Abstract
Marine bivalves are commonly affected by disseminated neoplasia of presumed hemocytic origin (i.e., hemic neoplasia and hemocytic neoplasia). Histopathology of 520 cultured hard clams (Mercenaria mercenaria) from Florida was performed for health surveillance over a consecutive 13-month period. Disseminated neoplasia was identified in 9 of 520 animals (1.7%). The neoplasia was characterized by the presence of large, round to oval, anaplastic cells within hemolymphatic vessels and sinusoids with variable infiltration into adjacent connective tissues of the visceral mass, mantle, foot, and/or adductor muscles. Frequent involvement and/or infiltration of the gill was also identified (5/9). Disseminated neoplasia in other species of clams, mussels, and cockles is considered a transmissible disease. At this time, it is unknown if these hard clams represent de novo development of the disease or potential transmission; however, this report expands both the geographic and host range for this condition.
Disseminated neoplasia of presumed hemocytic origin is one of the most commonly described neoplasms of invertebrates and has been reported in 16 marine invertebrate species, including 15 species of marine bivalves, as well as white shrimp (Litopenaeus setiferus). 8 Historically, this leukemia-like disease has gone by many names, including hemic neoplasia, hemocytic neoplasia, hemic sarcoma, hematopoietic neoplasia, and hemocytic leukemia; however, the more general moniker, disseminated neoplasia, appears to have been used with the most consistency in the literature. 1 The reported histologic features are similar across species and include the presence of large, round, anaplastic cells within hemolymphatic sinusoids, vessels, and connective tissues of the visceral mass, adductor muscle, foot, and mantle. 8 As the disease progresses, the neoplastic cells expand vessels and sinusoids and infiltrate tissues, resulting in subsequent organ dysfunction and ultimately death. 8
Transmissible tumors, such as Tasmanian devil facial tumors and transmissible venereal tumors in dogs, are well documented in veterinary literature. In addition, transmissible neoplasia has been documented in numerous marine bivalve species.2,3,5–9 Prior investigations, first in soft-shell clams (Mya arenaria) and since in multiple marine bivalve species, have demonstrated that this neoplastic disease results from the transmission of a neoplastic cell to a naive animal through the water column.2,5,7 The neoplastic cell lineages tend to be specific for different bivalve species; however, cross-species transmission can occur.5,6 To date, seven lineages of transmissible neoplastic hemocytes in eight marine bivalve species, including clams, mussels, and cockles, have been reported. 3 Considering the greater number of documented species, transmissible neoplasia appears to be more common in the marine environment than other terrestrial veterinary species, which has important implications given the economic and commercial value of these cultured species.
The purpose of this publication is to provide veterinary pathologists with a descriptive reference to aid in the recognition and diagnosis of disseminated neoplasia in marine bivalves, especially hard clams, particularly as bivalve transmissible neoplasia continues to expand its host range and/or geographic distribution.
A total of 520 cultured, approximately 2-year-old, hard clams (Mercenaria mercenaria) were examined for disease surveillance from January 2021 to February 2022. The hard clams were cultured on commercial farms located in two aquaculture use zones (AUZs) in the Gulf of Mexico off Cedar Key, Florida: Gulf Jackson AUZ to the west and Dog Island AUZ to the east. Hatchery-produced clam seeds were planted in coastal waters on submerged lands leased from the State of Florida. To protect the seed from predation during growout, clams were reared in bottom bags made of a polyester mesh material, which contained the clams and served as harvesting devices. The bags were staked to the bottom substrate, and naturally occurring sediments allowed the bag and clams to bury. Stocked at about 75 clams per square foot, the clams reached market size in 12–18 months depending on when the seed are planted seasonally. At the time of harvest, the clams were randomly selected and collected from growers for health assessments. Every month, 40 clams, 20 animals from each site, were submitted for postmortem examination and histologic interpretation (n = 520). Each animal was photographed and measured, and sterilely collected fresh tissue samples from the visceral mass were preserved in RNAlater (Thermo Fisher, United States) and saved frozen at −80°C.
Following at least 24 hours of fixation in 10% buffered formalin, multiple samples of gill, foot, mantle with mantle edge, adductor muscle, and/or visceral mass that included labial palps, esophagus, stomach with gastric shield, styles, intestine, rectum, anus, heart, kidney, ganglia, and gonads were submitted for each animal. Samples were routinely embedded in paraffin, and sections of formalin-fixed paraffin-embedded tissues were stained with hematoxylin and eosin according to routine laboratory procedures.
Of the 9 animals diagnosed with disseminated neoplasia, a mild (approximately 10%) decrease in visceral mass size was noted in 2 of the clams, and a marked (approximately 50%) decrease in visceral mass size with mantle edge recession was observed in 1 (Fig. 1). The surface of the visceral mass should be smooth and firm, and in this animal, the visceral mass was markedly reduced in size, soft, thin, and edematous. This is a nonspecific and subjective change that can suggest disease or potentially reproductive stage depending on severity. Similarly, there was recession of the mantle edge evidenced by the decreased distance between the mantle and shell edge, which has been reported in weak/sick bivalves. 7
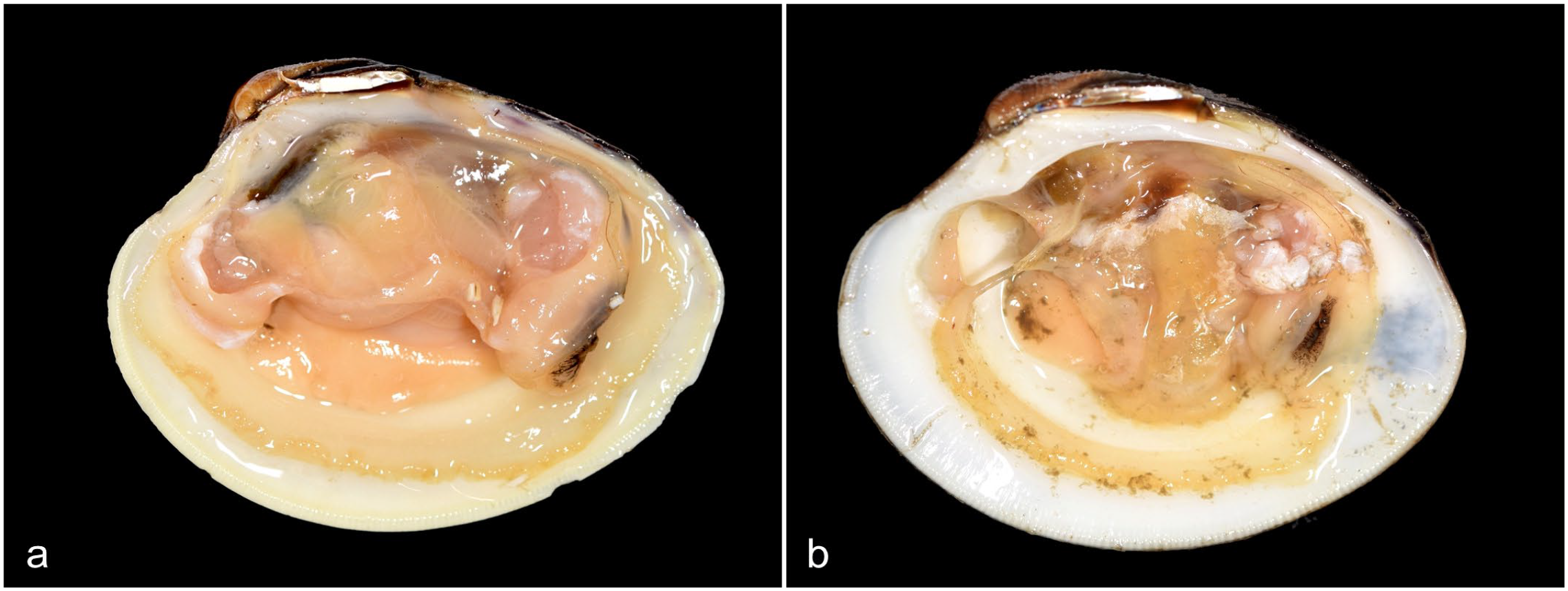
Images of normal and affected hard clams with the umbo (anterior) oriented to the left. (a) Unaffected male with expected visceral mass size. (b) Diseased male with disseminated neoplasia exhibits a markedly decreased visceral mass size and recession of the mantle.
Disseminated neoplasia was present in 9 from the 520 animals surveyed (1.7%). Six of these animals were female (6/9), and three were male (3/9). Most cases occurred during the coldest season in December and February (6/9). One case occurred in April (1/9), and 2 cases occurred in September and October (2/9). No cases were diagnosed during the summer. The significance of this seasonality is unclear, and a larger data set would be needed to deduce any casual relationship.
Different distributions of the neoplasia were identified (Fig. 2). In some cases, the neoplastic cells were in single or few hemolymphatic vessels and/or sinusoids without significant infiltration into connective tissues (Fig. 2). Presumptively, these milder lesions reflected an earlier stage of the disease. In more severe cases, the neoplastic cells multifocally expanded, infiltrated, and variably distorted the architecture of the connective tissues, gill, and/or labial palps (Fig. 2). Neoplastic hemocytes were arranged in dense sheets to loose cords. Individual neoplastic cells were approximately 2–4× the diameter of a normal hemocyte, round to oval, with variably distinct cellular borders and scant to moderate amounts of amphophilic cytoplasm. Nuclei were large, round to oval, and sometimes indented with coarsely clumped chromatin and indistinct nucleoli. Anisocytosis and anisokaryosis were moderate to marked with occasional binucleation and karyomegaly. Frequent mitotic figures were identified (up to 7 mitotic figures in ten 400× fields [2.37 mm2]).
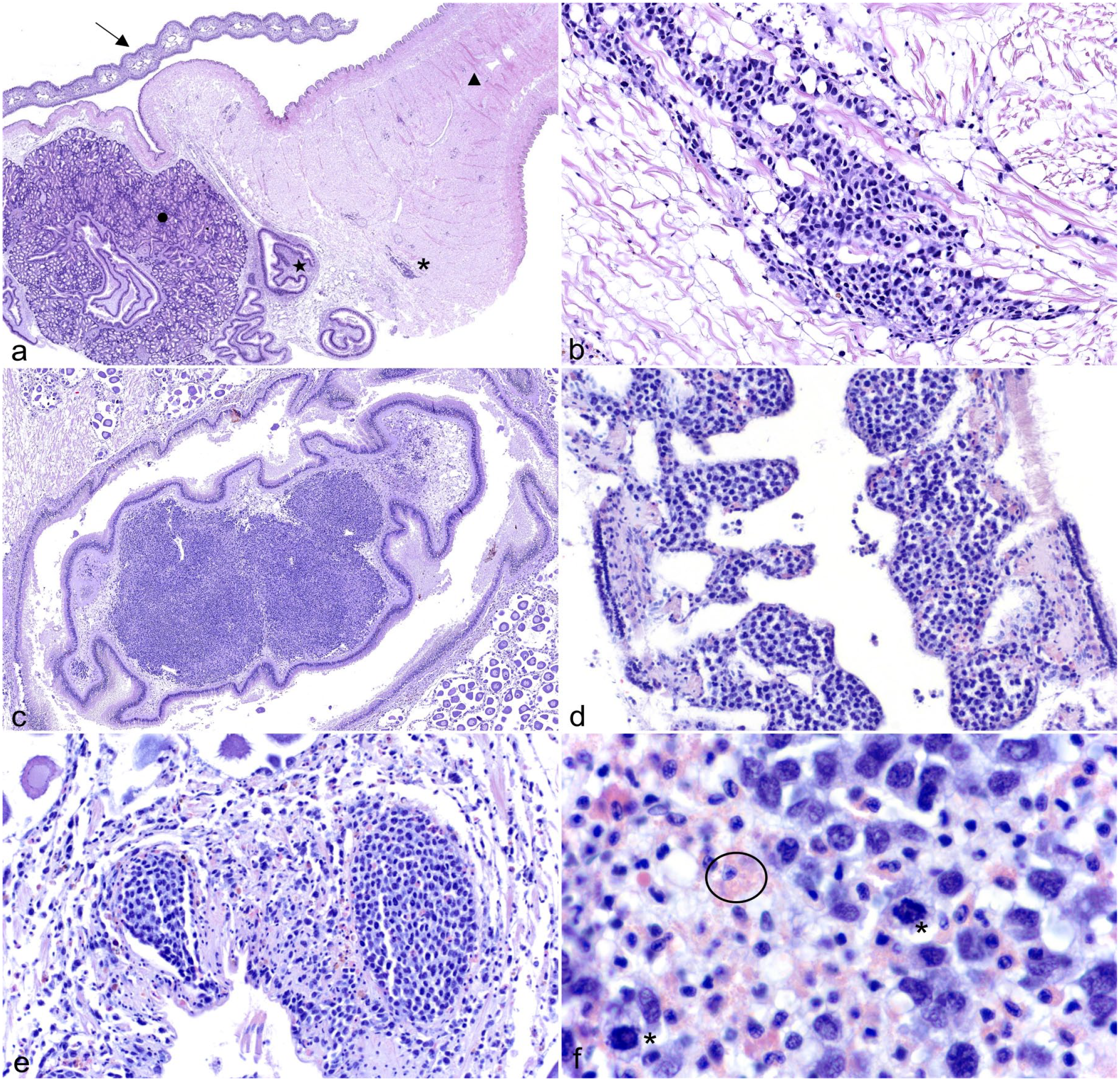
Histopathologic features of disseminated neoplasia in hard clams. Hematoxylin and eosin. (a) Transverse section through visceral mass to include gill, striated muscle of the gill (arrow), foot (triangle), descending intestine (star), and digestive diverticula (circle). Small aggregates of the neoplastic cells fill vessels and sinusoids and/or infiltrate into adjacent connective tissues (asterisk). (b) Connective tissue of visceral mass. Higher magnification of the neoplastic cells infiltrating the connective tissue, which corresponds to the asterisk in (a). (c) The connective tissue of the typhlosole in the descending intestine is infiltrated and expanded by the neoplastic cells forming dense sheets. (d) Gill. Branchial filaments are expanded by neoplastic cells. (e) Hemolymphatic vessels adjacent to female gonads. In more severe cases, there is partial to full occlusion of the vessels by the neoplastic cells. (f) High magnification of the neoplastic population. The neoplastic cells exhibit occasional mitosis (asterisk). Compare the nuclei of the neoplastic population to adjacent circulating granular hemocytes (circled).
All cases of disseminated neoplasia involved vessels and sinusoids (9/9), and in 5 of 9 cases, there was infiltration of connective tissues of the visceral mass and/or foot. Most cases also involved the gill, and in most cases, neoplastic cells infiltrated the gill (5/9). One case had renal involvement (1/9), and another case had infiltration to the labial palps (1/9).
Additional neoplastic findings included gonadal neoplasia of germ cell origin in 1 male and 1 female clam (2/520; 0.38%). Neither of the affected clam was concurrently diagnosed with disseminated neoplasia, and the gonadal neoplasia was localized to the gonads without vascular and/or sinusoidal involvement. Gonadal neoplasia has been previously reported in Mercenaria mercenaria from Florida. 4 The remaining findings were considered incidental and/or associated with common parasites, including metazoan larvae (224/520; 43%) and/or Chlamydiales-like organisms within digestive diverticula (4/520; 0.76%).
This report describes a neoplastic process morphologically similar to disseminated neoplasia reported in other species of marine bivalves. Increased mortality rates were not identified in the 2 sampled populations during the survey period, and the overall prevalence was low (9/520; 1.7%). Gross lesions were identified in 3 affected individuals, which included mantle recession, emaciation, and/or reduction in the size of the visceral mass. These changes are consistent with previously reported findings observed in bivalve cases of disseminated neoplasia.1,8 Concurrent gametogenesis via gonad atrophy and degeneration has also been previously reported. 8 Three of the 9 neoplastic cases had moderate to severe gonadal depletion; however, it is unclear if this represents physiologic atrophy following spawning or pathologic atrophy subsequent to the neoplasia.
At the time of this report, these findings are not a major concern for the clam industry in northwest Florida. To date, no significant problems subsequent to an infectious disease have been identified in culture clams from Florida waters; however, the cultured clam industry in Florida is of great economic and ecological importance, so continued investigation and surveillance is needed.
To the authors’ knowledge, this is the first histopathologic description of this neoplasia in hard-shell clams (Mercenaria mercenaria) off the coast of Cedar Key, Florida, which expands both the geographic and host species range of this disease. At this time, it is unclear if these cases are de novo development, the result of interstate transport of cultured clam seed from the Northeast United States, or potential transmission from wild populations. Samples from this work were saved for potential future genotyping, as has previously been reported with this tumor entity. In addition, investigation to confirm the origin of the neoplastic cells, the mode of transmission of this disease in this species, and to potentially determine the cause of neoplastic transformation may be warranted. Ultimately, this report provides a descriptive reference for veterinary pathologists to aid in the recognition and diagnosis of disseminated neoplasia in marine bivalve molluscs, particularly if this disease continues to expand its geographic location and/or host range.
Footnotes
Acknowledgements
The authors would like to thank the incredible histotechnologists at the University of Florida Histology Laboratory for their hard work and technical assistance. They would also like to thank Dr Roxanna Smolowitz for her initial consultation and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by faculty (JR) start-up funds at the University of Florida College of Veterinary Medicine.
