Abstract
Acute myeloid leukemia (AML) can infiltrate extramedullary tissues, such as the liver, spleen, and lymph nodes and can be difficult to differentiate from lymphoma in cytologic and histologic specimens. Our goal was to identify cytologic features that would support a diagnosis of AML in peripheral lymph node aspirates, for which we used the term extramedullary AML (eAML). Medical records of 23 dogs with a diagnosis of AML and archived lymph node aspirate smears from 2016 to 2024 were reviewed across 4 institutions. Inclusion criteria included ≥50% myeloid blasts plus differentiating myeloid cells in lymph node smears, confirmation of myeloid lineage by flow cytometric analysis, and complete medical records. Peripheral lymphadenopathy was the reason for presentation (9/23, 39%) or was found incidentally on physical examination (14/23, 61%). Most dogs were bi- or pancytopenic (18/23, 78%), with blasts identified in blood smears of 18 dogs (78%). Initial lymph node aspirate interpretations included hematopoietic neoplasia (8/21, 38%), AML (6/21, 29%), lymphoma (5/21, 24%), lymphoid hyperplasia (1/21, 5%), and granulocytic precursor infiltrates (1/21, 5%). On lymph node smear review, cytologic features supporting an eAML were differentiating granulocytes, blasts with myeloid features or promonocytes, dysplastic changes in myeloid cells, and retention of residual lymphocytes. The median survival was 22 days (range = 1-360 days), and 69% of 16 dogs given chemotherapy or glucocorticoids lived for 30 days or more. Our study highlights the importance of hemogram results and lymph node aspirate smear examination for morphologic features of myeloid differentiation to help diagnose eAML in lymph node smears.
Keywords
Acute myeloid leukemia (AML) is a hematopoietic neoplasm involving the clonal proliferation of immature myeloid cells in the bone marrow. There are many subtypes of AML, according to the World Health Organization (WHO) classification, with newer schemes based on defining genetic mutations,2,3,35 information that is currently lacking in dogs. AML in dogs is typically diagnosed by finding an overt leukemia (≥20% blasts) in blood smears or bone marrow aspirates.2,3,31,32 However, the leukemia can infiltrate extramedullary sites,1,8,12,16 where it can mimic lymphoma, which is a more common hematopoietic neoplasm. The WHO defines myeloid sarcoma as a subtype of AML that consists of an extramedullary proliferation of myeloid blasts, with or without differentiation, which partially or completely effaces the architecture of the infiltrated tissue. These extramedullary tumors may occur with or without peripheral blood or bone marrow involvement by the leukemia.1-3,35
A recent review article proposed the term “extramedullary” AML (eAML) for tumor infiltrates in nonhematopoietic tissue, instead of myeloid sarcoma or synonyms, such as chloroma. They also separated eAML into “synchronous” or “isolated” variants, with the term synchronous being applied to confirm AML in the bone marrow (also called intramedullary AML). Extramedullary AML occurs at 1% to 2% incidence in human patients newly diagnosed with AML, with synchronous eAML being the more common variant. 30
In humans with AML, commonly involved extramedullary anatomic sites are lymph nodes, soft or connective tissues, skin, central nervous system, gastrointestinal tract, and the mediastinum.1,10,14,26 There are 5 reported cases of AML in extramedullary tissues in dogs, including AML affecting the spinal cord and epidural space in 2 dogs, lymph nodes in 4 dogs (with partial or complete effacement of architecture on histopathologic examination in 2 dogs, fulfilling WHO criteria for myeloid sarcoma), and the mediastinum (with mass formation) in 1 dog. Neoplastic infiltrates were also identified in other tissues in some of the dogs, such as the liver, spleen, and kidney.8,16,34 In addition, a retrospective radiographic study highlighted the importance of considering AML as a differential diagnosis in dogs with detectable mediastinal masses, 12 although this report lacked histologic confirmation of tissue effacement; a WHO requirement for the diagnosis of myeloid sarcoma.2,3,35 Reports of myeloid sarcoma in other species include eosinophilic granulocytic sarcoma in a pig and a white rabbit4,25 and a myeloid sarcoma in the mediastinum of a common marmoset. 20
Because extramedullary involvement is not typically considered to be a clinical manifestation of AML, a diagnosis of AML is frequently overlooked when dogs are evaluated for organomegaly, such as peripheral lymphadenopathy. In our experience, a prevalence of blasts in aspirates from such nodes is frequently interpreted as lymphoma. In this retrospective study, we describe the clinical and clinicopathologic features, treatment, and outcome of dogs with AML that have concurrent peripheral lymph node involvement. We have chosen to use the term eAML, reserving a diagnosis of myeloid sarcoma for those dogs in which there was histologic confirmation of tissue effacement. Our goal was to determine if there are clinical or laboratory features that can assist clinicians and pathologists with recognizing an AML that is infiltrating or effacing peripheral lymph nodes.
Materials and Methods
Case Selection and Selection Criteria
Medical records of 23 dogs with a diagnosis of AML and concurrent lymph node involvement between January 2016 and January 2024 were reviewed across 4 institutions. The cases came from several sources: (1) archived cases at each institution in which lymph node aspirates with cytologic examination had been performed as part of routine diagnostic evaluation (n = 8) and (2) cases prospectively enrolled in a multi-institutional research study of AML that started in 2020 and was still ongoing at the time of conclusion of this study (February 2024) (n = 15). For the latter study, dogs with acute leukemia were prospectively enrolled for phenotyping the leukemia and were either direct referrals to the oncology or internal medicine service of the participating institution or samples from dogs that were submitted to institution 1 (Cornell University) from the primary veterinarian or an oncology service at a nonparticipating institution (outside submissions) (Supplemental Figure S1). Client consent for study participation was obtained. Predefined inclusion criteria were: (1) stained cytologic smears of a lymph node aspirate were available for review; (2) on review, the cytologic smears contained ≥50% myeloid blasts plus differentiating (immature and mature) myeloid cells, based on a reasoning that this cut-off reflected substantial infiltration of the lymph node; (3) a confirmed diagnosis of AML; and (4) availability of complete medical records, including treatment and outcome data. The diagnosis of AML was based on morphologic findings in cytologic or histologic smears of the lymph node, hematologic results, and flow cytometric- and cytochemical-based phenotyping of lymph node aspirates, bone marrow aspirates, or blood.
Case Data
The following data were retrieved from the medical records: patient signalment; history; clinical signs; physical examination findings at initial presentation, including the number, size, and location of enlarged peripheral lymph nodes; hematologic, biochemical, and urinalysis results on admission after referral or date of sample collection for inclusion in the AML research study; imaging results, cytologic results of initial and any repeat lymph node aspirates; bone marrow cytologic results; histopathologic results from surgical biopsies or post-mortem tissues; flow cytometric and cytochemical staining results, including flow cytometric results from initial lymph node aspirates; treatment information; and outcome data, including changes in lymph node size when documented in the record. When blood smears were available from samples submitted for the AML study, a blood smear examination was performed by 1 author (TS, Cornell University), and results were included in the summary of the hemogram findings. Because different analyzers and reference intervals were used across the study population, we applied reference intervals established for dogs at Cornell University (per American Society of Veterinary Clinical Pathology guidelines 13 ) to identify abnormal results.
Review of Smears From the Lymph Node Aspirates
Modified Wright’s-stained cytologic slides of lymph node aspirates from archived and prospectively collected cases were retrieved and reviewed by a clinical pathology resident and a board-certified clinical pathologist coauthor at each institution. The review included a 100-cell differential count to document the percentage of blasts, individual granulocytes (combining mature and immature forms), monocytes, lymphocytes (combining small and large cells), plasma cells, and macrophages. Only low numbers of neutrophils, eosinophils, and monocytes are seen in aspirates from normal lymph nodes28,37; therefore, we selected a ≥5% cut-off for reflecting increased numbers of these cells in the lymph node. The blasts were reviewed for morphologic features of myeloid differentiation31,32 (Supplemental Table S1), and the presence of immature or mature megakaryocytes and dysplasia in myeloid cells was noted.
Confirmation of the Diagnosis of Acute Myeloid Leukemia
A diagnosis of AML was confirmed by identifying myeloid lineage(s) for cells in lymph node aspirates or blasts in blood or bone marrow aspirates, using a combination of morphologic features (Supplemental Table S1), flow cytometric analysis, and cytochemical staining reactions. For all cases, flow cytometric analysis, using a panel of antibodies to lymphoid and myeloid markers (Supplemental Table S2), was performed on lymph node or bone marrow aspirates or peripheral blood at the time of referral or upon receipt of samples for the AML research study. In venous blood and bone marrow samples, we restricted our analysis to suspect tumor cells, ie, those with low to high forward scatter (FSC, size) and lower side scatter (complexity) than neutrophils. For CD34, positive (+) labeling were defined as ≥5% of gated suspect tumor cells, whereas ≥20% labeled cells was used for all other markers. 31 For lymph node aspirates, cells with low FSC and intermediate to high FSC were gated separately, when possible, reasoning that residual non-neoplastic lymphocytes may fall into the low FSC gate. Similar to the cytologic assessment of the nodes, ≥5% labeled cells was used to indicate an infiltrate of either CD34+ precursor cells or myeloid cells expressing one or more myeloid marker, including CD11b, CD11c, CD14, CD18, and CD4.15,31 We included CD4 as a myeloid marker as it is expressed by mature canine neutrophils as well as T lymphocytes. We have previously used CD4+ labeling in cells that are negative (-) for the T-cell markers, CD3 or CD5 or both, to identify AML. 31 Given that CD34+ cells in an acute leukemia are usually major histocompatibility complex II (MHCII)-,15,31,32 whereas CD34+ cells in B-cell lymphoma are MHCII+, 37 we subdivided CD34+ cells into CD34+/MHCII+ and CD34+/MHCII- subsets when possible. In addition, we quantified the percentage of CD14+ monocytes that were MHCII- when cells were co-labeled with both markers. Monocytes are normally MHCII+ but can be MHCII- in acute leukemia. 15 Flow cytometric analysis with a similar panel of antibodies was done by other laboratories for the prospectively enrolled cases in the AML study (Supplemental Figure S1). In addition, for prospectively enrolled cases, cytochemical staining for alkaline phosphatase, alpha-naphthyl butyrate esterase, chloroacetate esterase, myeloperoxidase, and Sudan black B were performed on samples if there were sufficient slides and tumor cells in the smears, as described previously.6,32 Cytochemical staining results were included if performed on the archived cases (Supplemental Figure S1). Cytochemical staining results consistent with AML included positive reactions for one or more of the stains in a pattern typical of myeloid cells27,31,32 (Supplemental Table S3).
Treatment and Outcome
Treatment protocols were divided into chemotherapy (with or without glucocorticoids), palliative (glucocorticoids only), and no treatment. The effect of treatment on lymph node size was reviewed when documented in the medical records.
Statistical Analysis
Data are descriptive and presented as median and range. Overall survival was calculated using Kaplan-Meier curves for all dogs from the time of diagnosis until death from any cause (GraphPad Prism version 10.2.3, GraphPad Software LLC). For dogs treated with chemotherapy or corticosteroids, overall survival was also calculated from the time of initiation of treatment until death from any cause. A log-rank analysis was used to assess differences in outcome according to treatment (chemotherapy vs corticosteroids). Dogs were censored from analysis if death had not occurred and if they were lost to follow-up at the time of analysis.
Results
Patient Demographics
The median age of the 23 dogs was 8 years (range = 1-14 years). Fourteen dogs were females, including 13 spayed and 1 intact dog, and the remaining 9 dogs were neutered males. Thirteen dogs were of large breeds (57%, ≥23 kg), and 5 each (22%) were of medium (10-22 kg) or small (≤10 kg) breeds. The large breed dogs included 4 golden retrievers, 2 goldendoodles, 2 Labrador retrievers, 3 mixed breed dogs, and 1 each of Rottweiler and pitbull terrier. The medium-sized dogs included 3 mixed breed dogs, 1 Catahoula leopard dog, and 1 German shorthaired pointer. Of the small breed dogs, there were 2 mixed breed dogs and 1 each of Maltese, miniature dachshund, and Yorkshire terrier (Supplemental Table S4).
Patient History and Clinical Signs
Peripheral lymphadenopathy was either part of the reason for presentation (9/23, 39%, including 2 dogs with enlarged lymph nodes that formed cervical or neck lumps) or found incidentally on physical examination (14/23, 61%). Information on the involved lymph nodes was available in 21 dogs, although the specific lymph nodes were not stated in 1 dog. Only 2 dogs had solitary lymph node enlargement (mandibular or prescapular), whereas the remaining dogs had multiple, usually bilaterally, enlarged nodes (median of 4 enlarged nodes). The prescapular (16/20, 80%), mandibular (14/20, 70%), and popliteal (10/20, 50%) nodes were the most commonly affected sites. Lymph node enlargement ranged from mild to marked (median = 2.9 cm, range = 1.2-6.0 cm). Lymph nodes were also described as being firm. Other presenting complaints included hyporexia or anorexia (9/23, 39%), lethargy (5/23, 22%), fever (4/23, 17%), weight loss (3/23, 13%), lameness (3/23, 13%), vomiting (3/23, 13%), coughing (2/23, 9%), and panting (2/23, 9%) (Supplemental Table S4).
Cytologic examination of lymph node aspirates had been previously performed on 21 dogs (91%), including all 8 archived cases and 13 prospectively enrolled cases in the AML study. In the remaining 2 dogs, lymph node smears were taken by the oncologist and submitted as part of the AML study, without prior assessment by another pathologist. The initial cytologic diagnosis in the 21 dogs consisted of not-otherwise-specified hematopoietic neoplasia (8/21, 38%), AML (6/21, 29%; 3 definitive, 3 presumptive), lymphoma (5/21, 24%), lymphoid hyperplasia (1/21, 5%), and a predominance of granulocytic precursors due to extramedullary hematopoiesis or myeloproliferative disease (1/21, 5%). Lymph node biopsies were performed on 2 dogs, with diagnoses of myeloid sarcoma (Supplemental Figure S2) and non-B non-T-cell lymphoma (Supplemental Table S5). Bone marrow aspirates were performed on 5 dogs, with cytologic diagnoses of hematopoietic neoplasia (2/5; suspect lymphoma or acute lymphoblastic leukemia), AML (2/5; 1 presumptive), and pockets of myeloblasts with dysplasia (1/5) (Supplemental Table S5).
Review of Lymph Node Cytologic Smears
On re-examination of the cytologic smears from the 23 dogs, blasts were identified in all samples (median = 62%, range = 18%-88%) (Supplemental Table S6 and Supplemental Figures S3-S10). The blasts were generally intermediate to large (10-20 µm), with moderate to high nuclear to cytoplasmic ratios (>2:1 nucleus: cytoplasm) and central to eccentric, round to oval nuclei with fine chromatin and prominent nucleoli (Fig. 1). Blasts frequently showed features of myeloid differentiation, including monocytoid nuclei, moderate amounts of light blue cytoplasm, and cytoplasmic granules (ranging from a pink granular blush to bright pink to magenta to purple mast cell-like granules) (Fig. 1a, b). In 15 cases (65%), blasts were accompanied by increased percentages of myeloid cells, including mature and immature neutrophils in 43% of dogs(10/23, median = 3%; range = 0%-56%), monocytes in 22% of dogs (5/23, median = 1%; range = 0%-27%), and, rarely, eosinophils in 9% of dogs (2/23, median = 0%; range = 0%-5%), in various stages of maturity (Fig. 1c, d). Promonocytes were included in the blast counts, and granular blasts were identified in 8 cases (35%) (Fig. 1a). In many cases, blasts were present among a background of lymph node constituents, including small and large lymphocytes (median = 23%; range = 1%-47%) with fewer plasma cells (median = 2%; range = 0%-12%), supporting an infiltrative population (Fig. 1c, d). Dysplastic changes were noted in monocytes and neutrophils in 10 cases (43%) (Fig. 1c). Dysplastic features included giant bands or monocytes, hypolobulation of neutrophil nuclei, variably sized nuclear lobes in neutrophils and monocytes, abnormally large monocyte nuclei, hypersegmented neutrophil nuclei, cells with overlapping features of neutrophils and monocytes, and features of toxic change in neutrophils. In 1 case (dog 5), aspirates were performed on both enlarged prescapular lymph nodes and the left popliteal lymph node, the latter of which was firm but not enlarged. Results from the prescapular node showed an infiltrate of cells with features consistent with AML, whereas the popliteal lymph node contained cells with cytologic features compatible with a T-zone cell expansion (intermediate lymphocytes with clumped chromatin and cytoplasmic tails). 29 Low numbers of T-zone cells were also seen in the smears from the prescapular lymph nodes. Flow cytometric analysis on the prescapular node aspirate from the dog was done by another laboratory 2 months before inclusion in this study and was consistent with AML and a concurrent T-zone cell expansion.
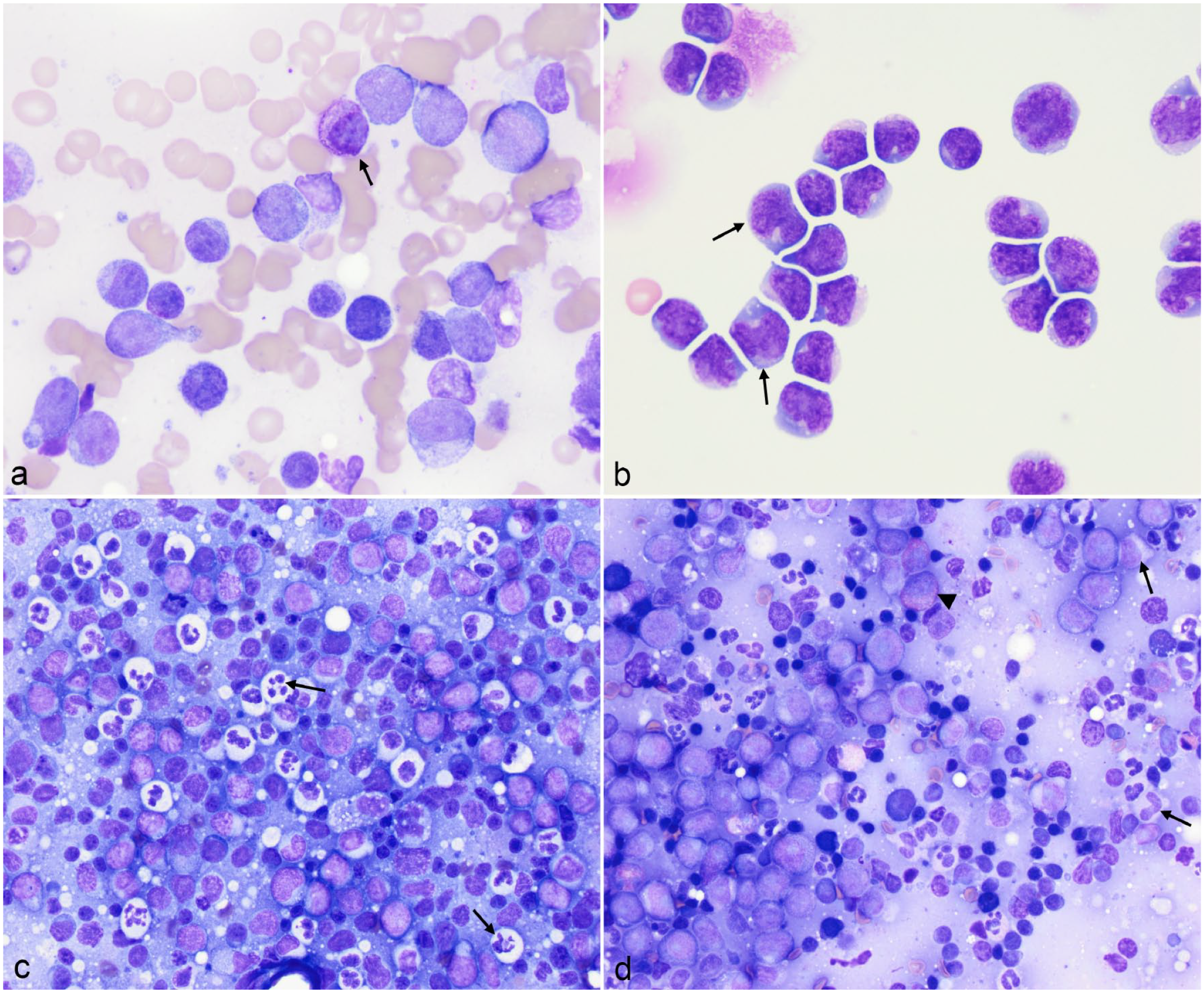
Cytologic patterns and morphologic features of blasts in smears of lymph node aspirates from dogs with extramedullary acute myeloid leukemia and lymph node involvement. (a) Blasts are the predominant cells and show features of myeloid differentiation, including fine magenta to purple granules (arrow) and increased amounts of medium blue cytoplasm. Dog 4. Modified Wright’s stain. (b) Blasts show myeloid differentiation with monocytoid nuclei (arrows). Dog 12. Modified Wright’s. (c) There are many neutrophils with dysplastic features, such as bizarre nuclear shapes (arrows), admixed with blasts and residual lymph node constituents, including small lymphocytes, plasma cells, and macrophages. Dog 5. Diff-quik stain. (d) There is an infiltrate of differentiating myeloid cells, including eosinophils (arrowhead) and neutrophils (arrows; myelocyte top right, band neutrophil lower right), with a high proportion of blasts and low to moderate numbers of residual small lymphocytes, supporting an infiltrative myeloid neoplasm. Dog 14. Modified Wright’s.
Hemogram, Biochemical, and Urinalysis Results
Most of the dogs were either bi- (9/23, 39%) or pancytopenic (10/23, 44%). Two dogs had no cytopenias, with mild other hematologic abnormalities (Supplemental Table S7). The most common hematologic findings were a normocytic, normochromic to hypochromic nonregenerative anemia followed by thrombocytopenia and neutropenia, with 48% (11/23) of dogs having a left shift in neutrophils. Toxic changes in neutrophils, specifically cytoplasmic basophilia and Döhle bodies, were seen in 4 dogs (17%). Circulating blasts, including granular forms, were identified in blood smears from 18 dogs (78%) and comprised between 1% and 82% of the differential cell count in 16 dogs (Fig. 2a, b and Supplemental Figures S11-S18). Eight dogs (35%) fulfilled WHO criteria of ≥20% blasts in blood for a diagnosis of AML.3,35 Nucleated red blood cells were counted during the differential leukocyte count in 9 dogs (39%) (median = 0; range = 0-24 nucleated red blood cells/100 white blood cells) (Table 1 and Supplemental Table S7). Morphologic abnormalities (dysplasia) were noted in 13 cases (57%) and were most prominent in neutrophils and monocytes (Fig. 2c, d, Supplemental Figures S13-S14) and similar to that described for the lymph node aspirates. Platelet dysplasia included abnormal granulation and giant forms. In addition, cells resembling micromegakaryocytes (small cells with round nuclei and small amounts of medium blue-gray cytoplasm with cytoplasmic blebs) were identified in 2 cases (Supplemental Figures S15-S16). Erythrocyte membrane blebs and bilobed nuclei in nucleated red blood cells were observed in 1 case.
Summary of selected hematologic findings in 23 dogs with acute myeloid leukemia with peripheral lymph node involvement.
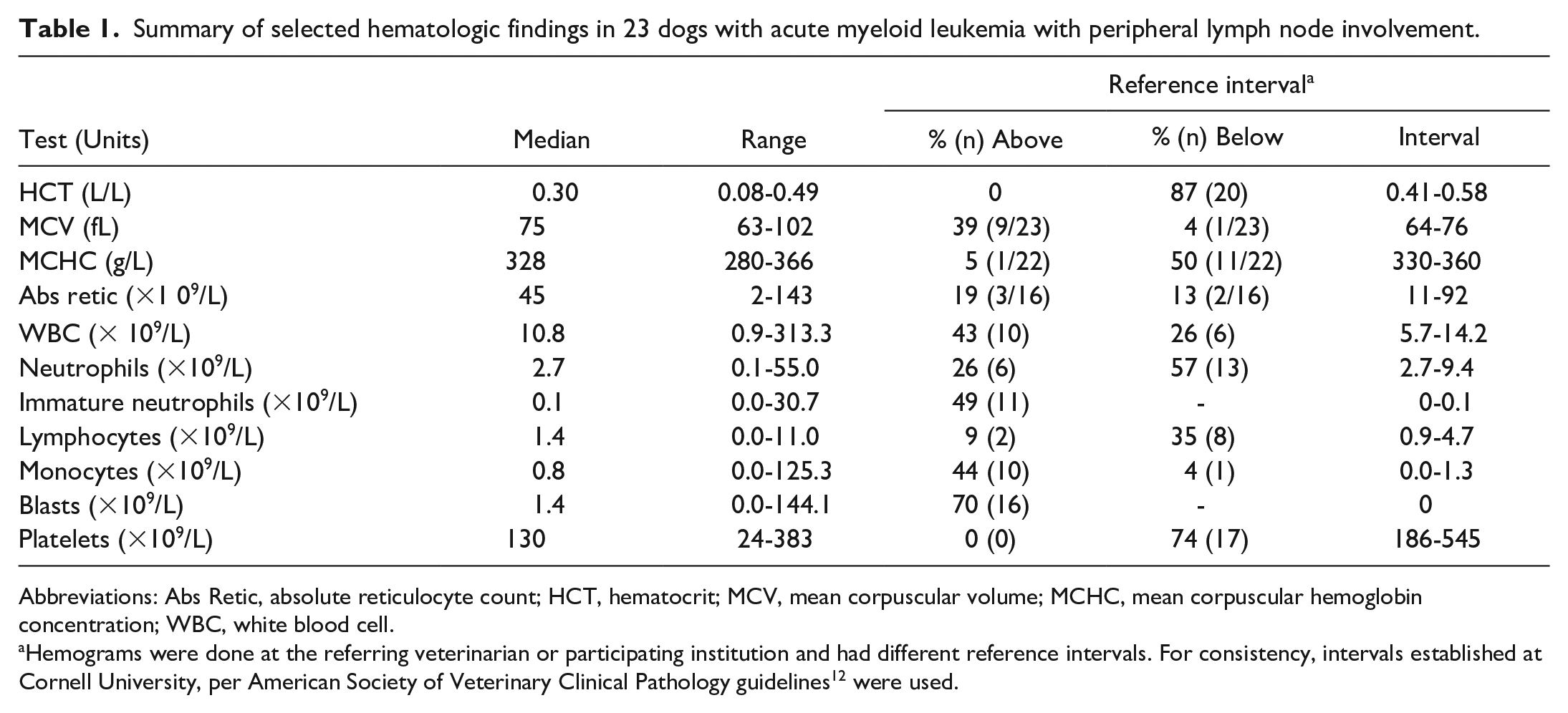
Abbreviations: Abs Retic, absolute reticulocyte count; HCT, hematocrit; MCV, mean corpuscular volume; MCHC, mean corpuscular hemoglobin concentration; WBC, white blood cell.
Hemograms were done at the referring veterinarian or participating institution and had different reference intervals. For consistency, intervals established at Cornell University, per American Society of Veterinary Clinical Pathology guidelines 12 were used.
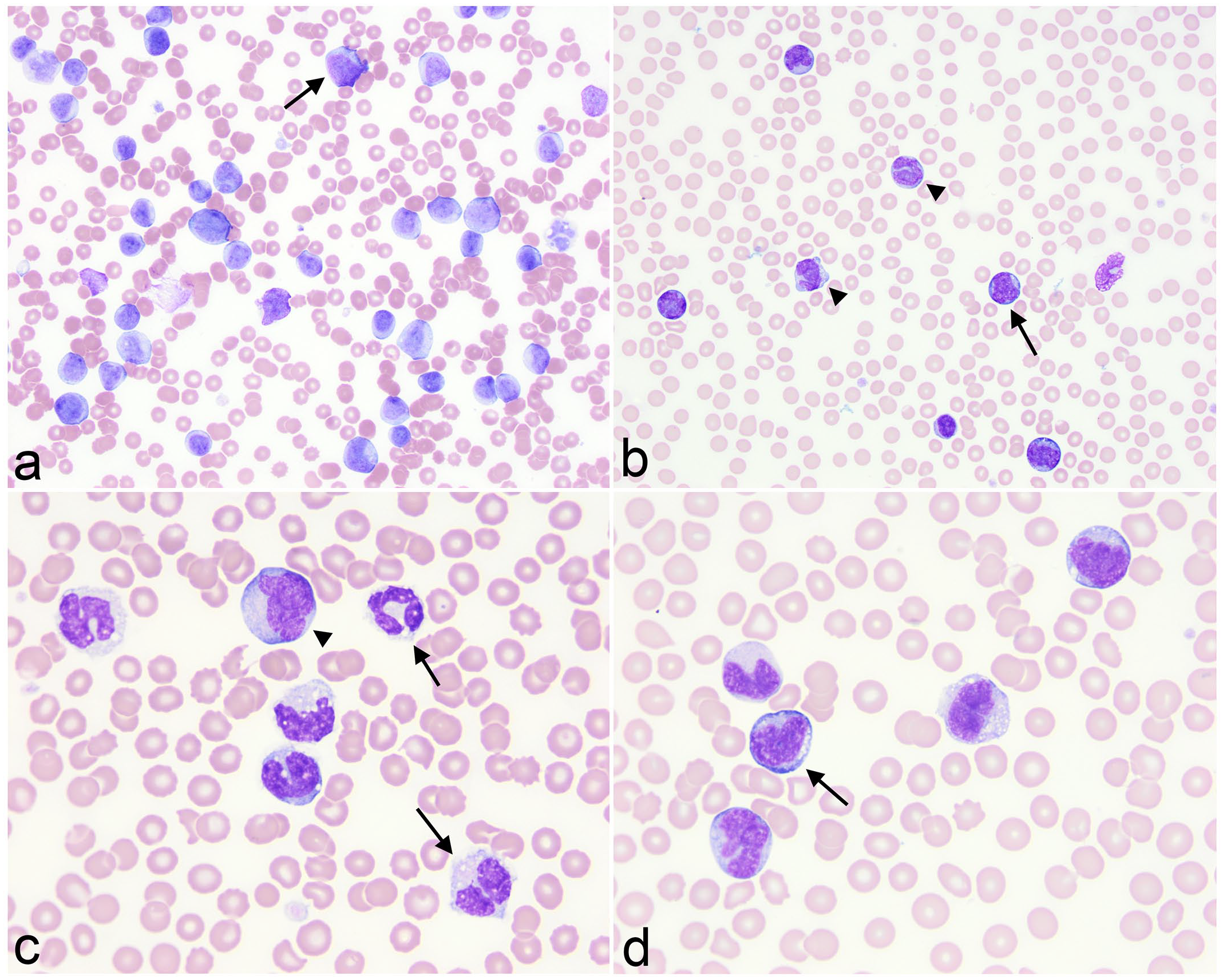
Circulating blasts and multilineage dysplasia in venous blood of dogs with extramedullary acute myeloid leukemia and lymph node involvement. Modified Wright’s stain. (a) Variably sized blasts (arrow) with fine chromatin comprise 72% of the differential count in a dog with marked leukocytosis (white blood cell count 184.4 × 109/L). The blasts have subtle features of myeloid differentiation, such as monocytoid nuclei. Dog 10. (b) There is a gradation between blasts (arrow) and mature monocytes (arrowhead), supporting monocytic differentiation. Dog 3. (c) Dysplastic changes in myeloid cells, which have overlapping features of neutrophils and monocytes (arrows), making them difficult to classify with certainty. The monocyte is mildly dysplastic, being larger than normal (arrowhead). Dog 2. (d) Prominent monocytic differentiation with mature monocytes, some of which are dysplastic with abnormal nuclear shapes, and a promonocyte (arrow). Promonocytes are included as blasts in WHO criteria of monocytic variants of acute myeloid leukemia.2,3,35 Dog 3.
Serum or plasma biochemical analytes were measured in 22 dogs (96%) and were within reference intervals in 3 dogs. The most common abnormalities were an increased alkaline phosphatase activity (13/22, 59%), hyperglobulinemia (6/22, 27%), and hypoalbuminemia (6/22, 27%) (Supplemental Table S8). A urinalysis was performed in 8 dogs, and no obvious abnormalities were detected.
Immunophenotyping and Cytochemical Staining
Flow cytometric analysis was performed on lymph node aspirates alone (12 dogs), blood alone (7 dogs), lymph node aspirates and blood (2 dogs), and bone marrow aspirates with and without concurrent blood samples (1 dog each). In the lymph node aspirates, there were ≥5% CD34+, typically MHCII-, cells in 9/14 dogs (64%) (Supplemental Table S9). Twelve dogs (86%) had myeloid infiltrates based on ≥5% positivity for myeloid antigens. In most dogs, the percentage of CD14+ cells exceeded the monocyte percentage in the differential count and these cells were frequently MHCII- (Supplemental Table S9). Cytochemical staining was performed on lymph node smears in 9 dogs, including 1 dog whose cells lacked myeloid markers on flow cytometric analysis. The blasts showed a range of positive reactions consistent with a myeloid lineage leukemia (Supplemental Table S9).31,32 Prior flow cytometric analyses performed in 5 dogs in the AML study were similar to the repeat results.
In the 10 blood samples, 5 (50%) had ≥5% CD34+ cells, which were mostly MHCII- in 3 dogs. The blast percentage in these 5 dogs ranged from 7% to 72% of the differential cell count. Since cells with myeloid antigens could be mature monocytes or segmented neutrophils, we compared the percentage of cells positive for myeloid antigens to the differential cell count. In 4/6 dogs with CD14+ cells; the percentage of CD14+ cells exceeded the monocyte percentage in the differential cell count. Two cases were CD18+ but CD14- (Supplemental Table S9). Cytochemical staining on blood smears supported a myeloid lineage for the blasts in all 8 cases in which the testing was performed. Flow cytometric results on paired blood and lymph node aspirates or paired blood and bone marrow were similar in the 4 dogs.
The flow cytometric and cytochemical staining results were used with the hemogram, other clinical pathology findings, and histopathologic analysis to finalize a diagnosis of AML in all cases, with 2 dogs having a diagnosis of myeloid sarcoma based on effacement of lymph node architecture on histologic examination (dogs 21, 22). In 5 dogs with bone marrow aspirates, the eAML was synchronous in 4 (90%) and isolated in 1 (10%) dog (dog 4) (Supplemental Table S5 and Supplemental Figure S19).
Imaging Studies
Thoracic radiography or abdominal ultrasonography was performed in 16 (70%) and 14 (61%) dogs, respectively. Abnormal thoracic findings were seen in 4 dogs and consisted of a mediastinal mass (3/16, 19%) and parenchymal lung disease (1/16, 6%). An aspirate of the mediastinal mass was performed in 1 dog (dog 11), and smears consisted of blasts similar to those seen in smears from the lymph node and bone marrow aspirates. The cytologic diagnosis was a probable hematopoietic neoplasm, suspect lymphoid in origin. Major findings on ultrasound examination were abdominal lymphadenopathy (10/14, 71%), splenomegaly (7/14, 50%), and hepatomegaly (6/14, 43%). None of these internal abdominal organs or masses was aspirated.
Treatment and Outcome
Seven dogs were euthanized within 1 day after diagnosis. Of the remaining dogs, 10 dogs received chemotherapy and glucocorticoids and 6 dogs were treated with glucocorticoids. Dogs were also given supportive care, such as gastric protectants or anti-emetics (11/16, 69%) and antibiotics (6/16, 38%). The 10 dogs were treated with different chemotherapeutic protocols, including 1 agent (2/10; prednisolone with doxorubicin or
Only 1 of the 6 dogs given corticosteroids had information in the medical record on lymph node size after starting therapy. The lymph nodes decreased from 2.9 to 1 cm within 1 month of treatment. However, the lymph node size then increased within a month and the dog became hyporexic and was humanely euthanized 45 days after initial diagnosis. The median survival in these 6 dogs was 22 days (8-90 days), which was not significantly different from dogs given chemotherapy (P = .055) (Fig. 3). Overall survival for all 23 dogs with eAML, including dogs euthanized upon diagnosis, was 22 days (range = 1-360 days), with 11/16 (69%) treated dogs living for 30 days or more.
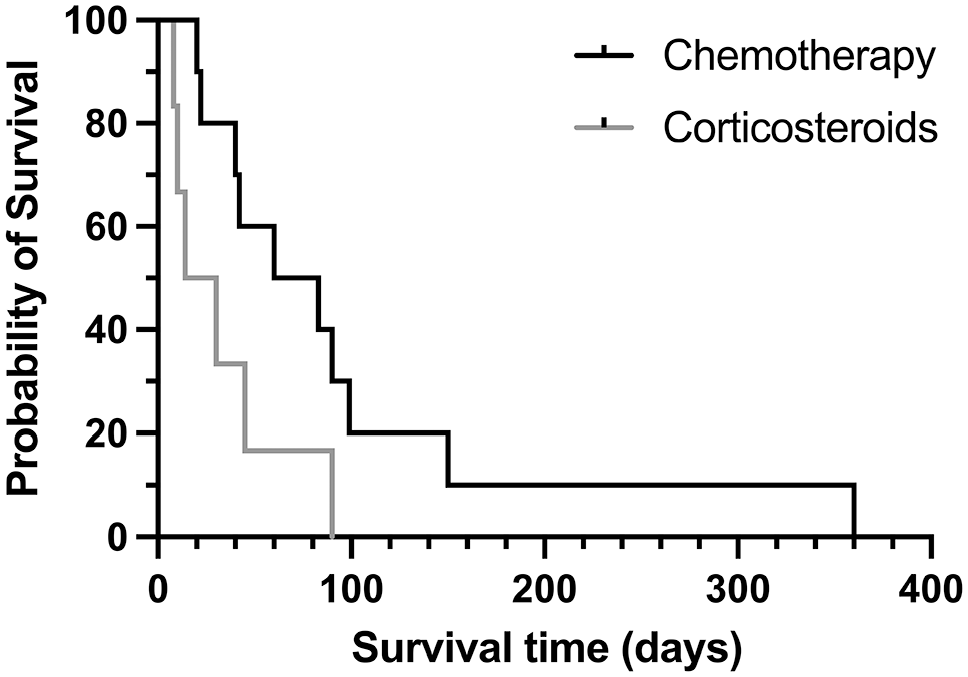
Kaplan-Meier survival curves for dogs with acute myeloid leukemia involving peripheral lymph nodes and treated with chemotherapy or corticosteroids. The curves were not significantly different (log-rank test, P = .055).
Discussion
Here, we present a case series of 23 dogs with AML where peripheral lymphadenopathy was the reason for presentation or an incidental finding on physical examination in dogs that were presented for other reasons, including wellness examinations or routine vaccinations. Our goal was to describe clinical and laboratory features that would be helpful in alerting clinicians and clinical pathologists as to the potential of AML in these dogs. Our results highlight the importance of contemporaneous hemogram testing and close scrutiny of blood smears and lymph node aspirates for features that yield clues as to the presence of a myeloid neoplasm. In dogs that are presented for lymphadenopathy, the main differential diagnosis is usually lymphoma, which is far more common than AML. The dogs that were presented with peripheral lymphadenopathy in this study had multiple enlarged lymph nodes and were mostly middle-aged or older (5-14 years), with no apparent sex predilection. In contrast, there was a slight male dominance in case series of myeloid sarcoma in humans.1,10,14,26,39 When documented in the medical record, lymphoma was the most common working diagnosis in these dogs. However, the presence of bi- or pancytopenia and circulating blasts on concurrent hematologic testing, common findings in dogs with acute leukemia,9,33 should alert a clinician as to the possibility of AML vs lymphoma in dogs with peripheral lymphadenopathy. Of the dogs in this study, 83% had such hematologic abnormalities, with circulating blasts being detected on scanning a blood smear in 1 dog with a solitary neutropenia, indicating that the hemogram and blood smears provide useful clues. However, 2 dogs with incidental lymphadenopathy lacked cytopenias or circulating blasts in blood, indicating that AML does not always manifest with the expected hemogram abnormalities. In a previous case series of 35 dogs with AML, several dogs lacked cytopenias or circulating tumor cells and 1 dog had a normal hemogram. 9 Similarly, 3 dogs with myeloid sarcoma had unremarkable hemograms at the time myeloid infiltrates were detected by flow cytometric analysis in aspirates of enlarged peripheral lymph nodes or pleural effusion (dog with a mediastinal mass). 34 Thus, normal hemogram results do not exclude a diagnosis of AML and may suggest an isolated versus synchronous eAML.
In the previous case series by Davis et al, 9 peripheral lymphadenopathy was documented in 76% of dogs with AML. In the current study, most dogs had multiple enlarged lymph nodes with only a few having solitary lymph node enlargement or bilateral involvement of 1 site (mandibular or prescapular lymph nodes). As previously reported, 9 lymph node enlargement was generally mild with only 23% of dogs having lymph nodes that were >3 cm in size. In addition, 24% of our cases had a diagnosis of lymphoma made from initial lymph node aspirates. It was only when additional phenotyping was done on the lymph node that a diagnosis of AML became apparent. Similarly, a cytologic diagnosis of lymphoma was made on an infiltrative AML in peripheral nodes of a dog in a 3-case series. 34 When imaging was performed, internal lymphadenopathy and hepatomegaly or splenomegaly were documented in 70% and 43% to 50% of cases, respectively. Similarly, enlargement of these organs was identified in 52% to 70% of dogs in the 35-dog case series. 9 Although not evaluated in this study, we speculate that the leukemia concurrently infiltrated these sites. Three dogs in this study also had mediastinal masses, with 1 dog having a diagnosis of “suspect lymphoid neoplasm” from a mass aspirate. Mediastinal masses occur in dogs with AML9,12,34 and, like lymph node aspirates, are often interpreted as lymphoma on cytologic and histologic assessment of the mass. Since the liver, spleen, and mediastinum are involved in hematopoiesis in the fetus and adult animal, and stem cells can normally home there, 30 it is not surprising that AML can infiltrate these sites. Taken together, these results support our contention that AML should be added as a differential diagnosis for dogs with peripheral lymphadenopathy (with or without internal organomegaly), particularly when mild to moderate in degree, and that smears of lymph node or other organ aspirates be examined for features supporting myeloid neoplasia.
Cytologic features that supported a myeloid neoplasm in the lymph node aspirates in this case series were (1) increased numbers of blasts with myeloid features, such as increased amounts of light blue cytoplasm and monocytoid nuclei; (2) granular blasts; (3) presence of differentiating granulocytes; (4) mature monocytes with monocytoid blasts or promonocytes; (5) dysplastic changes in mature or immature myeloid cells (granulocytes, monocytes); and (6) evidence of an infiltrative neoplasm with retention of residual lymph node constituents. Similar cytologic features with differentiating or dysplastic myeloid precursors and granular blasts were described for 2 other dogs with AML infiltrates in peripheral lymph nodes. 34 However, none of these findings alone are definitive for AML, making it difficult to conclusively diagnose AML in aspirates of organs based only on morphologic features. Several of the myeloid features were documented on the initial cytologic smear examination, prompting the use of the encompassing term of “hematopoietic neoplasia” or yielding a definitive diagnosis of AML in a few cases. We recommend using the nonspecific term of “hematopoietic neoplasia” when neoplastic cells have myeloid features, indicating the tumor could be of myeloid or lymphoid origin. As stated previously, cytopenias in a hemogram may also help sway the diagnosis to an acute leukemia versus lymphoma.
Flow cytometric analysis was a useful tool for identifying eAML in lymph node aspirates in this and another small case series. 34 Of the 23 cases herein, myeloid infiltrates were identified, with or without a population of CD34+/MCHII- cells, in the lymph node aspirates of all cases in which this analysis was done and included antibodies against myeloid antigens. Thus, flow cytometric detection of myeloid cells or CD34+/MHCII- cells in lymph node aspirates supports a diagnosis of eAML. However, CD34+/MHCII- cells can also be seen in acute lymphoblastic leukemia, 15 so this finding does not confirm an AML. The neoplastic cells in 10 dogs in this study were not CD34+, reinforcing that a lack of CD34+ cells does not preclude a diagnosis of AML in dogs. Similarly, extramedullary tumor cells in human patients with myeloid sarcoma may not be CD34+, 26 including monocytic variants. 7 Flow cytometric analysis on repeat lymph node aspirates were similar to the original flow results in 5 cases, indicating that different laboratories can provide the same results despite use of different antibodies and criteria for diagnosis of AML. 15 Flow cytometric analysis was not done on lymph node aspirates from all dogs, which is a limitation of the study. However, morphologic features of cells in the lymph node taken together with hemogram and flow cytometric results on blood or bone marrow supported eAML over another neoplasm in the nodes.
We defined a stringent cut-off of ≥50% myeloid cells in lymph node aspirates for this study to ensure that there were substantive infiltrates of AML in the nodes. In 2 other dogs with AML lymph node infiltrates, 53% to 71% of cells expressed myeloid markers in lymph node aspirates on flow cytometric analysis. 34 However, differences in the degree of infiltration may exist between nodes and lower levels of infiltrates do not preclude a diagnosis of eAML in lymph node aspirates. Given that extramedullary hematopoiesis is uncommon in lymph nodes, the finding of immature myeloid precursors in lymph node aspirates should prompt consideration of an acute or chronic myeloid neoplasm. An incorrect diagnosis of lymphoma is not unique to cytologic assessment but also occurs with histologic evaluation of biopsy specimens, as occurred with 1 dog in this case series. Similarly, misdiagnosis of myeloid sarcoma is also reported in human patients, where the neoplasm is difficult to differentiate from lymphoid tumors, particularly non-Hodgkin’s lymphoma.5,26 Infiltrates of AML in lymph nodes can be diffuse or localized to paracortical regions (encasing residual follicles) and medullary sinuses, and single-file patterns can be seen.19,26 The treatment protocols for lymphoma and AML can be different,9,18 highlighting the importance of a correct diagnosis to aid in selection of the most appropriate treatment.
We gave 2 cases (dogs 21 and 22) an antemortem diagnosis of myeloid sarcoma based on histologic evidence of effacement of lymph node architecture, aligned with WHO criteria: “myeloid sarcoma is a tumor mass consisting of myeloid blasts with or without maturation in an anatomical site other than the bone marrow. Infiltrates of any site in the body by myeloid blasts in leukemic patients are not classified as myeloid sarcoma unless they present with tumor masses in which the tissue architecture is (completely or partially) effaced.” 35 This definition remains unchanged in recent updates to WHO criteria and in the International Consensus Classification of AML.2,3 These extramedullary tumor masses in humans usually involve single sites, including lymph nodes, soft tissue and bone, skin, gastrointestinal and reproductive tracts, and mediastinum, but can affect more than 1 site.1,14,39 In 2 case series in humans, lymph nodes were the most common site of involvement.14,39 Myeloid sarcoma can occur in the absence of an overt leukemia in bone marrow (called “primary,” “de novo,” or “isolated”) or concurrently with intramedullary AML and may represent blast transformation of myelodysplastic syndrome or chronic myeloproliferative diseases.1-3,10,14,39 Another dog in this study (dog 13) would also fit the WHO definition of myeloid sarcoma, having effacement of multiple internal lymph nodes along with intramedullary AML on postmortem examination. In the remaining dogs, we could not distinguish between a myeloid sarcoma and infiltrative AML not effacing lymph node architecture because histologic evaluation of lymph nodes was not done.
The discrepancy in disease definition in the literature of myeloid sarcoma vs eAML affects comparison between studies. It should be noted that the authors that proposed the term eAML excluded sites of hematopoiesis, including the lymph node, liver, and mediastinum, restricting the definition of eAML to tumor infiltrates in nonhematopoietic tissue, such as the skin and central nervous system. They also excluded AML in body cavity fluids and localized noneffacing infiltrates (eg, gums) from the definition of eAML. 30 We considered these exclusions too restrictive in dogs and propose to adopt the term of eAML instead of myeloid sarcoma for dogs where the tumor is found in extramedullary tissues, including lymph nodes and the mediastinum, in cytologic smears of aspirates. Requiring the WHO definition of effacement of tissue architecture is unrealistic, given that histologic assessment of extramedullary tissues is not routinely done in dogs antemortem. We also recommend applying the modifiers “isolated” and “synchronous,” reserving the former term for cases in which there are <20% blasts in bone marrow, as seen in dog 5 in this study. Given that bone marrow aspirates are not routinely performed in dogs, ≥20% blasts in blood or fewer blasts with concurrent bi- or pancytopenia, as seen in most of the dogs in this study, could facilitate a presumptive diagnosis of synchronous eAML in dogs.
The mechanisms underlying AML infiltration of extramedullary sites are unknown. Various genetic mutations have been identified in human patients2,3,10,26,39 and gene sequencing of the extramedullary tumor and affected bone marrow has revealed discordancy in up to 1/3 of cases.11,17,24,36 How these genetic mutations may promote extramedullary infiltration is unknown, but there are data showing a worse prognosis in patients with different mutations in the extramedullary site compared to marrow. 39 Monocytic or myelomonocytic variants of AML constitute many of the eAML or myeloid sarcoma cases in humans21,30 and these are the most common subtypes of AML in dogs,31,32 which may explain the high percentage of reported cases of dogs with lymphadenopathy. 9 A subset of “cancer-initiating cells” expressing C1Q (one of the 3 subunits of the C1 component of complement) was uncovered in a patient with monocytic AML. These cells invaded extramedullary sites, particularly the skin and gastrointestinal tract, when xenotransplanted in immunodeficient mice. Tissue fibroblasts expressing the C1Q receptor promoted infiltration, persistence, and chemoresistance of the extramedullary tumor. 38 These results suggest that normal or abnormal homing mechanisms, facilitated by tumor-host cell interactions within the local microenvironment, govern the development of myeloid infiltrates in extramedullary tissues, but are unlikely to be unique to monocytic variants of AML.
It is difficult to compare survival between studies, given that owners may select chemotherapy, palliative therapy, or euthanasia, and different chemotherapeutic regimens are used to treat dogs with AML. In addition, reported median survival times usually include dogs euthanized after diagnosis due to the poor prognosis. Including the latter dogs, the overall median survival time in this study was 22 days, which is not that different from the 19 days reported in the previous 35-dog case series 9 but was slightly longer than 10 days in another case series of 38 dogs with different types of acute leukemia (B-cell, T-cell, undifferentiated, and AML) and follow-up information. 22 However, the 72 day median survival time was longer for those dogs given chemotherapy in this study compared to either of the previous case series (589 and 922 days, with the latter study including all types of leukemia not just AML). Using the Kaplan-Meier log-rank test, corticosteroid treatment was not associated with a significantly shorter survival in this study unlike that reported for the 35-case series (25 days). 9 The dog with longest survival time of 360 days had an isolated eAML and was given chemotherapy. Two other cases of isolated eAML or myeloid sarcoma in peripheral lymph nodes survived for 1 to 3 months after initial presentation or diagnosis, with corticosteroid treatment or delayed chemotherapy, 34 suggesting these dogs may have longer survival times. All 3 of these dogs eventually developed overt AML within 20 days to 9 months after the confirmed diagnosis of eAML. Progression to overt AML has been also reported in human patients with isolated myeloid sarcoma. 26 Both this and other case series9,18 show that individual dogs can survive for longer times than reported medians; however, we do not know the factors that dictate prognosis in dogs with or without extramedullary infiltrates. There is a need for improved prognostication in dogs with AML, given that genetic mutations can govern progression and prognosis of AML in humans.23,39 There are also discrepancies in survival in human patients with myeloid sarcoma. For instance, 1 study found no differences in survival in AML with or without myeloid sarcoma and lymph node involvement was not prognostic of survival with univariate analysis. 14 In contrast, another case series found that the presence of myeloid sarcoma conferred a poorer outcome. 10
Limitations of this study include the subjectivity and lack of standardization of cytologic assessment of lymph node aspirates. It can be difficult to impossible to reliably distinguish between blasts and immature cells of lymphoid or myeloid origin, emphasizing the need for immunophenotyping to determine cell lineage more definitively. Bone marrow was not aspirated in most of the cases. However, when there are ≥20% blasts and cytopenias in blood, phenotyping can be readily performed on blood and a bone marrow aspirate is not always justifiable. It would be worthwhile to evaluate bone marrow in dogs with mild or minimal cytopenias and no or low numbers of circulating blasts to distinguish between an isolated or synchronous eAML. This information may be important for prognostication, given that the dog with a cytologic diagnosis of isolated eAML had the longest survival in this study.
Supplemental Material
sj-docx-1-vet-10.1177_03009858241295397 – Supplemental material for Acute myeloid leukemia with peripheral lymph node involvement in dogs: A retrospective study of 23 cases
Supplemental material, sj-docx-1-vet-10.1177_03009858241295397 for Acute myeloid leukemia with peripheral lymph node involvement in dogs: A retrospective study of 23 cases by Jaspreet Kaur, Gabriella Diamantino, Katherine Morrison, Kristina Meichner, Nora L. Springer, Martha Hoffman, Dorothee Bienzle and Tracy Stokol in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the oncologists and veterinarians who submitted cases for inclusion in the prospective AML study, notably Dr Intile from North Carolina State University, Dr Regan from Sage Veterinary Hospital, and Dr Ewing from Angell Memorial Hospital. The authors also thank the owners who contacted the co-authors directly to include their dogs in the study or entered their dog information through Anitrials. The authors thank Carol Frederick, for generating webpages promoting the AML study and co-ordinating emails from Anitrial and other sources.
Author Contributions
JK wrote the manuscript, performed differential cell counts and cytologic assessment of the lymph node aspirates of cases submitted to institution 1, took and compiled images of cases at institution 1, and compiled medical record data in an excel spreadsheet from cases at institutions 1, 3, and 4. GD and KM performed differential cell counts and histologic and cytologic assessment of the lymph node and bone marrow aspirates or biopsies and blood smear examinations, took and compiled images, and compiled medical record data in an excel spreadsheet from cases at institution 2. KM and NLS performed differential cell counts and cytologic assessment of the lymph node aspirates and blood smear examinations of cases seen at institutions 3 and 4, respectively, and took images of cases at each institution. DB devised the study, edited the manuscript, and supervised GD and KM. MH performed cytochemical staining on prospective cases of acute leukemia. TS performed flow cytometric and statistical analysis, interpreted cytochemical staining, took and compiled images, performed differential cell counts and histologic and cytologic assessment of the lymph node and bone marrow aspirates or biopsies and blood smear examination, and co-wrote the manuscript with JK.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Riney Canine Health Center from Cornell University and Canine Health Foundation grant from the American Kennel Club (#2987).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
