Abstract
Histopathology of 61 captive American horseshoe crabs (HSCs; Limulus polyphemus) is reviewed. HSC organs evaluated histologically included body wall (chitin, epidermis, dermis, and skeletal muscle), hepatopancreas, gut, gonads, book gills, eyes, heart, brain, and coxal gland. In descending order, lesions were most frequently identified in compound eye, body wall, book gills, hepatopancreas, chitinous gut, nonchitinous gut, heart, and brain; lesions were not observed in coxal gland or gonads. Hemocytes (also called amoebocytes) surrounded infectious agents and occluded ulcers. Large hemocyte aggregates had a central eosinophilic coagulum (ie, hemocyte coagulum). Cutaneous ulceration (34/60 cases), branchitis (29/48 cases), and ophthalmitis (17/20 cases) were common lesions and consistently associated with fungi, which were invasive into subjacent tissues, and/or bacteria, which were usually superficial. Fungal culture was performed in 3 cases and isolated Fusarium spp., although fungal morphology varied and multiple fungal species may have been present. Presumptive green algae were associated with ulceration in 1 case with minimal to no inflammation. Presumptive cyanobacteria were identified within a biofilm overlying the gills in 4 of 48 cases and were not invasive. Multifocal, random hepatopancreatitis was identified in 16 of 57 cases, 10 of which were associated with bacteria. Metacercarial cysts were identified in 25 of 61 cases and associated with minimal to no inflammation. Depleted eosinophilic globules in hepatopancreatic interstitial cells were interpreted as decreased nutritional status in 12 of 57 cases.
American horseshoe crabs (HSCs; Limulus polyphemus) are arthropods representing the only extant member of the genus Limulus. 11 They are “living fossils” and have persisted for more than 200 million years. 11,16 HSCs are marine invertebrates that migrate to deep ocean waters in the winter and move to shallow waters to spawn in the late spring and early summer. Their habitat range is the Atlantic coast of North America. Known sources of mortality in wild populations include collection for biopharmaceutical production, natural predation, commercial harvest for bait, spawning habitat loss, pollution, bycatch, climate change, and collection of specimens for research, education, and aquaria. 11 These animals are used by the biopharmaceutical industry for an enzyme in their hemolymph called limulase amebocyte lysate (LAL), which detects minute quantities of endotoxin. Due to this property, the Food and Drug Administration requires that most pharmaceutical products are tested with LAL prior to market to screen for contamination. LAL is obtained by “bleeding” wild HSCs during spawning season, which incurs a mortality rate of 8% to 15% of males and 10% to 29% of females in wild populations. 1,16
The goal of this study is to describe the histologic findings of HSCs submitted to 3 diagnostic laboratories. In addition, normal histologic descriptions of HSC microanatomy are provided for comparison when necessary. The aim of this publication is to provide pathologists with references to improve their diagnostic acumen and recognize immune/tissue responses when evaluating HSCs histologically. In addition, it is the hope of the authors that publications describing diseases in captive HSCs (such as this) will lead to additional research on the care and management of these animals. Although multiple excellent publications are available on the care of captive HSCs 10,12 and their dissection, 2 histology, 5 and immune responses, 9 additional detailed studies are still needed.
Materials and Methods
Pathology archives of 3 institutions (National Zoo, Washington, DC; Johns Hopkins University, Baltimore, MD; and Northwest ZooPath, Monroe, WA) were searched for HSC cases. Microscopic slides from all cases were retrieved. All slides and necropsy reports were reviewed. To assist in microscopic evaluation of this species, normal histology of HSC was reviewed, 5 and a list of organs for examination was compiled (Table 1).
Organs for Histologic Evaluation in American Horseshoe Crabs (Limulus polyphemus).

Results
Archived tissues from 61 HSCs were examined, including 40 males and 11 females; sex was not determined for 10 cases. All animals were in captivity in zoos or aquaria in Maryland (n = 26); California (n = 12); Washington, DC (n = 9); Nevada (n = 5); Oklahoma (n = 2); Ohio (n = 2); Nebraska (n = 2); Louisiana (n = 1); Texas (n = 1); and an unspecified state (n = 1). The original source location for wild-caught animals was not available. The organs most commonly sampled in decreasing order included the body wall (60/61; 98%), hepatopancreas (57/61; 93%), gonads (51/61; 84%), gills (48/61; 79%), nonchitinous gut (42/61; 69%), chitinous gut (40/61; 66%), heart (20/61; 33%), and compound eye (20/61; 33%); rarely sampled organs include the brain (8/61; 13%) and coxal gland (4/61; 7%). When present in histologic section, the following organs most commonly had lesions: compound eye (17/20; 85%), body wall (50/60; 83%), gills (31/48; 65%), hepatopancreas (35/57; 61%), chitinous gut (20/40; 50%), nonchitinous gut (15/42; 36%), heart (7/20; 35%), and brain (1/8; 13%). No lesions were observed in the coxal gland or gonads. Bacterial (54/61; 88%), fungal (36/61; 59%), and parasitic (25/61; 41%) infections were common. Additional staining characteristics of bacteria were determined in 13 of 54 cases using special stains and revealed 10 cases with gram-negative rods, 4 cases with gram-negative cocci, 2 cases with gram-positive cocci, 1 case with gram-positive rods, and 1 case with acid-fast bacilli; 4 of 13 cases had mixed infections. Detailed histologic findings are available in Supplemental Table S1.
Inflammatory Response and Infectious Agents
The inflammatory cell of HSCs is the hemocyte. 4,9 Some texts refer to these cells as granulocytes or amebocytes. 10 The authors prefer the term hemocyte to denote a circulating immune cell (“-cyte”) in the hemolymph (“hemo-”). Hemocytes had abundant, brightly eosinophilic, cytoplasmic granules when they were in the vasculature (Fig. 1) but varied in the amount of cytoplasmic granules during an inflammatory response. In large inflammatory foci, the margins of the lesions were infiltrated by hemocytes with abundant cytoplasmic granules and round nuclei, while hemocytes closer to the centers of inflammatory foci frequently had few to no evident cytoplasmic granules and occasionally had elongate/oval nuclei. 9 For descriptive purposes, hemocytes with granules were called “granular hemocytes,” and those without granules and/or nuclear changes were called “reactive hemocytes.” Centers of large inflammatory foci were filled with amorphous, pale eosinophilic material (which represents coagulin gel) 9 admixed with a small amount of karyorrhectic debris; for descriptive purposes, this mix of hemocytes, coagulin, and necrotic cellular debris was called a “hemocyte coagulum” (Fig. 2). Rarely in the gills, large inflammatory foci had loose aggregates of plump spindle cells (Fig. 3). Infectious agents and cutaneous wounds were often embedded in and occluded by hemocyte coagula, respectively. Fungi were frequently seen invading from the external body wall or external surface of the gills and associated with a robust inflammatory response. Bacteria were commonly associated with small to moderately sized inflammatory foci randomly scattered throughout internal organs. Metazoan parasites frequently had minimal to no associated inflammation and are described further in each system below.
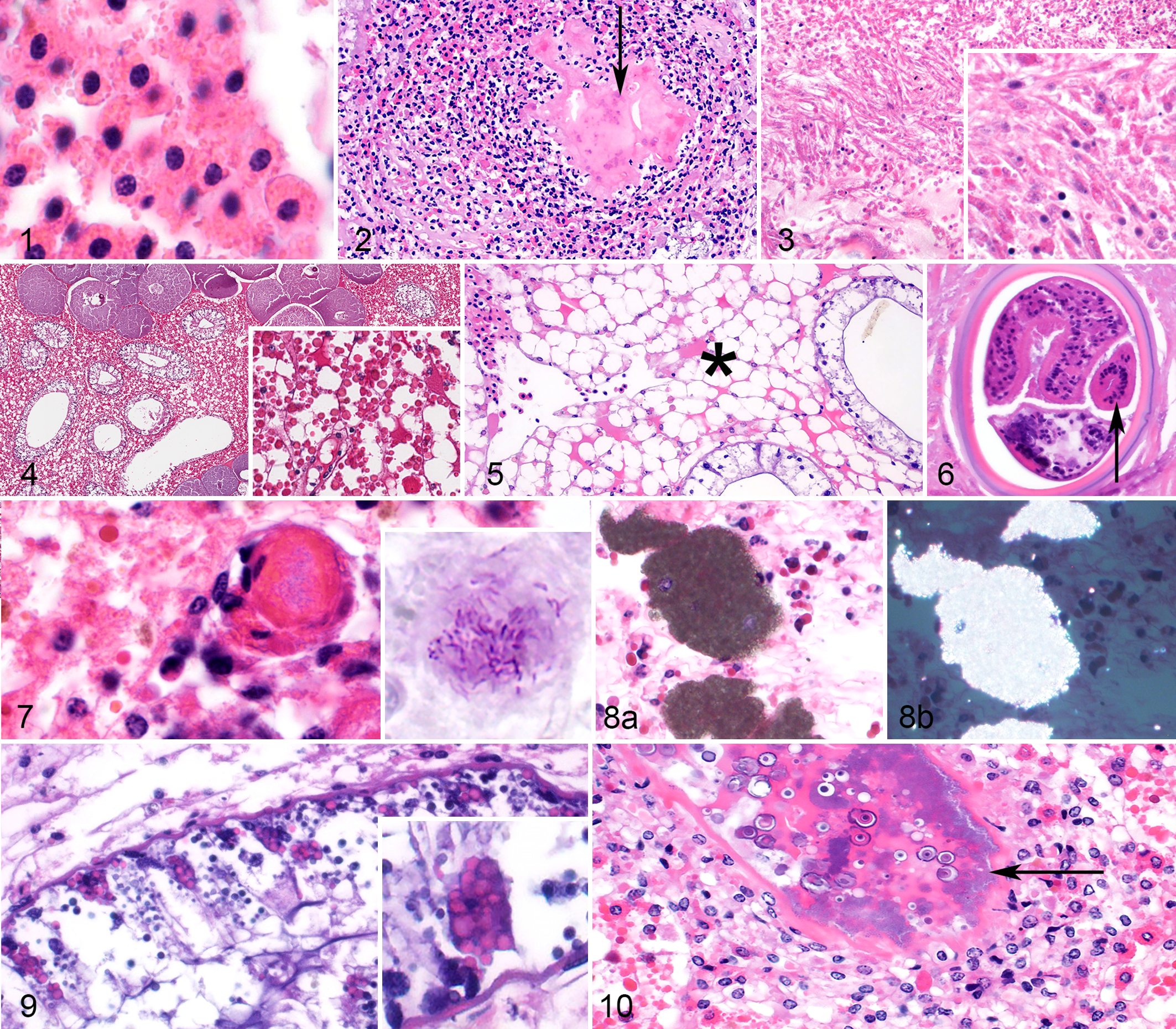
American horseshoe crabs (HSCs; Limulus polyphemus).
Hepatopancreas
The hepatopancreas occupies the majority of the prosoma (ie, largest and most anterior body segment) and was examined histologically in 57 cases. It interdigitated with skeletal muscle of the legs and the reproductive tissue (ie, testes or ovaries) and surrounded multiple hemocoel sinuses containing hemocytes (Fig. 4). The hepatopancreas comprises interstitial cells and collecting tubules. Interstitial cells (also called storage cells or reserve cells) 5 occupied the majority of the hepatopancreas and were interrupted regularly by collecting tubules.
Interstitial cells were polygonal and large with abundant, vacuolated cytoplasm; variable numbers of eosinophilic, spherical globules; and fewer, smaller basophilic globules that varied in size and shape. The amount of eosinophilic globules varied by case and was not uniform across different regions of the hepatopancreas. In some cases, eosinophilic globules were remarkably sparse (12/57 cases; 21%), and this was interpreted as eosinophilic globular depletion (Fig. 5). Hemocytes formed aggregates and coagula in the hepatopancreatic interstitium of 16 of 57 cases (28%). Inflammatory foci in the hepatopancreas were usually associated with identifiable infectious agents (see below). Encysted, variably degraded parasites resembling trematodes were identified in the hepatopancreatic interstitium of 22 of 57 cases (39%), as well as in many other tissues (see “Other Organs”). These cysts were 80 to 150 microns in diameter with a double wall and infolding of an acoelomate body that sometimes had recognizable suckers (Fig. 6). The immune response to these parasites ranged from small aggregates of granular hemocytes, to a combination of granular and reactive hemocytes, to a small amount of surrounding, compressed mesenchymal cells (possible fibrosis), to no identifiable host response. Bacterial infections in the hepatopancreatic interstitium were identified in 10 of 57 cases (18%); bacteria were associated with multifocal, randomly dispersed, small to moderately sized hemocyte aggregates and/or coagula. In 1 case, an acid-fast stain revealed intralesional acid-fast bacilli (Fig. 7). Fungal infections in the hepatopancreas were rare (2/57 cases; 4%) and always occurred at the margin of the organ due to extension of cutaneous infection through the body wall (see “Body Wall” section). Rarely (4/57 cases; 7%), the hepatopancreatic interstitium had granular, birefringent, brown crystalline material that was not associated with any identifiable host reaction and appeared both intra- and extracellular (Fig. 8).
Collecting tubules varied from 50 to 500 microns in diameter and were lined by simple, tall, columnar epithelial cells surrounded by a thin muscular network composed of circular and longitudinal striated muscle. 6 The 2 primary cell types in the tubule epithelium were light cells (also called acidophils) and dark cells (also called basophils) (Fig. 9). 5 Dark cells had secretory granules that were variable in number depending on the animal. Dark cells are analogous to the exocrine pancreas of mammals and secrete enzymes such as lipase, amylase, and trypsin; as such, their granules may be referred to as “zymogen granules.” 5 Light cells were much more numerous (approximately 4 times) than dark cells. Collecting tubules were sometimes distended with amorphous granular, basophilic material (presumed digestive contents) admixed with zymogen granules. Inflammation with peripheral granular and central reactive hemocytes surrounded and rarely infiltrated the muscular layer of tubules in 6 of 57 cases (11%), all of which had intratubular bacteria with effacement of tubular epithelium (Fig. 10).
Body Wall
Body wall was histologically identified in 60 animals. The cuticle was organized into 3 acellular layers: an external, thin, poorly staining epicuticle; a thick, chitinous exocuticle; and an inner, deeply eosinophilic endocuticle (Fig. 11). The epidermis was composed of melanized columnar cells. The dermis had large secretory glands that emptied through the outer cuticle.
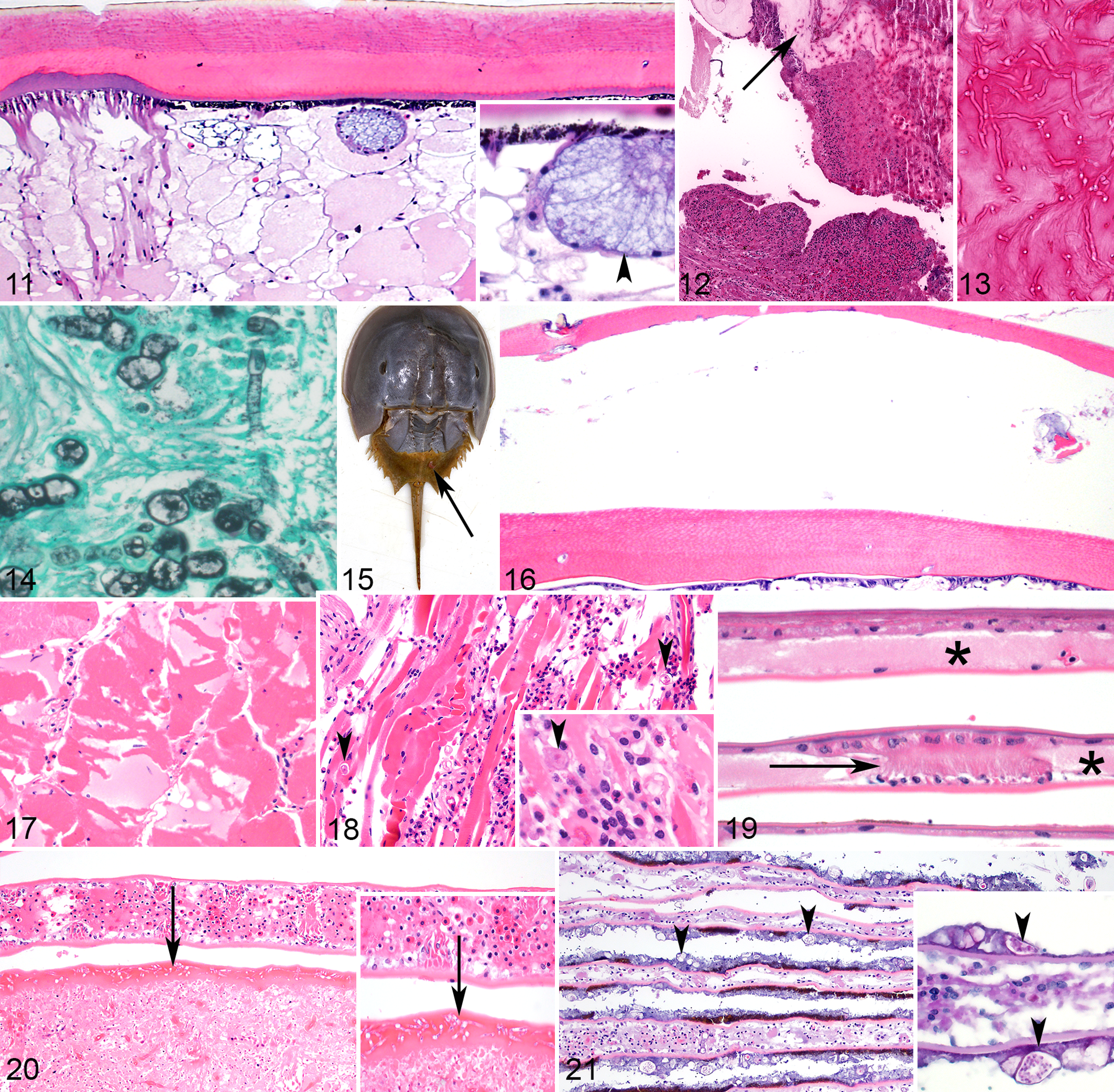
American horseshoe crabs (HSCs; Limulus polyphemus).
The most common lesions in the body wall were loss of a portion of the epicuticle (termed erosion), parasitic cysts, and ulceration of the cuticle and epidermis with inflammation in the dermis and surrounding cuticle (Fig. 12). Erosion without ulceration was identified in 10 of 60 cases (17%) and was invariably associated with superficial (ie, noninvasive) bacterial colonization without any appreciable inflammation. Ulceration was present in 34 of 60 cases (57%) and was consistently associated with infectious organisms; associated fungi were usually invasive and bacteria were usually superficial, suggesting that fungal agents were more likely causes of ulcerative lesions. Fungi had variable morphology. All were filamentous with septate hyphae. Branching was generally acute angle, and walls were either parallel or nonparallel. Size varied widely from approximately 6 to 30 microns in diameter (Fig. 13). Parasitic cysts, as described in the hepatopancreas, were identified in soft tissues of the body wall in 19 of 60 cases (32%).
An invasive algal infection was identified in the body wall in 1 of 60 cases (2%). Histomorphology of this infection was consistent with previous reports of green algal infection. 3,8 Algae had highly pleomorphic morphology with occasional septation, budding, and internal structure. Gomori methenamine silver stain revealed argyrophilic internal contents of the algae, but algal walls had minimal or no argyrophilia (Fig. 14). Algae were not associated with appreciable inflammation but appeared to cause defects in the cuticle as they were associated with ulceration and invaded into subjacent dermis and skeletal muscle. Superficial bacteria were also present in algal lesions.
Retention of molt was seen in 3 of 60 cases (5%) (Fig. 15). Histologically, an extra layer of cuticle (ie, previous molt) was overlying the body wall, which had a thin cuticle lacking normal layering and some chitinous debris between the old and new cuticle (Fig. 16). Retention of molt was also observed in the chitinous sections of the gut in those cases. Four cases had swollen myofibers with loss of cross-striations and occasional karyorrhectic nuclei (ie, rhabdomyolysis) in large sections of skeletal muscle without substantial inflammation (Fig. 17). Abundant hemocytic inflammation in the skeletal muscle with some myocellular necrosis (ie, myositis) was observed in 4 cases, 3 of which were associated with bacteria, and 1 was centered on presumptive amoeba, the latter of which were identified by their shape (amoeboid), spherical nucleus, size (20–35 microns), and cytoplasm with phagocytosed contents (Fig. 18). Superficially, the body wall was occasionally overlaid by arthropods, algae, protozoa, and other metazoan parasites, none of which were associated with inflammation or substantial cuticular changes.
Book Gills
Book gills were identified histologically in 48 of 61 cases (79%) and composed of lamellae that arose from opercula. Gill lamellae were arranged in 2 simple, flattened epithelial layers that enclosed a vascular lumen containing hemolymph. These epithelial layers were held apart by pillar cells (Fig. 19). 1 The most common branchial lesion was inflammation (29 of 48 cases; 60%). Hemocyte coagula were frequently multifocal and led to widening of the lamellae and sometimes ulceration of the lamellar tips (Fig. 20). Inflammation was variably associated with infectious agents, including bacteria in 21 of 29 cases (72%) and fungi in 19 of 29 cases (66%); no infectious agents were seen on HE-stained sections in 5 of 29 cases (17%) of branchitis, but special stains for infectious agents were not performed on branchial sections in those cases. Fungal and bacterial morphology were similar to those described above (see “Hepatopancreas” and “Body Wall”). Parasitic cysts embedded in connective tissue as described above were seen in the gills of 1 case (2%). Mats of bacteria, algae, fungi, and parasites (including arthropods, nematode larvae, and trematode larvae) occasionally formed on the surface of lamellae, were not associated with inflammation, and may have been incidental. In 4 of 48 cases (8%), these mats included algae consistent with blue-green algae (ie, cyanobacteria) characterized by 15- to 30-micron, spherical to indented algal cells with internal granules (Fig. 21). 8 Algae were consistently superficial and were not identified invading into gills.
Other Organs
Parasitic cysts as previously described were identified in a wide range of tissues in addition to those previously mentioned, including brain, chitinous gut, compound eye, heart, and nonchitinous gut. Lesions in the compound eye reflected the range of lesions seen in the body wall to include fungal and/or bacterial ophthalmitis with variable ulceration. Enteritis was identified in the chitinous gut in 4 of 40 cases (10%) and nonchitinous gut in 3 of 42 cases (7%) for which these organs were identified histologically. Bacteria were associated with enteritis in 5 of 7 cases (71%), 1 of which had acid-fast bacilli, and fungi were associated with enteritis in 1 of 7 cases, which was a mixed bacterial and fungal enteritis; infectious agents were not visibly associated with enteritis in the remaining 2 of 7 cases (29%). Lesions were not identified in the gonads or coxal gland in any case.
Culture Results
Bacterial or fungal culture results were reported in the necropsy records of 6 cases. Pseudomonas putrefaciens and Aeromonas hydrophila were isolated from hemolymph from cases 2 and 4, respectively. Micrococcus sp. was isolated from hepatopancreas from case 18. Fusarium spp. were isolated from gills from cases 14 and 15 and from an unspecified location from case 8.
Discussion
This report describes a range of histologic findings in American horseshoe crabs, including normal microanatomy and lesions. Most of the histologic lesions involved hemocytic inflammation. Hemocytes are the only immune cell of HSCs and are stored in vascular sinuses. 4,13 Under normal physiologic conditions, hemocytes have cytoplasmic granules. In pathologic conditions, hemocytes can discharge their cytoplasmic granules and aggregate around wounds, infectious agents, and foreign material to aid in the immune response. 4,13 Hemocyte granules contain a number of recognized antimicrobial substances, agglutinins, and factors to activate clotting. When clotting factors are released, this activates coagulen and results in the formation of an insoluble coagulin gel, also called a coagulum. This coagulum occludes wounds and engulfs and immobilizes microbes, which are subsequently killed by antimicrobial substances in the coagulum that are also released from hemocyte granules. 9 Over time, granular hemocytes infiltrate the periphery of the coagulum, release their cytoplasmic granules (ie, reactive hemocytes), and may become elongate with oval-shaped nuclei. This results in a healing wound with a central coagulum (which may contain foreign material and/or infectious agents) surrounded by reactive hemocytes that are further surrounded by marginal granular hemocytes. 4 Agglutinins released from hemocyte granules help to aggregate and isolate infectious agents, making them more susceptible to entrapment and phagocytosis, as well as opsonize foreign material. 9 After phagocytosis, bacteria are digested intracellularly by lysosomal enzymes. 13 In this case series, hemocyte coagula were frequently observed around infectious agents (ie, bacteria, fungi, and parasites), and hemocytes aggregated around and occluded ulcers. The hemocyte response ranged from small aggregates of granular hemocytes to large central coagula surrounded by a mix of granular and reactive hemocytes. In addition, coagula in the gills rarely contained spindle cells; the nature of these cells is unknown, although they may represent activated pillar cells or reactive hemocytes that have acquired a spindloid shape.
Hemocytic inflammation was almost always associated with bacterial or fungal infection. Fungal cultures were rarely performed and grew Fusarium spp. in all cases, which was consistent with the histology findings of relatively pleomorphic, branching, septate fungal hyphae. In a previous study focused on fungal lesions in HSCs, which included some animals in the current study, the associated fungi were commonly identified as Fusarium solani; Hortea werneckii and Stemphylium sp. were also isolated in fewer cases. 15 Considering how little research is published on the pathology and diseases of HSCs, fungal infections of the carapace and gills are relatively well documented. 10 Gross lesions in these organs can be striking, with deep cavitations, discoloration, and softening. Although fungal disease is reported only in captive animals, this may reflect the rarity of pathology reports in wild HSCs. According to available literature, fungal disease in HSCs seems to be distributed in the gills and carapace, as seen in these cases. Rarely, fungal infection was noted in the hepatopancreas in this series, and involvement of this organ was always via direct extension from an external lesion through the body wall (ie, extension of fungal dermatitis into subjacent hepatopancreas). This is in contrast to bacterial infections in the hepatopancreas, which were frequently scattered multifocally in the interstitium, suggesting hemolymphatic showering.
Multiple references list algae as a common infection identified histologically in HSCs; green algae, specifically, have been reported as the most common infectious agent in this species. 3,8 Green algal infection was rare in our series, identified in only 1 animal. Similarly, blue-green algae, or cyanobacteria, are reported as common causes of disease in HSCs, whereby algae invade the gills and lead to defects in osmoregulatory function and respiration. 8 Algae consistent with blue-green algae were observed on the gills in only 4 cases in this series, none of which had evidence of algal invasion into tissue. In all cases, the blue-green algae were present in a biofilm overlying the gills. Almost all of the HSCs in the previous studies that reported frequent green and blue-green algal infections were wild-caught from Cape Cod, Massachusetts, and housed in the same facility, 3,8 whereas the animals in the present study came from a wide array of facilities and states. Thus, it is possible that sampling bias in the previous reports led to overrepresentation of algal disease in HSC literature. Both previous and present studies used histology as the primary means of detecting algal infection. 3,8
A large number (41%) of cases had variably preserved encysted parasites distributed in the connective tissue of almost every organ, including the brain. The well-preserved parasites had typical features of metacercariae, which are typically spherical with a double cyst wall. Some trematode species with developing metacercariae in cysts have folded bodies within the cyst wall, and the suckers are variably evident in section. 7 Trematodes were tentatively identified as Microphallus limulus based on this morphology. M. limulus is a trematode that is very common in juvenile HSCs. 8 The first intermediate host is a snail, within which the parasite transitions from a miracidium to a sporocyst to a cercaria. The cercariae exit the first intermediate host and invade juvenile HSCs, encysting and forming metacercariae. Completion of the life cycle occurs if HSCs are consumed by birds. 5 In HSCs, metacercariae cysts of M. limulus form complex shells, which are approximately 200 microns in diameter, and filled with globular internal structure, as seen in this series. Encysted metacercariae of M. limulus are common in many types of connective tissue in HSCs and have also been reported in the eye and central nervous system, both of which were observed in this case series. Degenerate trematode cysts can often be found in adult HSCs. 5,14 M. limulus reportedly causes little host reaction, which is consistent with the findings reported here. 5
Evaluation of nutritional status in arthropods is a unique challenge for pathologists as these animals do not have white adipose tissue. The hepatopancreatic cells store lipids and may aid pathologists in estimating nutritional status. 5 In HSCs, eosinophilic globules are typically abundant in hepatopancreatic interstitial cells of well-fed animals and are especially abundant in the hepatopancreas around the heart, gut, and central nervous system. In starved HSCs, interstitial cells reportedly have reduced cytoplasm that ultrastructurally lacks large protein granules, has reduced numbers of small protein granules, and lacks glycogen. 6 In some cases in this series, eosinophilic globules within interstitial cells of the hepatopancreas were markedly sparse. The significance of this finding is unclear but is believed to represent decreased overall nutritional status. The amount of eosinophilic globules in interstitial cells appeared to vary somewhat in different regions of the hepatopancreas, which may complicate interpretation depending on the sampled location.
Histologically, lesions were most commonly identified in the compound eye and body wall. This is likely because these external organs are grossly visible, and lesions in these organs were the cause for euthanasia in many cases. The number of organs represented in sections varied widely among cases. The organs least commonly represented were the brain and coxal gland. An excellent reference on HSC dissection is available. 2 Such references undoubtedly improve the utility of necropsy and histologic examination for identifying disease and causes of death by helping prosectors collect a complete and representative tissue set. Further research is necessary on management techniques to reduce and mitigate diseases in these animals.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819859877 - Histologic Findings in Captive American Horseshoe Crabs (Limulus polyphemus)
Supplemental Material, DS1_VET_10.1177_0300985819859877 for Histologic Findings in Captive American Horseshoe Crabs (Limulus polyphemus) by Elise E. B. LaDouceur, Lisa Mangus, Michael M. Garner and Andrew N. Cartoceti in Veterinary Pathology
Footnotes
Acknowledgements
We thank pathologists previously or currently associated with Northwest ZooPath, National Zoo, and Johns Hopkins University who were primary reviewers for some of these cases, including Drs. Sarah Beck, Kathy Gabrielson, Joseph Mankowski, Richard Montali, Craig Morrell, Don Nichols, John Roberts, Nancy Stedman, John Trupkiewicz, Tabitha Viner, and Timothy Walsh. Thank you to Clinton A. Coates of Darnall Biomedical Learning Resource Center for assistance with obtaining reference materials. We also thank Dr. Carol Meteyer, Jill Arnold, and Dr. Leigh Clayton for collaborative efforts to recognize health and disease parameters of wild horseshoe crabs, which led to the conception of this study. Thank you to all of the veterinarians and animal care staff for managing the health of these animals and for caring enough about their well-being to investigate their diseases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this article are those of the author and do not reflect the official policy of the Department of Army/Navy/Air Force, Department of Defense, or US government.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
