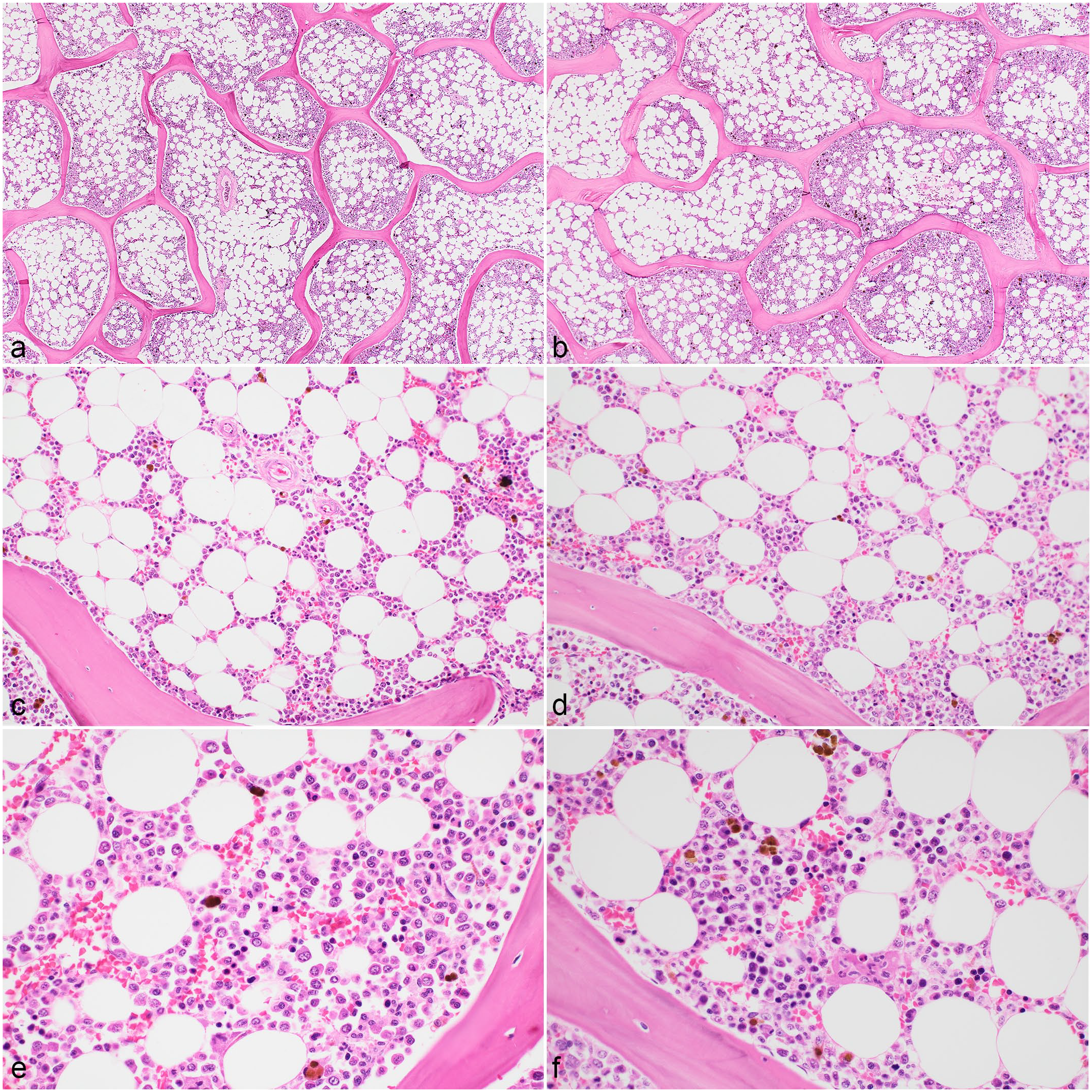
Histomorphology of sternal bone marrow sections from a dog. Histomorphology scores did not differ significantly between samples fixed with the different fixatives. All samples were decalcified with formic acid and fixed with (a, c, e) acid-zinc-formalin or (b, d, f) neutral-buffered formalin. Hematoxylin and eosin.
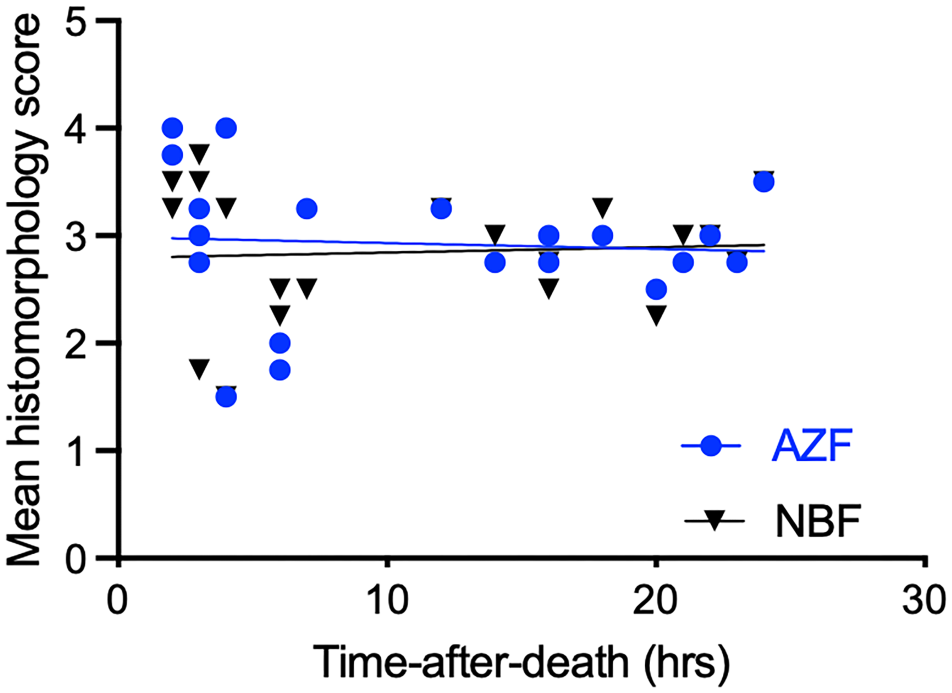
The mean histomorphology scores of 20 bone marrow samples prepared with 2 different fixatives were not significantly correlated with the time-after-death. AZF, acid-zinc-formalin; NBF, neutral-buffered formalin.
Histomorphology of Sections Prepared With Different Decalcifiers
The mean scores for 53 replicate samples of sternal BM decalcified with HCl, FA, or EDTA were 2.20 (SD = 0.500), 2.51 (SD = 0.487), and 2.76 (SD = 0.459), respectively (Fig. 4). A pair-wise test of the mean scores showed that the differences between the decalcifiers were statistically significant: HCl vs FA, P = .001; HCl vs EDTA was P < .001, and FA vs EDTA also P < .001 (Fig. 5). The most common attributes for lower morphologic scores in samples decalcified with HCl or FA were poor osteocyte preservation and trabecular bone fragmentation. The mean scores for the 3 decalcifiers were not significantly correlated with TAD: correlation coefficients were 0.0500 (P = .721), 0.214 (P = .122), and 0.192 (P = .167), for HCl, FA, and EDTA, respectively (data not shown). Similarly, the mean scores for decalcifiers were not significantly different between animals that were euthanized or died.
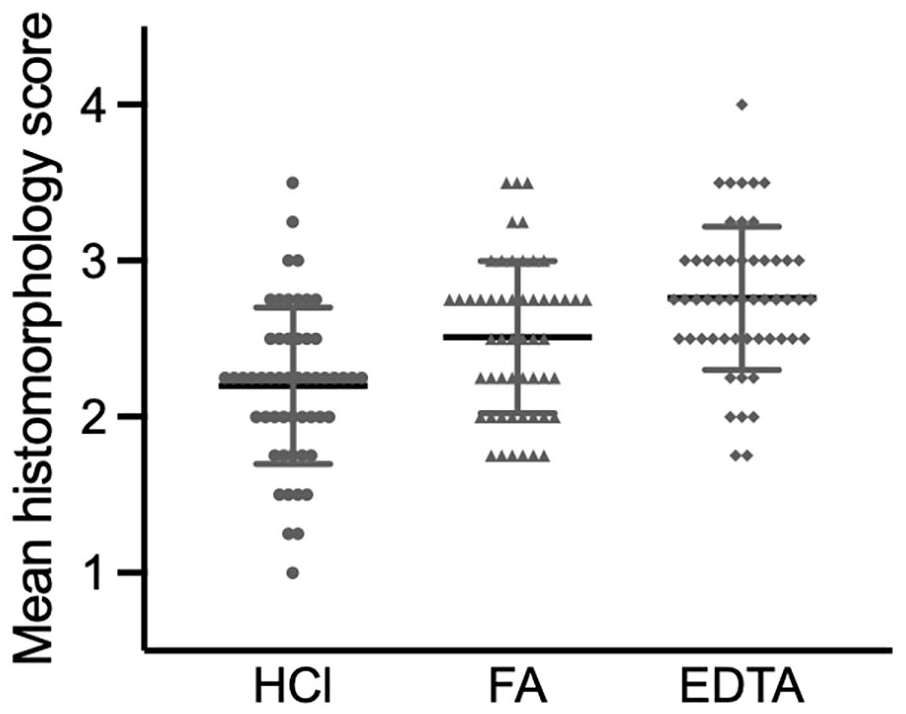
The mean ± standard deviation histomorphology scores for 53 bone marrow samples decalcified with hydrochloric acid (HCl), formic acid (FA), or EDTA. HCl vs FA, P = .001; HCl vs EDTA P < .001, FA vs EDTA P < .001.
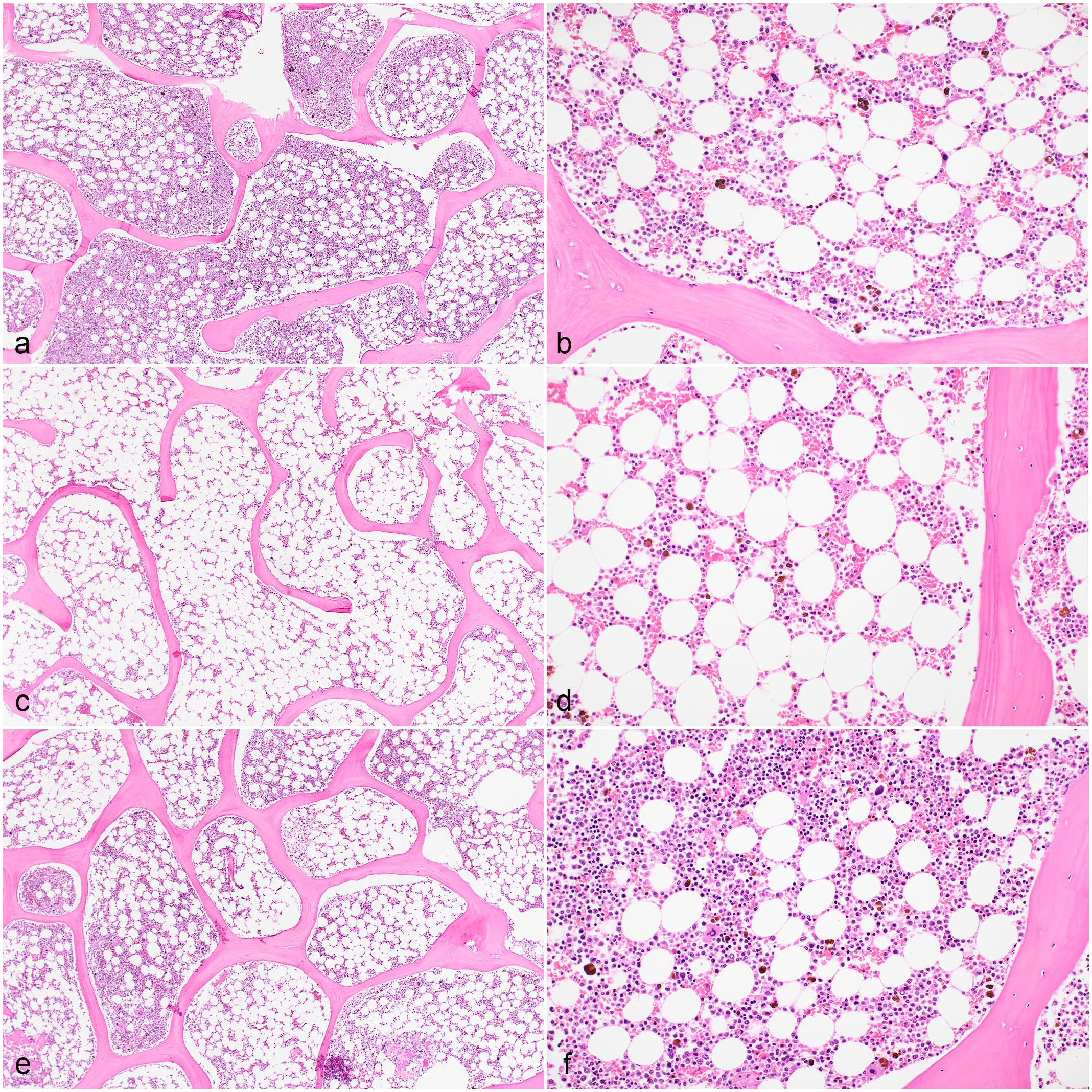
Histomorphology of sternal bone marrow sections from a dog. (a, b) Samples decalcified with hydrochloric acid (HCl) showed trabecular bone fragmentation, while (e, f) samples decalcified with EDTA had better bone and hematopoietic tissue preservation. All samples were fixed with acid-zinc-formalin and decalcified with (a, b) HCl, (c, d) formic acid, or (e, f) EDTA. Hematoxylin and eosin.
Effect of Fixation and Demineralization on DNA Amplification
Assessment of amplification results with descriptive contingency tables showed no significant relationship between the amplification scores and the different primer pairs (P = .163, Supplemental Table S3). The relationship between the amplification scores and the fixatives also was not significant (P = .069) (Supplemental Table S4). However, amplification scores of 2 or 3 were only obtained with EDTA decalcification, whereas only 2 of 72 amplifications of samples decalcified with either HCl or FA resulted any amplification, and only with a score of 1 (P < .001, Fig. 6 and Table 2).
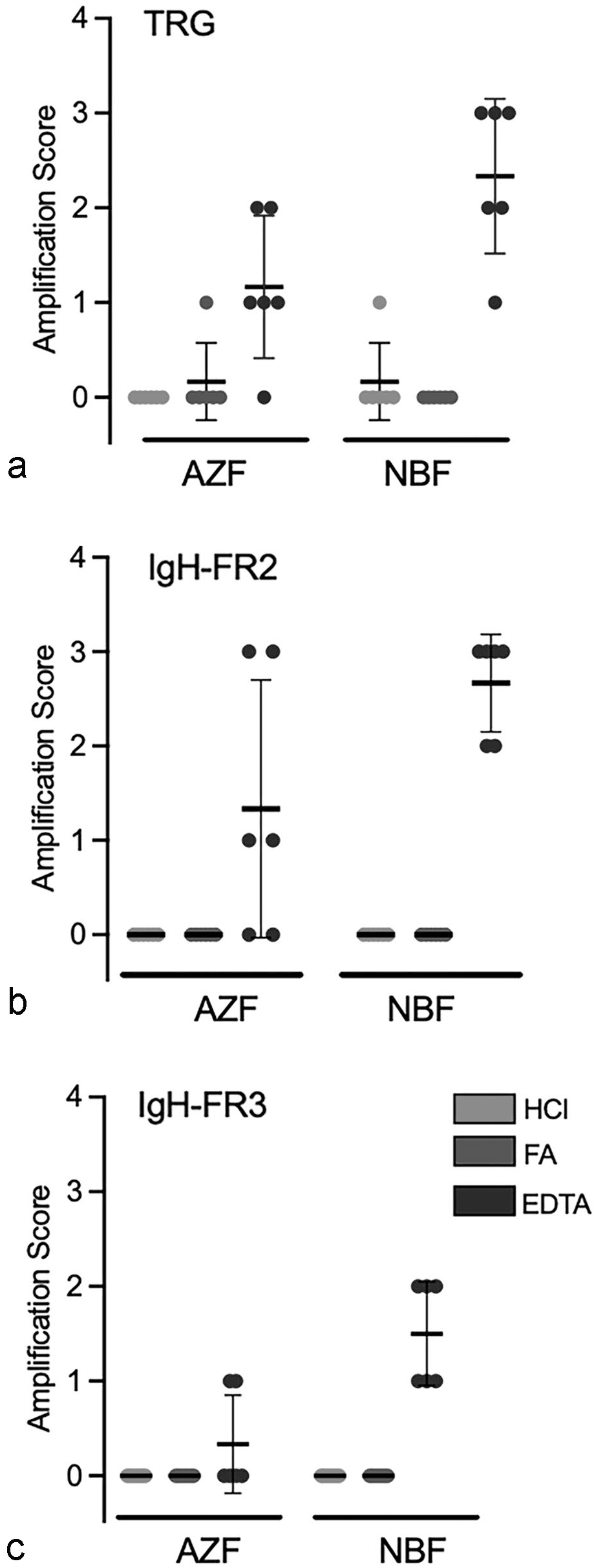
Amplification scores for bone marrow samples fixed with acid-zinc-formalin (AZF) or neutral-buffered formalin (NBF), decalcified with hydrochloric acid (HCl), formic acid (FA), or EDTA, and amplified with primers targeting (a) conserved regions of the T cell receptor gamma (TRG) and (b, c) the immunoglobulin heavy chain (IgH) genes. (b) IgH framework 2, FR2. (c) IgH framework 3, FR3. Amplification scores of samples decalcified with EDTA were significantly higher than those of samples decalcified with HCl or FA (P < .001).
Results of DNA amplification after treatment of bone marrow sections with 3 different decalcifiers.
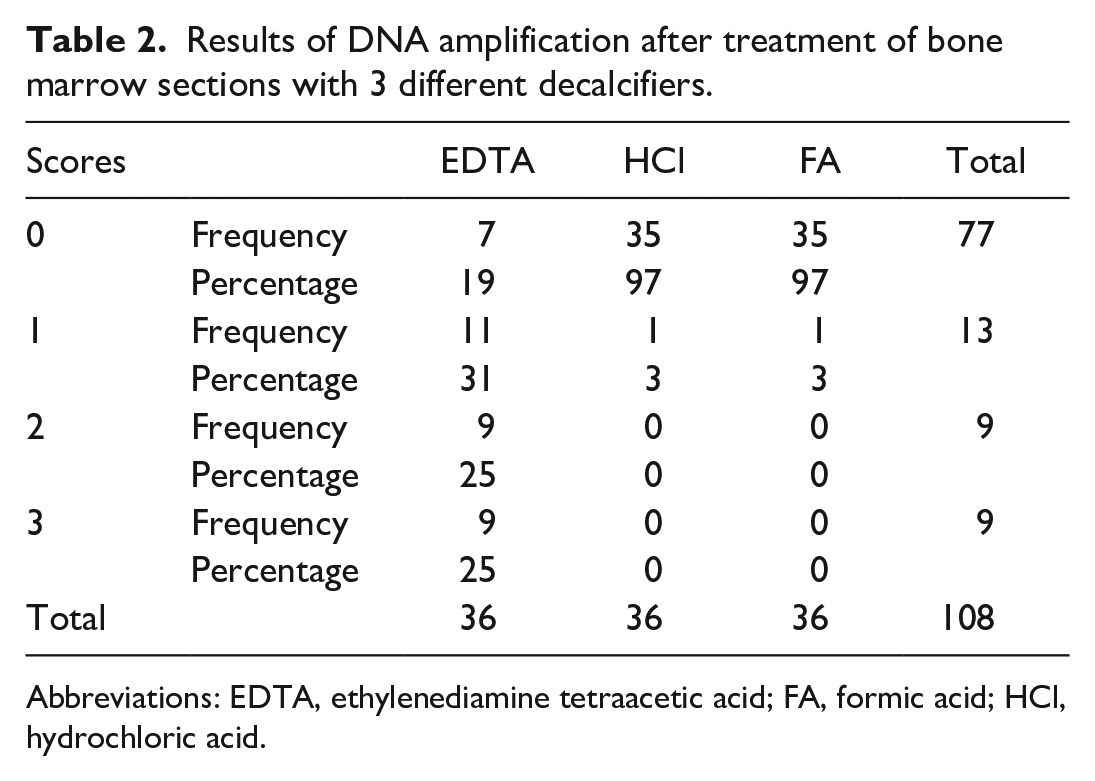
Abbreviations: EDTA, ethylenediamine tetraacetic acid; FA, formic acid; HCl, hydrochloric acid.
When amplification scores were transformed to binomial data for different fixatives and decalcifiers, and a logistic regression analysis was applied, there was a significant difference in the probability of obtaining any amplification (score 1–3) relative to none (score 0) between NBF and AZF. Including summative results for all decalcifiers, samples fixed with NBF had a probability of 21.83% (CI = 3.99, 65.23) to yield an amplification product, whereas samples fixed with AZF had a probability of 1.62% (CI = 0.12, 18.73) to yield an amplification product (P = .024). Samples decalcified with EDTA had a probability of 91.36% to result in DNA amplification, which was significantly more frequent (P < .001) than for samples decalcified with HCl or FA. The likelihood of amplification after either HCl or FA demineralization was the same (0.54%; P = 1.000; Supplemental Table S5).
Discussion
This study aimed to assess the effect of different fixation and demineralization methods on histomorphology and PCR-based DNA amplification. There was no significant histomorphologic difference between AZF and NBF. Decalcification with EDTA yielded higher histomorphology scores and profoundly higher amplification scores than acid-based decalcification. These results provide a guide for laboratories to optimize processing of BM samples.
The sternum was considered the most appropriate bone for sampling due to its elongate and approximately parallel-sided cuboidal shape, which allowed immobilization of individual sternebrae in the miter box and sawing of narrow adjoining replicate slices with limited risk to the operator or tissue. The method was suitable for obtaining 6 sequential slices from dogs as small as 5 kg of weight. The sternum consists of trabecular bone and marrow, is amenable for aspiration, and is readily accessible at postmortem examination. 9
Dogs with multiple conditions were included in this study to encompass variable ratios of hematopoietic to adipose tissue, and variable trabecular thicknesses. Although not measured, trabecular width was subjectively noted to be variable across samples, which likely reflects relative osteopenia in older animals. The TAD did not significantly affect the scores of samples fixed or decalcified by different methods. As the limit for sampling was set at a maximum of 24 hours after death, and carcasses were refrigerated until postmortem, it was unsurprising that there was no measurable effect within that time span. BM is a relatively stable tissue after death, with interpretations reported on samples collected beyond 140 hours postmortem. 18
AZF was hypothesized to yield sections with superior histomorphology and antigen preservation relative to NBF based on prior studies in other species. 23 However, here AZF and NBF yielded similar histomorphology scores, but NBF-fixation was more likely to allow PCR amplification than AZF fixation. Therefore, based on findings here, NBF is a suitable fixative for both histomorphology and subsequent PCR. The effects of AZF vs NBF with respect to immunohistochemical assays remain to be determined. Of note, samples in this study were handled following a standardized protocol with consistent incubation times, tissue movement during fixation (shaker), and incubation temperatures. Such standardized protocols may not be commonplace in laboratories and may have contributed to uniformly high-quality sections with relatively subtle effects of a particular fixative.
Demineralization with acids or chelating solutions removes calcium and other salts from bone while minimally altering hematopoietic and other soft tissue. Here, decalcification with EDTA yielded significantly higher histomorphology scores than either HCl or FA, and sections decalcified with FA had higher scores than those decalcified with HCl. Hydrochloric acid is a “strong” mineral acid with pH ≤0.8, whereas FA is considered a “weak” acid and typically has a pH of 2. 19 Decalcification with HCl requires 1 hour for tissues of a size similar to core biopsies, which is an attractive feature for facilitating an efficient laboratory workflow. Comparatively, decalcification with FA requires overnight incubation and EDTA requires 24-hour incubation. The efficiency of EDTA to chelate minerals depends on the pH of the solution, and replenishment with fresh EDTA of pH 7.0 should be performed after 12 hours. The effect of the decalcification method was most pronounced on PCR. Only 3% of samples fixed with AZF or NBF, and decalcified with either HCl or FA, generated any amplification (score of 1), whereas 81% of samples fixed with either AZF or NBF, and decalcified with EDTA, generated amplification scores of ≥1.
The PCR assays employed in this study are in use for investigating the presence of lymphocytes with clonally rearranged antigen receptor genes in dogs and are performed routinely on many different tissues, including marrow. 15 Adjoining BM samples were evaluated to minimize variations in the number of lymphocytes between samples. The targeted amplicons are of relatively small size, and fixation with formalin only moderately affected amplification efficiency. 15 The effect of different decalcification methods for these particular PCR assays has not been previously examined; however, starkly adverse effects of HCl and FA relative to EDTA decalcification have also been reported by others assessing human tissues. 16 In light of findings in this study, BM tissue that may require PCR-based tests subsequent to histologic evaluation should be decalcified with EDTA rather than acids.
There are limitations to this study. Postmortem rather than in vivo collected samples were analyzed. Analyzing adjoining sternal slices standardized sample composition; however, cores are not typically obtained in vivo from the sternum of dogs due to the limited size of individual sternebrae and the proximity to the thoracic cavity. Collecting 6 replicate core biopsies from the humerus or ilium from live dogs was not feasible; hence, relatively fresh postmortem sternal samples were used to maximize reproducibility. Different protocols for DNA amplification were not tested; therefore, the results should be considered applicable only with the described protocols. Nevertheless, amplicon length is a more significant determinant than primer sequence for PCR of decalcified tissue. 16 The effect of the different fixation and decalcification methods on IHC remains to be evaluated. The duration necessary for EDTA-based decalcification may potentially be shorter than the period employed in this study and should be further investigated to optimize workflow efficiency relative to sample quality. In particular, core biopsies from older dogs tend to have thinner trabecular bone, which is more readily decalcified than cortical bone. Ultrasonic treatment as a means to decrease the duration of EDTA decalcification has been described and may have potential for improved workflow. 6 Analysis of BM samples with clonally rearranged lymphocyte antigen receptor genes might yield different results than those from dogs without clonal rearrangements.
Conclusions
The formalin-based fixatives AZF and NBF yielded comparable BM histomorphology scores, but decalcification with EDTA resulted in better BM histomorphology scores than decalcification with FA or HCl. Amplification of DNA was more likely for NBF- rather than AZF-fixed samples, and much more likely for samples decalcified with EDTA rather than acids. Therefore, decalcification with EDTA should be employed for BM samples that may be further analyzed by PCR.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241257920 – Supplemental material for Effects of fixation and demineralization on histomorphology and DNA amplification of canine bone marrow
Supplemental material, sj-pdf-1-vet-10.1177_03009858241257920 for Effects of fixation and demineralization on histomorphology and DNA amplification of canine bone marrow by Gabriella M. L. Diamantino, Janet Beeler-Marfisi, Robert A. Foster, William Sears, Alice Defarges, William Vernau and Dorothee Bienzle in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff of the histopathology laboratory of the Animal Health Laboratory, University of Guelph, for their interest and skill in optimizing processing methods of bone marrow. We also thank David Marom and Kristy Harmon for their expert technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding for this research was from the Pet Trust Foundation, University of Guelph and the Natural Sciences and Engineering Research Council of Canada. GMLD received an OVC PhD Scholarship for doctoral studies.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
