Abstract
Insects play an important role in ecosystems. Changes in their abundance and biodiversity are of paramount interest, as there has not only been an alarming decline of insects important for ecosystem health throughout the past decades, but also an increase in insects detrimental for biomes. Furthermore, insects pose a threat to modern society as arbovirus-transmitting vectors. Therefore, detailed knowledge of insect staining characteristics could be beneficial as a basis for further studies, whether in the context of species conservation or control of insect pests. Thus, this study compared 14 histochemical stains for their usefulness in insects regarding nervous tissue, connective tissue components, mucins and polysaccharides, mineralization, and microorganisms. The study used formalin-fixed paraffin-embedded tissue sections of mammals (Equus caballus) and 2 dipterans (Culex pipiens biotype molestus, Drosophila melanogaster). Several histochemical stains were suitable for tissue assessment in insects and mammals, in particular for nervous tissue (Bielschowsky silver stain, luxol fast blue–cresyl violet) and polysaccharides (alcian blue, periodic acid–Schiff with and without diastase treatment, toluidine blue). Other stains proved useful for visualization of insect-specific organ characteristics such as Gomori’s reticulin stain for tracheoles in both dipteran species, Heidenhain’s azan for midgut-associated connective tissue, and von Kossa for mineral deposition in Malpighian tubules of C. pipiens biotype molestus. In summary, this study provides comparable insights into histochemical procedures in mammals and insects and their usefulness for histological assessment of C. pipiens biotype molestus and D. melanogaster.
Insects are an important subject of research as they play an immense role in ecosystems all over the world, 31 and are gaining increasing interest in the food industry as an alternative nutritional resource. 60 However, while climate change, loss of territory, and food sources along with pesticides have caused a worldwide decline in abundance and biodiversity of insects important for ecosystem health and economy,45,48,56 it has led to heightened populations of insects considered detrimental for biomes.56,63 Furthermore, insects pose a threat to modern society as arbovirus-transmitting vectors. 20 Increased risks of arbovirus transmission are a result of urbanization, globalization, and changing climatic conditions.20,65 Population size and habitat of various vector species are expanding due to involuntarily migration as stowaways, thus promoting spread of pathogens by introducing them to new areas. 37 The existing danger is also exacerbated by transmission of pathogens to native insect populations, which then represent potential vectors. 28
Mosquitoes are important vectors for transmission of various human and animal pathogens. The mosquito species Culex pipiens is one of the most widespread mosquitoes in the Northern Hemisphere 16 and known to transmit emerging diseases such as Rift Valley fever, 30 West Nile, and Usutu. 16 In contrast, fruit flies are one of the most intensely studied organisms, serving as a model for several aspects in research, including genetic and degenerative disorders, disease pathogenesis, and innate immune pathways.40,68
In mammals, histochemistry is a well-known tool for the analysis of different tissue types. Similarly, previous studies in insects have focused on histochemical staining to elucidate anatomical or physiological peculiarities.3,6,12,21,33,50,52,59 However, the class of insects is vast, and species within this class show great differences in their anatomy, physiology, and biochemistry. Unfortunately, comprehensive literature regarding staining properties for mosquitoes or fruit flies is scarce. The fat body of Anopheles spp. and Aedes aegypti was studied with periodic acid–Schiff (PAS) reaction.33,34 Another report indicates the usefulness of Gimenez staining for detection of the endosymbiont Wolbachia pipientis in cytological smears of ovaries from Culex quinquefasciatus. 41 Regarding fruit flies, 1 study describes alcian blue and PAS-positive mucins in the gastrointestinal tract of Drosophila melanogaster, 11 while another reports PAS-positive granules in their flight musculature. 12 Furthermore, the nervous system was evaluated using Bielschowsky silver stain. 38 To date, there has been no comparative study between insects and mammals or a survey of multiple stains explicitly for these 2 above-mentioned dipteran species, neither for healthy nor for diseased tissue. Therefore, detailed knowledge of insect staining characteristics could be beneficial as a basis for further studies, whether in the context of vector research or disease pathogenesis. 15 To enable such histological examination, 14 histochemical stains regularly used to evaluate mammalian tissues in routine diagnostic or research laboratories were comparatively assessed for their usefulness in insect species using healthy tissue from mammals and insects. Equine tissue served as representative mammalian positive control for the comparison of applied histochemical methods in both insects, because staining characteristics in horse tissues are well established. Regarding insects, 2 dipteran species were examined. The mosquito C. pipiens biotype molestus was chosen as a known vector for various pathogens such as arboviruses, while the fruit fly D. melanogaster was selected for its role as a model organism in research. Employed histochemical stains included (1) hematoxylin and eosin (H&E); (2) Bielschowsky silver stain, luxol fast blue–cresyl violet (LFB-CV), and phosphotungstic acid–hematoxylin (PTAH) for evaluation of nervous tissue; (3) elastica van Gieson (EVG), Gomori’s reticulin stain, Heidenhain’s azan, and Masson-Goldner for connective tissue; (4) alcian blue, PAS with and without diastase pretreatment, and toluidine blue for mucins and polysaccharides; (5) von Kossa silver stain for mineralization; and (6) Gimenez for microorganisms.
Materials and Methods
Animal Samples
A German field-collected C. pipiens biotype molestus mosquito line (courtesy of Department of Arbovirology, Bernhard Nocht Institute for Tropical Medicine, Hamburg, Germany) and a laboratory strain of the fruit fly D. melanogaster (kindly provided by Jean-Luc Imler, Institut de Biologie Moléculaire et Cellulaire; Université Luis Pasteur, Strasbourg) were reared at the Institute for Parasitology and Research Center for Emerging Infections and Zoonoses, University for Veterinary Medicine, Hannover. At least 6 female individuals per dipteran species (n = 6) served for histological assessment. With regard to the histochemical procedure and its adaptation to herein assessed dipteran species, more individuals were stained for Heidenhain’s azan and Masson-Goldner (n = 10); PTAH, Bielschowsky silver stain, and LFB-CV (n = 15); as well as H&E and Gimenez (n = 30). One male per dipteran species (n = 1) was stained with H&E for demonstration of gonads.
Collection and assessment of equine tissue samples as reference material (n = 1) was conducted during routine diagnostic necropsies at the Department of Pathology (University for Veterinary Medicine Hannover, Germany). Tissue samples taken included central nervous system (CNS; cerebrum, cerebellum, and brainstem), gastrointestinal tract (stomach, small and large intestine), kidney, liver, skeletal muscle, ovaries, and nasal conchae without significant pathological findings.
Tissue Processing
Prior to processing, insects were anesthetized with carbon dioxide. Equine and insect tissue samples were fixed in 10% neutral-buffered formalin for 24 hours and embedded in paraffin wax (formalin-fixed paraffin-embedded [FFPE]) before generation of 2 to 4 μm sections mounted on SuperFrost® Plus adhesion slides (Carl Roth GmbH & Co. KG, Karlsruhe, Germany). Subsequently, tissue sections were deparaffinized in xylene (Carl Roth GmbH & Co. KG) and passed through a descending alcohol series (isopropanol and 99.8% ethanol, Carl Roth GmbH & Co. KG) into distilled water. Finally, slides were submitted to various histochemical stains (Table 1), dehydrated in an ascending alcohol series, transferred to n-butyl acetate (Carl Roth GmbH & Co. KG), and mounted with Roti®Histokitt II (Carl Roth GmbH & Co. KG).
Histochemical staining qualities in insects and mammals.
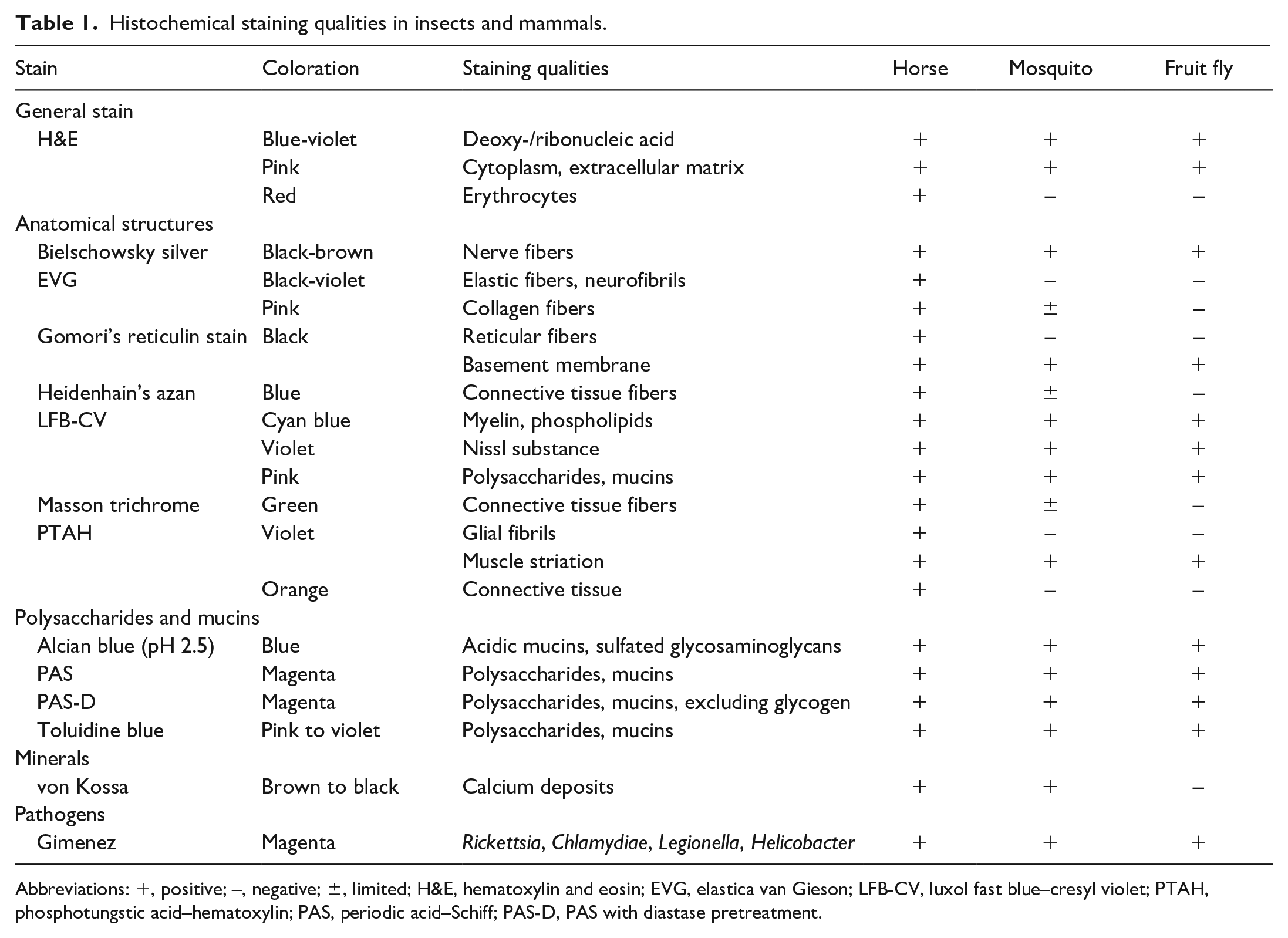
Abbreviations: +, positive; –, negative; ±, limited; H&E, hematoxylin and eosin; EVG, elastica van Gieson; LFB-CV, luxol fast blue–cresyl violet; PTAH, phosphotungstic acid–hematoxylin; PAS, periodic acid–Schiff; PAS-D, PAS with diastase pretreatment.
Histochemistry
A panel of 14 different histochemical techniques routinely used in mammals was compared with staining qualities in mosquitoes and fruit flies. Histochemical procedures are briefly described, while modifications and detailed protocols are provided as Supplemental Material. The number of individuals examined per dipteran species is given in parentheses for each histochemical stain.
All specimens were routinely stained with H&E (n = 30/species) as previously described using an automated slide stainer (Leica ST 4040; Leica Biosystems, Nussloch, Germany). 39
Evaluation of nervous tissue was conducted using Bielschowsky silver stain, LFB-CV, and PTAH. For Bielschowsky silver stain (n = 15/species), sections were treated in the dark at 37°C with 17% silver nitrate and thereafter with ammonium silver nitrate. This was followed by treatment with an ammonium developer solution. Sections were then washed with 0.3% ammonium hydroxide and finally fixed in 5% sodium thiosulfate. 39 LFB-CV staining (n = 15/species) was employed by 2 steps: (1) incubation of sections in 0.1% luxol fast blue followed by differentiation in 0.05% lithium carbonate and (2) staining with 0.1% acidified cresyl violet (CV) and differentiation in absolute alcohol. 39 PTAH staining (n = 15/species) involved incubation in potassium dichromate followed by treatment with potassium permanganate. Thereafter, sections were differentiated in 1% oxalic acid prior to staining with oxidized PTAH solution. 32
For assessment of connective tissue, EVG, Heidenhain’s azan, Gomori’s reticulin stain, and Masson-Goldner were employed. EVG staining (n = 6/species) was carried out by incubating sections with resorcin fuchsin. After differentiation in absolute alcohol, sections were stained with Weigert’s iron hematoxylin. Subsequent differentiation with 0.5% alcoholic hydrochloric acid was followed by staining with van Gieson’s saturated picric acid. 39 Applying Gomori’s reticulin stain (n = 6/species), sections were treated with 0.1% to 1% potassium permanganate, 2% ferric ammonium sulfate, and 10% ammonium silver nitrate followed by incubation with 10% neutrally buffered formalin and 0.1% gold chloride. Sections were then transferred to 1% to 3% potassium metabisulfite and finally fixed in 5% sodium thiosulfate. 39 For Heidenhain’s azan stain (n = 10/species), sections were incubated with azokarmin at 60°C followed by sequential treatment with 1% alcoholic aniline, 1% alcoholic glacial acetic acid, and 5% phosphotungstic acid. Finally, sections were incubated in azan working solution before being differentiated in absolute alcohol. 39 Masson-Goldner (n = 10/species) staining involved incubation in Weigert’s iron hematoxylin. Thereafter, sections were transferred to Ponceau-acid fuchsin-azophloxine and finally incubated with phosphotungstic acid–orange G. 39
Mucins and saccharides were evaluated using alcian blue, PAS, PAS with diastase pretreatment, and toluidine blue. Acidic mucopolysaccharides were visualized by alcian blue staining (n = 6/species) with a 0.1% alcian blue solution (pH 2.5) followed by a 0.1% nuclear fast red solution. 39 PAS reaction (n = 6/species) involved incubating sections in 1% periodic acid followed by Schiff’s reagent. Finally, sections were counterstained in Mayer’s hemalum. 39 Applying PAS with diastase pretreatment (n = 6/species), sections were preincubated in diastase for 60 minutes at room temperature. 39 Toluidine blue stain (n = 6/species) was carried out by applying a 0.1% toluidine blue solution (pH 3.5), which was produced with veronal-acetate buffer. 39
Staining method of von Kossa (n = 6/species) was applied according to the modification of Krutsay to visualize calcium minerals. 39 Therefore, sections were incubated in 3% to 5% silver nitrate solution under direct light and subsequently transferred to a 5% sodium thiosulfate solution before counterstaining with 0.1% nuclear fast red.
Gimenez staining (n = 30/species) was employed for detecting Rickettsia spp. in mosquito specimens. Thus, tissue sections were incubated in basic fuchsin, differentiated in 0.25% glacial acetic acid and finally counterstained with 0.2% fast green. 19
Evaluation of Results
Histochemical stains were examined by 2 pathologists using a light microscope (OLYMPUS BX53; Olympus Europa SE & Co. KG, Hamburg, Germany). Specimens were first evaluated using routinely H&E-stained sections followed by analysis of all other histochemical stains described above. Staining results of insect specimens were correlated to known staining properties in comparable equine tissue as a representative of mammals. In addition, unexpected, regularly occurring staining properties of insect tissue in individual histochemical procedures were classified as insect-specific, while inconsistent or variable staining was interpreted as unspecific background reaction. Histochemical staining images were acquired with the Olympus BX51 microscope using the Olympus DP72 digital microscope camera with oil immersion, the Olympus VS 200 Slide Scanner with oil immersion (Olympus, Deutschland GmbH, Hamburg, Germany) and the BZ-9000E microscope (Keyence Deutschland GmbH, Neu-Isenburg, Germany).
Results
Healthy tissue from 2 dipteran species, mosquito and fruit fly, was examined using 14 histochemical stains and compared with equine tissue as a mammalian reference. Histochemical results were grouped into general and specialized stains, with the latter classified according to their main target tissue type in mammals: nervous tissue, connective tissue, mucins and polysaccharides, minerals, and microorganisms. Results are summarized in Table 1. Insect-specific findings are briefly listed for each staining procedure in Table 2.
Further insect-specific histochemical staining qualities.
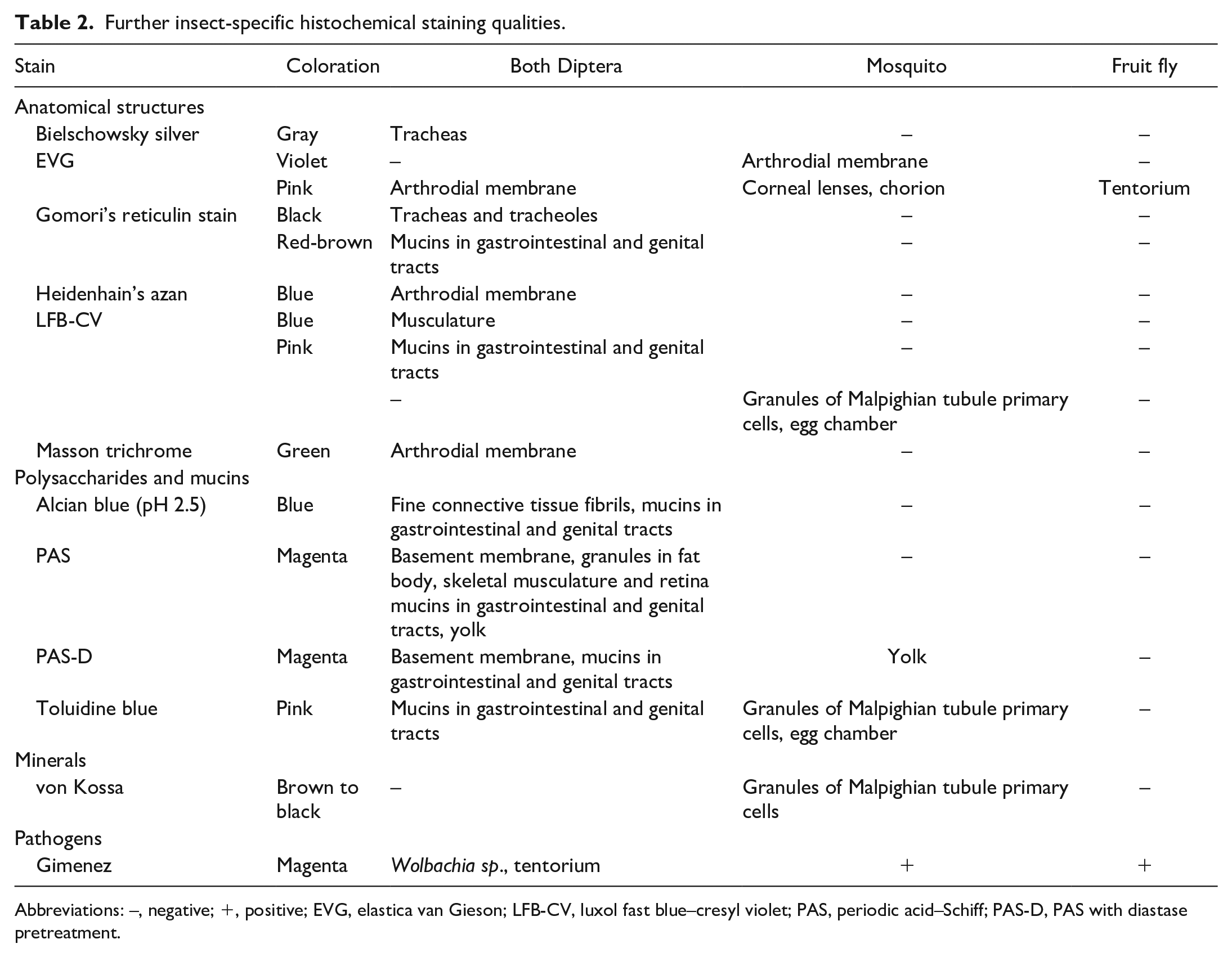
Abbreviations: –, negative; +, positive; EVG, elastica van Gieson; LFB-CV, luxol fast blue–cresyl violet; PAS, periodic acid–Schiff; PAS-D, PAS with diastase pretreatment.
H&E
In both dipteran species, the H&E staining results were similar to mammals (Figs. 1 and 2). Nuclei and nucleoli stained blue-violet, while cytoplasm as well as extracellular matrix were colored in varying shades and intensities of pink in all specimens examined. Equine tissue also showed red staining of erythrocytes. In contrast, no tissue in mosquitoes and fruit flies stained red.
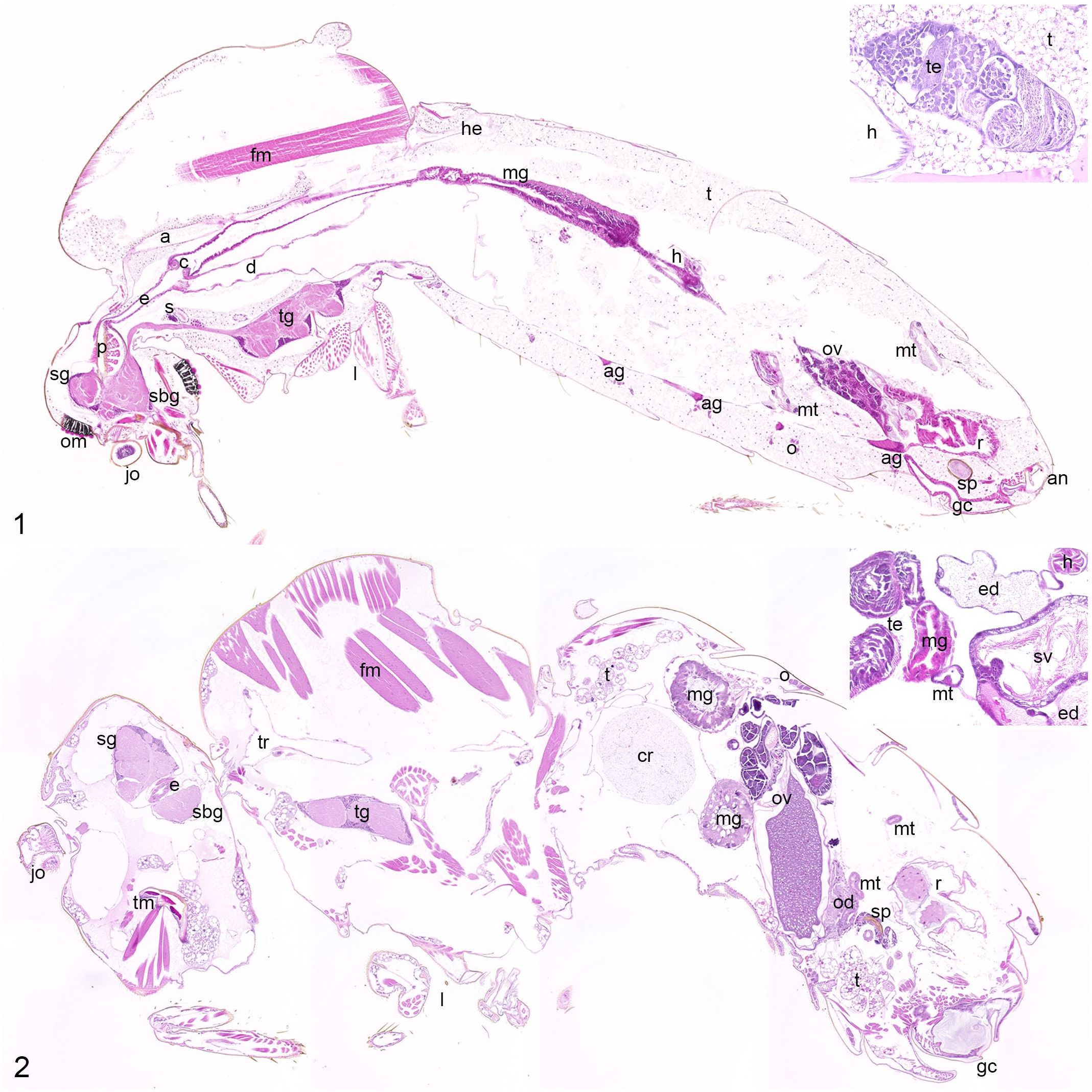
Normal anatomy of mosquito and fruit fly.
Stains for Nervous Tissue
The insect nervous system is decentralized and consists of several ganglia in head, thorax, and abdomen, interconnected by 2 parallel ventral nerve cords. The supra- and subesophageal ganglion together with the ventral nerve cord represent the counterpart of the mammalian CNS in insects. Each ganglion is composed of a cell-rich cortex and a centrally located neuropil surrounded by a dense extracellular matrix called neural lamella. 29
Bielschowsky silver stain visualized neuronal cell processes in brown to black in both dipteran species. Compared with equine nerve fibers showing an intense brown to black staining, mosquito and fruit fly samples often displayed a weaker impregnation (Figs. 3–5) accompanied by a variably intense background. Tracheae (fine insect respiratory tubes) 66 were visualized in gray, whereas the cuticle was stained black in both dipteran species (Supplemental Figs. S1–S7).
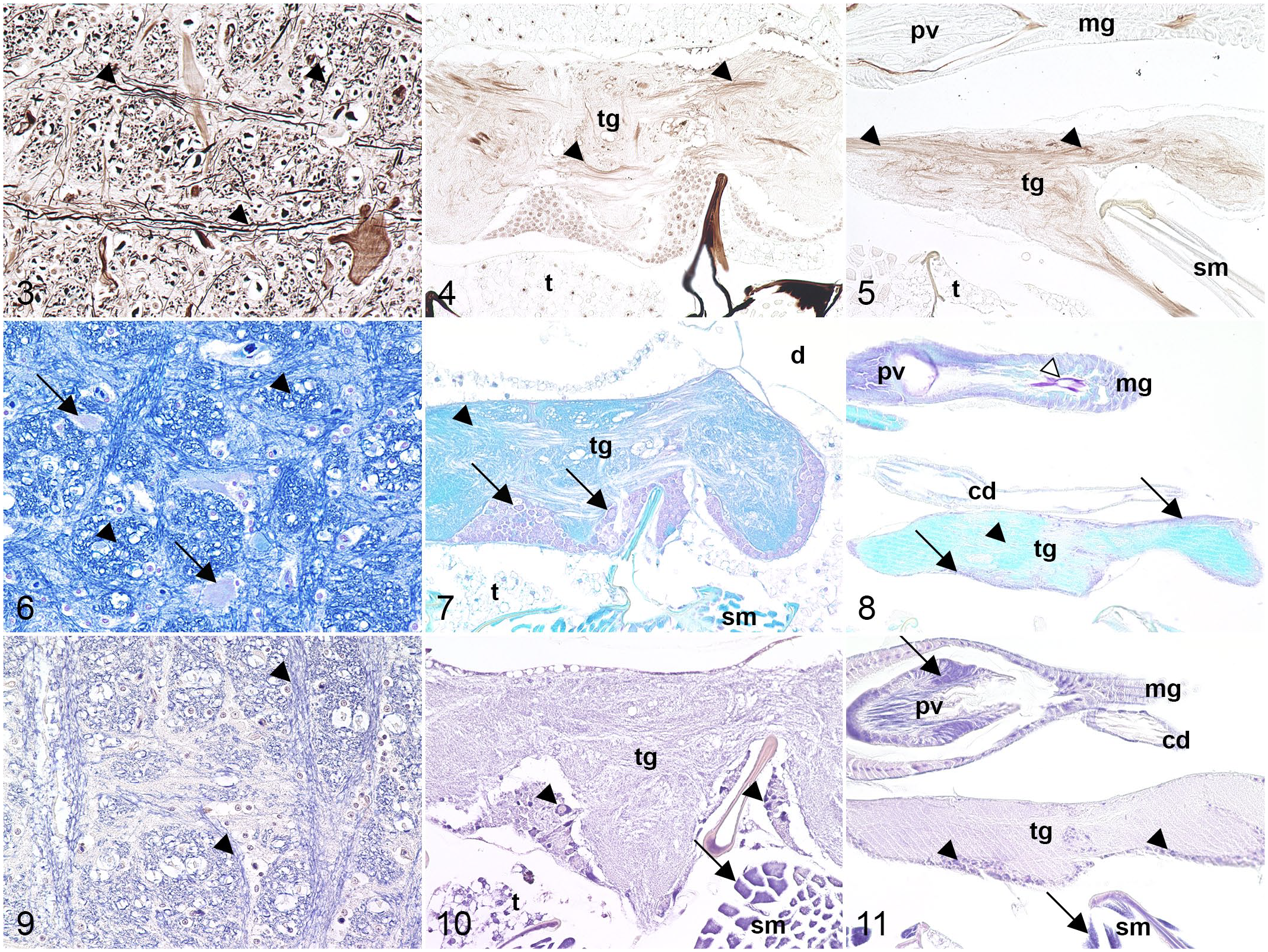
Comparative histochemical staining of nervous tissue in horse, mosquito, and fruit fly.
LFB-CV staining revealed similar characteristics in dipteran and mammalian tissue. Rough endoplasmic reticulum as well as nuclei were stained in violet in all species. The neuropil exhibited a variably pronounced blue coloration in both dipterans, while myelin stained intensely blue within equine samples (Figs. 6–8). Striated musculature was labeled in blue in mosquitoes and fruit flies. Intraluminal content in the intestinal and genital tracts in both dipteran species, along with cytoplasmic granules in the mosquitoes’ primary cells of Malpighian tubules, stained pink to violet (Supplemental Figs. S8–S13). Malpighian tubules consist of primary and stellate cells in mosquitoes and fruit flies. 43 They are a key element in osmoregulation, excretion, and absorption of ions and macromolecules in insect species, 43 similar to kidneys in mammals. 57
Glial cells occur in mammal and insect nervous tissue and share both functional and anatomical characteristics. 25 However, while PTAH-stained glial fibrils were violet in equine nervous tissue (Fig. 9), PTAH did not stain glial cells or their processes in either dipteran species. Cortical cells of body ganglia infrequently stained violet in mosquitoes and fruit flies and, consequently, lacked reliable reproducibility (Figs. 10 and 11). Striated, smooth, and heart muscle fibers were highlighted in violet in both dipterans. PTAH staining also allowed visualization of muscular cross-striation similar to equine skeletal musculature (Supplemental Figs. S14–S21).
Stains for Connective Tissue
Connective tissue in insects is sparse and acellular. 4 Location-dependent, it consists of 3 components: an amorphous, extracellular matrix consisting of neutral and acidic polysaccharides (basement membrane), and less often fine filaments, or collagen-like filaments. 4 Of the latter 2, Diptera only possess fine fibrils. 5 Other connective tissue types mainly found in insects are (1) located in midgut and reproductive tract, 7 (2) nonspecified elastic fibers, 14 and (3) the elastic protein resilin. 1
EVG staining, applied for demonstration of collagen and elastic fibers in mammals, was of limited success for highlighting connective tissue in mosquitoes and fruit flies. Equine samples displayed pink collagen fibers and black-violet elastic fibers, while midgut-associated connective tissue was stained pale pink in mosquitoes. In contrast, no equivalent was observed in fruit flies (Figs. 12–14). However, arthrodial membranes (flexible connections between cuticle segments) 62 were pink in both dipteran species. In addition, corneal lenses and chorion (outer shell of insect eggs) 9 in mosquitoes and the tentorium (a cuticle ingrowth in the head of insects) 58 in fruit flies were pink. The arthrodial membrane connecting the last thoracic and first abdominal sclerite (rigid cuticle segments) 62 stained violet in mosquitoes (Supplemental Figs. S22–S28).
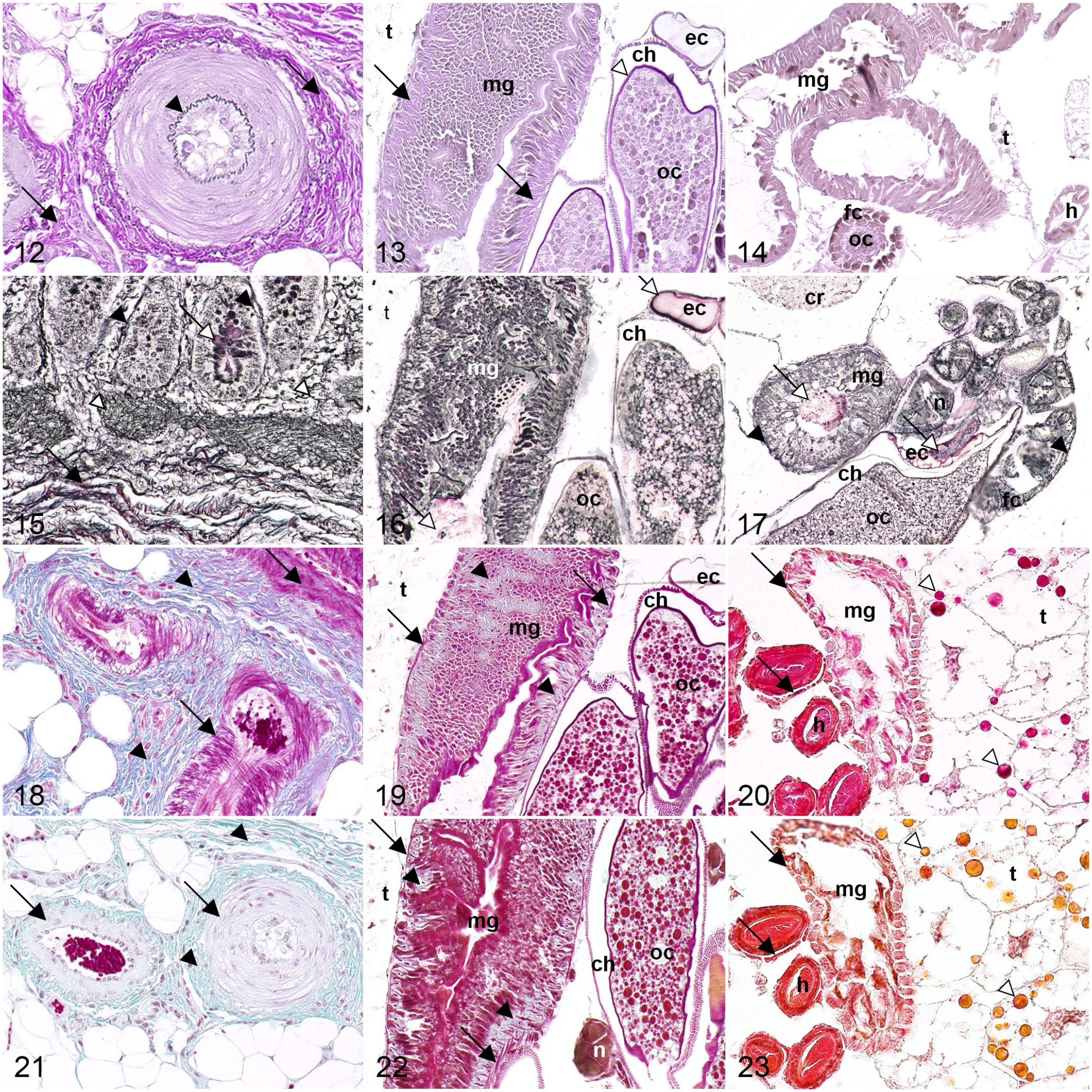
Comparative histochemical staining of connective tissue in horse, mosquito, and fruit fly.
Gomori’s reticulin stain was not suitable for assessing connective tissue fibers in mosquitoes or fruit flies as observed with black-stained reticulin and red-brown collagen fibers in equine samples. However, the basement membrane was successfully visualized in black in all specimens investigated. Equine intestinal goblet cells were stained red-brown, whereas intraluminal contents of the gastrointestinal (mid- and hindgut) and genital (egg chamber and gonoduct) tracts in mosquitoes and fruit flies stained red-brown (Figs. 15–17). Both dipteran species further displayed black-stained nerve fibers, cuticle, tracheae, and tracheoles throughout the body (Supplemental Figs. S29–S37).
Heidenhain’s azan was only of limited success for differentiation of muscle and connective tissue. Equine tissue showed intensely blue-stained collagen and reticulin fibers, while mosquitoes exhibited pale blue connective tissue in midgut and reproductive tract. No corresponding staining was observed in fruit flies (Figs. 18–20). Arthrodial membranes and apodemes (cuticle ingrowths serving as attachment points for muscles)58,62 stained pale blue in both dipterans. Remaining tissue displayed varying shades of pink in all species, including musculature and egg yolk, but also cytoplasmic granules in trophocytes of fruit flies (Supplemental Figs. S38–S45). Trophocytes, in addition to oenocytes, are a cell type found in the fat body of mosquitoes and fruit flies. 2 The metabolism of this organ includes storage of lipids, proteins, and glycogen 2 and is therefore reminiscent of both liver and adipose tissue in mammals. 27
Similar to azan, Masson-Goldner stained connective tissue in mosquitoes and fruit flies only to a limited extent. While samples from horse displayed pale green collagen and reticulin fibers, only mosquitoes showed faint green connective tissue in the gastrointestinal and reproductive tracts. No equivalent green staining of connective tissue was present in fruit flies (Figs. 21–23). Arthrodial membranes and apodemes were colored light green in both dipteran species. Remaining tissue exhibited different shades of red, including musculature, or orange, such as cytoplasmic granules in trophocytes or egg yolk in mosquitoes and fruit flies (Supplemental Figs. S46–S53).
Stains for Mucins and Saccharides
Alcian blue (pH 2.5) stains acidic polysaccharides such as proteoglycans (sulfated mucins) or sialomucins (carboxylated mucins), 54 which were detected in blue in all specimens examined. Equine samples exhibited an intense blue coloration of intestinal goblet cells and a light blue staining of collagen fibers (Fig. 24). In mosquitoes and fruit flies, intraluminal contents of the gastrointestinal (mid- and hindgut) and genital (egg chamber and gonoduct) tracts stained blue (Figs. 25 and 26). Fine connective tissue fibrils located below the integument were stained blue in both dipteran species (Supplemental Figs. S54–S60).
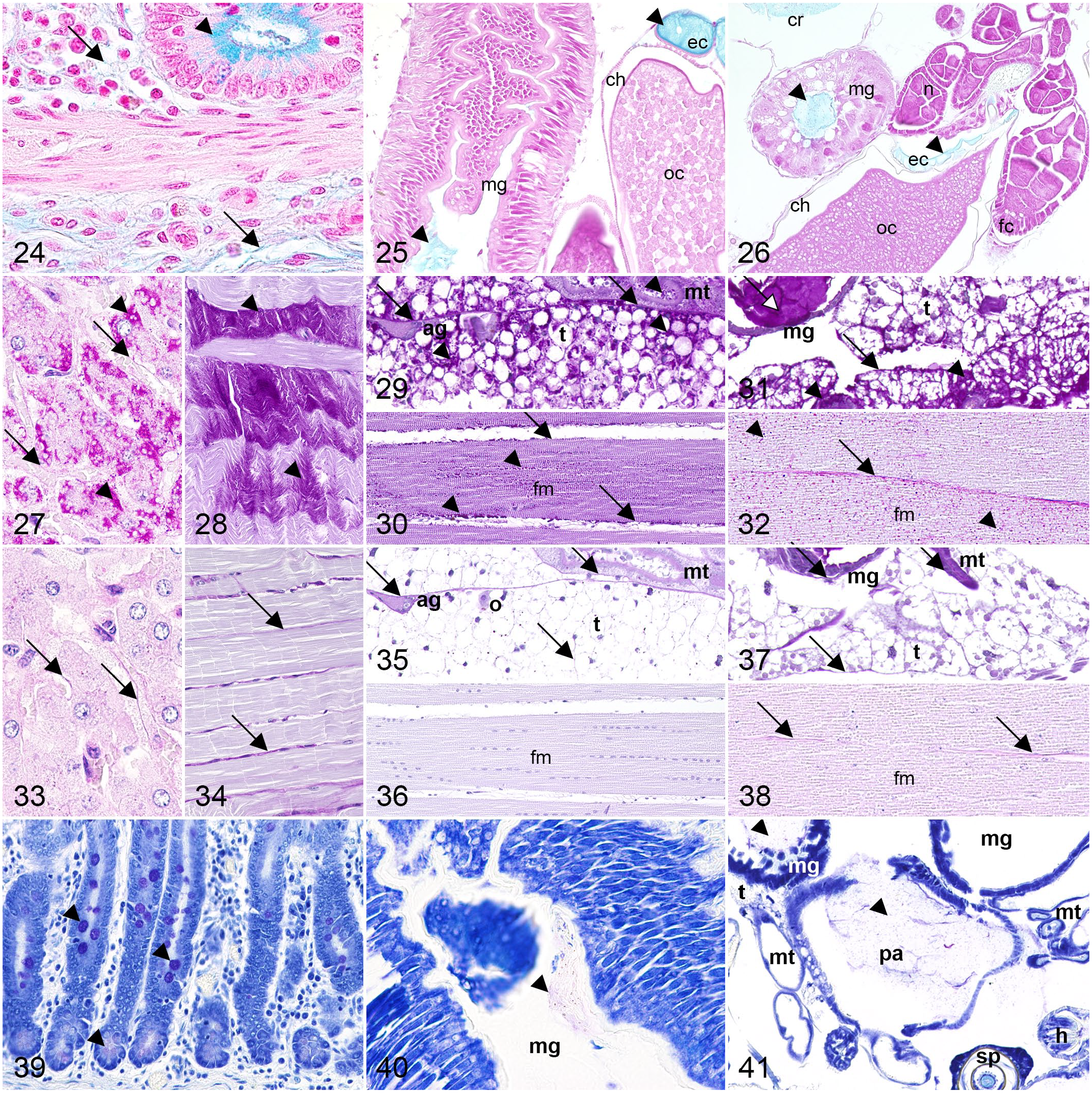
Comparative histochemical staining of polysaccharides in horse, mosquito, and fruit fly.
PAS with and without diastase pretreatment was applied for visualizing glycogen and other neutral polysaccharides, including mucins. In addition to the basement membrane, cytoplasmic granules in trophocytes stained magenta similar to equine hepatocytes using the PAS reaction. Also, striated muscle contained magenta-labeled sarcoplasmic granules in all species. While equine goblet cells stained magenta, dipterans exhibited magenta-stained intraluminal contents of mid- and hindgut as well as genital tract (egg chamber, gonoduct). Mosquitoes and fruit flies further displayed magenta-stained yolk, while only mosquitoes showed magenta salivary gland secretions and cytoplasmic granules in Malpighian tubule primary cells (Figs. 27–32, Supplemental Figs. S61–S68).
Applying PAS reaction after diastase pretreatment, cytoplasmic granules in striated muscle cells, equine hepatocytes, and dipteran trophocytes lost their magenta staining. Similar results were obtained for granules within primary cells of Malpighian tubules of mosquitoes, whereas yolk of fruit flies was only diastase-sensitive to a limited extent. In contrast, basement membrane had diastase-resistant magenta staining in all species examined. In addition, equine intestinal goblet cells, intraluminal contents of midgut, hindgut, and genital tract of both dipteran species, salivary gland secretions, and yolk of mosquitoes had diastase-resistant magenta staining (Figs. 33–38, Supplemental Figs. S69–S76).
The cationic dye toluidine blue demonstrated polyanionic glycoproteins (sulfated and carboxylated mucins) through its metachromatic properties similar to CV in the combined LFB-CV stain. 13 Employing toluidine blue, pink to violet staining was observed across all species examined. Pale pink- to violet-stained intraluminal contents of mid- and hindgut were visible in both dipterans, whereas equine tissue exhibited pink- to violet-stained intestinal goblet cells (Figs. 39–41). Pink-stained cytoplasmic granules were seen in Malpighian tubule primary cells of mosquitoes along with pink intraluminal contents of the egg chamber. Fruit flies exhibited pink contents in the gonoduct (Supplemental Figs. S77–S83).
Stains for Minerals
Von Kossa silver stain visualized the presence of calcium salts in brown to black in mosquito and equine tissue. In equine tissue, black-stained calcium deposits were visible in non-decalcified nasal conchae. In mosquitoes, cytoplasmic granules were stained brown to black in Malpighian tubule primary cells. Interestingly, no staining of calcium salts was observed in fruit flies (Figs. 42–44, Supplemental Figs. S84–S88).
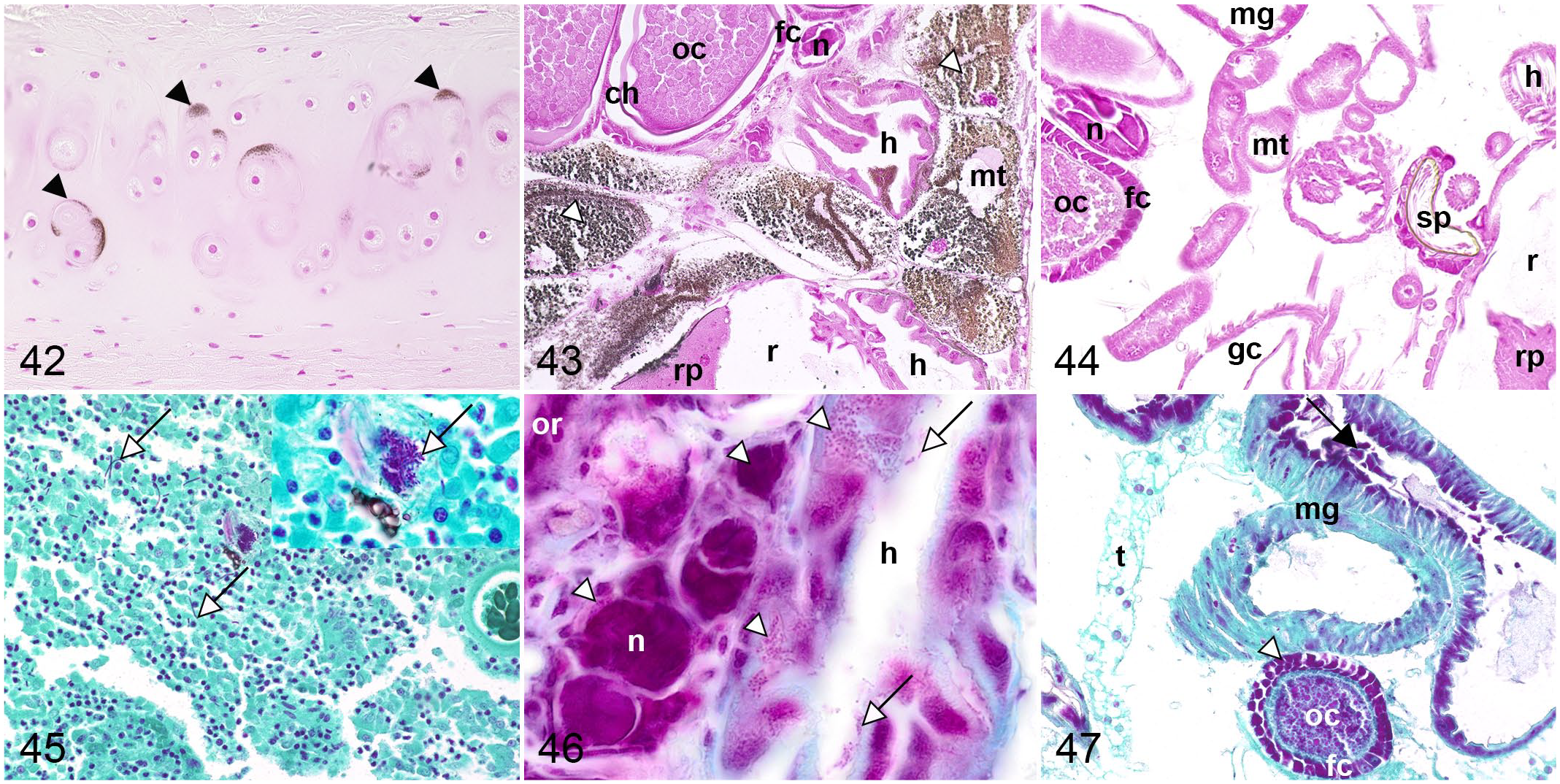
Comparative histochemical staining of minerals and selected microorganisms in horse, mosquito, and fruit fly.
Stain for Microorganisms
Application of Gimenez staining, commonly used for detection of bacteria in tissue samples, stained microorganisms in magenta in equine and both dipteran species. In equine tissue, microorganisms were only detected to a limited extent within the intestinal lumen. In contrast, apart from the aforementioned microorganisms within intestinal lumina, both dipterans showed magenta, coarse to finely granular cytoplasm in ovaries, mosquitoes in hindgut epithelium, and fruit flies in cortical ganglion cells (Figs. 45–47). Similar staining patterns were displayed in salivary gland, cardia/proventriculus, and midgut epithelium of mosquitoes and fruit flies. Regardless of microorganism detection, nuclei in dipterans and mammals, goblet cells in equine, tentorium and Malpighian tubules in mosquitoes and fruit flies, and oenocytes and yolk in fruit flies were irregularly and homogeneously stained in magenta (Supplemental Figs. S89–S96).
Discussion
Insects play an important role in ecosystems all over the world. 31 In recent years, not only an alarming decline in beneficial insects has been observed but also a simultaneous increase in insects harmful to the environment, economy, and public health.21,45,48,56,63 Therefore, detailed knowledge of insect staining characteristics could be beneficial as a basis for further surveys, whether in the context of species conservation, control, or vector research.
Histochemical staining plays an important role in morphologic investigations for diagnostic or research purposes visualizing physiological tissue structures and alterations. As for mammals, H&E staining provided a detailed overview of microscopic anatomy facilitating differentiation between cellular and extracellular tissue components. Thus, HE staining represents a valuable routine stain in mosquitoes and fruit flies as observed for equine tissue.
Insect ganglia are composed of a cortex with numerous cell bodies (neurons and glial cells) and a centrally located neuropil with cell processes (dendrites, unmyelinated axons, and glial cell protrusions) and synapses. 29 Bielschowsky silver stain and LFB-CV highlighted the structure of nervous tissue in mammals and insects, with Bielschowsky silver stain allowing the tracing of individual cell processes. Using CV in combined LFB-CV staining, nuclei as well as rough endoplasmic reticulum of neurons and cortical ganglion cells could be visualized in all species. Interestingly, LFB-CV stained neuropil in mosquitoes and fruit flies uniformly blue comparable with equine samples despite the lack of myelin in the aforementioned dipteran species. 24 Therefore, staining of insect neuropil was interpreted as reactivity of luxol fast blue with other phospholipids. 51 PTAH staining did not highlight glial fibrils as seen in equine tissue, nor did it result in consistent or specific staining of cortical ganglion cells in mosquitoes and fruit flies. 32 Therefore, PTAH is not recommended for visualization of glial cells in insect species.
Connective tissue of Diptera mainly consists of the basement membrane that lines epidermis, muscle, gut, fat body, glands, respiratory and reproductive organs, occurs as part of the neural lamella, 3 or fine banded fibrils. 5 Moreover, a few insects possess modified connective tissue that is unique to them, such as in the midgut or ovarioles of mosquitoes, 7 rendering classical mammalian histochemical techniques for connective tissue less diagnostically conclusive. Gomori’s reticulin stain and PAS, but not PTAH, are useful to evaluate the basement membrane in mammals and insects investigated, based on the formation and labeling of free aldehydes of its carbohydrate-rich matrix. 35 Alcian blue visualized fine fibrils multifocally below the integument in both dipterans with a stronger presentation in fruit flies. In contrast, Gomori’s reticulin stain, Heidenhain’s azan, Masson-Goldner, and PTAH failed to highlight fine fibrils in either dipteran species investigated. However, EVG, Heidenhain’s azan, and Masson-Goldner visualized the midgut-associated connective tissue in C. pipiens biotype molestus, with Heidenhain’s azan and Masson-Goldner also staining connective tissue associated with the reproductive tract, which was previously described in A. aegypti. 7 This staining pattern indicates a loose protein network within modified connective tissue of mosquitoes, as observed in mammals, allowing large-molecule dyes to penetrate these tissue fibers. 23 At the same time, no comparable staining pattern was observed in the gastrointestinal or reproductive tracts of fruit flies. Unlike in mammals, EVG was not useful to demonstrate elastic fibers in mosquitoes or fruit flies, possibly due to their different structure and composition.1,5,14
As in mammals, Heidenhain’s azan, Masson-Goldner, and LFB-CV stained musculature in mosquitoes and fruit flies. Therein, the pink or red staining with the anionic dyes (Heidenhain’s azan and Masson-Goldner) indicates a dense protein network within the dipteran musculature, 23 which can also be seen in proteinaceous granules in trophocytes or egg yolk of insects. However, PTAH was best at highlighting musculature, as previously reported, 6 given that striated, smooth, and heart musculature were ascertainable. As this dye also binds to Z-bands, 47 cross-striation of muscle fibers was most obvious with PTAH in equine and dipterans, which renders it particularly interesting for the evaluation of myopathies in all species investigated.
Glycans and carbohydrates play a crucial role in structure, metabolism, and physiology of biological systems as well as in disease. 61 While some of these molecules are highly conserved across species, others vary immensely within and between species. 61 Interestingly, despite their high specialization with marked interspecies differences, acidic polysaccharides were detected with alcian blue in the gastrointestinal tract of both dipterans comparable with stomach and intestine in mammalian species.11,55 Acid mucins visualized intraluminally in the genital tract of mosquitoes and fruit flies indicate secretory activity of epithelial cells analogous to that of mammals. 26
PAS reaction revealed neutral polysaccharides in several organs in equine and dipteran samples. The most prominently highlighted cell types in dipterans were trophocytes and striated muscle cells, which lacked magenta staining with PAS reaction after previous diastase pretreatment. This is comparable with mammalian hepatocytes and muscle cells and characterizes the stained substance as glycogen, 18 which is stored as an energy source in trophocytes and striated muscle cells of insects analogous to mammals.2,12,42 Together with Heidenhain’s azan and Masson-Goldner staining, which present protein granules within trophocytes, one could possibly use the PAS reaction to draw conclusions about the nutritional status of endangered insects or the reduced reproductive capacity of arbovirus-infected vectors. Interestingly, glycogen was also detected within Malpighian tubules of mosquitoes and fruit fly yolk, indicating nutrient storage.9,10 The structures that retain the magenta color with PAS staining after diastase pretreatment of the dipterans investigated here corresponds to those of mammals. Neutral mucins were predominantly present in epithelial cells, such as salivary glands,49,53 gastrointestinal tract,11,55 or genital tract. 26 Moreover, neutral polysaccharides were detected in egg yolk of both dipteran species, especially mosquitoes, representing carbohydrate-rich nutritional components for offspring. 9
Toluidine blue and CV revealed polyanionic glycoproteins in gastrointestinal and genital tracts similar to alcian blue in both dipteran species. Metachromatic staining was further detected within Malpighian tubule primary cells of mosquitoes, with CV and especially toluidine blue indicating species-specific intracellular molecules such as sulfated glycoproteins. 44
Von Kossa staining visualized mineralized tissue within equine nasal turbinates, while no comparable reaction pattern was observed in mosquitoes or fruit flies. This was attributed to the lack of mineralized bone in insects, which instead possess a sclerotized and chitin-containing exoskeleton 62 that is not demonstrable with von Kossa stain. In addition, insects do not react to tissue damage with deposition of calcium salts as regularly seen in mammals, but with melanization, enclosing damaged tissue. 40 However, von Kossa stained cytoplasmic granules of Malpighian tubule primary cells in mosquitoes, but not in fruit flies. Principally, von Kossa stain exchanges calcium in carbonates or phosphates for silver ions that are consecutively reduced to metallic silver. 36 As calcium concrements have been described in primary cells in mosquitoes, 8 the detected granules were interpreted as calcium deposits. However, the stain does not only react with calcium ions but also with inorganic salts. 36 Components such as carbonate, sulfate, or chloride 44 are likewise present in Malpighian tubules, so it cannot be completely ruled out that the reaction visualized anionic aggregates.
Gimenez staining is known to visualize a variety of bacteria, including Rickettsiae. 19 This stain successfully detected microorganisms in intestinal lumina of all species examined. Interestingly, bacteria were detected intracellularly in multiple organs in mosquitoes and fruit flies. As both C. pipiens biotype molestus and D. melanogaster are naturally infected with the intracellular bacterium W. pipientis, which belongs to the order Rickettsiales, these microorganisms might represent Wolbachia. This bacterium infects over 40% of all insect species 22 and mainly relies on vertical transmission. Therefore, density is usually highest within the reproductive system, 64 but they also occur in extragonadal tissue such as head and body ganglia17,46 in both dipterans, and in hindgut of C. pipiens biotype molestus as observed in the present study. Still, it cannot be excluded that other microorganisms besides W. pipientis are detected within samples investigated. Especially positive signals detected within the gastrointestinal lumen could be related to the physiological microbiome. Presence of W. pipientis has not been described in organs like salivary gland, cardia/proventriculus, or midgut of mosquitoes and fruit flies within previous studies. However, in this investigation, a coarse, magenta-stained cytoplasm was observed in salivary gland, midgut, or cardia in both dipterans. This observation requires further investigation to determine the specificity of this signal, while variable and homogeneous staining of nuclei, tentorium, Malpighian tubules, and yolk needs to be considered as nonspecific. Therefore, Gimenez staining should be evaluated critically and rather serve as an initial screening tool for FFPE material of Wolbachia-infected insects.
The dipteran integument is composed of multiple substances, including chitin, proteins, and glycoproteins. 62 Visualization was achieved by multiple stains in mosquitoes and fruit flies. Thus, H&E and PAS visualized neutral polysaccharides such as chitin, while LFB-CV indicated lipids, and Heidenhain’s azan and Masson-Goldner staining suggested dense protein cross-links. Arthrodial membranes and apodemes were distinguishable in mosquitoes and fruit flies using EVG, Heidenhain’s azan, and Masson-Goldner, denoting a rather loose composition of protein cross-links in these areas. 23 In addition, not only the cuticle but also tracheae and tracheoles were displayed by silver impregnation according to Gomori’s reticulin stain and Bielschowsky silver stain, which is due to argentaffin properties of the cuticulin layer of the cuticle. 67 In particular, Gomori’s reticulin stain allowed visualization of fine, delicate tracheole branching within multiple tissue structures in both mosquitoes and fruit flies.
A general but critical aspect is possible overstaining of insect species due to a prolonged incubation time in histochemical dyes or insufficient differentiation in consecutive solutions, especially when applying Gimenez and LFB-CV. Consequently, critical assessment of staining results, as well as a frequent control under the microscope during the staining procedure, is highly recommended to prevent such discoloration. Further details regarding staining techniques can be found in Supplemental Material.
In summary, this study provides insights into comparative staining characteristics in mammals and dipterans using various histochemical techniques, and addresses their usefulness for histological studies in insects. Multiple stains proved to be valuable tools for assessing C. pipiens biotype molestus and D. melanogaster samples. Several histochemical stains applied were suitable for tissue assessment in insects as well as mammals, in particular for nervous tissue (Bielschowsky silver stain, LFB-CV) and polysaccharides (alcian blue, PAS with and without diastase treatment, toluidine blue). Some stains proved useful for visualization of insect-specific organ characteristics such as Gomori’s reticulin stain for tracheoles in both dipteran species. Others were only applicable for 1 species like Heidenhain’s azan for midgut-associated connective tissue or von Kossa for mineral deposition in Malpighian tubules of C. pipiens biotype molestus. Histochemical staining techniques are of great importance in the study of insects, whether in the context of species conservation, control of insect pests, or monitoring of food resources. In addition, histochemical studies may provide further insights into the growing field of vector research and enable detailed investigations of insects as part of pathogenetic studies of known and emerging vector-borne diseases.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221088786 – Supplemental material for Histochemical staining techniques in Culex pipiens and Drosophila melanogaster (Diptera) with a comparison to mammals
Supplemental material, sj-pdf-1-vet-10.1177_03009858221088786 for Histochemical staining techniques in Culex pipiens and Drosophila melanogaster (Diptera) with a comparison to mammals by Katharina M. Gregor, Stefanie C. Becker, Fanny Hellhammer, Kathleen Schön, Wolfgang Baumgärtner and Christina Puff in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Julia Baskas, Petra Grünig, Hella Heidtmann, Valery Kozalla, and Caroline Schütz for their excellent support.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Katharina M. Gregor received financial support by the Grimminger-Stiftung für Zoonoseforschung, Stuttgart, Germany. Kathleen Schön’s position is funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, 398066876/GRK 2485/1).
Data Availability Statement
The data that support the findings of this study are included within the main document or supporting supplemental material. The remaining data, which are not included in the main document or supplemental material, are available from the corresponding author upon reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
