Abstract
Although neoplasia has been documented in invertebrates, it has not been reported in scorpions. This report describes presumed hemocytic neoplasia in 2 scorpions: a >3-year-old, female emperor scorpion (Pandinus imperator) and a >4-year-old, male, Asian forest scorpion (Heterometrus sp.). The emperor scorpion had a 1-month history of body wall swelling separating the exoskeleton of the caudal opisthosoma. At necropsy, this corresponded to a white mass in the caudal coelom. The forest scorpion was found dead and processed whole for histology, at which point multiple masses were identified in the coelom and invading skeletal muscle. Histologically, both masses were composed of sheets of hemocytes with round to oval nuclei; eosinophilic, periodic acid Schiff-positive, cytoplasmic granules; mild cellular atypia; and low mitotic rates. Features of inflammation (e.g., melanization and nodulation) were not observed. These masses were diagnosed as a hemocytoma (emperor scorpion) and a hemocytic sarcoma (forest scorpion), possibly of plasmatocyte origin.
Neoplasia has been documented in many invertebrates, most commonly in mollusks and insects.14,20 Reported invertebrate neoplasms include carapacial wall tumors, papillomas, carcinomas, and hemocytic neoplasms.14,20 Hemocytic neoplasms, also called “hemic tumors,” are the most commonly documented neoplasms in invertebrates. The cell of origin is often assumed (not proven) to be hemocytic based on widespread dissemination in the hemolymphatic sinuses and/or the morphologic similarity of neoplastic cells to hemocytes, which are the only immune cell of arachnids. 14
Neoplasia has not been reported in scorpions, and only a single case is reported in arachnids, which was an epithelial oviductal neoplasm in a spider. 12 The lack of reports of neoplasia in scorpions may be due to low incidence in this species, carcass disposal without histologic evaluation, and/or pathologist unfamiliarity with arachnid anatomy and histopathology, including basic tissue reactions to insult, such as inflammation and wound repair. 14 When evaluating invertebrate neoplasms, there are added challenges that may confound accurate diagnoses, including understanding tumor biology, the use of cell-identification tools (e.g., immunohistochemistry), and standardized classification.
Emperor scorpions (Pandinus imperator) are listed in the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) list due to the threat of overexploitation for the pet trade as well as habitat destruction.1,4 Asian forest scorpions (Heterometrus sp.) are common in zoos and as exotic pets but are also harvested for food or medicinal purposes; they are not CITES listed.13,16 The study describes hemocytic neoplasia in two scorpions.
Case 1 was a >3-year-old, multiparous, female, emperor scorpion. Case 2 was a >4-year-old, male, Asian forest scorpion. Both scorpions were housed in public displays for 3–4 years in individual enclosures with carefully monitored temperature and humidity; they had annual veterinary examinations and weekly husbandry checks. Case 1 was fed a weekly diet of adult house crickets (Acheta domesticus) and large locusts (family: Acrididae). Case 2 was fed a weekly diet of dubia roaches (Blaptica dubia), house crickets (Acheta domesticus), and superworms (Zophobas morio). Case 1 had a swelling on the right lateral coelom that separated the tergites (dorsal exoskeleton) from the sternites (ventral exoskeleton) of the caudal opisthosoma (Fig. 1a); the animal was found dead 1 month later. At necropsy, upon removal of the carapace, a well-demarcated, soft, pale tan to white mass measuring approximately 1 cm in diameter was identified in the right lateral coelom that corresponded to the coelomic swelling (Fig. 1b). The mass was pedunculated and adhered to the region where we would expect ovarian tissue to be located (confirmation of this origin was not possible histologically). The midgut diverticula, Malpighian tubules, gonad, book lungs, eyes, gut, and mass were collected and fixed in 10% neutral buffered formalin for ≥24 hours. Case 2 was found dead with no premonitory signs. The carcass was fixed whole without gross examination of internal structures; no external gross lesions were seen. After formalin fixation for ≥24 hours, the whole body was placed in Davidson’s solution for 72 hours, then transverse sections were made through the carcass, and sections were placed in 70% ethanol for 24 hours. In both cases, tissues were processed routinely for histology, sectioned at 5 µm, and stained with hematoxylin and eosin, Gram stain, and periodic acid-Schiff (PAS) reaction. Case 1 was also stained with Ziehl-Neelsen stain, and case 2 was stained with Gomori methenamine silver.
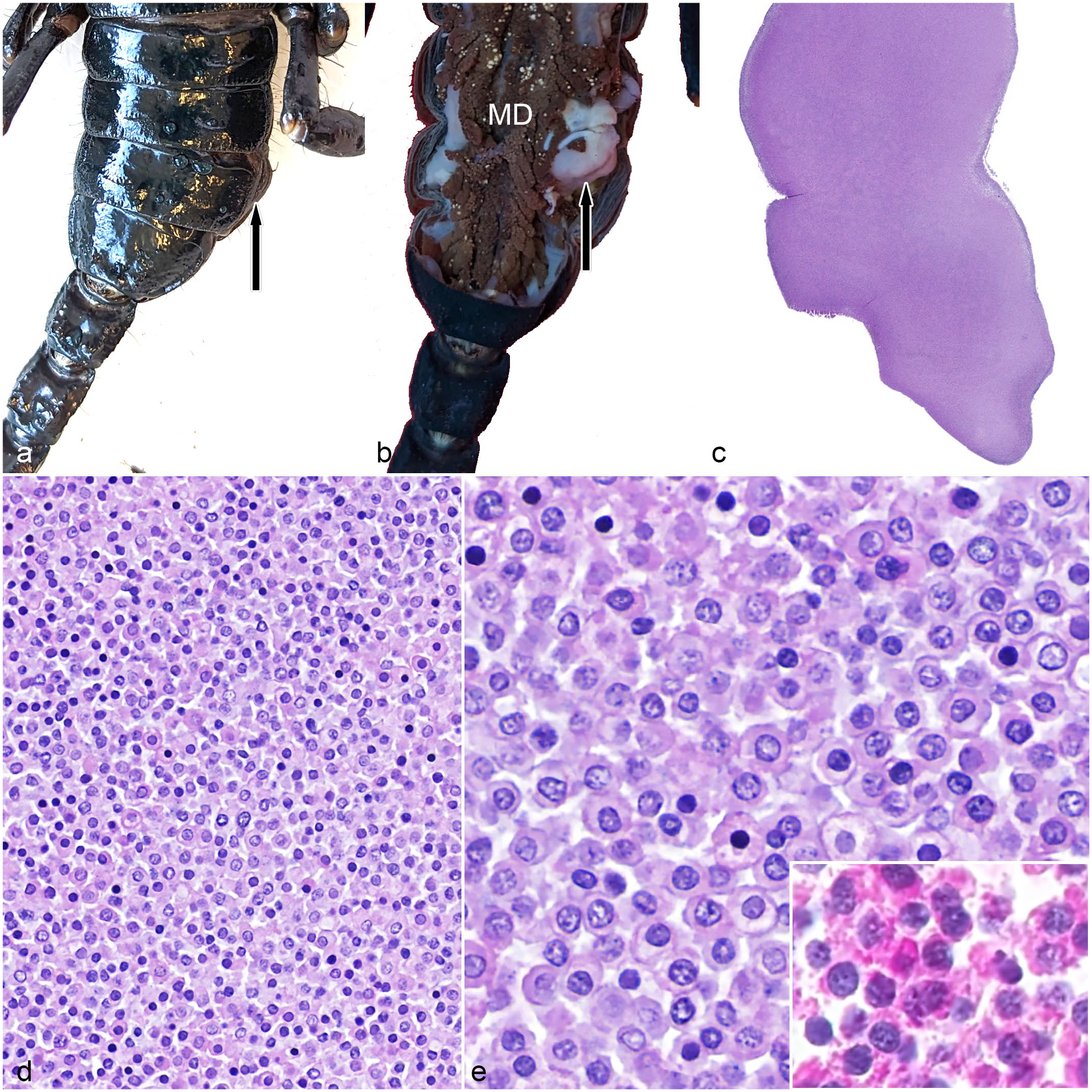
Hemocytic neoplasm in an emperor scorpion (Pandinus imperator). (a) Gross image showing swelling on the right side of the opisthosoma (arrow). (b). Gross image with the carapace removed showing a white mass (arrow) in the opisthosoma coelomic cavity that corresponds to the swelling seen in (a). MD, midgut diverticula. (c) The neoplasm is homogeneous and blue with minimal associated stroma. Hematoxylin and eosin (HE). (d) Neoplastic hemocytes form sheets of densely packed cells. HE. (e) Neoplastic hemocytes are round with a moderate amount of pale eosinophilic cytoplasm that is highly granular on periodic acid-Schiff reaction (inset). Nuclei are round with stippled chromatin. HE.
In case 1, there was a thinly encapsulated pedunculated neoplasm within the caudolateral aspect of the opisthosomal coelom (Fig. 1c–e). In case 2, multiple, discrete, unencapsulated, densely cellular, neoplastic masses with multifocal areas of central coagulative necrosis expanded the dorsal portion of the coelom in the prosoma and in the ventral and midline portion of the opisthosoma and infiltrated the adjacent striated muscle of the body wall (Fig. 2a–c). In both cases, the neoplasms were composed of dense sheets to packets of monomorphic, round to rarely oval, neoplastic hemocytes with minimal associated stroma (Figs. 1d, e and 2b, c). Neoplastic hemocytes ranged from 8 to 15 µm in diameter with distinct cell borders; moderate amounts of eosinophilic, finely granular cytoplasm; round to oval (to reniform in case 2) nuclei with finely stippled to dense chromatin; and indistinct nucleoli. Occasionally, neoplastic cells had many distinct eosinophilic intracytoplasmic granules. Anisocytosis was mild. Anisokaryosis was mild to moderate. Binucleation was rare. The mitotic count was 1 (case 1) and 4 (case 2) per 2.37 mm2. Neither melanization nor inflammation was identified in the neoplasms. In case 2, neoplastic cells had random, individual cell necrosis (i.e., hypereosinophilic cytoplasm and a pyknotic nucleus). Striated muscle invaded by neoplastic cells had multifocal degeneration and necrosis in case 2. In both cases, PAS stain revealed numerous PAS-positive granules within neoplastic hemocytes. Gram (cases 1 and 2), Gomori methenamine silver (case 2), and Ziehl-Neelsen (case 1) stains were negative for infectious agents within the neoplasms. Other histologic findings in both cases included atrophy of midgut diverticula (as evidenced by a paucity of eosinophilic cytoplasmic globules). In addition, case 1 had focal hemocytic inflammation with nodulation in the wall of the oviduct; the cause for nodulation was not identified, and the neoplasm was not seen invading this tissue. Case 2 had rare erosion with melanization of the arthrodial membranes and cuticle and multifocal hemocytic pneumonia with a mixed bacterial biofilm. The remaining organs were histologically unremarkable.
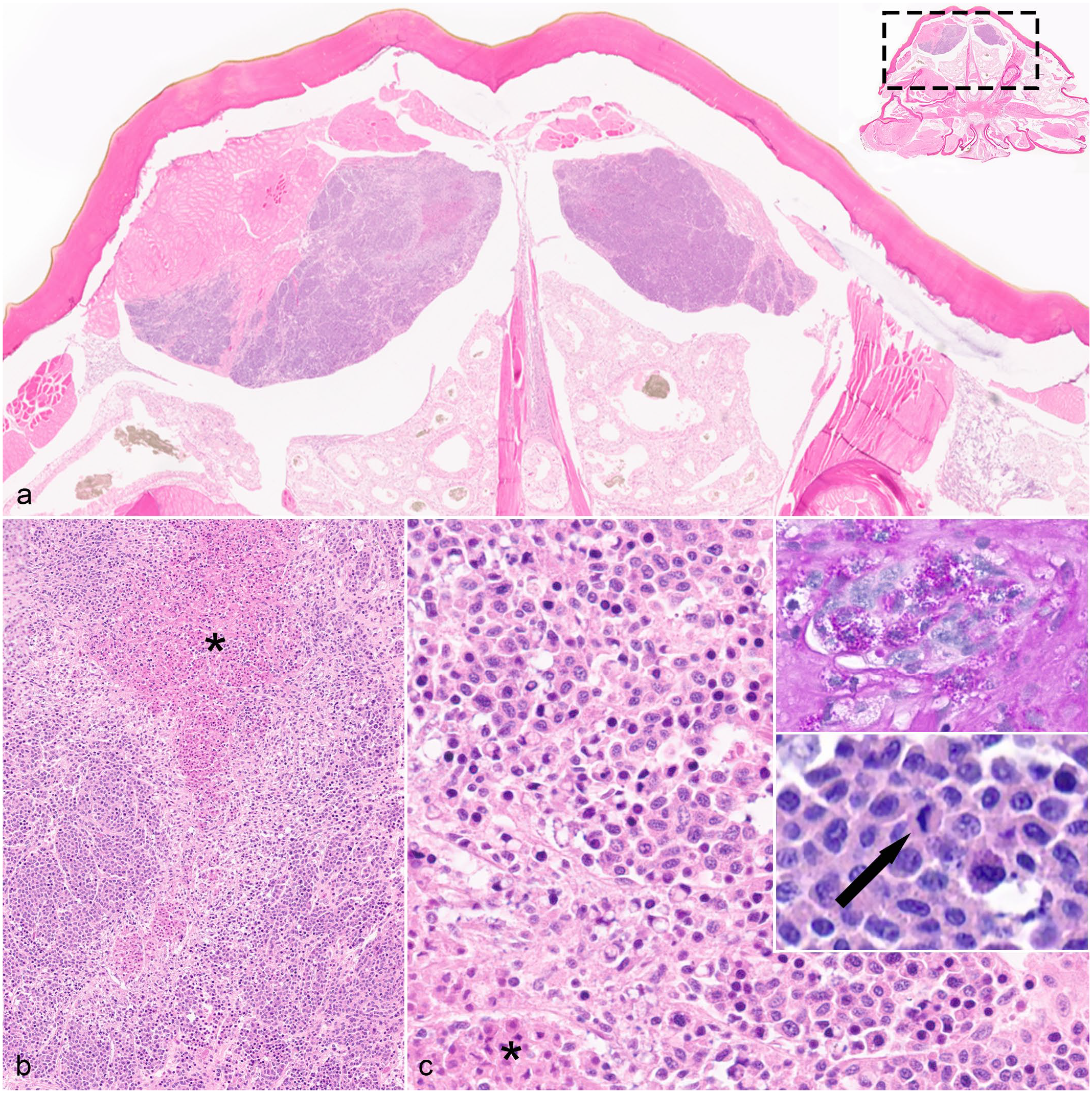
Hemocytic neoplasm in an Asian forest scorpion (Heterometrus sp.). (a) Neoplastic cells form multiple masses in the dorsal aspect of the prosomal coelomic cavity. Neoplastic masses infiltrate skeletal muscle and have central areas of necrosis. Hematoxylin and eosin (HE). Inset: Subgross image of a complete cross-section of the prosoma with dashed rectangle showing where image was taken. (b) Neoplastic cells form sheets to nodules with minimal associated fibrous stroma. There are multifocal regions of coagulative necrosis (asterisk). HE. (c) Neoplastic cells are round to be irregularly shaped with round to reniform nuclei. They have a moderate amount of cytoplasm that occasionally has abundant, deeply eosinophilic granules. There is coagulative necrosis of small foci of neoplastic cells (asterisk). HE. Top inset: Periodic acid-Schiff (PAS) reaction shows numerous, cytoplasmic, PAS-positive granules. Bottom inset: Neoplastic cells have mitotic figures (arrow). HE.
This study describes presumed hemocytic neoplasia in two scorpions. Neoplasia is rarely described in arachnids, with only one case in the literature.12,14,20 In the previously reported arachnid neoplasm, the oviductal neoplasm was composed of palisading and stratified columnar cells; this is inconsistent with the neoplasms reported here, which were composed of sheets of round to spindle cells. 12 In a retrospective study of arachnid pathology, inflammation with infection was the most common histologic lesion, and neoplasia was not identified. 5 Hemocytic neoplasms are one of the most commonly described neoplasms in invertebrates and are reported in multiple species of bivalve mollusks, white shrimp, Drosophila, and a California king crab.2,3,8,10,15,19 In these reports, hemocytic neoplasms formed masses or were disseminated in the hemolymphatic sinuses. The masses had neoplastic cells that were equal to or up to twice as large as circulating hemocytes. Neoplastic hemocytes were round to spindeloid with variable PAS-positive granules, similar to the cells reported here. In most cases of invertebrate hemocytic neoplasia, the cellular origin was assumed based on morphologic similarities to mature hemocytes.8,14
Hemocytes are the immune effector cells of arachnids.6,7 Hemocytes are responsible for phagocytosis, inflammation (capsule formation with either encapsulation or nodulation), synthesis of antimicrobial peptides, and activation of enzymatic cascades to include phenoloxidase activity and melanization. Some studies report four types of hemocytes in scorpions: plasmatocytes, granulocytes, cyanocytes, and prohemocytes; other sources indicate additional types of less numerous hemocytes.7,9,17 Electron microscopy and biochemical analysis are the predominant methods used to differentiate arachnid hemocytes; cytology with light microscopy is less commonly performed.9,17 Detailed histologic characterization of hemocytes has not been performed in scorpions, making it difficult to definitively determine the specific hemocyte cell of origin in these neoplasms. Extrapolating from cytologic reports, neoplastic hemocytes reported here may represent plasmatocytes. Plasmatocytes are the most abundant hemocytes and are round to irregularly shaped, 15–30 µm in diameter cytologically, and have empty or granular cytoplasm. 9 The neoplastic hemocytes reported here were smaller (range from 8 to 15 µm), possibly due to formalin shrinkage and lack of cellular spreading on histology. Granulocytes were another consideration for the origin neoplastic cells but are consistently round and even larger (20–40 µm in diameter cytologically) than the neoplastic cells reported here. Cyanocytes were considered an unlikely source as these are large cells (50 µm in diameter on cytology) without granules. Prohemocytes were considered unlikely as they have scant cytoplasm. 9
Differentiating inflammation from neoplasia is essential to accurately diagnosing hemocytic neoplasia in invertebrates. Key histological features of inflammation in arachnids include hemocytic infiltration, nodulation, and melanization.6,7 Hemocytic infiltration is characterized by aggregates of hemocytes that can be either granulated or nongranulated and, if nodulation occurs, are surrounded by bands of elongated, degranulated hemocytes, similar in histologic appearance to granulomas in vertebrates.6,7 With chronicity, these nodules may develop a core of degranulated hemocytes with central necrosis.6,7 Melanization is part of the innate immune response and may be seen with or without hemocytic inflammation. 6
Both scorpion cases presented here had large sheets of hemocytes with mild cellular atypia and low mitotic activity, but without some classic scorpion features of inflammation, specifically nodulation and melanization. This is consistent with hemocytic neoplasia. The neoplasm in case 1 was most consistent with a benign neoplasm (i.e., hemocytoma), and case 2 was consistent with a malignant neoplasm (i.e., hemocytic sarcoma). Case 2 had features of malignancy that were not present in case 1, notably invasion of the adjacent skeletal muscle, multiple masses (possibly caused by metastasis), and central areas of coagulative necrosis, likely caused by rapid growth of the neoplasm leading to decreased hemolymph perfusion. The neoplasms were the most substantial histologic findings in both cases.
Case 2 had multiple lesions that may have been secondary to the neoplastic masses. This included muscular degeneration and necrosis, which may have been secondary to regional trauma/compression to the muscle from the neoplasm. There was also a mixed bacterial biofilm overlying pulmonary lamellae with hemocytic pneumonia. Pneumonia may have been secondary to immunosuppression, potentially from neoplasia or advanced age. Pneumonia, particularly bacterial pneumonia, is a commonly reported lesion in arachnids and is often believed to occur secondary to systemic disease. 5
Lesions in the midgut diverticula are commonly reported in arachnids and were seen in both cases. 5 Midgut diverticula, also known as digestive ceca, are a complex network of blind-ended branches of the midgut. 11 Atrophy of midgut diverticula has been documented in arachnids during times of decreased nutritional status or starvation. 11 In these cases, midgut diverticular atrophy was considered secondary to neoplasia, likely due to an increased metabolic demand coupled with decreased caloric intake. 18
In conclusion, neoplasia is rarely reported in arachnids.12,14,20 Discerning inflammation from hemocytic neoplasia can be a diagnostic challenge and is critical to accurate diagnoses. These scorpions had presumed hemocytic neoplasia. A major limitation to this study and other reports of invertebrate neoplasia is the lack of available immunohistochemical antibodies to definitively identify the cells of origin.
Footnotes
Acknowledgements
Thank you to Dr. Megan Schreeg (The Ohio State University College of Veterinary Medicine) for her groundbreaking work in arachnid pathology that paved the way for this manuscript. Thanks also to Tyrone Capel and the ectotherm team at ZSL for detailed records and images of case 1. Thank you also to Tatiane Watanabe for her contributions while at North Carolina State University College of Veterinary Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily reflect the official policy of the Department of Defense, or the U.S. Government.
