Abstract
Leukemia is broadly divided into acute and chronic lymphocytic and myeloid types based on the proportion of blasts, morphology of cells, and expression of specific antigens on neoplastic cells. Classifying leukemia in horses can be challenging if blasts predominate and since few antibodies to identify cell types are available. The objective of this study was to describe in detail the clinical and pathologic features of acute leukemia in horses. Twelve horses ranging from 0.2 to 25.9 years of age were diagnosed with acute leukemia. Six cases were classified as acute lymphocytic leukemia (ALL) based on predominance of blasts, lack of granulocytic or monocytic differentiation, and detection of CD3, CD20, and/or CD79a antigens by immunohistochemistry. Six other cases were classified as acute myeloid leukemia (AML) with myelomonocytic (n = 4), basophilic (n = 1), and eosinophilic (n = 1) differentiation based on > 20% bone marrow blasts and partial leukocytic differentiation. Reactivity with antibodies to Iba-1/AIF-1, CD172a, and CD163 was determined for all cases of AML. Eleven horses had thrombocytopenia, 10 had neutropenia, 8 had anemia, all had blasts on blood films, and none had leukocytosis. Ten horses had increased serum acute phase proteins. Bone marrow cellularity ranged from 30% to 100%, and the proportion of blasts ranged from 80% to 100% and 30% to 60% in ALL and AML, respectively. Horses were severely ill at diagnosis and euthanized within days or weeks. Unique features of acute leukemia in horses compared to other species were variable lymphocyte antigen expression (ALL) and frequent inflammation (ALL and AML).
Keywords
Leukemia is a neoplasm of hematopoietic cells that is broadly divided into lymphocytic and myeloid types. Lymphocytic leukemia is further classified into either acute or chronic based on the size and type of neoplastic cells. 42 Degree and duration of illness, presence or absence of lymphocytosis, and severity of cytopenia also differentiate acute and chronic forms. Specific subtypes of leukemia may be recognized by expression of cell antigens identified with immunoassays such as immunohistochemistry (IHC), flow cytometry, or immunocytochemistry. In humans, myeloid neoplasms are categorized into acute myeloid leukemia (AML), myeloproliferative neoplasm (MPN, formerly called chronic leukemia), myelodysplastic syndrome (MDS), and MDS/MPN based on specific clinicopathologic criteria. 4 Since similar entities are also recognized in animals, this classification scheme has been adapted for veterinary medicine. 44 Application of morphologic, immunologic, cytogenetic, and/or molecular assays identifies many additional subtypes within each of the human AML classifications, and such subtypes often have unique biological behavior and prognosis; in some instances, therapies are available that specifically target protein products of particular mutations. 14 There are few assays to subclassify leukemia in animals, but consideration of the proportion of blasts, clinical history, laboratory findings, and neoplastic cell morphology generally allows categorization into acute leukemia, MPN, MDS, or MDS/MPN. Some genetic lesions have been identified in leukemia of animals. 40 Distinction of acute lymphocytic leukemia (ALL) from some types of AML can be difficult since morphologically indistinguishable blasts may predominate in both.
Diagnostic criteria, prognosis, and response to therapy of different types of leukemia are ill defined in animals, in particular in horses. There are several case reports of leukemia in horses, including lymphocytic 2,12,29,37 and myeloid leukemia, 7 –11,18,19,24,34,35,39,41 and one recent series including 6 cases of ALL and AML. 5 Both acute and chronic lymphocytic leukemia have been reported. 12,29,37 As in other species, ALL in horses was characterized by rapid onset of severe clinical illness, marked cytopenia, and short survival, whereas horses with chronic lymphocytic leukemia had indolent illness, massive lymphocytosis, and extensive tissue infiltrates. Acute myeloid leukemia in horses was subclassified most often as myelomonocytic based on cytochemical reactivity and/or electron microscopic features, and it manifested with severe illness, profound cytopenia, and short survival. 7 –10,35,41 Only 1 case of MPN (chronic granulocytic leukemia) and 1 case of MDS have been reported. 15,24 Individual case reports inconsistently included macroscopic, histopathologic, and immunohistochemical findings and outcome; therefore, diagnostic and prognostic criteria of leukemia in horses remain incompletely described.
The objectives of this study were to describe clinical and pathologic findings in 12 horses with acute leukemia and to investigate the utility of additional antibodies to leukocyte antigens to characterize neoplastic cells.
Materials and Methods
Cases
Records of the Animal Health Laboratory and Ontario Veterinary College Health Sciences Centre databases between 2000 and 2016 were searched for equine cases with a diagnosis of leukemia on cytology and/or histopathology of bone marrow and a complete medical record. Blood and bone marrow cytology slides, histopathology slides, formalin-fixed paraffin embedded blocks, laboratory test results, and hematology analyzer cytograms, if available, were retrieved. Data tabulated from medical records were signalment, history, clinical signs, treatment, and outcome.
Laboratory Assessment
Blood and bone marrow aspirates were collected into ethylenediamine tetra-acetic acid tubes. All blood samples were run in an automated analyzer (Technicon H2, n = 1; Advia 120 or 2120, n = 11; both Siemens, Oakville, ON, Canada), and 8 bone marrow aspirates were also run in the Advia. Slides of blood and bone marrow were stained with a modified Romanowsky stain. Sternal bone marrow core biopsies (T-handle Jamshidi® needle; BD, Mississauga, ON, Canada) and postmortem tissue samples were fixed in neutral buffered formalin, processed routinely, sectioned at 3 μm (bone marrow) or 5 μm (other tissues), and stained with hematoxylin and eosin.
Cytopathology and Histopathology
All blood films and cytology and histopathology slides were reviewed. Manual nucleated cell differential cell counts (100 and 500 cells for blood and bone marrow films, respectively) were performed, morphologic abnormalities were noted, and for bone marrow aspirate slides, the ratio of granulocytic and monocytic cells to erythrocytic cells (M:E) was determined. If cells were identified as lymphocytes or blasts, they were not included in the M:E ratio. Blasts were defined by presence of a round to oval or angular nucleus, moderate to high nuclear to cytoplasmic ratio, single or multiple nucleoli, and lack of cell differentiation into granulocytes, monocytes, or rubricytes. Cells were assessed for dysplastic features such as nuclear to cytoplasmic maturational asynchrony, absent or uneven lobulation of megakaryocyte nuclei in cytology preparations, granulocyte nuclear hypolobation, and mitoses in neutrophils with pink granular cytoplasm typical of band and segmented neutrophils. 22,45 On bone marrow sections, cellularity was determined as the proportion of intertrabecular spaces composed of hematopoietic relative to adipose tissue, the average number of megakaryocytes per intertrabecular space was enumerated, cell morphology and presence of iron stores were assessed, and the M:E ratio was estimated.
Classification of Neoplasms
Horses were considered to have leukemia if there was uni- or multilineage cytopenia for ≥ 7 days and on ≥ 2 complete blood cell counts (CBCs), if blasts were identified on blood or bone marrow films, and if clinical and imaging assessments failed to identify a focus of inflammation and/or infection. Leukemia (ALL or AML) was further diagnosed as acute if the proportion of such cells in bone marrow exceeded 20%. Neoplasms were classified as AML based on morphology, if a continuum of differentiation of blasts into neutrophils, monocytes, eosinophils, or basophils was apparent. Neoplasms without cell differentiation were investigated by IHC as detailed below. Gross postmortem and microscopic lesions were recorded, and microscopic infiltrates of neoplastic cells in nonhematopoietic tissues were noted.
Immunohistochemistry and Histochemistry
If neoplastic cells lacked features of differentiated leukocytes on cytology or histopathology, IHC was performed on formalin-fixed paraffin-embedded hematopoietic tissue sections with antibodies to CD3 epsilon (CD3), CD20, and CD79a. These antibodies have previously been validated with tissues from various animal species, including horses. 16,32 Additional antibodies tested were directed against ionized calcium-binding adapter molecule-1/allograft inflammatory factor-1 (Iba-1/AIF-1), CD34, myeloperoxidase (MPO), CD172a, CD163, and equine granulocytes, as summarized in Table 1. Antibody to Iba-1/AIF-1 has been reported to label all macrophage populations and spermatids in mice, 26 phagocytic macrophages and microglia in horse brain, 13 and neoplastic myelomonocytic cells and B lymphocytes in horse bone marrow. 5 Molecule CD34 is expressed by hematopoietic precursor cells and nonhematopoietic stem cells. Antibodies reactive with fresh but not formalin-fixed equine tissue have been identified. 17 Antibodies against MPO and equine granulocytes were suggested by the manufacturer to be reactive with formalin-fixed epitopes on maturing granulocytes of horses and several other species. Equine monocytes and granulocytes express CD172a, and CD163 is expressed on monocytes and phagocytic macrophages. 21,43 All antibodies were tested at concentrations and with antigen retrieval as recommended by the manufacturer on sections of normal equine bone marrow, and normal canine lymph node and bone marrow. Signals were considered specific if they recapitulated the tissue labeling as described previously for the species against which the antibody was raised, and if negative cells were clearly evident within control sections. If initial results were negative, at least 1 higher and 1 lower dilution of antibody was also tested, and antigen retrieval at both pH 6.0 and 9.0 was attempted. Normal control tissues were included in each batch of sections from cases, and for each control and case sample, a section with and without primary antibody was included. Bound antibodies were detected with peroxidase-labeled polymer (EnVision; Dako, Mississauga, ON, Canada) and Nova Red chromogen, or with avidin-biotin alkaline phosphatase complexes and Vector Red substrate (Vector Laboratories, Burlingame, CA, USA).
Antibodies Used for Immunohistochemistry to Identify Neoplastic Equine Leukocytes.
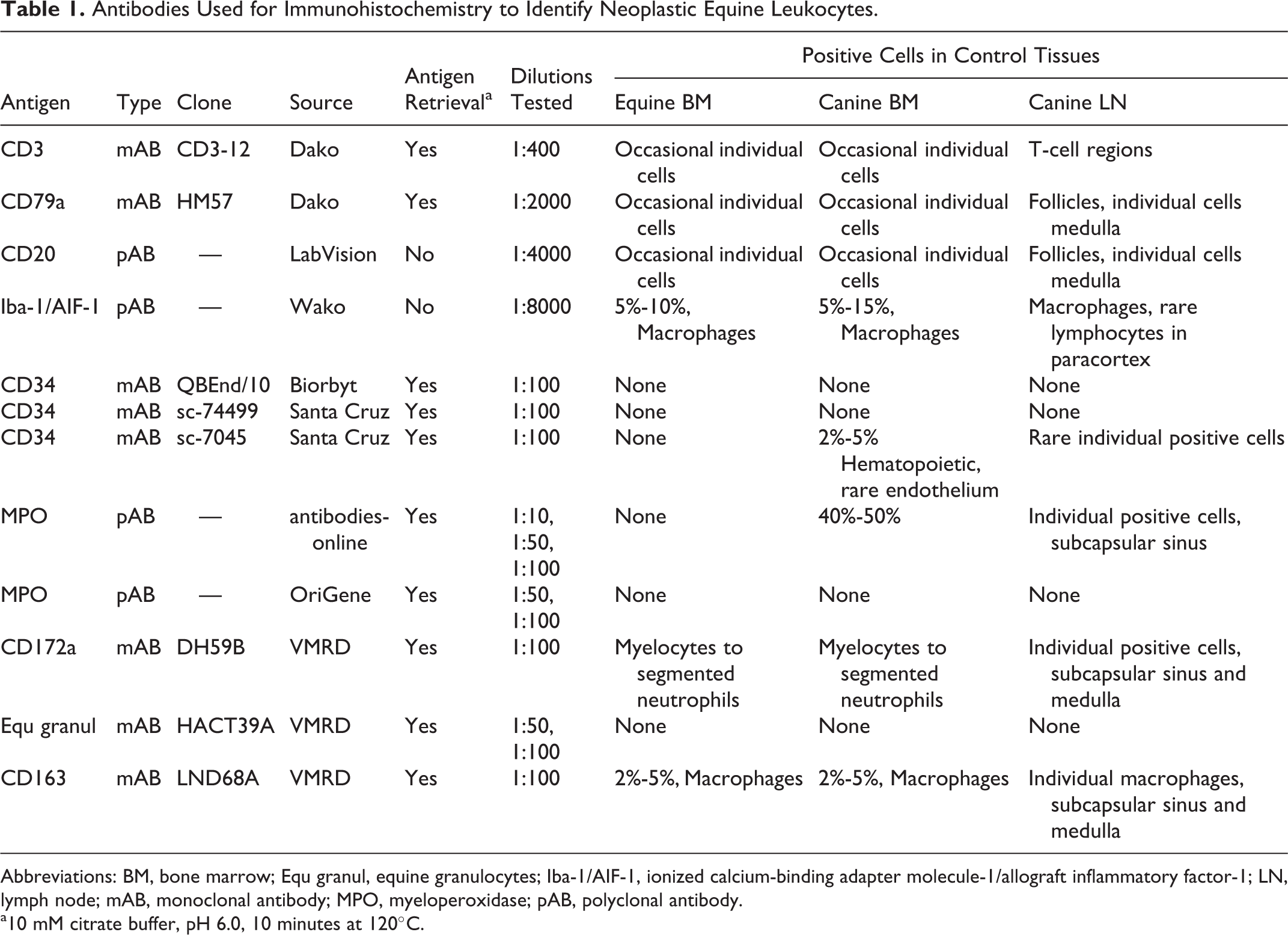
Abbreviations: BM, bone marrow; Equ granul, equine granulocytes; Iba-1/AIF-1, ionized calcium-binding adapter molecule-1/allograft inflammatory factor-1; LN, lymph node; mAB, monoclonal antibody; MPO, myeloperoxidase; pAB, polyclonal antibody.
a10 mM citrate buffer, pH 6.0, 10 minutes at 120°C.
Neoplasms from horses that consisted of blasts without differentiation were considered to be ALL if ≥ 20% of blasts expressed lymphocyte antigens (CD3, CD20, or CD79a). Cases of ALL were further subclassified as T-acute lymphocytic leukemia (T-ALL) if ≥ 20% of blasts expressed CD3 and ≤ 5% expressed CD20 or CD79a, and as B-acute lymphocytic leukemia (B-ALL) if ≥ 20% of blasts expressed either CD20 or CD79a, and ≤ 5% expressed CD3. Neoplasms were considered to be AML if ≤ 3% of blasts expressed CD3, CD20, or CD79a and if differentiation into granulocytes or monocytes was evident. All cases of AML were assessed by IHC with antibodies to Iba-1/AIF-1, CD172a, and CD163. In cases of AML with eosinophilic or basophilic differentiation, sections were also prepared with Luna and toluidine blue stain, respectively.
Results
Signalment and Clinical Findings
Twelve horses with a diagnosis of leukemia and complete medical records were identified. Two were intact males (including 1 colt), 3 were geldings, and 7 were females (including 1 filly). Breeds consisted of Thoroughbred (n = 4), Warmblood (n = 3), Standardbred (n = 3), Percheron (n = 1), and 1 crossbred horse. One mare (case 7) was approximately 5 months pregnant at the time of diagnosis, and 1 mare (case No. 10) had foaled 4 months prior to diagnosis. The mean and median age at diagnosis was 7.2 and 3.6 years, respectively (Table 2). The reported duration of clinical illness ranged from a few days to a few weeks. The most common clinical abnormalities were fever (n = 11), tachycardia (n = 9), anorexia (n = 8), tachypnea (n = 6), distal limb or ventral edema (n = 6), spontaneous bleeding (n = 6), peripheral mild to moderate lymphadenomegaly (n = 5), icterus (n = 5), and dehydration (n = 4).
Clinical Findings in Horses With Acute Leukemia.
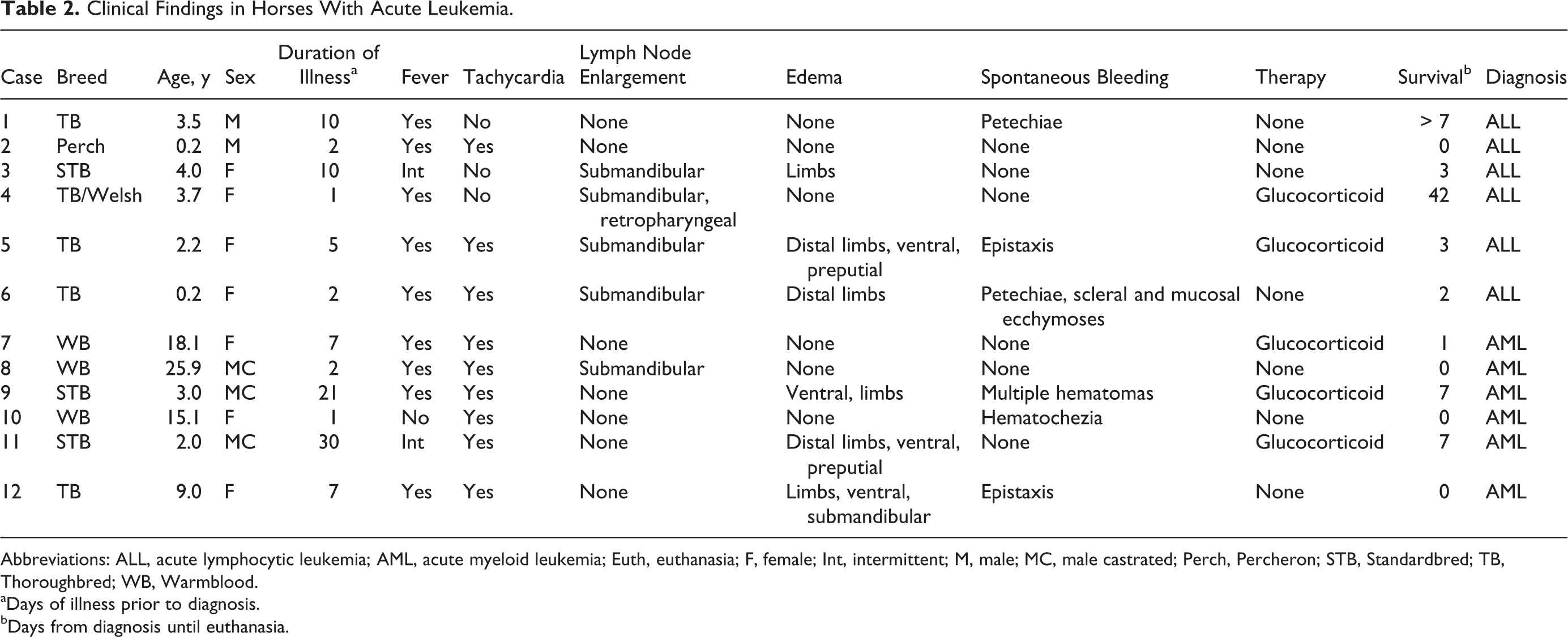
Abbreviations: ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; Euth, euthanasia; F, female; Int, intermittent; M, male; MC, male castrated; Perch, Percheron; STB, Standardbred; TB, Thoroughbred; WB, Warmblood.
aDays of illness prior to diagnosis.
bDays from diagnosis until euthanasia.
Laboratory Findings
Results of the CBC at diagnosis identified thrombocytopenia (n = 11), neutropenia (n = 10), and/or anemia (n = 8). Each horse had more than 1 cytopenia, and only 1 had very mild neutrophilia (Table 3). The degree of thrombocytopenia was severe (< 50 × 109/L) in all except in 1 case, and the degree of anemia was severe (hematocrit ≤ 0.15 L/L) in 4 cases. Two horses had microcytosis and 1 horse had macrocytosis. Mean corpuscular hemoglobin concentration (MCHC) was increased in 1 case. Red blood cell distribution width, a numerical index of erythrocyte anisocytosis, was increased in 5 horses with anemia. No erythrocyte morphologic abnormalities other than agglutination were present. Erythrocyte agglutination (clusters of > 5 erythrocytes) was present in 5 cases with ALL, and 3 of these were anemic. Polychromatophilic cells were not identified by review of blood films (automated reticulocyte counts were not performed). Blast cells were identified in blood in each case and made up 10% to 40% of leukocytes; these blast cells were approximately 20 to 35 μm in diameter, had round to oval or angular large nuclei, and lacked cytoplasmic differentiation. In 3 cases of AML, intermediate or late stage rubricytes, sometimes in mitosis, were noted on blood films. Nucleated erythrocytes are rare in horse blood. Dysplastic cells other than rubricytes with nuclear–cytoplasmic asynchrony, such as lack of nuclear condensation despite fully hemoglobinized cytoplasm, were not identified. A mild left shift without toxic change was present in case Nos. 6 and 10. Serologic testing for equine infectious anemia virus was performed in 7 cases and was negative.
Laboratory Findings in Horses With Acute Leukemia.
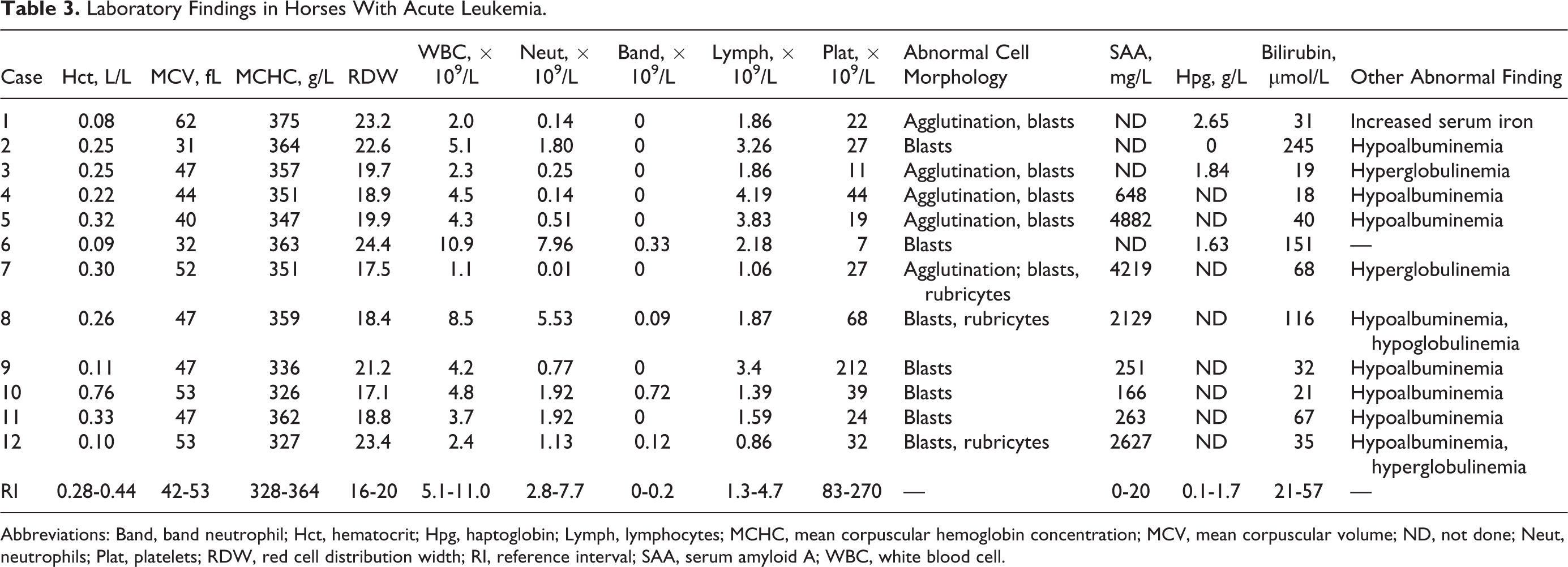
Abbreviations: Band, band neutrophil; Hct, hematocrit; Hpg, haptoglobin; Lymph, lymphocytes; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; ND, not done; Neut, neutrophils; Plat, platelets; RDW, red cell distribution width; RI, reference interval; SAA, serum amyloid A; WBC, white blood cell.
The most frequent abnormality on serum biochemical testing was an increase in acute phase protein concentration. The laboratory initially used haptoglobin (Beckman Coulter AU480 Chemistry Analyzer Haptoglobin Assay) and later serum amyloid A (SAA, Eiken SAA Turbidometric Assay on a Cobas 6000 c501 Analyzer) concentration to indicate acute inflammation in horses. In 10 cases, 1 of these was increased, and in 4 cases where SAA was determined, concentration was increased > 100 fold. Hypoalbuminemia occurred in 8 horses, and bilirubin concentration was mild to moderately increased in 5 cases. Other laboratory abnormalities occurred in fewer than 2 horses each.
Bone Marrow Findings
Antemortem bone marrow biopsies were available for 10 cases. Bone marrow aspirates contained none or only few particles in 9 cases, although bone marrow films were highly cellular (Table 4). The percentage of blasts enumerated by cytology ranged from 32% to 98%, and the M:E ratio varied from 1:1 to 12:1. On histopathology, all cases of ALL had cellularity > 80%, except case 1, in which the cellularity varied from 30% to 100% across the core biopsy section. In bone marrow of AML cases, cellularity ranged from 35% to 100%, and cells were generally tightly packed against each other. Iron stores were rare or absent in all cases of ALL. Myelofibrosis was not identified in any bone marrow section.
Bone Marrow Findings in Horses With Acute Leukemia.
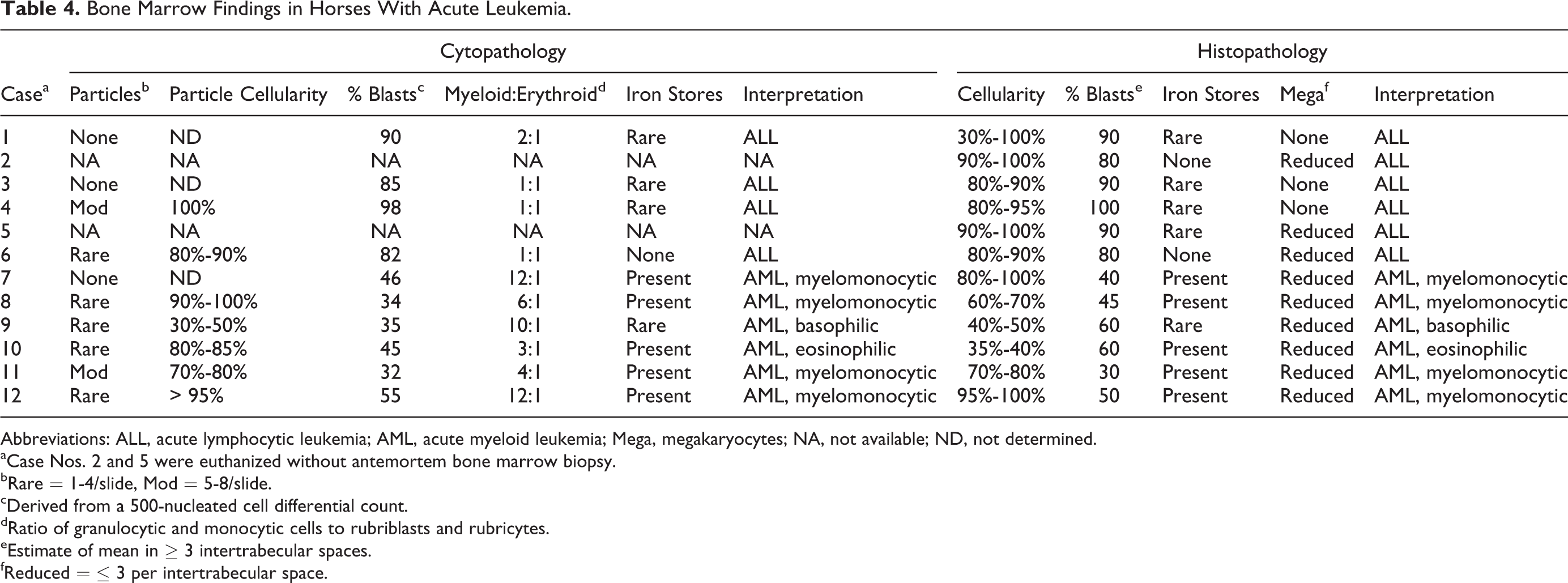
Abbreviations: ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; Mega, megakaryocytes; NA, not available; ND, not determined.
aCase Nos. 2 and 5 were euthanized without antemortem bone marrow biopsy.
bRare = 1-4/slide, Mod = 5-8/slide.
cDerived from a 500-nucleated cell differential count.
dRatio of granulocytic and monocytic cells to rubriblasts and rubricytes.
eEstimate of mean in ≥ 3 intertrabecular spaces.
fReduced = ≤ 3 per intertrabecular space.
Classification
In 6 cases, hematopoietic tissue consisted predominantly of blasts without differentiation into granulocytes or monocytes. Based on expression of lymphocyte antigens on ≥ 20% of blasts, these cases were classified as ALL. Of these, 5 cases were subclassified as B-ALL, and 1 case (No. 2) could not be subclassified since approximately equal proportions of cells expressed CD3 or CD20/CD79a (Table 5).
Gross, Histopathologic, and Immunohistochemical Findings in Horses With Acute Leukemia.
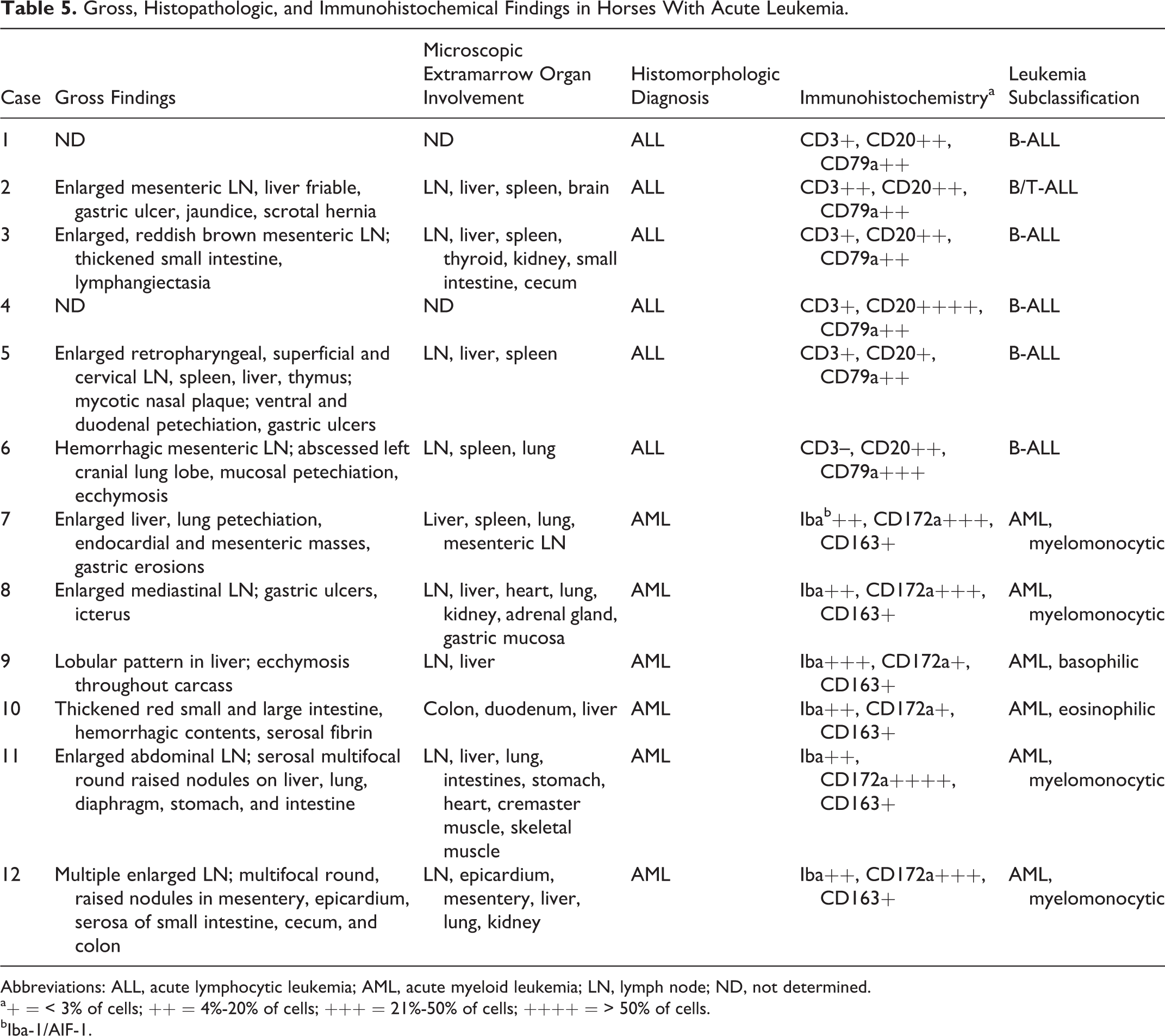
Abbreviations: ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; LN, lymph node; ND, not determined.
a+ = < 3% of cells; ++ = 4%-20% of cells; +++ = 21%-50% of cells; ++++ = > 50% of cells.
bIba-1/AIF-1.
Partial or complete differentiation into monocytes and neutrophils was apparent in 4 cases of AML, and these were subclassified as myelomonocytic (Table 4). In 1 case, each differentiation into eosinophils or basophils was apparent, and these were accordingly subclassified as eosinophilic and basophilic AML.
Immunohistochemistry
Protocols for CD3, CD20, and CD79a IHC were previously established and yielded expected results with control tissues. 32 Iba-1/AIF-1 is considered a pan-macrophage marker in mice, 26 the antibody to Iab-1/AIF-1 bound to equine microglial cells 13 and labeled cells with macrophage and dendritic cell morphology in normal canine lymph node and equine bone marrow (data not shown). In addition, Iba-1/AIF-1 was also detected in rare lymphocytes in the paracortical T-cell–rich region of dog lymph node sections and in some blasts in equine bone marrow.
Three antibodies against CD34 were tested. Monoclonal AB sc-7045 yielded expected results with canine bone marrow sections (Table 1) but did not label any cells in equine bone marrow (data not shown). Other antibodies to CD34 also did not have reactivity with equine bone marrow.
Two different polyclonal antibodies to MPO were tested, and neither labeled cells in equine tissues, although reactivity with canine granulocytic cells was observed with 1 antibody (Table 1). Antibody to CD172a reacted with canine and equine macrophages, monocytes, myelocytes, metamyelocytes, and band and segmented neutrophils but not blasts. Antibody to CD163 reacted with macrophages in all tissues. Monoclonal antibody to equine granulocytes did not label any cells in equine bone marrow sections.
Acute Lymphocytic Leukemia
In all cases of ALL, bone marrow cellularity ranged from 30% to 100%, and blasts made up ≥ 80% of cells. In all cases of ALL, blasts were identified on blood smears (Fig. 1, Supplemental Fig. S1). The blasts typically made up 10% to 40% of leukocytes in blood and lacked peroxidase activity as assessed with the Advia hematology analyzer (data not shown). Cells with similar appearance predominated in bone marrow (Figs. 2, 3, Supplemental Figs. S2–S4). Bone marrow polychromatophils were rare or absent in all cases of ALL, and dysplasia was not identified.
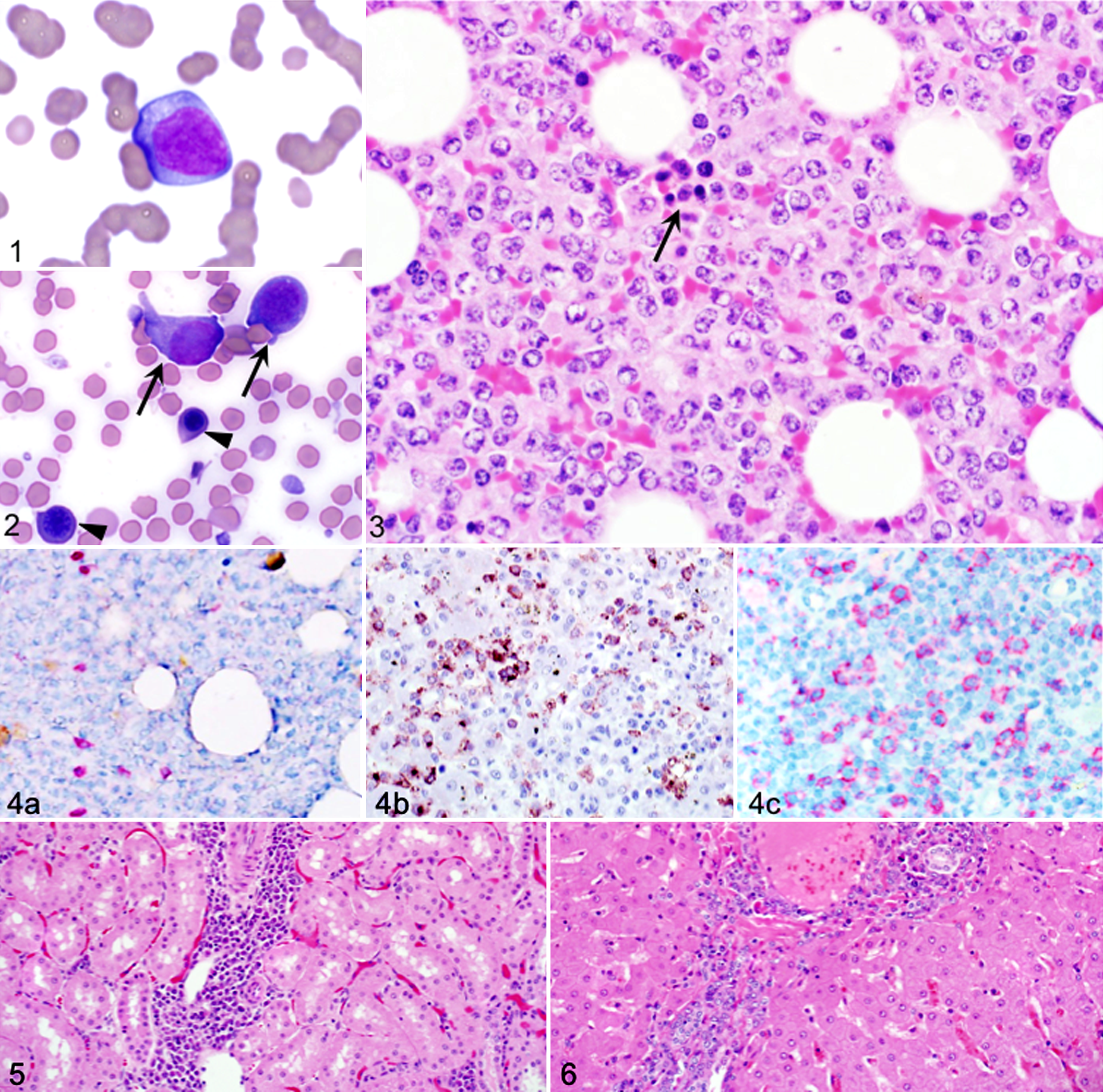
Expression of cytoplasmic or membranous lymphocyte antigens on blasts was not uniform, ranging from approximately 20% (Fig. 4a–c) to > 50% (Supplemental Figs. S5, S6). In some cases, the proportion of blasts expressing the B-cell markers CD20 or CD79a was similar (Fig. 4b, c), whereas in other cases, either CD20+ or CD79a+ cells were more numerous. Five of 6 cases of ALL were subclassified as B-ALL, and 1 case could not be subclassified since the proportion of T-cells and B-cells was similar (Table 5).
Four cases of ALL underwent autopsy. Gross masses were not identified, but in each case, there was microscopic infiltration of splenic red pulp and multiple nonhematopoietic tissues by neoplastic cells, and in some cases, there was mild organ enlargement (Table 5, Figs. 5, 6). Cell death among neoplastic cells—characterized by nuclear fragmentation and condensation (likely apoptosis)—was apparent in some cases of ALL. Despite near-complete replacement of hematopoietic tissue with blasts in all cases of ALL, none had leukocytosis (Supplemental Figs. S3, S4).
Information on the outcome was available for 5 of 6 horses with ALL. Three cases were euthanized within 3 days of diagnosis due to deteriorating clinical condition. Case No. 1 was lost to follow-up but after diagnosis was not registered to race again in an Ontario racing jurisdiction. Glucocorticoid treatment was initiated in case Nos. 4 and 5, and these were euthanized 42 and 3 days after diagnosis, respectively. Serial CBCs during therapy showed no hematologic improvement in case No. 4.
Acute Myeloid Leukemia
All cases of AML had blasts on blood smears, and in 3 cases, rubricytes with dysplastic features or in mitosis were noted (Fig. 7). Bone marrow aspirates were moderately to highly cellular, yielding numerous individual cells on films but only few particles. Blasts similar to those in blood were frequent in bone marrow (Fig. 8). In 4 cases, grossly the bone marrow cavity had irregular patches of red marrow interspersed by fatty tissue (Fig. 9). Bone marrow sections showed an increased proportion of blasts (30% to 60% of all cells). Blasts did not form a homogeneous population that expanded to cause myelophthisis; rather, the blast cells were accompanied by a reduced but persistent population of band and segmented neutrophils, megakaryocytes, and rubricytes (Figs. 10, 11). There was a paucity of polychromatophils on bone marrow cytology preparations in all cases. All horses with AML had marked cytopenia of at least 2 cell lines, despite the presence of bone marrow precursor cells.
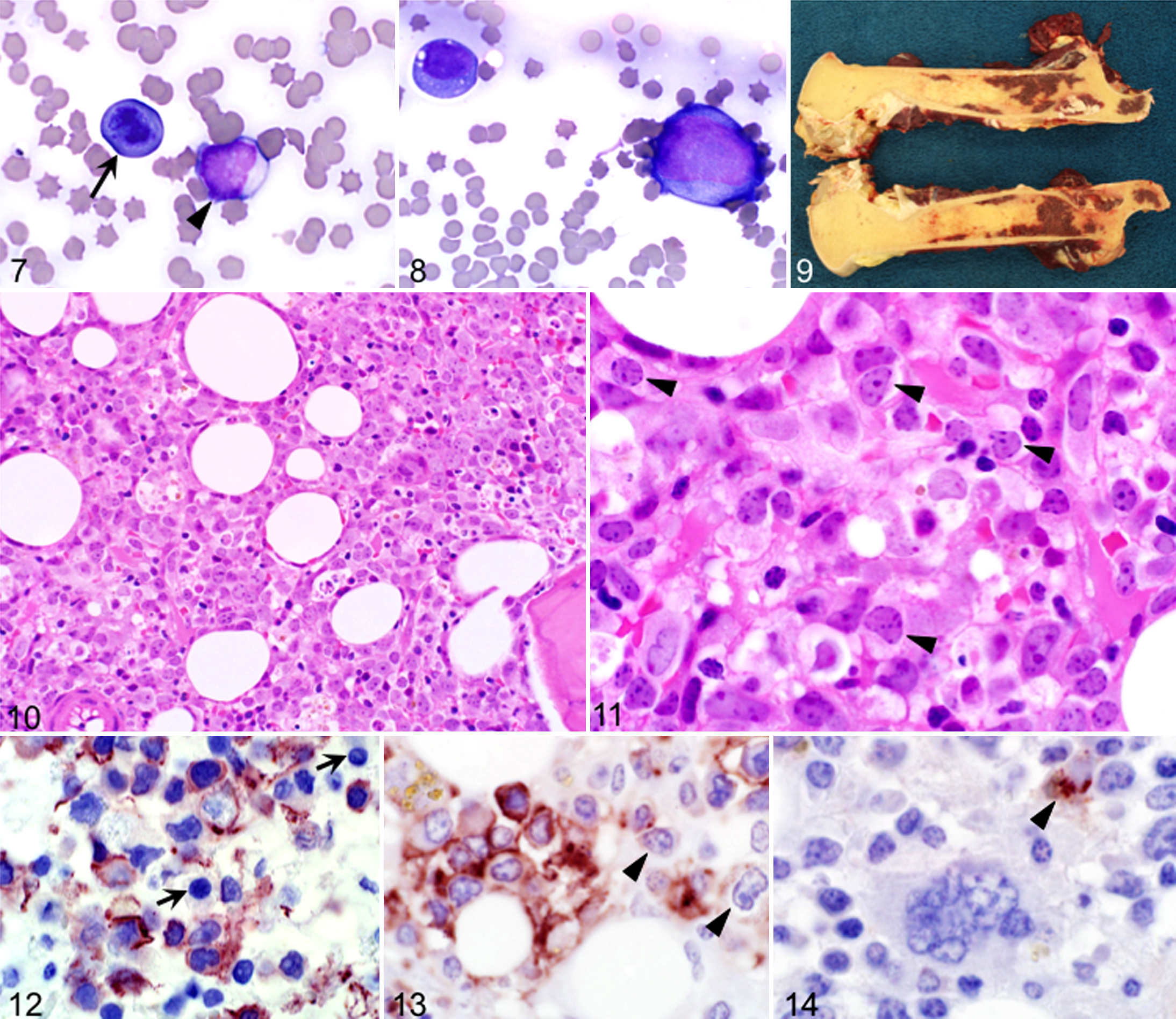
Immunohistochemically, in AML with myelomonocytic differentiation, 20% to 30% of blasts had membranous expression of Iba-1/IAF-1 (Fig. 12), granulocytic cells ranging from myelocytes to segmented neutrophils expressed CD172a (Fig. 13), and rare single cells had cytoplasmic aggregates positive for CD163 (Fig. 14).
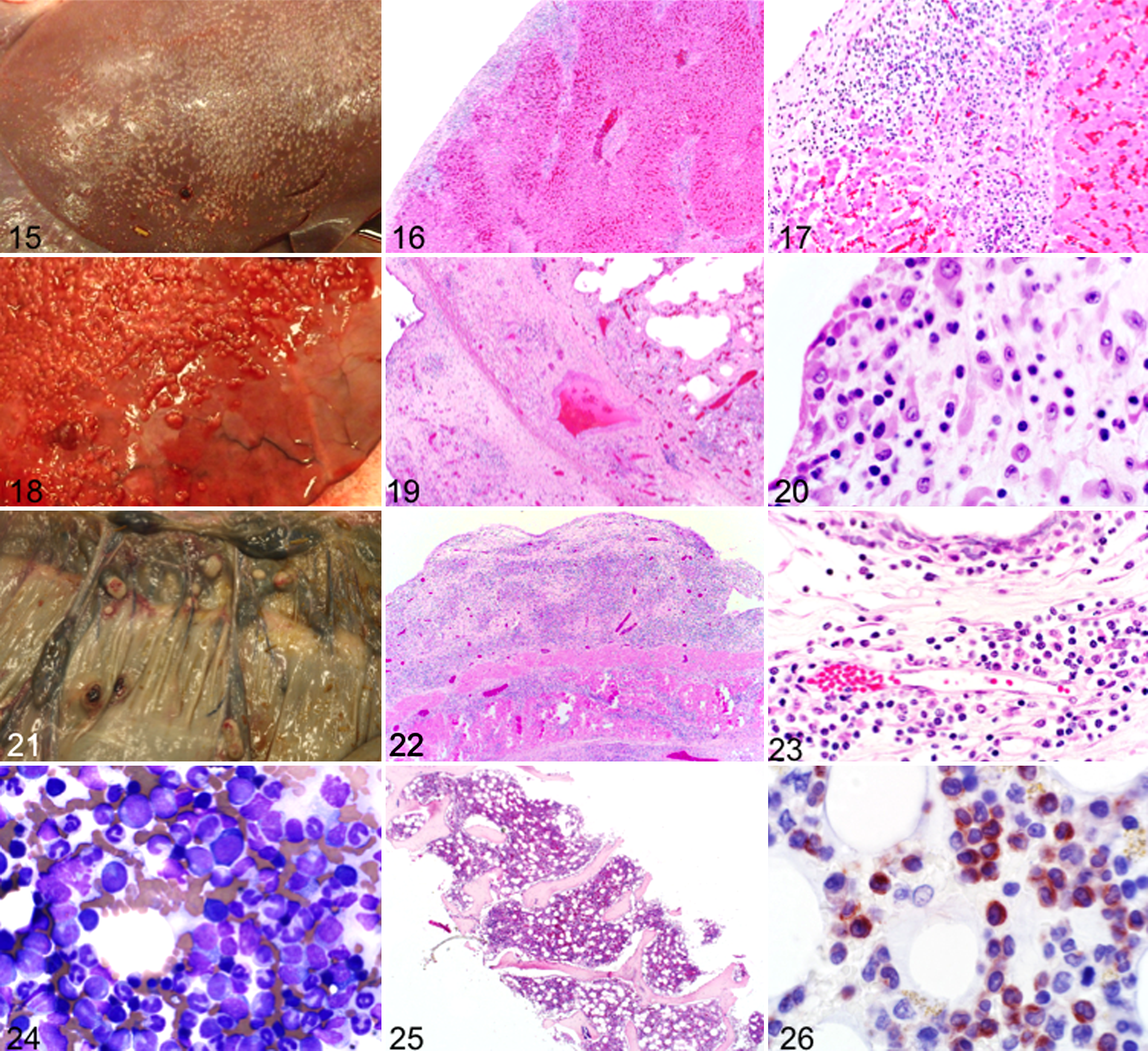
Case No. 11 of AML with myelomonocytic differentiation had unusual miliary nodules on the visceral surfaces of the peritoneum and pleura (Fig. 15). Histologically, the nodules consisted of a mixed population of hematopoietic cells including blasts that extended into hepatic parenchyma (Figs. 16, 17) and pulmonary interstitium (Figs. 18–20). Nodules in the mesentery were more discrete, protruding from the underlying tissues (Figs. 21–23). The bone marrow had an increased proportion of blasts with partial neutrophilic differentiation, scattered early stage rubricytes (Fig. 24), and near-complete paucity of megakaryocytes (Fig. 25). Some medium-size cells with round nuclei (myelocytes) and most differentiated granulocytes expressed CD172a (Fig. 26). Case No. 12 also had miliary nodules on visceral surfaces of the peritoneum and pleura. Bone marrow findings were similar in all cases of AML with myelomonocytic differentiation.
Case No. 9 of AML with basophilic differentiation had rare atypical myelocytes and metamyelocytes with cytoplasmic granules in blood (Supplemental Fig. S7) and bone marrow films (Supplemental Fig. S8). Blasts made up approximately 40% of the sternal bone marrow biopsy section (Supplemental Fig. S9), but metaphyseal bone marrow collected from the femur postmortem 2 days later was more cellular and included a greater proportion of blasts (Supplemental Fig. S10). On IHC, a moderate number of blasts expressed Iba-1/IAF-1 (Supplemental Fig. S11), whereas only rare cells were positive for CD172a (Supplemental Fig. S12). Fine metachromatic granules were present in approximately 10% of cells with toluidine blue staining (Supplemental Fig. S13).
Case No. 10 of AML with eosinophilic differentiation had neutropenia, severe thrombocytopenia, hypoalbuminemia, and severe tachycardia. On bone marrow aspirate and core biopsy slides, blasts were approximately 40%, and myelocytes and metamyelocytes with eosinophilic cytoplasmic granules (prominent with Luna staining) were approximately 30%. Erythrocyte precursor cells were slightly reduced relative to leukocyte precursors.
Three horses with AML were treated with glucocorticoids and euthanized after 1 to 7 days, whereas 3 others were euthanized within 24 hours of diagnosis due to deteriorating condition.
Discussion
This study describes clinical and pathologic features of acute leukemia in horses. Leukemia is a rarely reported disease in horses of unknown prevalence. Too few cases were accrued for this series to identify specific risk factors. However, all horses with ALL were younger than 4 years, which is similar to humans, where ALL is most often a pediatric cancer. 20 Horses with AML were of variable age. Two mares in this study were pregnant at or prior to diagnosis of AML, as also previously reported in another horse. 39 Concurrence of leukemia and pregnancy is likely coincidental in horses and is also considered coincidental in humans. 36
The clinical presentation of ALL and AML in horses was very similar and consisted of a relatively short duration of nonspecific illness characterized by anorexia and intermittent or persistent fever. Half of the cases had edema and petechiation or spontaneous bleeding, but there was no obvious correlation of spontaneous bleeding with the degree of thrombocytopenia. Peripheral lymph nodes were more frequently enlarged in horses with ALL than AML, but overall, lymph node enlargement was only mild to moderate in any case.
Results of CBCs indicated more than 1 cytopenia in each horse. Cytopenia is uncommon in horses except for neutropenia in systemic bacterial infections such as salmonellosis. Such infections are typically accompanied by hemodynamic instability, fever, and neutrophil toxic changes. Neither hemodynamic instability nor neutrophil toxicity was present in horses in this study. Most cases of thrombocytopenia in horses are associated with administration of drugs that are thought to function as haptens or with infections such as equine infectious anemia virus, piroplasmosis, and anaplasmosis. 3,31 Anemia of chronic inflammation is also uncommon in horses, and if a source of severe inflammation or infection is not identified, hemolymphatic neoplasia should be considered as a differential diagnosis. Erythrocyte agglutination was observed on the blood smear of 5 horses, but presence of agglutination did not correspond to more severe anemia or hyperbilirubinemia. Thus, anemia was probably the result of altered or reduced erythropoiesis and/or shortened erythrocyte lifespan. Paraneoplastic autoimmunity is considered to arise from malignancy-induced change in positive selection, negative selection, antigen expression, or antigen processing and presentation in the thymus, and it results in an altered circulating mature T-cell repertoire. 30 A range of neoplasms, including ALL and AML, has been associated with autoimmune hemolytic anemia (AIHA) in humans. 30 It is plausible that similar and more widespread disruption of immune regulation contributes to AIHA in leukemia and lymphoma horses. 32
Two horses < 3 months of age had microcytosis, which was considered most likely due to age-dependent limited iron availability. 27 Five horses with anemia had increased erythrocyte anisocytosis, and 1 also had an increased MCV. These changes most likely reflected accelerated or altered erythropoiesis with an increased proportion of young or macrocytic erythrocytes in circulation, respectively. The MCHC was increased in 1 horse that also had erythrocyte agglutination and severe anemia. Increased MCHC was most likely due to presence of intravascular hemolysis. One horse (case No. 10) had polycythemia, neutropenia, and thrombocytopenia. The bone marrow in this case had an increased proportion of blasts and eosinophil precursors and was classified as AML with eosinophilic differentiation. Polycythemia may have been due to splenic release of stored erythrocytes or ectopic erythropoietin production. 25
Most horses in previous reports of acute leukemia had neutropenia, 7,10,11,18,29,35,39,41 although high white blood cell counts and overt leukemia have also been reported. 8,9,11,34 Similar to previous reports, in this study even horses with neutropenia and/or leukopenia had blasts on blood films, albeit often in very low number. This finding emphasizes the importance of blood smear morphologic evaluation in horses with cytopenia. Myelocytes and metamyelocytes may be observed as a result of extreme left shift and sepsis, but anemia or thrombocytopenia is not usually present in those conditions.
The high frequency of increased acute phase protein concentration was an unexpected finding, suggesting that acute leukemia in horses is often associated with inflammation. Most horses also had hypoalbuminemia, which could be because albumin is a negative acute phase protein or because of blood loss. Polyclonal hyperglobulinemia, increased acute phase proteins, fever, and AIHA also occur in horses with lymphoma. It is unclear whether these changes are due to paraneoplastic autoimmunity, production of pro-inflammatory cytokines, or a combination thereof, but they may constitute a peculiar response of horses to hemolymphatic neoplasia.
Antemortem bone marrow aspirates and core biopsies were obtained in 10 cases, and sections of bone marrow from postmortem examination were reviewed in the remaining 2 cases. Most bone marrow aspirates contained few or no particles but were nevertheless highly cellular. This suggests that the neoplastic cells had altered adhesive properties and were less likely to be part of the hematopoietic niche. 23,38 Reduced expression of molecules mediating adhesion to endothelium and bone may have contributed to the lack of cohesive bone marrow particles in aspirates and also to variable cellularity in the core section in case No. 1. The proportion of blasts was ≥ 80% in all cases of ALL but more variable in AML. Hence, although > 25% bone marrow blasts is considered a diagnostic criterion of ALL in humans, this threshold was far exceeded by all equine cases of ALL. 42 The proportion of normal bone marrow hematopoietic cells in cases of ALL was low but generally with a normal M:E ratio. In cases of AML, the proportion of myeloid cells (including myeloblasts) consistently outnumbered erythroid precursor cells, but in no case was there complete myelophthisis by a homogeneous blast population.
Although there was no or only mild-to-moderate enlargement of lymph nodes, spleen, and liver at postmortem examination, neoplastic infiltrates were noted microscopically in many organs. Splenic red pulp contained dense sheets of lymphocytes in all cases of ALL and cell populations similar to those in bone marrow in cases of AML. The extent of organ involvement did not distinguish ALL from AML, although serosal, capsular, or mesenteric nodules were noted in only 3 cases of AML.
It was challenging to subclassify most cases of ALL as T-cell or B-cell type by IHC since there was expression of both T-cell and B-cell antigens in each case, and often < 50% of cells were labeled with any antibody to lymphocyte antigens. In 4 cases, only approximately 20% of lymphocytes expressed CD3 or CD20/CD79a. Variable expression of antigens associated with T and B lineage has also been described in horses with lymphoma, and markers such as CD79a, CD20, and PAX-5 that are widely used to identify B-cells have yielded discrepant results. 32,33 Since none of the antibodies were raised specifically against equine antigens, there may be suboptimal antibody cross-reactivity. This is especially true for CD79a, which, in the authors’ experience, generally labels weaker than CD20 in the horse and is prone to off-target labeling. Trabecular bone marrow is decalcified prior to paraffin embedding, which may also negatively affect epitope preservation. Definitive identification of a neoplastic population that makes up a minority component of a mass requires proof of clonality, in vitro immortality, or transplantability of those particular cells, which remains to be performed for lymphocytic neoplasms of horses such as T-cell–rich B-cell lymphoma and ALL. 1,16 Hence, it is suggested from findings in this study that assignment of lymphocyte lineage in horses with currently available IHC markers is ambiguous and that generation of additional antibodies reactive with formalin-fixed horse epitopes or whole-exome sequencing assays for cell-specific antigen receptor gene rearrangement should be explored. Flow cytometry of bone marrow aspirates might have been helpful to characterize lymphocytes with an expanded range of antibodies but was not performed. Flow cytometric analysis of AML bone marrow aspirates would have been challenging due to the mixed nature of cell populations and lack of cell type-specific antibodies.
Categorization of acute leukemia as AML was based on criteria of > 20% blasts in bone marrow and evidence of partial leukocyte differentiation. However, different from many cases of AML in humans and dogs, none of the horses with AML had unequivocal myelophthisis, but rather some erythropoiesis and thrombopoiesis also persisted. Differentiation into cells resembling maturing granulocytes or monocytes was appreciable in each case and formed the basis for distinction of AML from ALL. Antibody to Iba-1/IAF-1 appeared to label myelomonocytic precursor cells and macrophages; however, in some instances, cells with round nuclei and lacking cytoplasmic vacuoles or granules were also labeled by Iba-1/IAF-1 antibody, suggesting that these might be myeloblasts. Although considered specific for macrophage-lineage cells and spermatids in other species, antibody to Iba-1/IAF-1 labeled a few cells in the T-cell–rich areas of dog lymph node sections, suggesting that the epitope recognized by this antibody is expressed variably in different species. Antibody to CD172a, signal regulatory protein-α, labeled some macrophages and all maturing neutrophils from myelocytes to segmented neutrophils and was helpful for outlining such cells in bone marrow. This antibody did not label eosinophilic or basophilic precursor cells. Antibody to CD163, a scavenger receptor for hemoglobin/haptoglobin complexes, labeled cells with macrophage morphology in canine and equine tissues. 43 Only single (likely nonneoplastic) phagocytic macrophages were labeled in neoplastic bone marrow. Several antibodies to CD34 and MPO were tested but did not label cells in equine tissues when tested at multiple different dilutions and with different antigen retrieval methods. This suggests that the equine MPO and CD34 epitopes differ from those of other species. An antibody raised against equine granulocytes (clone HACT39A) also did not react in FFPE tissues.
Several cases of AML were diagnosed after short or peracute illness. Anemia in conjunction with a coagulopathy that could not be attributed to thrombocytopenia was the main clinical feature in the case of basophilic AML (case No. 9). 19 The coagulopathy was thought to be due to an acquired secondary hemostasis defect, possibly related to heparin release from basophilic leukemia cells as described in humans. 6 Case No. 10 presented with severe acute illness, neutropenia, thrombocytopenia, hypoalbuminemia, and erythrocytosis and was euthanized 4 hours after presentation due to rapidly declining clinical condition, despite supportive therapy. Erythrocytosis was likely due to splenic contraction and/or hemodynamic changes. Ectopic production of erythropoietin by lymphoma tissue has been described in a horse but was not investigated in this case. 25 In the cases of AML with basophilic and eosinophilic differentiation, the diagnosis was based on the same criteria as for other cases of AML, but the clinical presentations likely in part reflected an excess of anticoagulant released from neoplastic cells (case No. 9) and hemodynamic instability due to internal fluid shifts, intestinal tumor infiltration, or paraneoplastic neuropathy (case No. 10). 28
Chemotherapy is used to treat acute human and canine leukemia but would be costly in horses and has unproven efficacy. 10,14,39,41 Return to athletic or reproductive performance would appear unlikely. Treatment of ALL or AML with glucocorticoids was attempted in 5 cases but was unrewarding. Future studies incorporating additional diagnostic approaches, in particular reagents and assays to characterize and distinguish reactive and neoplastic lymphocytes, would likely be helpful.
Horses with acute leukemia in this case series had fever, inflammation, and severe cytopenia and lacked leukocytosis. Blasts were uncommon on blood smears but could be identified in each case. Neither clinical findings nor routine laboratory tests allowed distinction of ALL from AML. Cases of ALL differed from AML by absence of granulocytic or monocytic cell differentiation in ALL. Leukemia in horses likely includes additional subtypes that cannot be clearly distinguished with currently available limited reagents for immunohistochemical, cytogenetic, or molecular analysis.
Footnotes
Authors’ Note
A manuscript concerning clinical aspects of case No. 9 has been published as a case report in the Canadian Veterinary Journal, 57(10):1067–1071.
Acknowledgements
We thank Susan Lapos of the Histopathology Laboratory and Dr Josepha DeLay from the Animal Health Laboratory for assistance with immunohistochemical assays.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
