Abstract
The objective of this study was to evaluate the effect of different fixatives and demineralizers on immunohistochemical (IHC) detection of antigens in bone marrow (BM). Sternal BM samples were collected within 24 hours of death from dogs with spontaneous disease, fixed with acetic acid-zinc-formalin (AZF) or 10% neutral-buffered formalin (NBF) for 24 hours, and decalcified with formic acid, hydrochloric acid, or ethylenediaminetetraacetic acid (EDTA) for 1, 12, or 24 hours, respectively. Immunohistochemical sections for CD3, CD20, CD34, CD204, FLT3, and factor VIII–related antigen (FVIII-rAG) were scored for background, signal intensity, and percent positive cells by 4 independent raters. Some endothelial but not hematopoietic cells were CD34 immunopositive, precluding further assessment. Immunopositive BM cells ranged from 1.3% to 18.3% (CD3), 1.3% to 22.3% (CD20), 0.5% to 22.5% (CD204), and 0% to 17.6% (FLT3). Background scores were similar for all antigens and fixatives except for CD3, for which NBF yielded lower scores than AZF. The signal intensity for all IHC assays was similar for AZF and NBF. The percentage of FLT3- and FVIII-rAG-immunopositive cells was slightly higher in AZF- than NBF-fixed samples. Different demineralizers yielded similar results for all IHC assays except higher background for CD20/EDTA. Signal intensity was higher for CD204 in samples demineralized with EDTA rather than acids. Higher percentages of CD204 and FLT3 immunopositive cells were noted with EDTA relative to acid demineralization. Overall, with the use of stringently standardized pre-analytic and processing protocols, all IHC assays yielded acceptable results, and differences between protocols were considered minor.
Keywords
Bone marrow (BM) is assessed to investigate hematologic abnormalities, and suspected infection or neoplasia of hematopoietic tissue. Since BM is comprised of soft hematopoietic tissue and mineralized bone, fixation and demineralization are required prior to paraffin-embedding and sectioning of the tissue. The methods for processing BM usually reflect established preferences, a desired turn-around-time, and perceived suitability for subsequent immunohistochemical and nucleic acid analyses. While recommendations are in place for standard processing of human BM samples for immunohistochemistry (IHC), there is variability in implementation.13,41,42,50 A universal approach for fixation and decalcification of BM samples in dogs remains to be established.
The types of fixative and demineralizer (also called decalcifier since the majority of mineral in bone is calcium) are considered important for IHC since they can affect antigen preservation. Although neutral-buffered formalin (NBF) is most widely used, other fixatives such as acetic acid-zinc-formalin (AZF), Bouin’s, and mercury-containing fixatives, including Zenker’s and B5 solution, are also used.2,19,26 Formaldehyde preserves tissues through the formation of adducts such as methylol, Schiff base, and 4-imidazolidinone, and cross-links via methylene bridges, which averts degradation of biomolecules.37,39 The addition of zinc to formalin reduces cross-linkage and denaturation of proteins relative to NBF, and reduces hydrolysis in the presence of weak acids. 49 As a result, AZF may accelerate decalcification without adversely affecting tissue structures or antigen preservation. 26
Bone contains hydroxyapatite composed primarily of insoluble calcium and phosphorus salts. Thus, samples containing trabecular or cortical bone need to be demineralized. 6 Standard demineralizing solutions are organic acids such as formic acid (FA), inorganic acids such as hydrochloric acid (HCl), and chelating agents such as ethylenediaminetetraacetic acid (EDTA). Hydrochloric acid results in rapid decalcification but can lead to tissue deterioration and loss of antigenicity. 22 Formic acid removes minerals more slowly but is considered to better preserve antigenicity compared with HCl. 24 Relative to either acid, EDTA requires more time to remove minerals but is superior for preserving antigenicity and nucleic acid integrity.4,5,9,34 Complete and even decalcification is essential for sectioning BM samples since retained mineralized areas preclude artifact-free sectioning. 50
Fixatives and demineralizers can cross-link proteins, which may create steric hindrance for antibody-antigen interaction. In turn, this can affect IHC results and potentially subsequent molecular analyses. Immunohistochemistry is used to identify specific cells or structures in tissue samples and, in BM, may be used to identify lymphoid cells, to distinguish T cells from B cells, to identify epithelial cells, to characterize histiocytes, and to detect organisms with tropism for hematopoietic tissue.1,30,35,41,46 Variability in BM trephine biopsy procurement, preparation, processing, and interpretation has contributed to variability in diagnosis and classification of hematopoietic diseases in humans. 15 To address the need for standardization, the International Council for Standardization in Hematology formed a Working Party to develop guidelines of best practices for “Standardization of Bone Marrow Specimens and Reports.” 19 Despite these guidelines, differences persist in fixative and decalcifier, incubation time, temperature, tissue agitation, and end-point assessment.38,41
Immunohistochemistry is a cornerstone for characterizing hematopoietic neoplasms in humans, and also for determining eligibility for targeted therapies to block growth factor receptors or for immune checkpoint inhibitors. 12 Immunohistochemistry for CD3, CD20, PAX-5, CD79a, CD204, and IBA-1 has been used to characterize cells in canine BM.44,45 CD3 is specific for T cells, while CD20, PAX-5, and CD79a are expressed in B cells. CD34 is an adhesion molecule on endothelial precursor cells, some mature endothelial cells, hematopoietic stem cells, and hemangiosarcoma cells. 14 Measurement of BM microvessel density with CD34 IHC was prognostically useful in human myeloid neoplasms. 40 CD204 is a scavenger receptor typically expressed by histiocytes and has been suggested to label functionally distinct subsets of tumor-associated macrophages in canine malignant mammary tumors and oral melanomas.27,29 Fms-like tyrosine kinase 3 (FLT3, CD135) is a receptor that is mutated and overexpressed in canine leukemias, mammary carcinomas, and mast cell tumors, and internal tandem duplications in FLT3 are associated with poor outcome in humans with acute myeloid leukemia (AML).20,32,43,44 Factor VIII–related antigen (FVIII-rAG), previously termed von Willebrand factor, is expressed by endothelial cells, megakaryocytes, megakaryocyte precursors, and platelets; is present in plasma; and functions in primary hemostasis. Detection of FVIII-rAG has been used to characterize megakaryocytic leukemia in dogs.7,28 In circulation, FVIII-rAG stabilizes factor VIII, and following endothelial injury, factor VIII is activated, dissociates from FVIII-rAG, and contributes to the intrinsic coagulation pathway. 36
In this study, we compared the effects of different fixation and demineralization methods on IHC of canine BM. We hypothesized that fixation with AZF and decalcification with EDTA would yield superior results compared with NBF and acid decalcification, respectively.
Materials and Methods
Samples
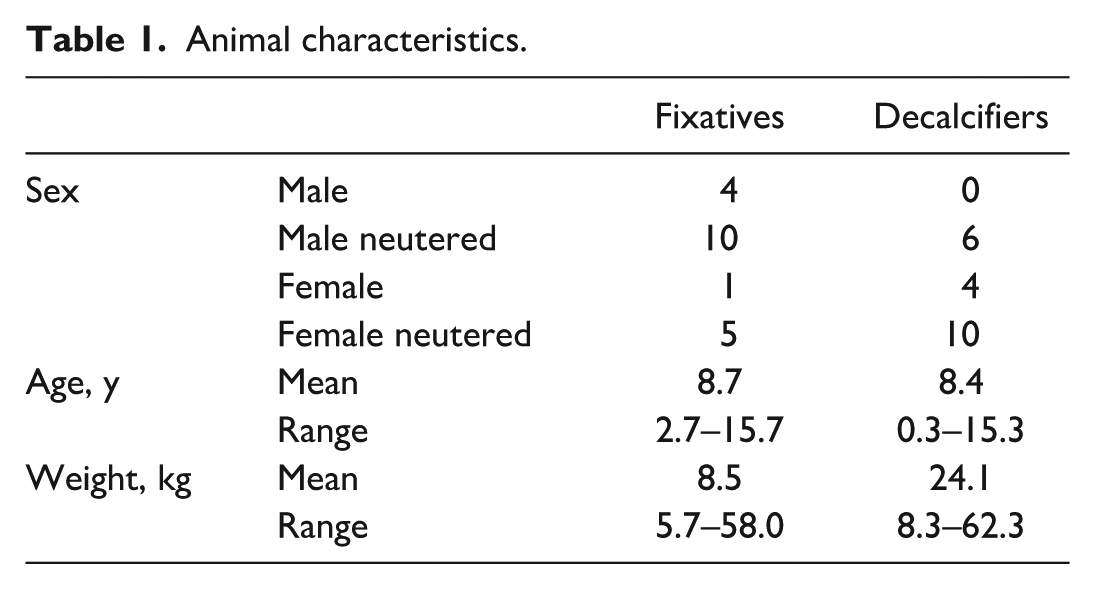
Samples were collected from 40 dogs that had either been euthanized (n = 33) or died (n = 7) due to spontaneous disease and were submitted for postmortem examination. The inclusion criteria were weight ≥5 kg and initiation of the autopsy within 24 hours of death. The dogs ranged in age from 0.3 to 15.7 years and weighed between 5.7 and 62.3 kg (Table 1). None of the animals were diagnosed with primary hematopoietic disease or lymphoma. Sample collection was exempted from animal use approval by the Animal Care Committee at the University of Guelph since BM assessment is a standard component of an autopsy. Duplicate BM samples from 20 dogs were fixed with AZF and NBF and decalcified with FA. Triplicate samples from another 20 dogs were fixed with AZF, and decalcified with HCl, FA, or EDTA. In total, 100 slides from 40 animals were assessed by 4 raters. Samples were collected from the sternum and placed in a miter box to saw adjoining transverse slices 0.2 to 0.3 cm in thickness, as previously described. 9
Animal characteristics.
Fixation and Demineralization
The samples were fixed overnight in freshly prepared AZF or 10% NBF (Leica Biosystems, Concord, ON, Canada) and demineralized with FA (Cal-Ex II Fixative/Decalcifier, Fisher Scientific, Ottawa, ON, Canada; 10.6% FA, 7.4% formaldehyde, 1% methyl alcohol demineralization solution), HCl (SurgiPath Decalcifier II, Leica; 7% HCl), or freshly prepared 14% EDTA solution for 1, 12, or 24 hours at room temperature on a shaker, respectively, as previously described. 9 The samples were then rinsed with tap water for 30 seconds, re-placed into the original fixative, routinely processed by dehydration, embedded in paraffin, sectioned at 2 to 3 µm, and stained with hematoxylin and eosin (Animal Health Laboratory, University of Guelph).
IHC Assays
Immunohistochemistry was performed with an automated instrument (Autostainer; DAKO/Agilent, Mississauga, ON, Canada) on sections that were incubated at 60°C for 12 to 18 hours, deparaffinized, hydrated in a sequence of isopropanol alcohols ranging from 100% to 70%, and then placed in deionized water. 10 Endogenous peroxidase activity was quenched (Dual Endogenous Enzyme Block; DAKO) for 15 minutes at room temperature. Antigen retrieval was performed by placing slides in citrate buffer (pH 6) in a decloaker (Biocare Medical, Pacheco, California) at 95°C or by enzymatic antigen retrieval (Proteinase K; DAKO). Slides were left to cool to room temperature for 20 minutes, and nonspecific binding was blocked by incubation with a serum-free solution containing 0.25% casein and 0.015 mol/L sodium azide in saline. Previously titrated antibodies to CD3, CD20, CD34, CD204, FLT3, and FVIII-rAG were then applied at room temperature (Table 2). Thereafter, slides were rinsed, incubated with anti-mouse or anti-rabbit peroxidase-labeled polymer (EnVision+ HRP; DAKO) for 30 minutes at room temperature, and rinsed again. Bound antibodies were visualized by exposure to NovaRed (Vector Laboratory, Burlingame, California) chromogen, and slides were counterstained with hematoxylin. All IHC runs were performed in batches, and each batch included multi-organ canine positive control sections, and negative control sections with omission of the primary antibody.
Immunohistochemistry parameters for canine bone marrow.

Abbreviations: na, not applicable; HIER, heat-induced epitope retrieval at 95°C and pH 6; FVIII-rAG, factor VIII–related antigen.
Microscopic Assessment
The IHC slides were evaluated by 4 assessors who were blinded in regard to sample identity, fixation, and demineralization. Immunohistochemical slides were scored for nonspecific background labeling (0 = none/faint; 1 = mild; 2 = moderate; 3 = marked); signal intensity (0 = none/faint; 1 = mild; 2 = moderate; 3 = marked); and percent positive cells (averaged from 5 fields viewed at 400× magnification; 0.237 mm2). Background labeling was identified as brownish color in areas devoid of hematopoietic cells or associated with adipocytes, whereas specific labeling was associated with hematopoietic cells. For FVIII-rAG, the proportion of immunopositive megakaryocytes (identified as large cells with multi-lobulated nuclei) among all megakaryocytes was recorded.
Statistical Analysis
Data were analyzed with Statistical Analysis Software (SAS, version 9.4; Cary, North Carolina), and differences were considered significant at P < .05. To examine the effect of fixative and decalcifier on the IHC signal and nonspecific labeling, in a Friedman’s randomized complete block design, animals were considered “blocks.” Before conducting Friedman’s test, the scores among raters were averaged, and for decalcifiers, pairwise comparisons were conducted. PROC FREQ was used to conduct the analyses. For signal intensity and nonspecific labeling, weighted multi-rater Fleiss kappa (SAS MAGREE) and pairwise kappa (PROC FREQ) tests between raters were performed for each treatment (fixative and decalcifiers) and each IHC assay. The kappa tests included tests of bias that evaluated whether raters tended to disagree more in one direction than another direction (a test of symmetry).
The proportion of immunopositive cells was treated as binomial-like data because the data were estimated proportions and were constrained to be between 0% and 100%. A logit transform was applied with a bias correction of 0.25 using PROC MIXED. Bias correction reduces bias and accommodates responses of 0% or 100%. 47 Residual analysis was conducted to evaluate the assumptions of analysis of variance. This involved a formal assessment of residuals for normality using Shapiro-Wilk, Kolmogorov-Smirnov, Cramer-von-Mises, and Anderson-Darling tests. Residuals were plotted against the model’s predicted values and other variables to identify outliers, unequal variances, or other parameters. The responses were modeled with a fixed effect of treatment (fixative or decalcifier) and random effects of the rater, treatment*rater, animal, rater*animal, and treatment*sample. Approximate 95% confidence intervals for parameter estimates were generated. The raw data from this study are available by contacting the corresponding author.
Results
Samples
Bone marrow samples were obtained from dogs with a range of conditions that resulted in euthanasia or spontaneous death (Supplemental Table S1). Twenty-two dogs had cancer with hemangiosarcoma as the most frequent diagnosis (n = 4), and 7 had infectious diseases.
Tissue Characteristics of IHC Results
The mean proportion of immunopositive cells for all fixatives and decalcifier sections combined ranged from 1.3% to 18.3% for CD3, 1.3% to 22.3% for CD20, 0.5% to 22.5% for CD204, and 0% to 17.6% for FLT3 (Fig. 1). CD3 and CD20 immunopositive cells were scattered individually, and round or round-angular in shape (Fig. 2a, b). Cells immunolabeled for CD3 were generally smaller and rounder than CD20 immunolabeled cells, and CD3 immunopositivity was noted throughout the cytoplasm and along the membrane. Immunolabeling for CD20 was most intense along the cell membrane and extended faintly into the cytoplasm. Only endothelial cells of small vessels in some sections, but not hematopoietic cells, were immunopositive for CD34 (Fig. 2c), which precluded analysis of CD34 IHC. Cells positive for CD204 typically had an irregular or elongated shape with cytoplasmic extensions that intertwined among hematopoietic cells, and intermediate size nuclei with open chromatin (Fig. 2d). The cells were immunopositive throughout the cytoplasm and along the cytoplasmic membrane. Cells positive for CD204 were most often located amidst clusters of maturing granulocytic and erythrocytic cells (Fig. 2d). Macrophages containing hemosiderin were inconsistently CD204 positive. Cells positive for FLT3 were scattered individually, irregularly shaped, and had large nuclei with up to 3-fold size variation. Strongest immunopositivity was along the cytoplasmic membrane or in cytoplasmic extensions interspersed among hematopoietic cells (Fig. 2e). A variable proportion of megakaryocytes and platelet aggregates (present in some sections), and the luminal aspect of endothelial cells, were immunopositive for FVIII-rAG (Fig. 2f). Immunopositivity was granular, variably intense, and sometimes included the entire cytoplasm and sometimes just the cytoplasmic membrane. Some megakaryocytes recognizable by size and morphology lacked FVIII-rAG immunoreactivity in samples prepared with either fixative or decalcifier.
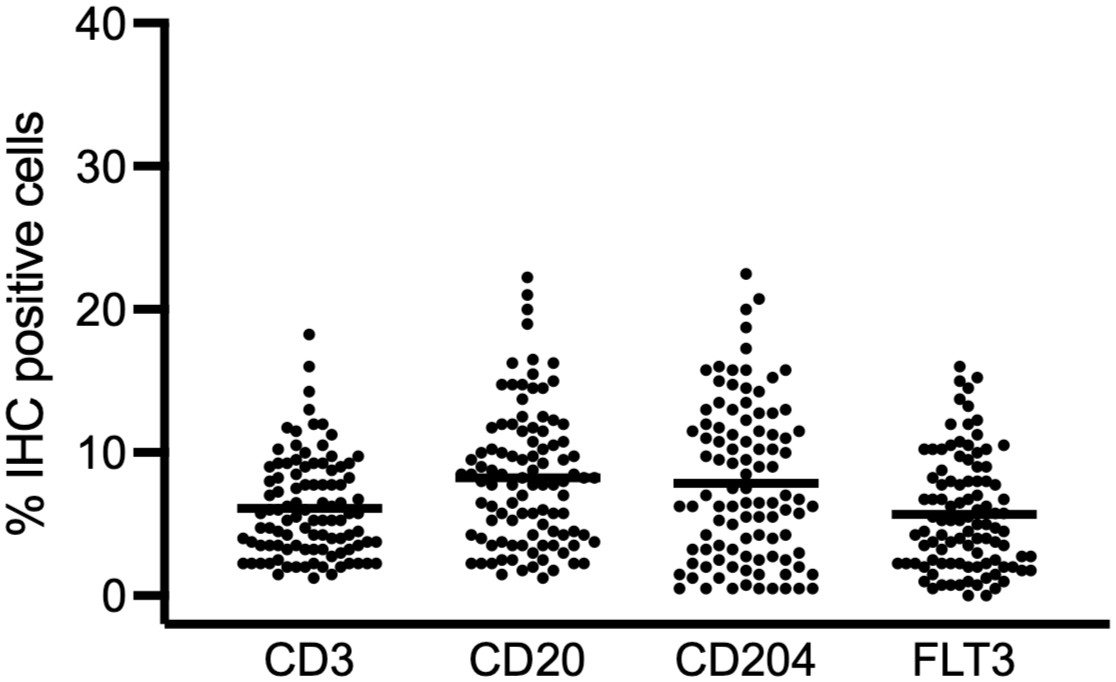
Combined results of the percent of cells that were immunohistochemically (IHC) positive for CD3, CD20, CD204, and FLT3 in canine bone marrow fixed with either acid-zinc-formalin or neutral-buffered formalin, and demineralized with either hydrochloric acid, formic acid, or EDTA (n = 100). The graph represents all results combined with the mean indicated by the line.
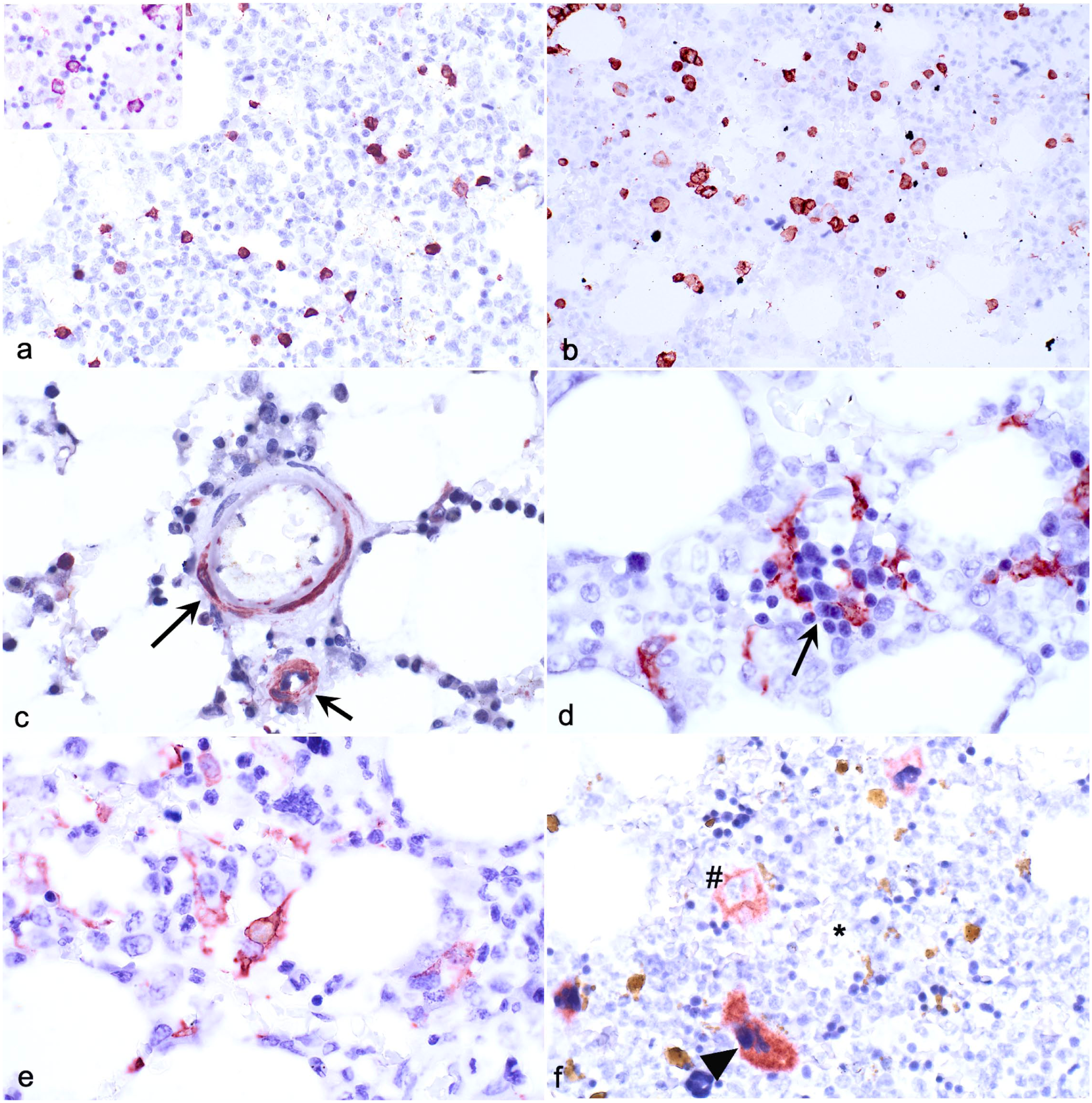
Immunohistochemical assays applied to canine bone marrow fixed with acid-zinc-formalin and demineralized with formic acid. Case 78. (a) CD3 immunopositive cells are small, round, and individually dispersed, and immunoreactivity is throughout the cytoplasm and along the cell membrane. Inset (upper left): higher magnification. (b) CD20 immunopositive cells are larger than CD3 cells and are individually dispersed. Immunoreactivity is concentrated along the cell membrane. (c) Vascular endothelial cells are immunopositive for CD34 (arrows). (d) CD204 immunopositive cells are irregularly shaped and often interdigitate among rubricytes of different maturational stages (arrow) or immature granulocytes. Immunopositivity was throughout the cytoplasm and along the cytoplasmic membrane. (e) FLT3 immunopositive cells have variable shapes and sizes, frequent cytoplasmic extensions, and are individually dispersed. (f) Immunopositivity for factor VIII–related antigen varies in intensity and distribution. Some megakaryocytes are intensely immunopositive throughout the cytoplasm (arrowhead), some are faintly immunopositive along the cytoplasmic membrane (#), and some are immunonegative (*).
Effect of Fixatives on IHC Assessment
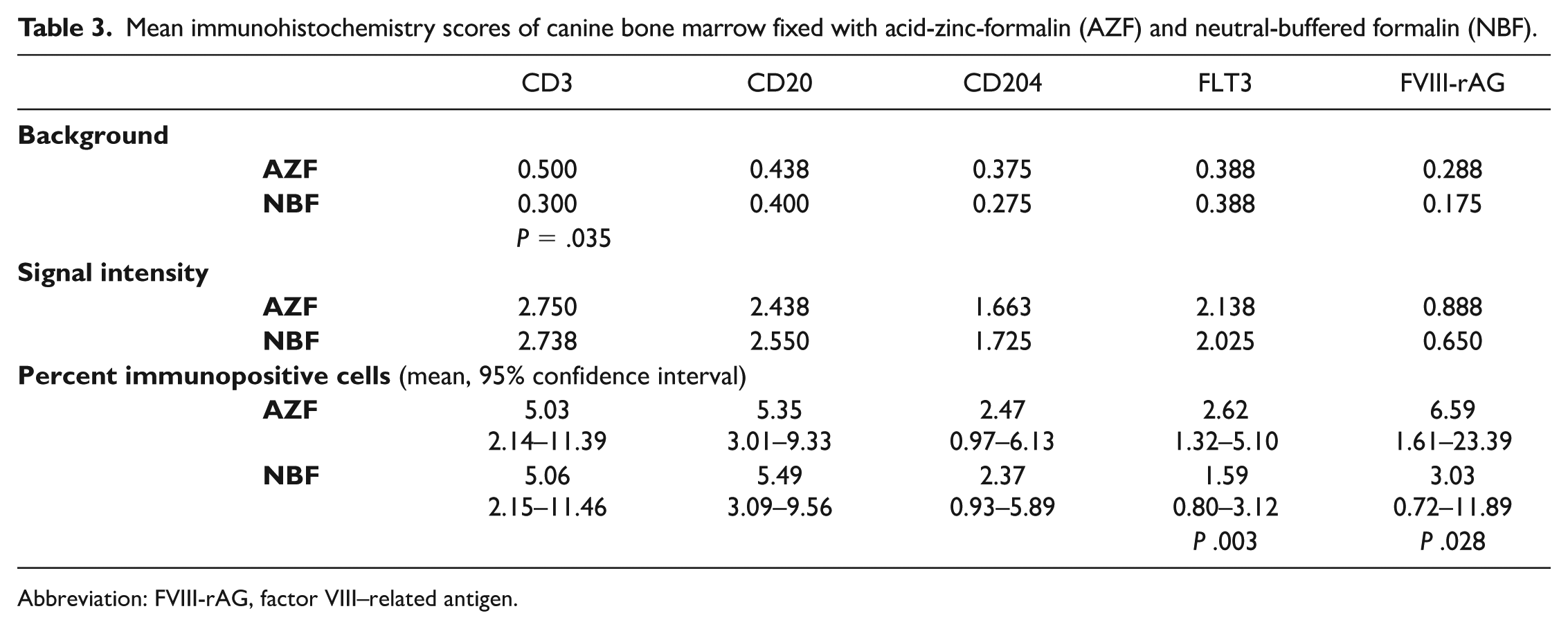
Duplicate BM samples were fixed with NBF and AZF and decalcified with FA. Background scores were lower for NBF relative to AZF for CD3 (P = .035; Table 3) and did not differ significantly between fixatives for CD20 (P = .248), CD204 (P = .206), FLT3 (P = .739), and FVIII-rAG (P = .206, Fig. 3). Bias was detected for some combinations of antigen and fixative. Weighted kappa values ranged from 0.041 to 0.101, indicating slight agreement among raters for fixatives and IHC markers (Supplemental Table S2). 17 Weighted kappa values could not be calculated for FLT3/AZF and FVIII-rAG /AZF due to multiple “0” scores.
Mean immunohistochemistry scores of canine bone marrow fixed with acid-zinc-formalin (AZF) and neutral-buffered formalin (NBF).
Abbreviation: FVIII-rAG, factor VIII–related antigen.
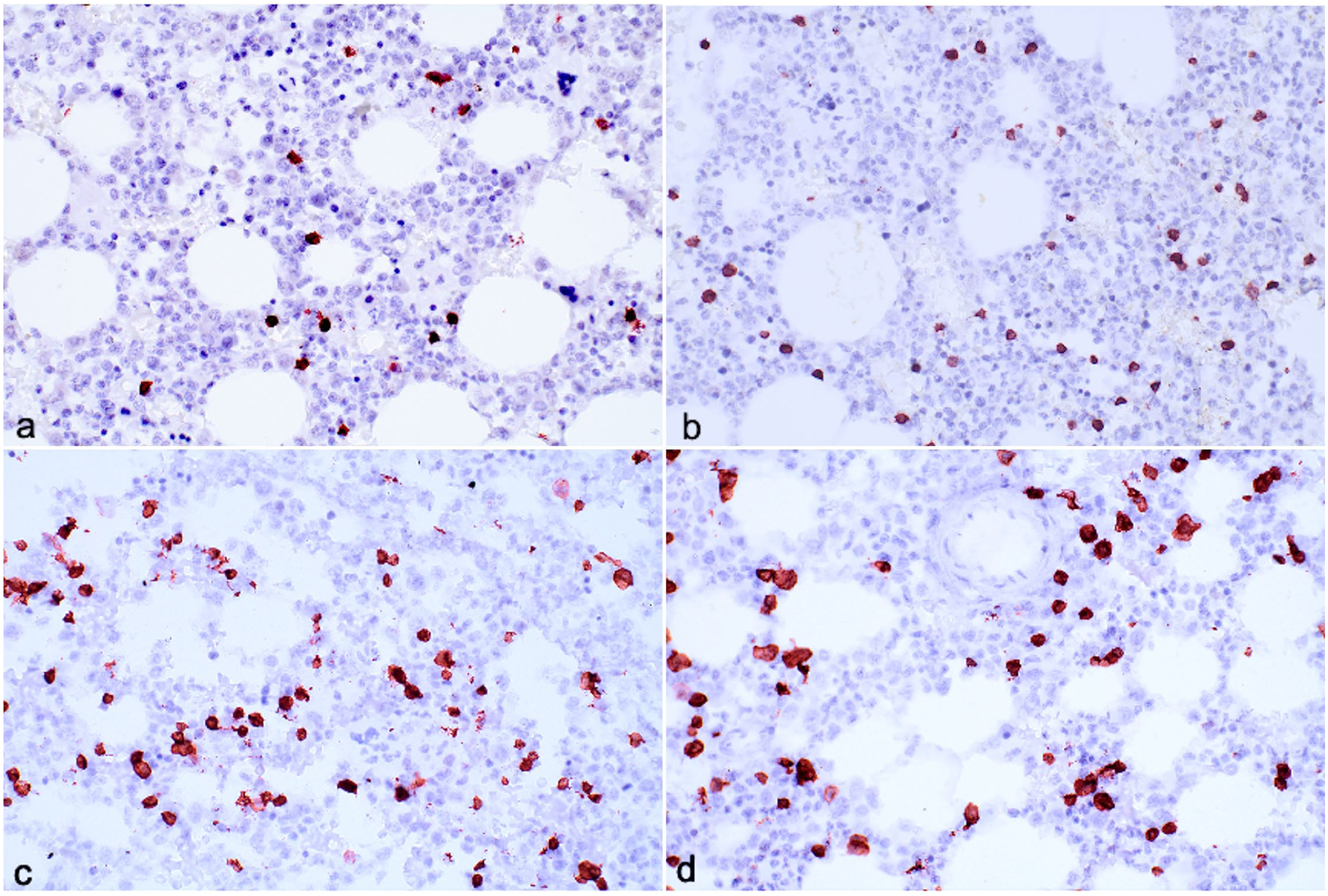
Immunohistochemical assays applied to canine bone marrow fixed with acid-zinc-formalin (AZF) (a, c) or neutral-buffered formalin (NBF) (b, d) and demineralized with formic acid. Case 4. (a, b) Fixation with AZF yields higher (0.58) CD3 immunohistochemistry background scores than fixation with NBF (0.45). (c, d) Background scores do not differ significantly between AZF (0.43) and NBF (0.42) for CD20.
The scores for IHC signal intensity did not differ between AZF and NBF for any marker: CD3 (P = .480), CD20 (P = .109), CD204 (P = .617), FLT3 (P = .564), and FVIII-rAG (P = .109, Table 3). Some bias among raters was detected, and weighted kappa values ranged from 0.016 to 0.175 (Supplemental Table S3). 17
Logistic regression analysis showed no differences in the percentage of immunolabeled cells in samples fixed with AZF or NBF for CD3 (P = .940), CD20 (P = .773), and CD204 (P = .673, Table 3). The percentage of FLT3- and FVIII-rAG immunolabeled cells was higher for AZF than NBF (P = .003 and P = .028, respectively), resulting in the odds of identifying FLT3- and FVIII-rAG immunopositive cells being 67 and 126% higher in tissues fixed with AZF relative to NBF.
For analysis of covariance, random sources of variation with P < .250 and residual covariant parameters were retained. The residual analysis for all markers revealed unequal variance, which needed to be accommodated based on the Akaike information criterion. Raters (CD3, CD20) and individual animals (CD204, FLT3, FVIII-rAG) contributed most variation (Supplemental Tables S4–S8).
Effect of Decalcifiers on IHC Assessment
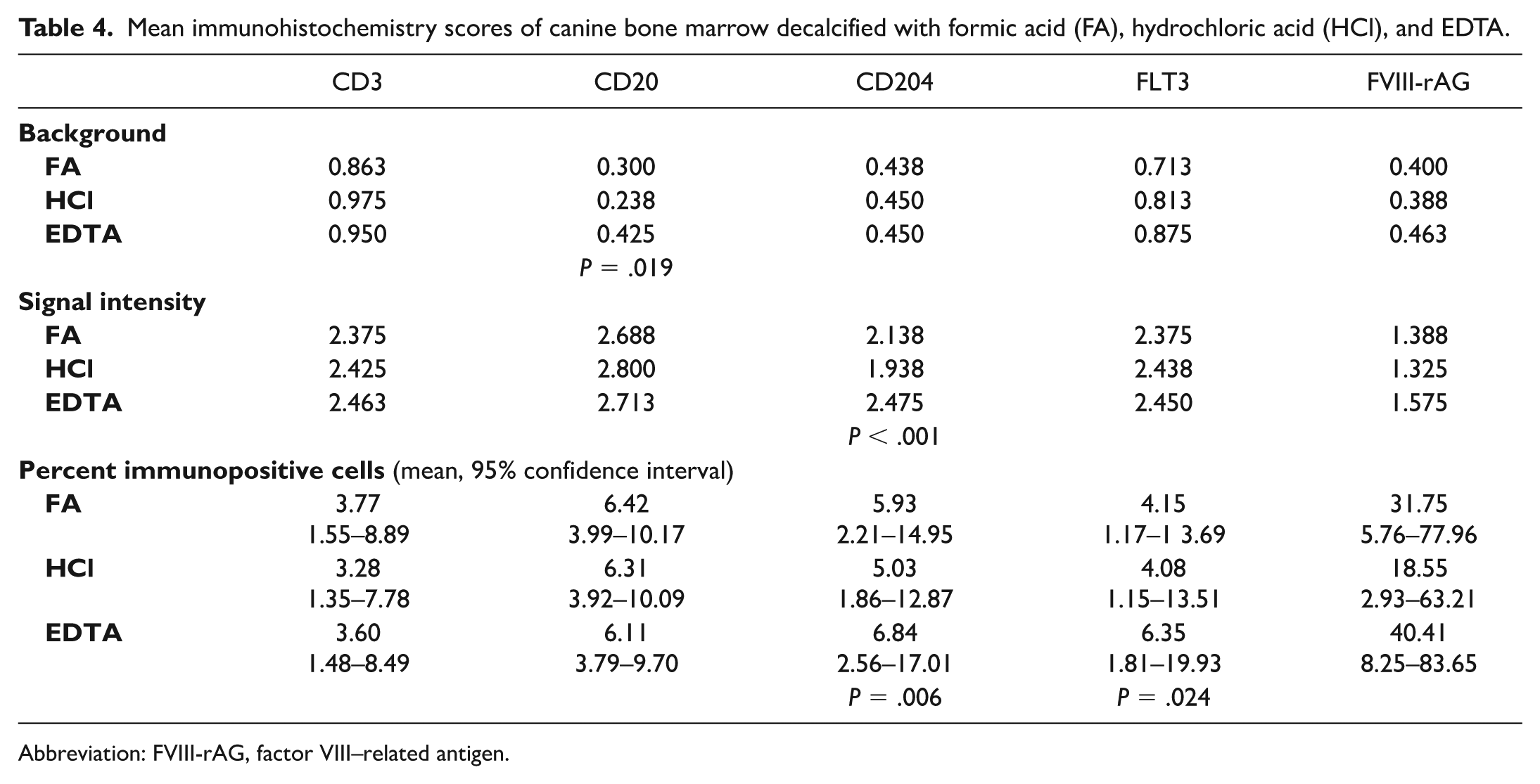
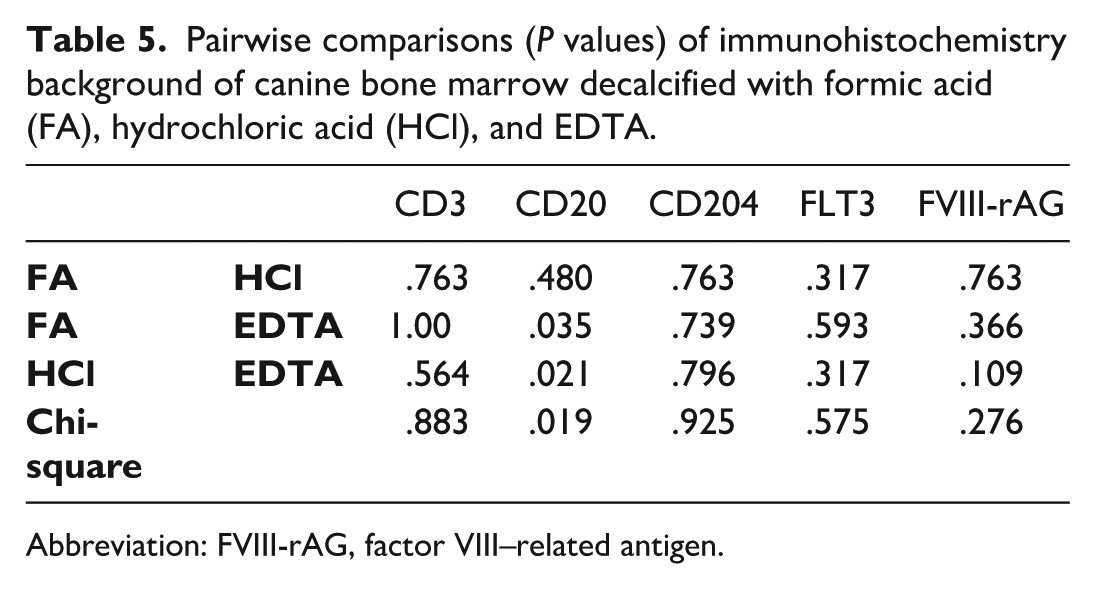
Triplicate BM samples were fixed with AZF, and then decalcified with FA, HCl, and EDTA. Decalcification of BM with FA, HCl, or EDTA yielded similar background scores for CD3 (P = .883), CD204 (P = .925), FLT3 (P = .575), and FVIII-rAG (P = 0.276, Table 4). Scores for CD20 differed between decalcifiers (P = .019) with highest background scores for EDTA (Table 4, Fig. 4). Pairwise comparisons showed that the main differences were between FA and EDTA (P = .035) and HCl and EDTA (P = .021, Table 5). There was bias among raters for several IHC markers and decalcifiers, and weighted kappa values ranged from 0.001 to 0.228 (Supplemental Table S9).
Mean immunohistochemistry scores of canine bone marrow decalcified with formic acid (FA), hydrochloric acid (HCl), and EDTA.
Abbreviation: FVIII-rAG, factor VIII–related antigen.
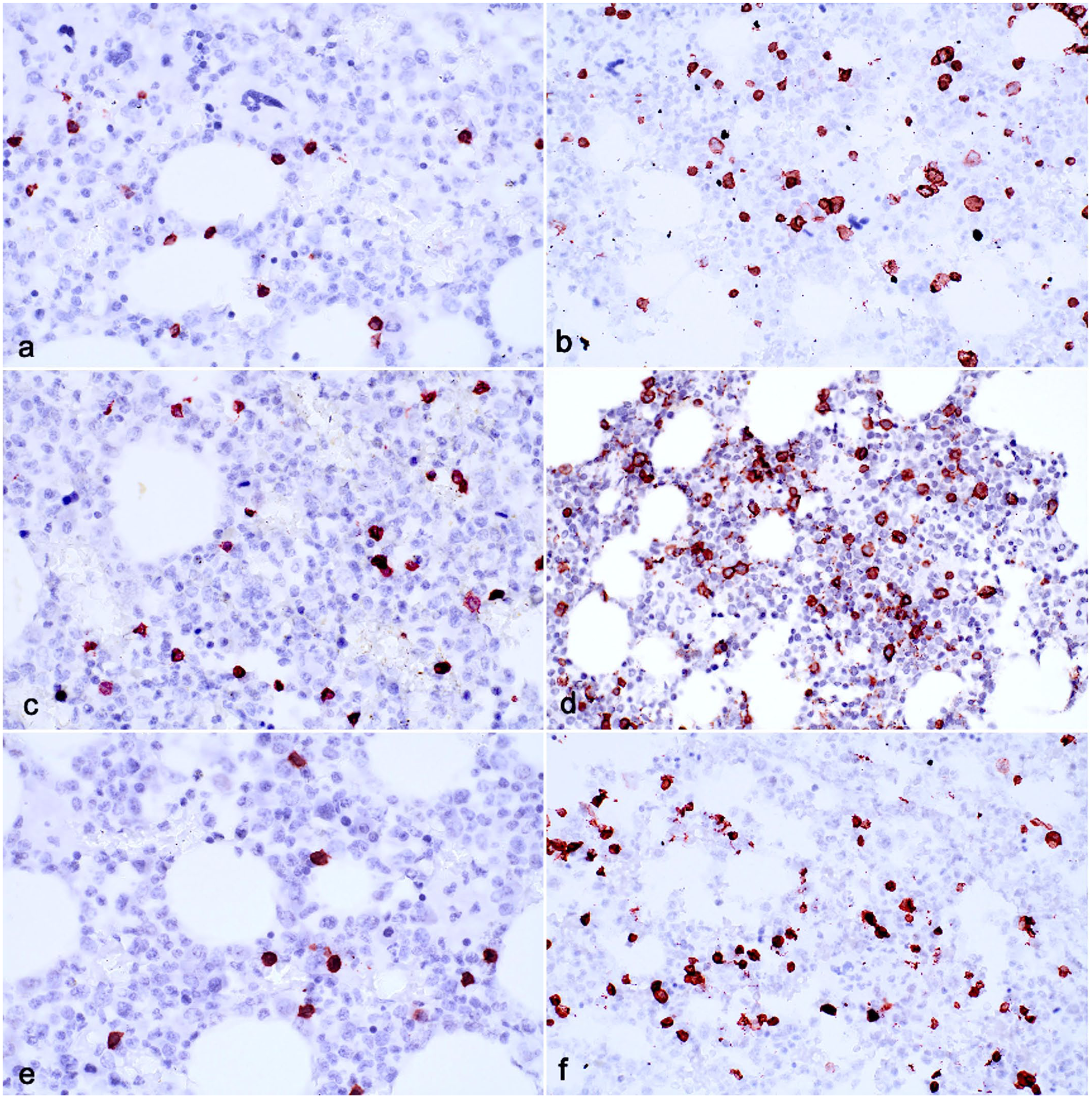
Immunohistochemical assays applied to canine bone marrow fixed with acid-zinc-formalin and decalcified with formic acid (a, b), hydrochloric acid (c, d), and EDTA (e, f). Case 13. Scores for background are similar for CD3 (a = 0.88, c = 0.90, e = 0.92, respectively) but for CD20, formic acid and hydrochloric acid decalcification yielded lower background scores than EDTA (b = 0.31, d = 0.25, f = 0.41).
Pairwise comparisons (P values) of immunohistochemistry background of canine bone marrow decalcified with formic acid (FA), hydrochloric acid (HCl), and EDTA.
Abbreviation: FVIII-rAG, factor VIII–related antigen.
Signal intensity did not differ between decalcifiers for CD3 (P = .892), CD20 (P = .507), FLT3 (P = .274), and FVIII-rAG (P = .278) but differed for CD204 (P <.001) where EDTA yielded higher scores (Table 4). For CD204, each pairwise comparison of decalcifiers indicated differences (Table 6). Bias was noted among raters for several IHC marker—decalcifier combinations (Supplemental Table S10). Weighted kappa values ranged from 0.103 to 0.269. 17 Logistic regression analysis showed no significant difference in signal intensity for CD3 (P = .253), CD20 (P = .809), and FVIII-rAG (P = .211).
Pairwise comparisons (P values) of immunohistochemistry signal intensity of canine bone marrow decalcified with formic acid (FA), hydrochloric acid (HCl), and EDTA.
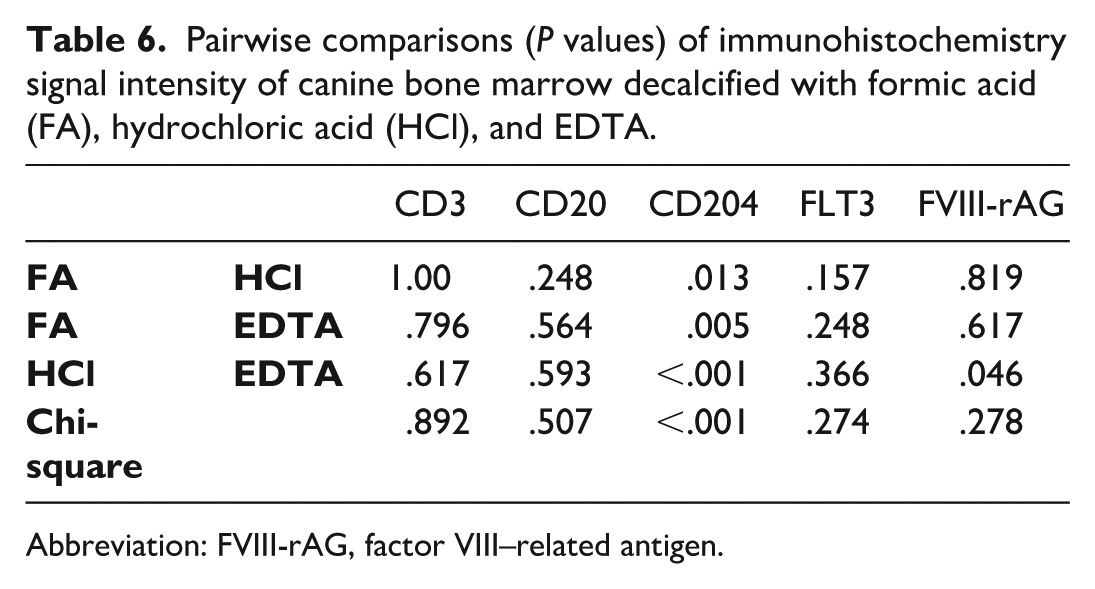
Abbreviation: FVIII-rAG, factor VIII–related antigen.
The percentage of CD204 and FLT3 immunopositive cells was higher in sections decalcified with EDTA relative to FA or HCl (P = .006 and P = .024, respectively; Table 4). The odds of identifying CD204 immunopositive cells in EDTA-decalcified sections were 39% higher than the odds in HCl-decalcified sections (Supplemental Table S11), and the odds of FLT3 immunopositive cells were 46% higher for EDTA- relative to HCl-decalcified sections (Supplemental Table S12).
The residual analysis for all IHC markers showed unequal variance, which was adjusted according to the Akaike information criterion. Rater was the main source of variation for CD3, CD204, and FLT3, while individual animal was the main source of variation for CD20 and FVIII-rAG (Supplemental Tables S13–S17).
Discussion
Fixatives vary in their effect on IHC, and BM demineralization with acids or EDTA has the potential to further alter antigen-antibody interactions.2,16,23 In this study, the effect of 2 commonly used fixatives and three decalcifiers on background immunolabeling, signal intensity, and percentage of positive cells was assessed by IHC with antibodies suitable for characterizing hematopoietic tissue. Unique strengths of this study were that all 40 samples originated from dogs with spontaneous disease, that they were processed in a standardized manner, and that they were assessed by four independent raters.
Antibodies reactive with lymphocytes were tested since it is frequently important to differentiate lymphoid from nonlymphoid hematopoietic cells in BM, and the percentage of IHC-identified lymphocytes in marrow of adult dogs with non-hematolymphoid disease has not previously been reported. A similar proportion of CD3 and CD20 immunopositive lymphocytes was identified, and the cells were present individually with infrequent clustering. This information may be helpful for distinguishing BM infiltration in lymphoma from the pattern and frequency of BM lymphocytes in dogs with other spontaneous diseases. 31 The functional properties of B and T cells in the BM of dogs without hematolymphoid disease are unknown. CD34 was included since it can be useful to diagnose acute leukemia. 11 However, only vascular endothelial cells were immunopositive, suggesting that BM from dogs without acute leukemia does not contain detectable CD34 immunopositive hematopoietic cells. An IHC assay for CD34 may nevertheless be useful to evaluate marrow vascularity. 40 Cells positive for CD204 are considered to be histiocytes, which include macrophages and dendritic cells (DCs) that are not all equally immunopositive for CD204. 8 Bone marrow is a common site for involvement by histiocytic sarcoma in dogs, and knowledge of the frequency of CD204 positive cells in non-neoplastic BM may be helpful to improve the detection of histiocytic sarcoma. 25 Cells positive for CD204 were detected in each sample, and most were elongated cells with long processes and relatively small nuclei, reminiscent of DCs. Marrow DCs are considered to be a unique subtype whose role in immunity and neoplasia is increasingly understood.21,33 Detection of FLT3 by IHC in canine BM has not previously been reported but since FLT3 is mutated and/or overexpressed in human and canine leukemia, IHC detection is likely to be useful for comprehensive assessment of BM biopsies. 44 FLT3 is expressed in normal human BM by stem cells and lineage-committed early precursors and is more highly expressed in leukemic cells. 43 In canine BM, cells positive for FLT3 generally had large nuclei, and scant cytoplasm, and included cells with cytoplasmic protrusions. Although the identity of the hematopoietic cells that normally express FLT3 is not well-described, consistent with a role in signal transduction, immunopositivity was concentrated along the cell membrane. An IHC assay for FVIII-rAG was included since detection of FVIII-rAG can be helpful to identify morphologically atypical megakaryocytes and their precursors. Immunopositive cells included immature megakaryocytes with rounded or hypolobulated nuclei and mature megakaryocytes with hyperlobulated nuclei. Granular immunoreactivity in the cytoplasm but not cell membrane was typical. Immunolabeling of megakaryocytes was often sparse or absent, and in some sections concurrently immunolabeled and unlabeled megakaryocytes were noted. While immunolabeling of intravascular platelet aggregates and the luminal surface of mid-size vessels was consistent with the distribution of FVIII-rAG, it is unclear why megakaryocyte labeling was variable. The finding may relate to intermittent production of FVIII-rAG, similar as reported in humans, and additional markers such as CD42b and CD61 are required to identify all megakaryocytes. 16
Immunohistochemical results varied slightly by fixative and antibody. Background was similar for all markers, except CD3 IHC yielded slightly lower scores with NBF than AZF fixation. The signal intensity and the percentage of positive cells were similar for both fixatives, except a slightly higher number of FLT3-positive cells and FVIII-rAG-positive megakaryocytes was noted with AZF fixation. The difference in positive cells was likely a type I error where an effect is detected that is not really present (false positive). Analysis showed some bias among raters. Bias in microscopic assessment may arise from variable tissue response to fixation and demineralization, the size of tissue available, the choice of area to be examined, the individual perception and interpretation of signal and background intensity, and other sources. 18 It was attempted to reduce bias in this study by standardized tissue processing, assessment of all samples by 4 independent raters, and prior calibration of assessment using 10% of all sections. Other approaches to reduce bias could consider automated rather than manual image analysis, and 3-dimensional rather than 2-dimensional tissue investigation. Limitations to such approaches are the variable size of sections of BM available from small relative to large dogs; the need for an initial, usually manual, annotation algorithm to exclude non-hematopoietic tissue (bone, fat, intravascular space) from analysis; and the need to distinguish the different colors of iron-containing pigments from antibody-linked chromogens. Such manipulations were considered to introduce more subjectivity than visual scoring. 3
The decalcification method had no effect on IHC background or signal intensity except that the acidic decalcifications yielded less background than EDTA for CD20 IHC, and that EDTA decalcification yielded a better signal than acids for CD204 IHC. The differences in both variables were small. The proportion of CD204- and FLT3-positive cells was slighter higher in sections decalcified with EDTA than with acids, which was also considered to reflect a likely type I error.
Veterinary pathology archives contain mostly paraffin blocks with tissues initially fixed in NBF. Decalcification methods do not appear to be homogeneous across laboratories though FA appears to be more commonly used for BM core biopsies than EDTA or HCl, while samples with abundant bone are more often decalcified with HCl. 48 The duration of fixation and decalcification, agitation, replenishment of decalcifier, and other factors can also influence IHC.2,16,23 Results from this study suggest that tissues containing bone; fixed in either AZF or NBF; and decalcified with FA, HCl, or EDTA may be comparably suitable for IHC if standardized protocols are employed. 23
Several IHC assays described here were novel for use in canine BM and may be considered for routine panels. Our results do not allow for an unequivocal recommendation of a universally optimal IHC protocol. There were subtle differences between fixatives, decalcifiers, and antibodies, which suggests that optimized protocols should be adjusted for each antibody. The previous finding that AZF fixation and EDTA decalcification yielded superior histomorphology and profoundly better DNA PCR amplification compared with NBF, FA, and HCl, respectively, suggests that the combination of AZF and EDTA is likely the best overall yet still practical combination. 9 Outcomes for BM samples collected at autopsy may not be readily transferable to in vivo collected biopsies since the latter are generally of much smaller size, and therefore more quickly fixed and decalcified.
Other limitations in this study are that different fixation and decalcification times were not assessed though they are likely important variables for IHC. While all IHC assays were initially assessed for an optimal antigen retrieval method and antibody incubation time, IHC platforms vary in their accommodation of different chromogens, signal amplification methods, and incubation times. Furthermore, not all possible combinations of fixation and decalcification methods were assessed since 6 adjoining, thick, transverse slices could not be obtained from the sternebrae of smaller dogs. Hence, it is possible that, for example, NBF fixation in combination with certain decalcifiers may yield better IHC results than AZF fixation. Prior evidence regarding the effect of specific fixation and decalcification methods on IHC is limited; therefore, while findings from this study offer some guidance on IHC for BM, optimization may require adjustments by individual laboratories.
Conclusions
For canine autopsy-collected BM samples, the formalin-based fixatives AZF and NBF yielded comparable IHC results for most markers. Overall, decalcification with EDTA resulted in higher IHC scores than decalcification with FA or HCl. Differences between variables were modest, and various combinations of fixative and decalcifier may be expected to yield useful IHC results.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858261426547 – Supplemental material for Effects of fixation and demineralization on immunohistochemical assessment of canine bone marrow
Supplemental material, sj-pdf-1-vet-10.1177_03009858261426547 for Effects of fixation and demineralization on immunohistochemical assessment of canine bone marrow by Gabriella M. L. Diamantino, Janet Beeler-Marfisi, Robert A. Foster, Gabrielle Monteith, William Sears, Alice Defarges and Dorothee Bienzle in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr. Josepha Delay for expert advice on IHC, and Laura Austin and Erin Specker from the histopathology laboratory (Animal Health Laboratory, University of Guelph) for technical expertise.
Supplemental material for this article is available online.
Author Contributions
GMLD designed and performed the experiments; GMLD, JB-M, RAF, and DB contributed to experimental design and performed microscopic interpretations; GM and WS reviewed the experimental design and performed the statistical analysis; AD contributed to experimental design; DB obtained funding for this study; and GMLD and DB wrote the manuscript with contributions from the other authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Pet Trust Foundation, University of Guelph, and the Natural Sciences and Engineering Research Council of Canada. GMLD is the recipient of a doctoral fellowship from the Ontario Veterinary College.
