Abstract
Histopathologic data of millipedes are scarce. Little is known about health and disease of these invertebrates despite their exhibition at zoological institutions and use in ecotoxicological studies. In a retrospective study of 69 zoo-housed giant African millipedes (Archispirostreptus gigas) submitted between 2018 and 2021, most deaths occurred during midwinter and in 2021. The most common lesion was inflammation (n = 55; 80%). Necrosis was seen concurrently in 31 (45%) millipedes and of these, bacteria (20; 29%) and fungi (7; 10%) were detected in lesions. Inflammation was seen in the head/collum (20; 29%), hemocoel (16; 23%), and appendages (9; 13%), specifically in perivisceral fat body (42; 61%), gut (16; 23%), tracheae (26; 38%), skeletal muscle (24; 35%), and ventral nerve (17; 25%). Inflammatory cell types and patterns included agranular hemocytes (61; 88%), granular hemocytes (39; 57%), and nodulation/encapsulation (47; 68%) often accompanied by melanization. The oral cavity or gut (ingestion), spiracles (inhalation), or cuticular defects were considered plausible routes of bacterial entry. Metazoan parasites (adult nematodes: 2, 3%; trematode ova: 2, 3%; and arthropods: 1, 1%) were associated with gut necrosis and inflammation in 5 millipedes. In addition, adult nematodes were noted in the gut of 4 millipedes without lesions. Neoplasia was not detected in any millipedes. Speculatively, environmental factors may have predisposed to disease, as most deaths occurred during winter months. Disease surveillance of millipedes is critical to optimize husbandry practices in zoo populations and investigate potential impacts of environmental degradation and climate change on wild millipedes.
Subphylum Myriapoda includes classes Symphyla, Pauropoda, Chilopoda (centipedes), and Diplopoda (millipedes). Diplopods (millipedes) are characterized by 2 pairs of legs on most body segments. There are currently over 12,000 described millipede species, but classifications remain highly controversial and commonly undergo revisions. 2 Interestingly, while the term “millipede” translates to “a thousand feet,” it was only in 2020 that the first true millipede with 1,306 feet was discovered in Western Australia (Eumillipes persephone). 20 Millipedes are found on every continent except Antarctica and are extremely important detritivores, the third-most following earthworms and termites. 14 Although millipedes can be highly destructive agricultural pests, they are still considered keystone species in their ecosystems and are used as indicator species in ecotoxicology studies, 6 evaluating soil contamination and other environmental pollutants.9,14,36
Millipedes have varied morphologies and sizes, some with vibrant colors, and are generally docile, making them ideal for zoological exhibits and educational encounter programs. They may be housed successfully in mixed species exhibits as they are nonaggressive, and most are herbivorous. 4 The largest extant species is the giant African millipede (Archispirostreptus gigas), with a natural range spanning the forests of East Africa. A. gigas are a particularly popular species in zoological collections and are increasingly being kept as nontraditional pets due to their size (12–15 inches long and 2.5 inches diameter) and relatively long lifespan (5–7 years).
Understanding gross features and microanatomy of millipedes is crucial to enhancing diagnostic evaluations. Standardization of trimming and appropriate tissue processing protocols are also important to optimize microscopic data from these and other invertebrates. Millipede anatomy consists of a tube of gut surrounded by the perivisceral fat body (PVB; digestive system), Malpighian tubules (excretory system), spiracles and tracheae (respiratory system), skeletal muscle, and nerves, all encased in a cuticle composed of an epidermis, endocuticle, exocuticle, and epicuticle.4,25 The cuticle has numerous body rings composed of a hard calcareous and chitinous tergite with or without peripheral adornments (paranota). The body cavity consists of a hemocoel that is divided into three sinuses: pericardial (dorsal), perivisceral, and perineural (ventral). The gut has three parts: foregut, midgut, and hindgut, each with specific enzymatic and chemical digestive functions. 37 Myriapods, including diplopods (millipedes), have both tracheal systems and circulating hemocyanin in contrast to other terrestrial arthropods and most metazoans that use one or the other. 8 Millipedes have 2 pairs of legs on most body rings (diplosegments) except the first 5 body rings which have 1 set of legs (haplosegments). The tergite on body ring 1 is an elongated shield called the collum. The eyes are arranged into compound structures called ocelli. They have a pair of antennae, and ventral mouthparts composed of the gnathochilarium (mandible) and maxilla. Each body segment has a gland and an associated ozopore through which they secrete defensive substances. Legs are attached to the body rings ventral and centrally at the sternite. Also located on the sternites are stigma (spiracles) which are the openings of the dense tubular respiratory system (tracheae). Males have specialized reproductive legs at the seventh body segment called gonopods. The gonads for both sexes are located within body segment 2.
Like other invertebrates, the immune cells of millipedes are referred to as hemocytes, which circulate within hemolymph among hemolymphatic vessels and sinuses, and include different phenotypes and mechanisms of action. 17 Primary hemocyte types found in hemolymph are prohemocyte (progenitor cells), plasmatocytes, and granulocytes.17,26 Like vertebrate immune cells, hemocytes can be activated and differentiated depending on the stimulus or insult and carry out phagocytosis, wound healing, hemolymph clotting, and microbicidal functions. 17 Histologically, granulocytes appear as granular hemocytes, which are round with eccentric to central oval nuclei and eosinophilic granular cytoplasm.17,25 Activated plasmatocytes are referred to as agranular (or hyaline) hemocytes, and function as phagocytic cells with homogenous, foamy, or debris-laden cytoplasm, and lack cytoplasmic granules.17,25 Important processes of invertebrate inflammation are nodulation, encapsulation, and melanization. Nodulation occurs when small microbes such as bacteria infect the hemocoel and are surrounded by nodular aggregates of hemocytes that over time degranulate and deposit melanin around the infectious agent.17,26 With chronicity (1–2 days), the hemocytes form peripheral concentric bands of flattened, spindloid cells that surround the nodule of inflammation. Encapsulation occurs with pathogens that are too large to be phagocytosed and is otherwise similar to nodulation. 25 As offending agents are often not detected in the section, these inflammatory aggregates can be more generally termed “nodulation/encapsulation.” Melanization, which can occur within foci of nodulation/encapsulation or in areas of infection or tissue destruction, represents accumulation of brown melanin pigments produced by hemocyte enzymatic activity.17,26
Information regarding millipede health and disease is limited. There are several reports describing presumably commensal relationships with various metazoans including paraphagic or phoretic mites (order: Mesostigmata), 38 gastrointestinal nematodes (superfamilies: Rhigonematoidea, Thelastomatoidea, Diplogastridae, Rhabtidae, and Ransomnematoidea),3,4,19,24,29,38 and fungi (order: Laboulbeniales).30,33,34 Order Laboulbeniales is a highly specialized group of fungi that lives only on arthropods with high species specificity and parasitism appears benign to the host. However, some fungal species penetrate the host cuticle, which is speculated to open pathways for other pathogens. 30 The few reports on truly parasitic metazoans and fungi include Gordius spp. (parasitic nematodes that use millipedes as their definitive hosts and may manipulate host behavior), Brachycybe spp. (a fungus lethal to millipedes), Arthrophaga myriapodina (an entomophthoralean fungus that induces predeath climbing behavior), and Myriophora spp. (parasitic phorid flies that use healthy millipedes as hosts for larval development).5,12,13,18 Some mites are purported to transmit pathogens such as viruses and bacteria between invertebrate hosts, 27 although viral infections causing disease or mortality in millipedes have yet to be officially documented. 4
There are reports of Puerto Rican white and giant African millipedes with midgut ulcers and transmural hemocytic inflammation associated with rhigonematid nematodes, resulting in digestive gland atrophy and thin body condition. 26 Other documented millipede diseases appear to be husbandry-related, and many involve cuticular desiccation. 4 Millipedes are extremely prone to desiccation for many reasons including low chitin and high calcium content of their cuticles and from fluid losses through their gas exchange system (open spiracles), gastrointestinal tracts, and during reproduction. 7 Wrinkled cuticles in young growing millipedes are thought to be due to defects in the chitinous part of the cuticle or abnormal calcification, both of which have may be due to environmental and husbandry conditions including diet (lack of calcium, excessive protein), substrate (low pH), or the environment (suboptimal humidity, organic chemicals). 4 Additional diagnostic investigations of millipede diseases are required to better understand husbandry pitfalls and potential challenges millipedes may face with climate change and altered ranges of infectious agents and parasitic organisms.
This study includes a census of histologic lesions seen in a zoo-housed population of a popular millipede species over a 3-year period and provides recommendations to optimize diagnostic evaluations in millipedes for future disease surveillance.
Materials and Methods
A comprehensive database search for giant African millipede submissions to the Zoological Pathology Program (University of Illinois) was conducted. Giant African millipede submissions came from a single zoological institution (Fort Wayne Children’s Zoo) from December 2018 to December 2021. Mortality counts were available for most of this period (January 2019–December 2021). Millipedes were group-housed in clear plastic storage totes with either clear or solid-colored latching lids. No additional lighting was provided other than the room’s fluorescent lighting, which reflected natural light cycles. Substrate included peat moss and 1–2 pieces of cork bark to provide shelter. Diet included fresh apple, sweet potato, papaya, and various leafy greens (kale, endive, collard greens, and romaine lettuce) and was offered twice weekly. No additional potable water sources were provided. Temperatures and humidity were logged daily for each tote (Inkbird thermohygrometer). Temperatures ranged from 75°F to 80°F depending on substrate depth, seasonality, and tote placement near air-conditioning units. Substrate was misted as needed to maintain average 70% to 85% humidity. Tote cleanings were minimized to decrease disturbances to the millipedes.
Throughout the year, totes containing millipedes were transported to classrooms for 4-week intervals as part of an educational program. The temperature and humidity were not logged during transport though totes were transferred in insulated coolers containing hot water bottles in a temperature-controlled vehicle. When on program, students recorded data for their millipedes on a care sheet, including temperature, humidity, diet, and activity (mainly signs of molting). Instructors checked the care sheet every week to make sure the temperature and humidity were in safe range; however, instructors did not provide this information to the zoo unless a millipede required a veterinary evaluation or died prior to the end of the program. When totes were returned to the zoo, these individuals entered a quarantine period and were placed in new totes. Tote set up included a rinse using “Amphibian water” (reverse osmosis) with alkaline buffer and Replenish (Food Lion); drying; and placement of 2–3 inches of fresh peat moss. Millipedes used in the educational programs prior to August 2021 were born at the zoo and required to be adult-sized for enrollment, thus estimated to be 2–3 years old. The largest (adult) millipedes were not used in educational programs and were kept at the zoo to maintain the breeding colony. Millipedes acquired from other institutions in 2021 arrived as adults and ages were not known. The historically observed lifespan of millipedes at the zoo was approximately 5 years.
Millipede submissions were received immersion fixed in 10% neutral-buffered formalin, either whole or in 3 to 4 fragments. Gross exams were not performed prior to formalin fixation, though rare clinical notes were included with submission. During the study period, trimming was standardized to capture specific anatomical structures in histologic sections (Fig. 1a). Fixed millipedes were trimmed either transversely (Fig. 1b) or longitudinally (Fig. 1c) at the head and first to second body rings for evaluation of mouthparts, brain and ganglia, salivary gland, tracheae, and foregut. In some cases, longitudinal sections encompassed several body rings to include the foregut to midgut junction, (Fig. 1c) and rarely, the gonopods (males) were distinguishable (Fig. 1c, inset). Typically, transverse sections were made along the length of the body at second body ring to see gonads (male, Fig. 1d and female, Fig. 1e), the seventh body segment for cranial midgut and additional reproductive tract, and between approximately 50th–70th body rings to visualize additional midgut, PVB, Malpighian tubules, hemocoel, ventral nerve, and heart (Fig. 1g, heart not in field). The hindgut and rectum were represented with a longitudinal section at the end of body (Fig. 1f). Dechitinization was achieved by washing the sections in water for 20 minutes, then submerging in Perenyi’s solution (4 parts 10% nitric acid, 3 parts 100% ethanol, 3 parts 0.5% chromic acid) until chitin softened adequately (maximum 3 days). Specimens were then routinely processed, embedded, sectioned at 5 µm, and stained with hematoxylin and eosin. Brown and Hopps Gram stain was utilized in one case. Histopathology reports and slides were examined to confirm original diagnoses and cross-referenced with appropriate invertebrate microanatomy, immunology, and terminology.17,25,26 Each lesion and microscopic finding were tallied and categorized.
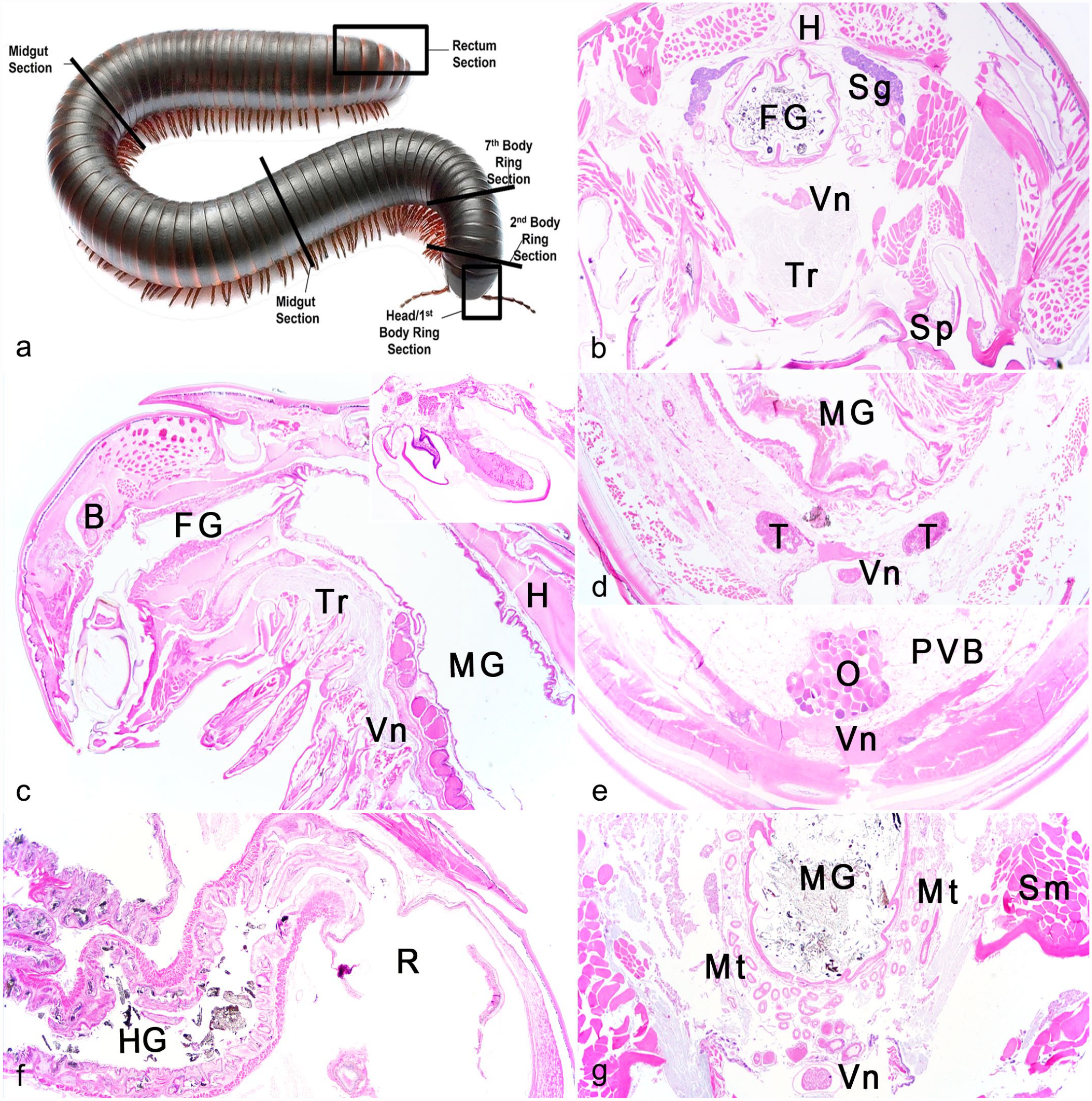
Suggested standard trimming for representative sections of millipedes.
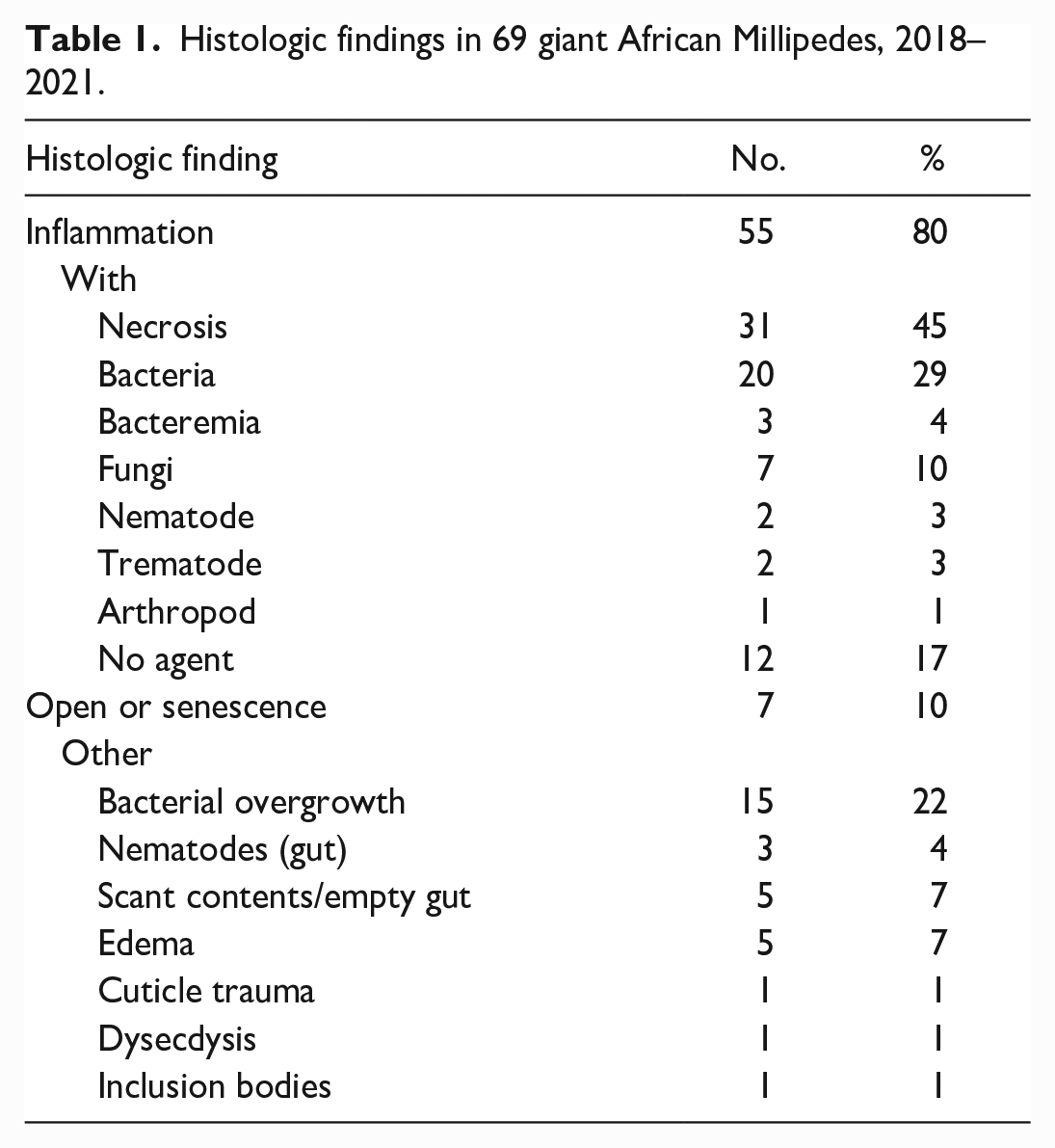
Histologic slides were initially examined by board certified pathologists and reevaluated together with the archived case history and original histology report to confirm lesions, interpretations, and diagnoses. Lesions and findings were tallied and categorized as follows: inflammation with concurrent necrosis, intralesional bacteria, fungi, or no agent; bacteremia (bacteria detected within hemolymphatics); open or senescence; and other based on prevalence and relevance to morbidity, mortality, or group health (Table 1). Inflammation was further characterized by cell type (agranular or granular hemocytes) and pattern including nodulation/encapsulation with or without melanization and location (body part and tissue/organ; Table 2).
Histologic findings in 69 giant African Millipedes, 2018–2021.
Characterization of inflammation in 69 giant African millipedes, 2018–2021.

Results
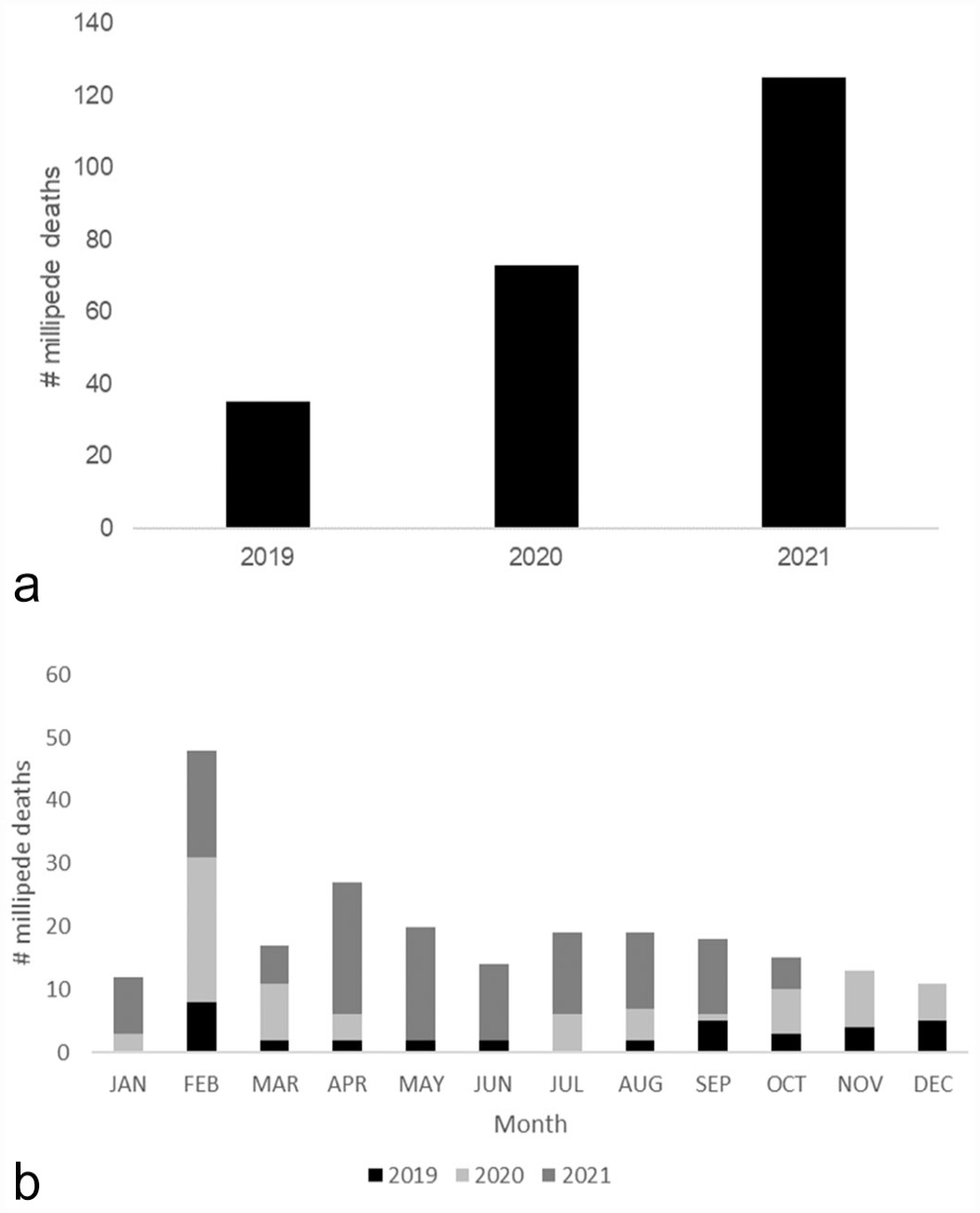
From January 2019 to January 2022, a total of 233 millipedes died or were euthanized. Millipede mortality was not logged in 2018; only 2 millipedes were submitted for histologic evaluation that year in December. Sixty-nine (approximately 30%) of these deceased millipedes were submitted for histologic examination after spontaneous death (65; 94.2%) or euthanasia (4; 5.8%). Euthanasia was performed by isoflurane overdose via chamber induction (soaked cotton ball in small sealable plastic bag) until all movement ceased, even with stimuli, before placing in formalin.
From 2019 to 2021, over half of deaths occurred in 2021 (125; 54%; Fig. 2a) and in all years, the majority of deaths occurred in February (48; 21%; Fig. 2b). In 2021, millipede deaths occurred steadily throughout the spring and summer months with a die-off occurring in May. Each millipede was reported to be an adult at time of death. Sex was recorded in some cases and was determined by detecting gonopods on the seventh body ring (male) or normal legs (female). The case histories consisted of 26 (38%) females, 17 (25%) males, and 26 (38%) of unknown sex. Histologically, 33 (48%) were confirmed as males and 30 (43%) as females, and the sex was not confirmed in 6 (9%) individuals due to lack of gonadal tissue present in examined sections. Gross lesions were rarely included in case histories, due to general lack of complete dissection and macroscopic evaluation. Reported gross lesions prior to fixation included softening of the cuticle, generalized pallor, focal white cuticular exudate, and large presumably ruptured ovarian follicles.
The most common lesion found in this group of zoo-housed millipedes was inflammation (55; 80%), which was accompanied by necrosis in 31 (45%) individuals. Intralesional bacteria or fungi were detected in areas of inflammation in 20 (29%) and 7 (10%) millipedes, respectively, while 12 (17%) millipedes did not have evident infectious agents. Inflammation was associated with adult nematodes in 3 (4%) cases and trematode ova in 2 (3%) cases.
Types of inflammation were characterized by cell type (agranular, granular, or both) and pattern including nodulation/encapsulation and noncapsular melanization (ie, melanization that was not associated with nodulation/encapsulation). Agranular hemocytic inflammation (46; 67%) was more common than granular hemocytic inflammation (16; 23%), although over 50% of millipedes had both agranular and granular hemocytic infiltrates. Nodulation was seen in 47 (68%) millipedes and characterized by nodular aggregates of hemocytes with or without concentric bands of attenuated to spindloid cells and central brown granular pigments (melanization). In 4 (6%) millipedes, severe inflammation and necrosis, either in the hemocoel or appendages, contained extracellular accumulations of brown pigment interpreted as noncapsular melanization.
The PVB (42; 61%) was the most commonly inflamed tissue/organ with varying severity, inflammatory cell types, and patterns. Inflammation of the PVB was predominantly seen in millipedes with inflammatory lesions and/or infections in other tissues/organs including the oral cavity/foregut, midgut, and hemocoel. In severe cases, the PVB was multifocally to regionally obscured by dense sheets of agranular hemocytes with or without granular hemocytes (Fig. 3a). In 7 millipedes, inflammation was limited to the PVB, consisting of few scattered foci of nodulation/encapsulation, and noncapsular melanization (Fig. 3b). Although infectious agents were not noted in many of these lesions, an infectious etiology was considered likely based on inflammation pattern (nodulation/encapsulation and noncapsular melanization). Two millipedes had 40 × 50 µm oval trematode ova with brown shells and internal eosinophilic material among mixed hemocytic inflammation and nodulation in the PVB. One individual had suspect intranuclear inclusion bodies in epithelial cells of the PVB, with adjacent multifocal nodulation/encapsulation (Fig. 3c). Some PVB epithelial cells were swollen with increased colorless cytoplasm, interpreted as degeneration. Additional investigations into possible viral infection were not conducted; however, other causes of inclusions were considered, including cytoplasmic invagination or nuclear accumulation of degenerative products.
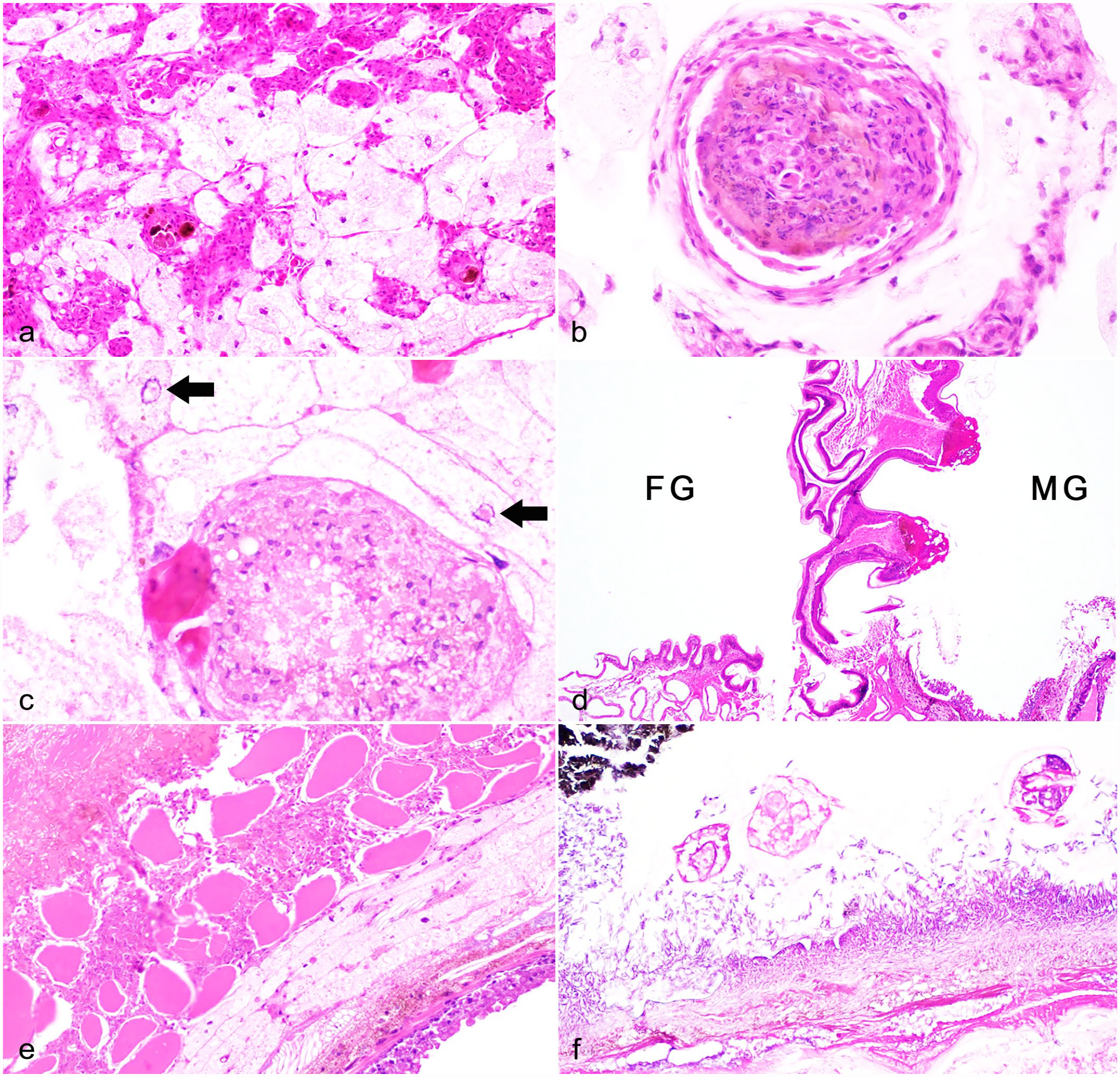
Inflammatory lesions in giant African millipedes. Hematoxylin and eosin.
Inflammation affected the head region in 20 (29%) millipedes, and often surrounded the oral cavity or foregut, regionally encompassing the tracheae, ventral nerve, and skeletal muscle, and rarely involving the salivary gland (2; 3%). Inflammation in the head occasionally extended into proximal body rings and was associated with erosions/ulcers in the oral cavity or foregut-midgut junction (Fig. 3d) or inflammation and debris within spiracles (Fig. 4a).
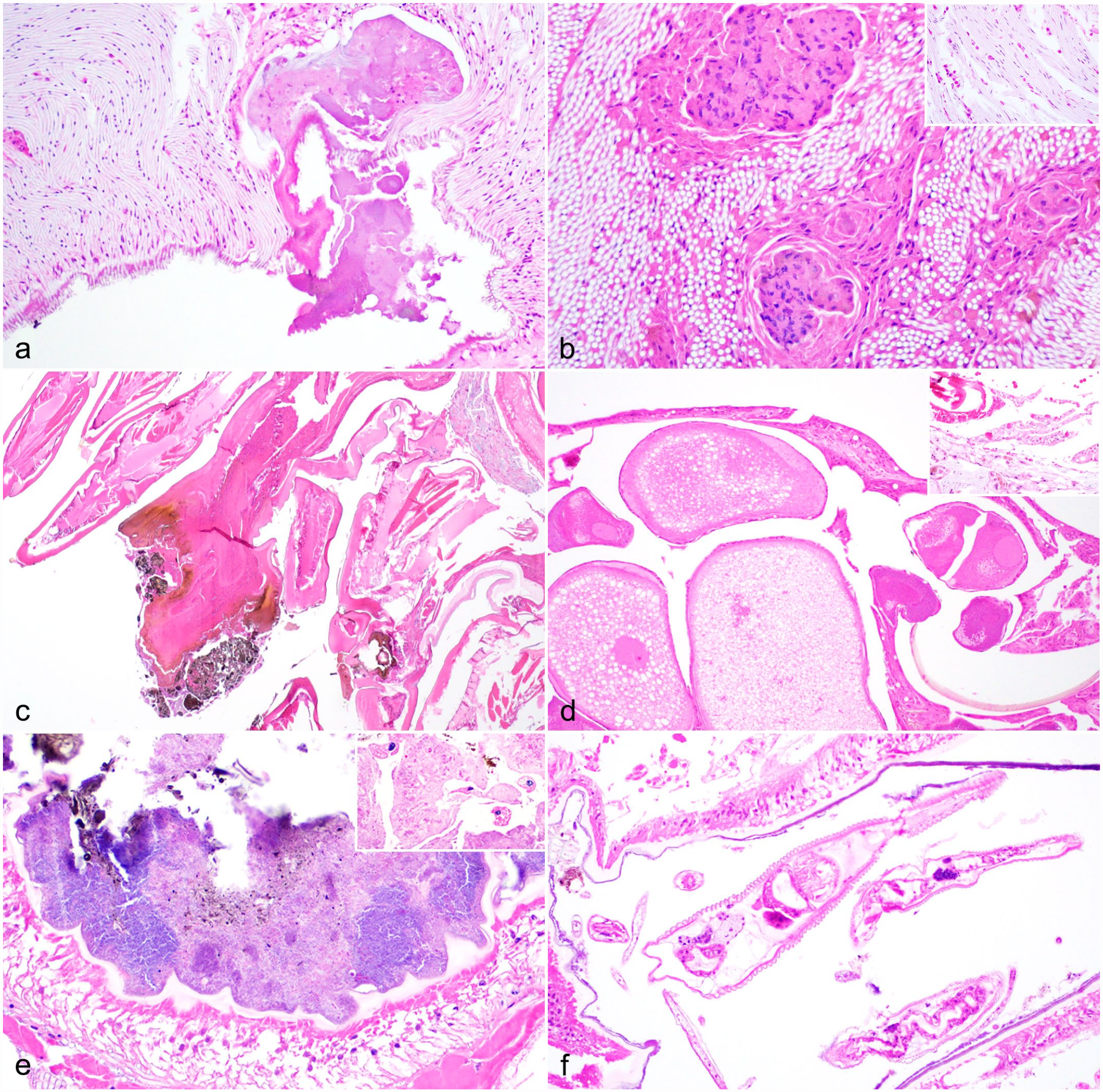
Inflammatory and other lesions in giant African millipedes. Hematoxylin and eosin.
The hemocoel was affected in 16 (23%) cases secondary to gut inflammation and necrosis with and without perforation/rupture. In severe cases, inflammation and necrosis spanned the hemocoel, encompassed portions of the PVB and surrounding skeletal muscle, and extended to the endocuticle (Fig. 3e). In many of these cases, the midgut had segmental to regionally extensive, mucosal to transmural necrosis with hemocytic inflammation and intralesional bacteria (16) and/or fungi (7; Fig. 3f). Fungal hyphae were typically 3–5 µm in diameter, nonstaining to basophilic, parallel-walled, and irregularly septate with acute angle branching and occasional colorless bulbous swellings. Fungi were not further characterized or identified by culture or molecular analyses. Metazoan parasites were noted along the mucosal surfaces in 3 affected millipedes and were characterized as nematodes (2) or small arthropods (1); the latter of which had a faintly yellow chitinous exoskeleton and short appendages with skeletal muscle (Fig. 3f). Nematodes were up to 200 µm in diameter with a coelomyarian musculature, cuticular ridges, and digestive and reproductive tracts; larval forms had lateral alae and vacuolated lateral cords (Fig. 4f). One millipede had abundant digesta at the gut rupture site, thus obstruction was considered as a possible inciting factor.
Few millipedes (3; 4%) had bacteremia as evidenced by bacteria within hemolymphatics vessels or aggregates of bacteria among tissues throughout the body. One individual had multisystemic necrosis associated with bacteria that lacked substantial inflammation. Gram stains revealed mainly Gram-negative short bacilli among necrotic debris and lining the necrotic gut mucosa together with fungi and nematodes; the gut was considered a possible origin of bacterial sepsis. Bacteria were not further characterized or identified by culture or molecular analyses.
Inflammation in the tracheae (26; 38%) and ventral nerve (17; 25%) was typically seen in millipedes with lesions in the head and more proximal body rings and was associated with lesions in either oral cavity/mouth, foregut-midgut junction (Fig. 3d), or spiracles (Fig. 4a). Inflammation in the tracheae ranged from multiple foci of nodulation/encapsulation (Fig. 4b) to minimal granular hemocytic inflammation (Fig. 4b, inset). Similarly, skeletal muscle had varying degrees of inflammation (24; 35%) that was an extension of or collateral damage from primary disease processes such as head or appendage lesions or gut necrosis and hemocoelic inflammation (Fig. 3e).
Ten (14%) millipedes had cuticular lesions, most (9) of which affected the appendages (Fig. 4c). Two millipedes had focal necrosis and hemocytic inflammation spanning the epidermis and all layers of the hard cuticle (epi-, exo-, and endocuticle) and subjacent muscle of the appendage with superficial melanization (Fig. 4c); one millipede had extension of inflammation into the perineural sinus.
In two female millipedes, hemocoel inflammation, presumably originating in the gut, extended into and encompassed portions of the ovaries with degeneration of oocytes (Fig. 4d). A single female millipede had gross and histologic evidence of ovarian rupture with free oocytes and bright red-magenta globular yolk material in the coelom with minimal inflammation, suggestive of acute yolk-induced hemocoelic inflammation (Fig. 4d, inset).
In addition to inflammation, there were other potentially significant findings detected in this group of millipedes, though some findings were seen concurrently with inflammation or other lesions. Bacterial or fungal overgrowth in the gut was noted in 15 (22%) millipedes, and was represented by dense mats or aggregates of monomorphic bacteria lining the gut mucosa with typically intact chitin (Fig. 4e). Rare luminal ciliates were noted in the gut of 2 millipedes and were presumed to be incidental commensal organisms (Fig. 4e, inset). These microorganisms were not further characterized.
Overall, metazoan parasites were detected in 8 (12%) examined millipedes. Five (7%) millipedes had few nematodes in the gut lumen (Fig. 4f) with (2) or without (3) associated gut inflammation and necrosis. One millipede with transmural gut necrosis had small arthropods along the necrotic mucosal surface in addition to myriad bacteria and fungi (Fig. 3f). Trematodes were only seen as ova associated with PVB inflammation in 2 millipedes. Metazoan parasites were not further characterized.
Empty guts or scant digesta were found in 5 (7%) millipedes, and often these individuals had inflammatory lesions elsewhere in the gut or PVB. Eosinophilic fluid within the skeletal muscle and hemocoel was noted in 2 (3%) of the millipedes and was interpreted as edema. Notably, these individuals had other changes consistent with autolysis, so the significance of this finding was uncertain. Dysecdysis was diagnosed in only one millipede with fragmented layers of exocuticle loosely attached to the outermost layers of cuticle. Another individual had cuticular fungal colonization without associated necrosis or inflammation. Acute cuticular trauma was reported in one millipede with exudation of white material.
Many millipedes (7; 10%) lacked inflammation or other substantial microscopic changes and were diagnosed as “open” or “senescent” based on presumed advanced age (approximately 5 years old), with supportive findings such as presumed atrophy of muscle or fat bodies or small, inactive ovaries, if present (Fig. 1e).
Discussion
This retrospective case series reports histologic lesions encountered in a zoo-housed population of giant African millipedes, members of the class Diplopoda. During the study period, different trimming and tissue processing techniques were attempted to optimize diagnostic evaluations. Based on these results, standardized trimming (Fig. 1) and processing using Perenyi’s solution for dechitinization are recommended for millipede histology to reduce section fragmentation; capture specific tissues/organs in section; and master invertebrate microanatomy. Furthermore, clinical histories, age approximation, and gross findings should be conveyed to pathologists to better correlate microscopic findings to potential inciting or predisposing factors such as husbandry, diet, or environmental parameters.
Millipedes were under managed care, and as such were not exposed to known malignant parasites, predators, or typical environmental stressors. Notably, most millipedes died during February (midwinter) and although temperature and humidity were closely monitored in the millipede habitats, temporary or minute changes in these parameters could have predisposed to disease. Millipedes were transported to schools for educational programs throughout the year and a breeding colony was maintained at the zoo. Although efforts were made by zoo and school staff to limit environmental stressors while transporting or on loan in the classroom, intermittent anomalies in husbandry, diet, or climate could have occurred and played a role in increased mortalities, particularly during colder times of year.
The ages of millipedes at time of acquisition (prior to 2021) were not documented, thus specific ages of many millipedes in this study were unknown. However, all submitted millipedes were adult-sized and those known to be born at the zoo were likely at least 1–2 years old at time of death based on their observed rate of growth and maturity. Most millipedes died in 2021 and based on zoo census, no new millipedes were added to the colony, either by acquisition or breeding; therefore, increased deaths during this time could be related to attrition. Of the microscopically examined millipedes (approximately 30% of total deaths), “open” or “senescence” was diagnosed in 7 (10%) based on a lack of lesions and/or signs of reproductive activity, such as small ovaries, or subjectively small or atrophic tissues (PVB, muscle). Nevertheless, significant lesions were found upon histologic evaluation of the remaining submitted millipedes.
Most histologic lesions represented inflammatory processes in examined millipedes, some of which had evidence of bacterial and/or fungal infections. In some cases, lesions were identified in the oral cavity, gut, spiracle, or cuticle, providing a possible route of entry for infectious agents. Bacteria and fungi were not further characterized via culture or molecular analyses; thus, genus and species of these microorganisms remain unknown.
Interestingly, bacterial overgrowth was noted in the gut of over 20% of millipedes, many of which had intact chitin overlying the mucosal epithelium, indicating a benign process and possibly postmortem proliferation of normal gut microflora. However, some millipedes with midgut necrosis and inflammation had abundant monomorphic luminal bacteria elsewhere in the gut, which could represent dysbiosis. Since millipedes are essentially mid and hindgut fermenters, dysbiosis resulting from inappropriate diet, environment factors, stress, or underlying disease could have a significant impact on the overall health of the individual. The secretions of each part of the gut have been studied in detail to understand organic matter transformation, animal-microbial interactions, and compartmentalization of digestion, and these secretions may be investigated further in the context of millipede health.21,31,32,37
Indeed, there were several millipedes in this population that had histologic evidence of gut necrosis and hemocoelic inflammation with bacterial invasion and dissemination. Large numbers of fungal organisms often accompanied bacteria in these lesions and could represent postmortem proliferation and opportunistic infection versus true pathogens. Extensive and severe inflammation in the hemocoel was almost exclusively related to midgut lesions, with mucosal to transmural necrosis and inflammation, which often affected and obscured surrounding tissues/organs including the PVB, Malpighian tubules, and ovaries in females. Male gonads were also likely affected but the reproductive tract was often obscured by inflammation in examined sections. For the most part, midgut lesions were severe, mucosal epithelium was necrotic or absent (ulceration), and the gut wall was smudged and hypereosinophilic (necrosis and degeneration). At the time of histologic examination, it was not possible to discern the initial insult, though there were often myriad bacteria and/or fungi colonizing the necrotic gut wall and extending into surrounding tissues. In a few of these millipedes, bacterial sepsis was purported based on presence of bacteria in necrotic lesions or within hemolymphatics vessels. There was a single millipede with a grossly evident cuticular defect with exudation of white material, purported as a focal penetrating wound, thus an alternative pathogenesis.
Metazoan parasites, specifically nematodes, were seen in gut lumens of millipedes, with and without gut lesions, and appeared morphologically similar. Although many nematode species have been documented in millipedes as commensal and even symbionts,22,24 there are rare reports of rhigonematoid nematodes associated with gut lesions. 26 Speculatively, it is possible that some nematodes in these millipedes either incited or contributed to gut injury through mucosal damage or potentially harboring pathogenic bacteria or fungi. Alternatively, nematodes may have been incidentally residing in gut lumen and other injurious stimuli (eg, bacterial toxins, fungal virulence factors, mechanical trauma, chemical irritants, or endogenous stimuli) caused mucosal necrosis and subsequent bacterial and fungal colonization, proliferation, and invasion. Interestingly, small metazoans originally described as nematodes had histologic features most consistent with arthropods (Fig. 3f). Like bacteria and fungi, the significance of metazoans to gut lesions remains unclear; therefore, additional studies and continued surveillance are needed to determine pathogenicity of these microorganisms and parasites.18,22,24,26,27
A very common site of inflammation was the PVB. This organ is thought to be analogous to chloragogen tissue in annelids and is the main storage system for glycogen, lipid, protein, and uric acid. 21 Inflammation in the PVB ranged from minimal to severe with various types and patterns, and in most millipedes, there were lesions in other tissues. Considering the importance of PVB functions, severe inflammation and damage to this organ likely caused debilitation and demise in some millipedes. Of note, one millipede had PVB epithelial cells containing rare intranuclear inclusions. Although these inclusions were suspicious for viral infection, additional investigations were not pursued. Considering viral-induced disease has not yet been documented in this species, 4 and the PVB had substantial inflammation and degenerative changes, viral infection was thought to be unlikely. Future studies should include diagnostic assays to better evaluate potential viral etiologies in millipedes.
Inflammation in the head (collum) and proximal (first to second) body rings was seen in roughly 30% of examined millipedes and typically involved either the oral cavity, foregut, or spiracles with extension into surrounding tissues such as the tracheae, ventral nerve, and skeletal muscle. The inciting cause of these lesions was not always evident, although speculatively, ingested or inhaled infectious or other potentially damaging agents were considered.
The appendage lesions (9; 13%) were comprised of damage to the cuticle and variable inflammation of adjacent or subjacent tissues like the skeletal muscle and perineural sinus, surrounding the ventral nerve, and spiracles and tracheae. Histologically, it was difficult to discern the initial injury or predisposing lesion for appendage necrosis, inflammation, and melanin accumulation; however, defects in cuticle formation or maintenance could be involved. 4 More thorough clinical and gross examinations are required to correlate any cuticle abnormalities to microscopic lesions in the cuticular components, appendages, and internal tissues and organs, and to cross-reference these data to environmental or dietary anomalies, if pertinent. Notably, softening of the cuticle and pallor were noted as gross findings in only few millipedes, and dysecdysis was histologically diagnosed in one millipede. As for the oral cavity and gut, food or substrate components or inadvertent contaminants (eg, diatomaceous earth) with the potential for injury were speculated as a plausible source for some of these lesions.
On initial review, characterization of lesions in general, and specifically inflammatory lesions, was confounded by differing terminology across pathologists as well as suboptimal sections of some millipede specimens. With attempts to standardize trimming and optimization of processing protocols (dechitinizing solution including decalcifying agents), histologic sections were easier to evaluate and interpret. Some diagnoses were subjective (eg, bacterial overgrowth versus dysbiosis) or may have been determined based on sectioning artifacts (eg, empty gut). Review of these cases highlighted discrepancies in knowledge of invertebrate microanatomy and immunology. Standardization of histologic sections and descriptions was paramount to creating a concise case series that may serve as a baseline for future studies in zoo-housed millipedes.
Diplopod pathology should be more thoroughly investigated as they are the third-most important detritivores (after termites and earthworms) 14 and are keystone species in many terrestrial ecosystems, often serving as a bioindicators for the evaluation of pollutants in the environment.9,21 Millipedes are also widely studied models in ecological, engineering, and medical fields with many publications utilizing them for research on body segment development, morphology and locomotion dynamics, 10 analysis of secretions for antimicrobial and other industrial and medical properties,1,11,15,16,23,28,35 soil ecotoxicology, 6 and gut microbiota and enzymatic processes for organic matter transformation.14,37 Importantly, millipedes are found in zoo settings, involved in outreach and educational programs, and are now considered a nontraditional pet; therefore, interest in optimizing husbandry and veterinary care for these invertebrates will become increasingly relevant. Better understanding of health and disease of millipedes will positively impact these and related invertebrate species in the natural world and in managed care settings.
Footnotes
Acknowledgements
Special thanks to Jenny McFarland and the husbandry staff, and veterinary staff at Fort Wayne Children’s Zoo for providing husbandry information, case histories, and mortality data. Thanks to faculty and resident pathologists of Zoological Pathology Program (ZPP) for initial case evaluations, and big thanks to University of Illinois Veterinary Diagnostic Lab (VDL) histology lab technical staff for invertebrate slide preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
