Abstract
Canine oral malignant melanoma (COMM) is the most common neoplasm in the oral cavity characterized by local invasiveness and high metastatic potential. Hypoxia represents a crucial feature of the solid tumor microenvironment promoting cancer progression and drug resistance. Hypoxia-inducible factor-1α (HIF-1α) and its downstream effectors, vascular endothelial growth factor A (VEGF-A), glucose transporter isoform 1 (GLUT1), C-X-C chemokine receptor type 4 (CXCR4), and carbonic anhydrase IX (CAIX), are the main regulators of the adaptive response to low oxygen availability. The prognostic value of these markers was evaluated in 36 COMMs using immunohistochemistry. In addition, the effects of cobalt chloride–mediated hypoxia were evaluated in 1 primary COMM cell line. HIF-1α expression was observed in the nucleus, and this localization correlated with the presence or enhanced expression of HIF-1α-regulated genes at the protein level. Multivariate analysis revealed that in dogs given chondroitin sulfate proteoglycan-4 (CSPG4) DNA vaccine, COMMs expressing HIF-1α, VEGF-A, and CXCR4 were associated with shorter disease-free intervals (DFI) compared with tumors that were negative for these markers (P = .03), suggesting hypoxia can influence immunotherapy response. Western blotting showed that, under chemically induced hypoxia, COMM cells accumulate HIF-1α and smaller amounts of CAIX. HIF-1α induction and stabilization triggered by hypoxia was corroborated by immunofluorescence, showing its nuclear translocation. These findings reinforce the role of an hypoxic microenvironment in tumor progression and patient outcome in COMM, as previously established in several human and canine cancers. In addition, hypoxic markers may represent promising prognostic markers, highlighting opportunities for their use in therapeutic strategies for COMMs.
Canine oral malignant melanoma (COMM) is the most frequent malignant oral tumor in dogs, accounting for 30% to 40% of all oral malignancies. 4 COMM is a solid tumor arising from neoplastic transformation of mucosal melanocytes and is characterized by aggressive behavior, extensive local invasiveness, and metastatic potential, resulting in poor prognosis.3,54
Hypoxia represents one of the most crucial microenvironmental features in solid tumors 49 and has been associated with invasiveness, angiogenesis, vasculogenic mimicry, and response to therapy in several cancer types, including melanoma.10,19 Hypoxia and subsequent acidification of the tumoral microenvironment are reported to promote cancer progression and drug resistance, 38 and contribute to immunotolerance of cancer cells, conferring resistance to immunotherapy.1,11,15,24,47 Hypoxia-inducible factor-1α (HIF-1α) is the primary regulator of the adaptive response to low oxygen availability.20,22,27 Upon stabilization and migration to the nucleus, HIF-1α acts as transcription factor for several hypoxia-regulating elements. Hypoxia-regulating elements, in turn, induce and modulate various processes, including glycolysis, angiogenesis, cell migration, invasion, metastasis, and chemoresistance.11,43,48,49,51 Vascular endothelial growth factor A (VEGF-A) is a key effector of the hypoxic response, which stimulates angiogenesis that provides nutrients and oxygen to proliferating cancer cells. 35 Moreover, hypoxia stimulates cell homing and migration through chemokine-mediated stimuli. C-X-C chemokine receptor type 4 (CXCR4) is overexpressed in various human cancers, including melanoma, and primarily contributes to tumor growth, angiogenesis, metastasis, and therapeutic resistance.8,31
Cancer cells undergo metabolic reprogramming, exhibiting a highly glycolytic phenotype, which is associated with elevated expression of glucose transporter isoform 1 (GLUT1). 12 The metabolic shift increases lactate production. Lactate is then exported into the extracellular space, leading to microenvironment acidification and carbonic anhydrase IX (CAIX) induction.
Hypoxic biomarkers have been extensively investigated in human cancers and have been associated with prognosis and therapy resistance.18,28 Despite the increasing knowledge on the role of hypoxia in tumors, to date only a few studies are available in dogs. 30 Hypoxia has been reported as a frequent condition occurring in COMMs in which HIF-1α activation induces the transcription of GLUT1 and CAIX. 30 A recent study showed that the hypoxia-regulated miRNAs, miR-210 and miR-301, are differentially expressed in primary and metastatic canine melanoma cell lines, and metastatic cells are more resistant to hypoxia stimuli than primary tumor cells.17,50 In addition, it has been demonstrated, by the expression of HIF-1α, that COMM tumor cells invading the bone are under hypoxic conditions, and this may explain a poorer efficacy of radiotherapy in dogs with bone lysis. 33
Based on these premises, and considering the aggressiveness and therapy resistance of COMM, 37 the aim of this study was to unveil the prognostic value of HIF-1α and the associated hypoxia-response proteins, GLUT1, CXCR4, CAIX, and VEGF-A, in COMM. In addition, the study aimed to examine the activation of HIF-1α and its biological alterations after inducing an hypoxic state in a metastatic COMM cell line.
Material and Methods
Sample Collection and Clinical Data
A retrospective study was performed on 36 COMMs diagnosed at the Department of Veterinary Sciences of the University of Turin in the period between 2005 and 2018. The cases were retrieved through a database search using the following keywords: “canine,” “oral cavity,” and “malignant melanomas.” Preoperative clinical tumor staging4,55 was assessed by means of total body computed tomography in 24 cases, or skull and 3 views chest radiographs and abdominal ultrasound in 12 cases. The primary tumor was removed by en bloc excision (mandibulectomy, maxillectomy, or lip-cheek excision followed by reconstruction), with regional (ipsilateral or bilateral) lymphadenectomy.
Dogs were clinically staged based on tumor size, regional lymph node involvement, and metastasis (TNM) according to Bergman.3,4 Sixteen out of the 36 COMMs with a chondroitin sulfate proteoglycan-4 (CSPG4) immunohistochemical score ≥3/8 received CSPG4 DNA electro-vaccination according to the Good Clinical Practice guidelines for animal clinical studies.29,40,45,58 The remaining 20 cases were treated with surgery alone. Both the Ethics Committee of the University of Turin and the Italian Ministry of Health approved the trial (0004230-20/02/2018-DGSAF-MDS-P).
Follow-up for the nonvaccinated dogs consisted of clinical evaluation and thoracic radiographs performed every 3 months during the first year and then every 6 months for a minimum of 2 years. Dogs receiving CSPG4 DNA electro-vaccination were clinically evaluated monthly with thoracic computed tomography.
Histological and Immunohistochemical Analysis
Formalin fixed and paraffin-embedded histologic sections of the tumors were stained with hematoxylin and eosin. Diagnoses were performed independently by 3 observers (LM, SI, and CG). In case of disagreements, a consensus was reached using a multiheaded microscope.
Specimens were classified according to the World Health Organization (WHO) guidelines 54 and evaluated for prognostic parameters in canine melanocytic neoplasia according to the current literature. Briefly, nuclear atypia was assessed according to what has been described by Spangler and Kass. 57 A threshold value of 30% atypical nuclei was considered. Mitotic count was obtained by counting the number of mitoses in 2.37 mm 2 , considering the regions of highest mitotic activity and avoiding areas of ulceration, necrosis, and inflammation. The mitotic count cutoff value was ≥4 according to Smedley et al. 54 Pigmentation was assessed subjectively as described by Smedley et al, 54 evaluating all available sections and dividing cases into those with ≥50% and those with <50% pigmented cells. Ki67 was determined as the average number of positive neoplastic cell nuclei per 1 cm2 optical grid reticle at 400x magnification/40x objective (5 grid areas counted) in the highest labeling area with a cutoff value of 19.5.
Immunohistochemistry was performed on 4-µm thick paraffin sections. After blocking peroxidase activity (0.3% hydrogen peroxide in deionized water for 30 minutes) and heat-induced antigen retrieval (30 minutes with citrate buffer at 98°C, pH 6.0), sections were incubated with primary antibodies Ki-67, HIF-1α, CAIX, CXCR4, GLUT1, and VEGF-A overnight at 4°C (Supplemental Table S1); the detection was performed using the Vectastain Universal Quick Kit (Vector Laboratories, Inc.). Immunolabelled slides were randomized and masked for blinded examination, which was performed by 3 independent pathologists (SI, ML, and LA). In case of disagreements, a consensus was reached using a multiheaded microscope.
Antibodies specificity for HIF-1α, CXCR4, VEGFA, GLUT1, and CAIX was assessed on positive and negative tissues (canine mammary carcinomas, renal carcinoma, skin, and testis), as shown in Supplemental Fig. S1.
Immunohistochemical evaluation was performed by a semiquantitative method, evaluating both the intensity and the percentage of positive cells using scoring systems previously published (Supplemental Table S2).25,29,30,39,44,55
Cell Line and CoCl2 Treatment
A canine malignant melanoma cell line (OLGA), previously established by the authors, 46 was cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum, 1% glutamine, 100 µg/mL penicillin, and 100 µg/mL streptomycin at 37°C and 5% CO2. The OLGA cell line was cultured in chemically induced hypoxia for 12, 24, and 48 hours using a concentration of 200 µM cobalt chloride (CoCl2; Sigma Aldrich, 15862-1ML-F). 42
Western Blot Analysis
Proteins from the OLGA cell line, treated with 200 µM of CoCl2 for 12, 24, and 48 hours or untreated (dimethyl sulfoxide [DMSO] alone), were extracted in lysis buffer (1% Triton X-100, 10% glycerol, 50 mM Tris, 150 mM sodium chloride, 2 mM EDTA, pH 8.0, and 2 mM magnesium chloride) containing Protease Inhibitor Cocktail (P8340 Sigma). Twenty micrograms of total proteins were separated on a 10% SDS-PAGE gel and transferred onto a 0.2 µm pore-size nitrocellulose membrane (Thermo Fisher Scientific Inc.). After washing, membranes were incubated in Tris-buffered saline with 10% bovine serum albumin at room temperature for 1 hour and then incubated overnight at 4°C with HIF1-α and CAIX antibodies; α-tubulin was used as a housekeeping protein (Supplemental Table S1). Horseradish peroxidase-conjugated secondary antibodies were diluted at 1:15 000. The chemiluminescence substrate (Clarity ECL Substrate; Bio-Rad Laboratories) was used to produce the light signal, acquired with CL-XPosure films (Thermo Fisher Scientific Inc).
Immunofluorescence
To investigate the effects of CoCl2 on HIF1-α nuclear translocation and CAIX expression, immunofluorescence against HIF1-α and CAIX was performed on OLGA cells exposed to the highest CoCl2 concentration (200 µM) for 12, 24, and 48 hours. Briefly, 2 × 104 cells were plated in 8-well chamber slides (Lab-Tek II Chamber Slide System; Nalge Nunc International Corporation) until 70% confluence. After treatment, cells were fixed with methanol:acetone (1:1 proportion) for 30 seconds. After washing 3 times with Tris-HCl (0.1 M, pH 7.6), cells were blocked with 10% normal goat serum for 1 hour at room temperature and then incubated overnight at 4°C with antibodies to HIF-1α (1:100) and CAIX (1:200). After washing with Tris-HCl, cells were incubated with a fluorescent secondary Alexa488-conjugated goat anti-rabbit IgG antibody (1:500 dilution, ThermoFisher) and anti-mouse IgG (1:500) for 1 hour in the dark. Subsequently, cell nuclei were stained with DAPI (0.5 µg/ml in Tris-HCl, Sigma-Aldrich, USA) for 10 minutes, washed 3 times with Tris-HCl, and then the sections were covered with mounting medium (PermaFluor, Thermo Scientific) and kept overnight in the dark. Fields were randomly selected by microscope TCS SP8 (Leica Microsystems CMS GmbH Mannheim, Germany) and z-stacks of 10 nm were acquired. Z-stacks were then processed directly with the Leica LAS-X software to produce maximum projection images of each field. To detect colocalization of different fluorochromes, fluorescent signals in different channels were merged to produce multicolor images. Image acquisitions were performed with a resolution of 1024 × 1024 pixels with a 200 Hz sampling frequency.
Statistical Analyses
Fisher exact test or the χ2 test was used to test possible associations among immunohistochemical marker expression and clinical and histopathological data. Disease-free interval (DFI) was calculated as the time between surgery and the first detection of metastases and/or local recurrence, while overall survival (OS) was defined as the period from the day of surgery to the patient’s death due to a cancer-related cause. The survival functions of the DFI and OS were estimated with the Kaplan-Meier method, and the comparison of survival function was done by means of the log-rank test. Dogs that died from unrelated causes, were lost to follow-up, or were still alive at the end of the study were right-censored. Univariate and multivariate Cox models were fitted to verify the effects of hypoxia biomarkers on DFI or OS. In multivariate analysis, we considered the following parameters: HIF-1α, CXCR4, VEGFA, GLUT1, and CAIX immunohistochemical expression. Statistical significance was set at a .05 level. Because the clinical samples belonging to stage I were censored, this stage was omitted. GraphPad Prism 8 (GraphPad Software, San Diego, California) and R statistical software (R Core Team, 2018) were used for statistical analysis.
Results
Patient and Tumor Characteristics
A total of 36 dogs with COMM met the inclusion criteria. Clinical and follow-up data, and histopathological and immunohistochemical diagnostic features are provided in Tables 1 and 2, respectively. Survival analysis (Fig. 1) showed that dogs with clinical stage IV had a worse prognosis compared with dogs with clinical stages II and III (P = .002). Similarly, vaccinated dogs had a significantly higher OS time compared with nonvaccinated dogs (P < .001; Fig. 2). Although not significant, we found that COMMs with a total absence of melanin or less than 25% pigmented neoplastic cells were associated with a lower DFI when compared with tumors with >50% pigmented neoplastic cells (median DFI: days 180 vs 210 days, respectively, P = .06). The OS was not significantly associated with the amount of pigmentation (median OS: 235 days vs 778 days, P = .08), and none of the other histopathological parameters showed prognostic significance.
Clinical features of dogs included in the study.
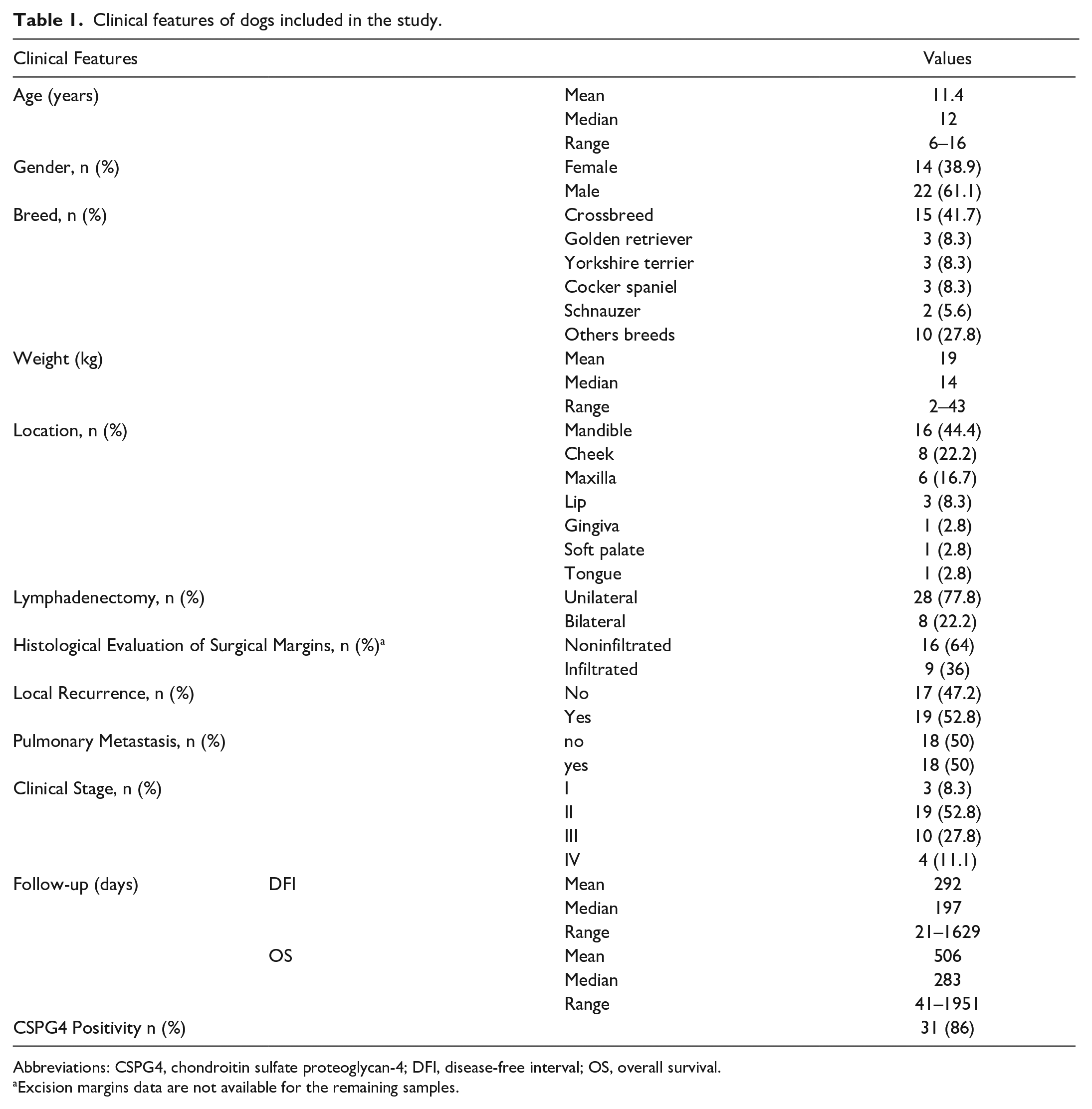
Abbreviations: CSPG4, chondroitin sulfate proteoglycan-4; DFI, disease-free interval; OS, overall survival.
Excision margins data are not available for the remaining samples.
Histopathological and immunohistochemical diagnostic features of canine oral malignant melanomas.
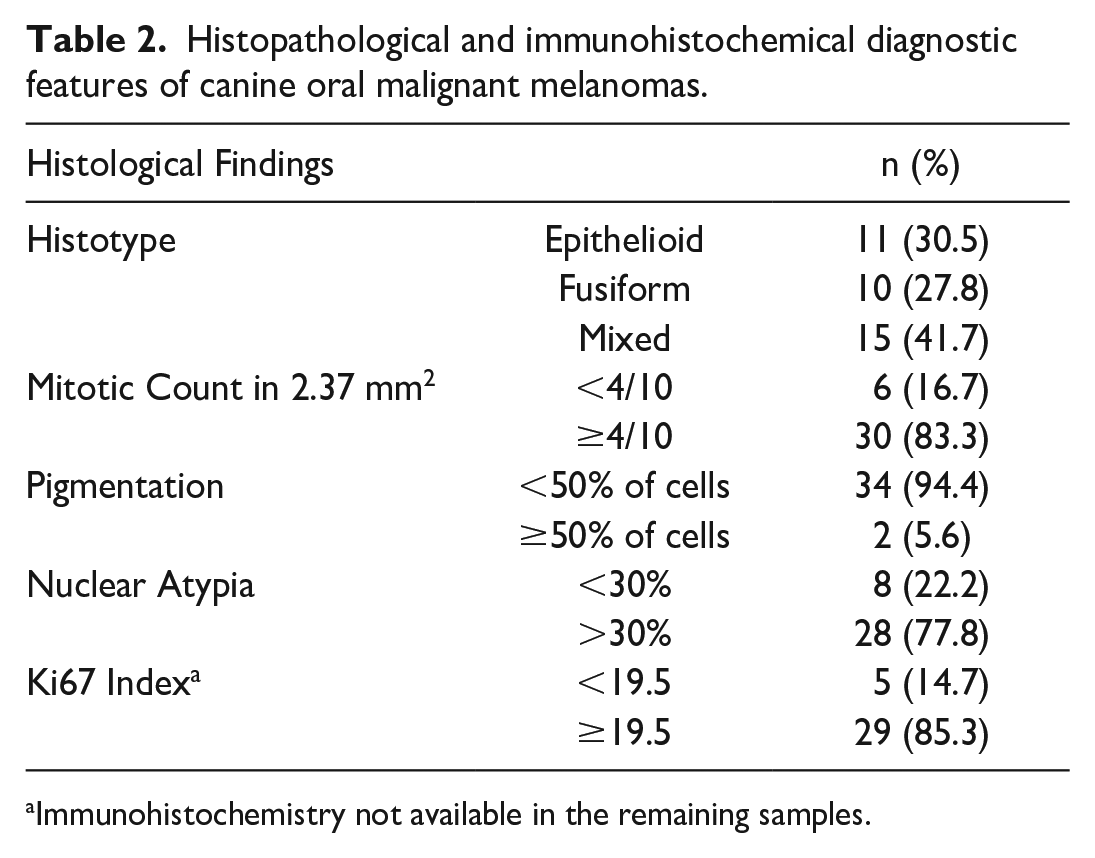
Immunohistochemistry not available in the remaining samples.
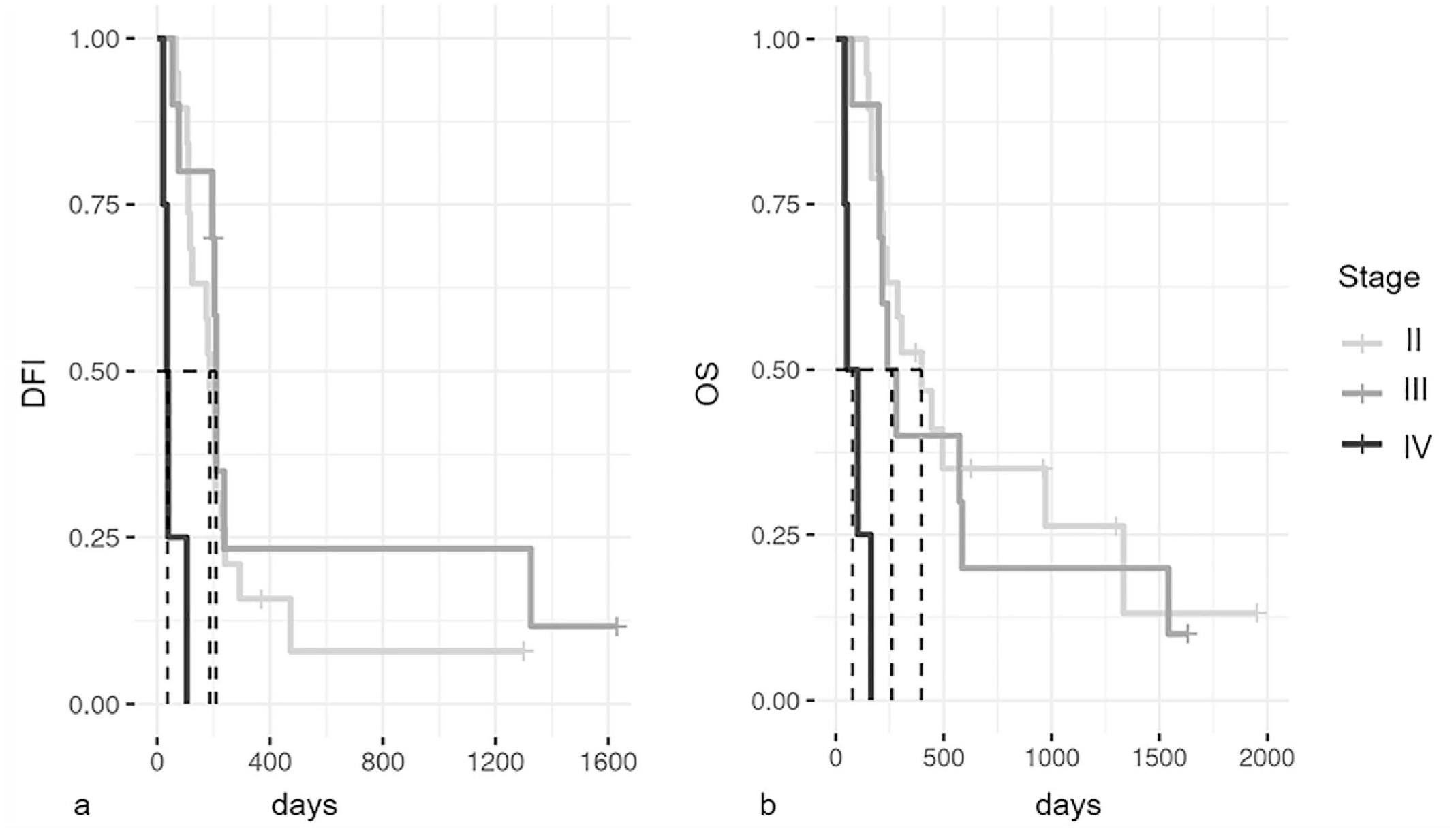
Kaplan-Meier survival curves for stages II, III, and IV canine oral malignant melanomas.
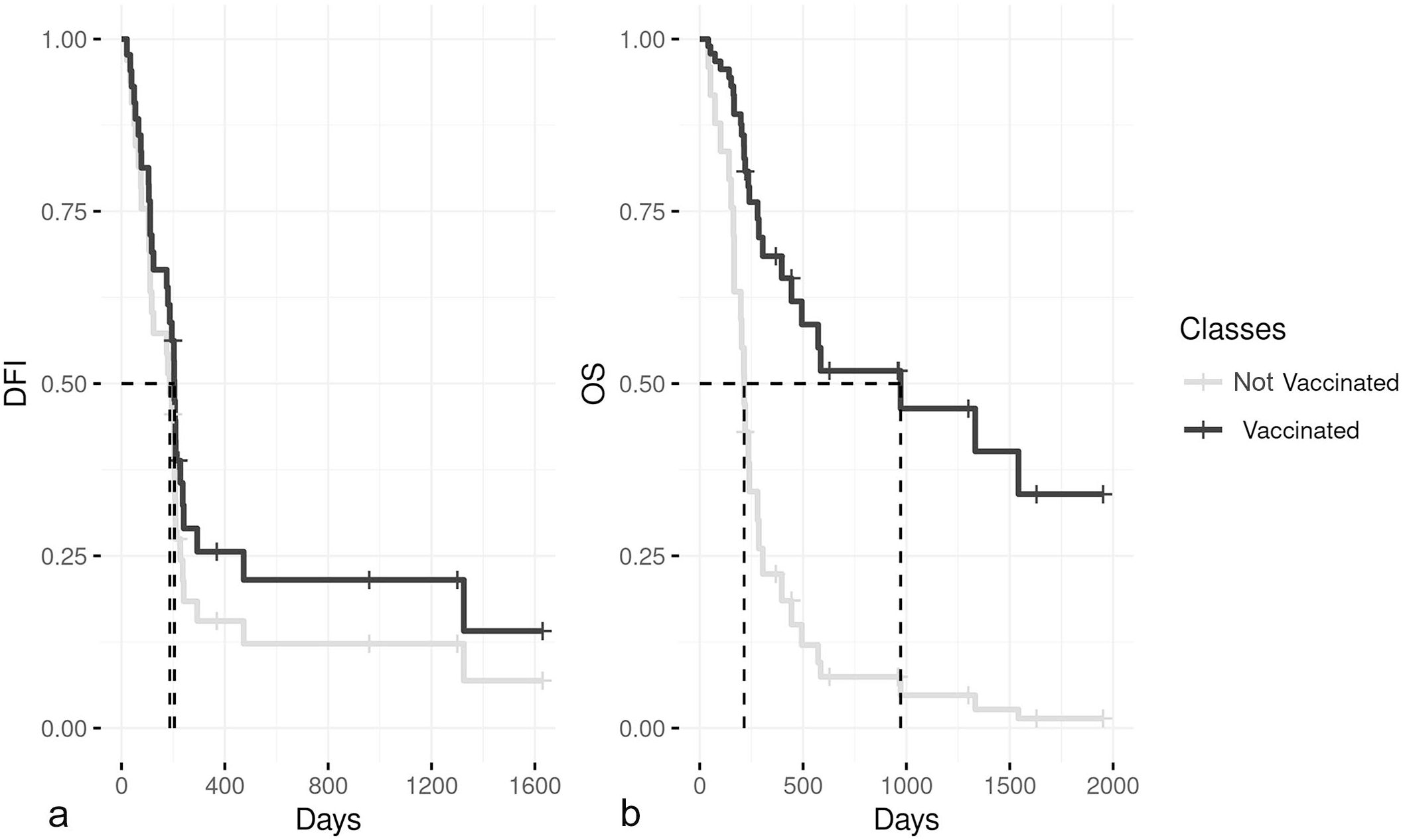
Kaplan-Meier survival curves of dogs with canine oral malignant melanomas treated with or without chondroitin sulfate proteoglycan-4 (CSPG4) DNA vaccination.
Overexpression of Hypoxic Markers Negatively Affects Prognosis in Dogs Receiving Immunotherapy
The immunohistochemical scores for HIF-1α, CAIX, GLUT1, CXCR4, and VEGF-A are summarized in Table 3. Representative images are shown in Fig. 3. The CAIX score was associated with the HIF-1α score (P = .001). In addition, we found a statistical association between CXCR4 and Ki-67 index (P = .046; Table 4). Multivariate analysis revealed that dogs receiving immunotherapy and overexpressing HIF-1α, VEGF-A, and CXCR4 had a significantly shorter DFI (median = 111 days) compared with dogs with the same treatment that were negative for all 3 markers or only positive for 1 or 2 markers (median = 204 days; P = .03; Fig. 4), whereas no significant differences were observed for OS.
Immunohistochemical (IHC) scoring of hypoxia-related markers.
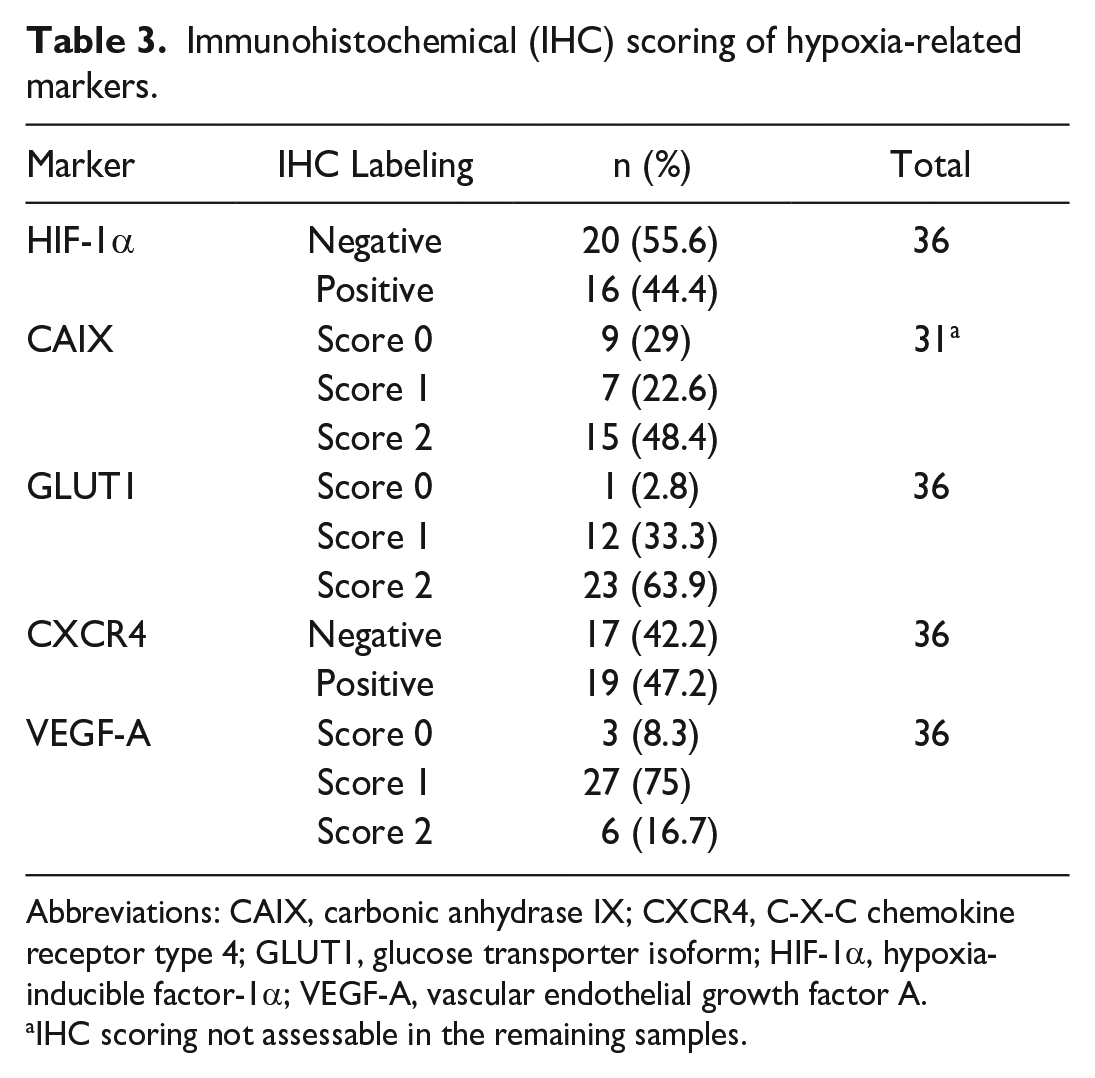
Abbreviations: CAIX, carbonic anhydrase IX; CXCR4, C-X-C chemokine receptor type 4; GLUT1, glucose transporter isoform; HIF-1α, hypoxia-inducible factor-1α; VEGF-A, vascular endothelial growth factor A.
IHC scoring not assessable in the remaining samples.
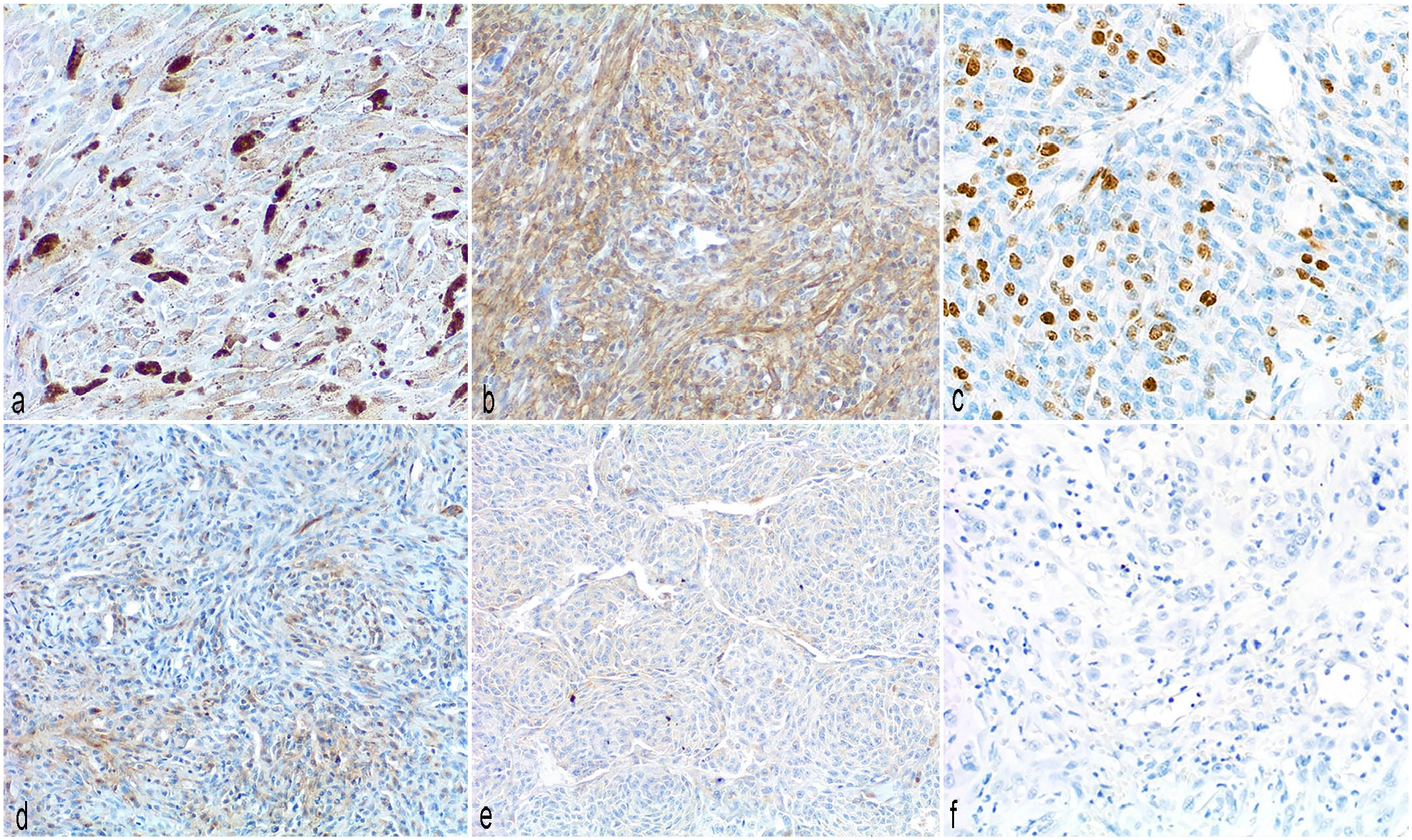
Immunohistochemistry (IHC) for hypoxia-associated factors in canine oral malignant melanomas, dog.
Contingency tables of significantly associated hypoxia immunohistochemical markers.
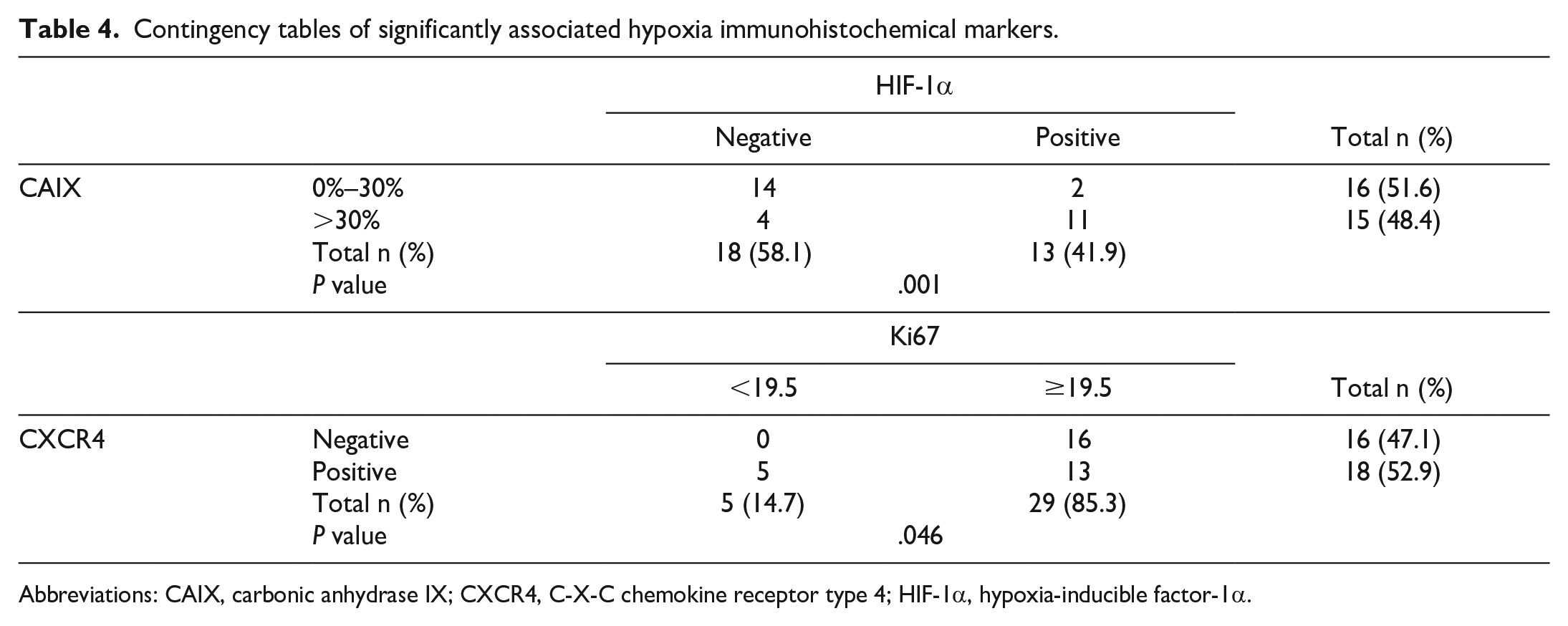
Abbreviations: CAIX, carbonic anhydrase IX; CXCR4, C-X-C chemokine receptor type 4; HIF-1α, hypoxia-inducible factor-1α.
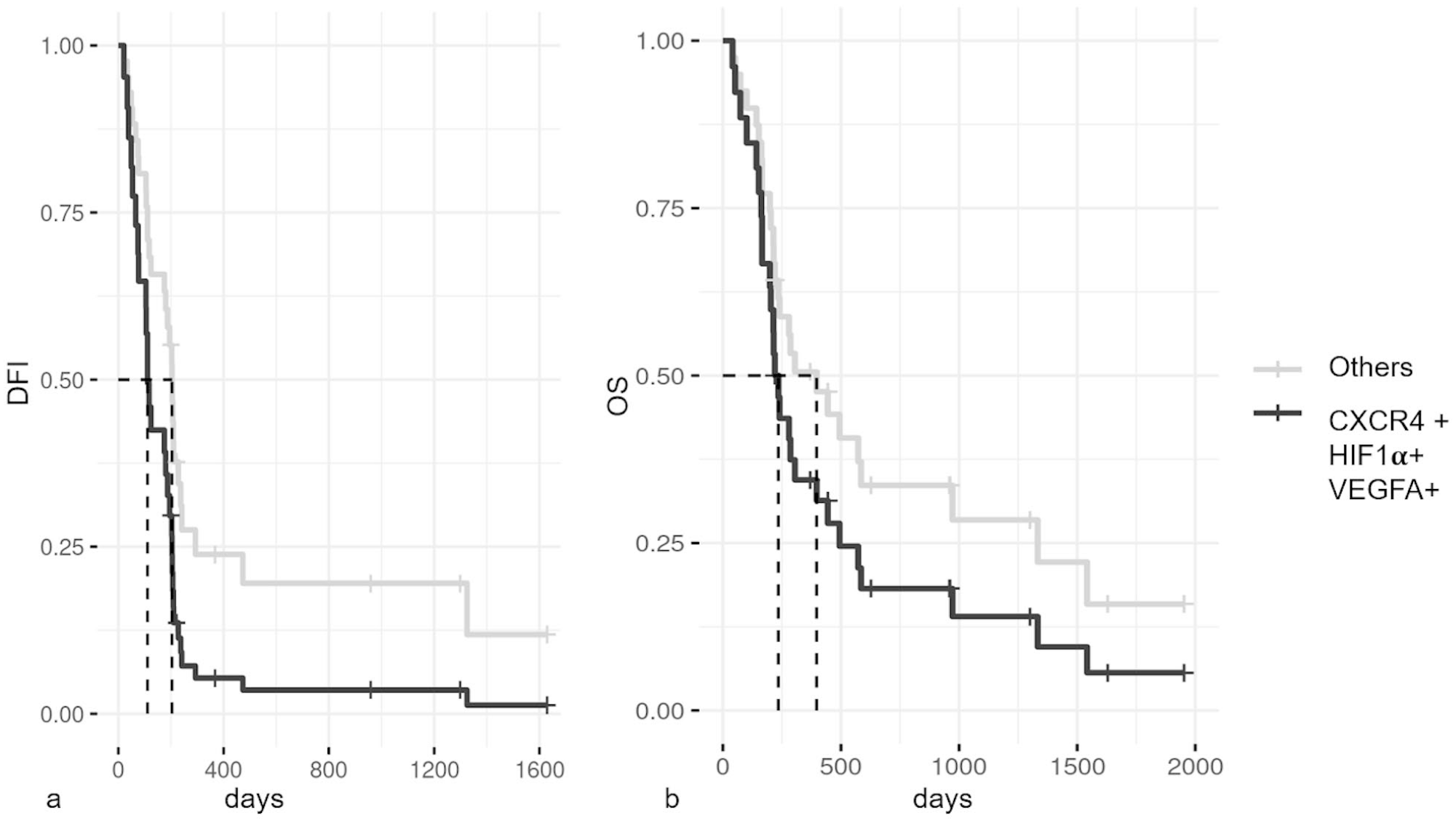
Kaplan-Meier survival curves of dogs with canine oral malignant melanomas treated with chondroitin sulfate proteoglycan-4 DNA vaccination that either co-express hypoxia-inducible factor-1α (HIF-1α), C-X-C chemokine receptor type 4 (CXCR4), and vascular endothelial growth factor A (VEGFA) or lack the expression of at least one of the proteins (others).
Multivariate analysis in nonvaccinated patients revealed no statistically significant differences in OS or DFI between dogs with concomitant overexpression of HIF-1α, VEGF-A, and CXCR4 and dogs negative for the markers or expressing only 1 or 2 markers.
CoCl2 Treatment Induces HIF-1α Accumulation and Nuclear Translocation
Distinct bands corresponding to HIF-1α (120 kDa) and CAIX (58 kDa) proteins were more pronounced in the Olga cell line treated with CoCl2 at 12 and 24 hours (Fig. 5). In contrast, the expression of these proteins was either negative (HIF-1α) or very low (CAIX) in untreated cells (Fig. 5).
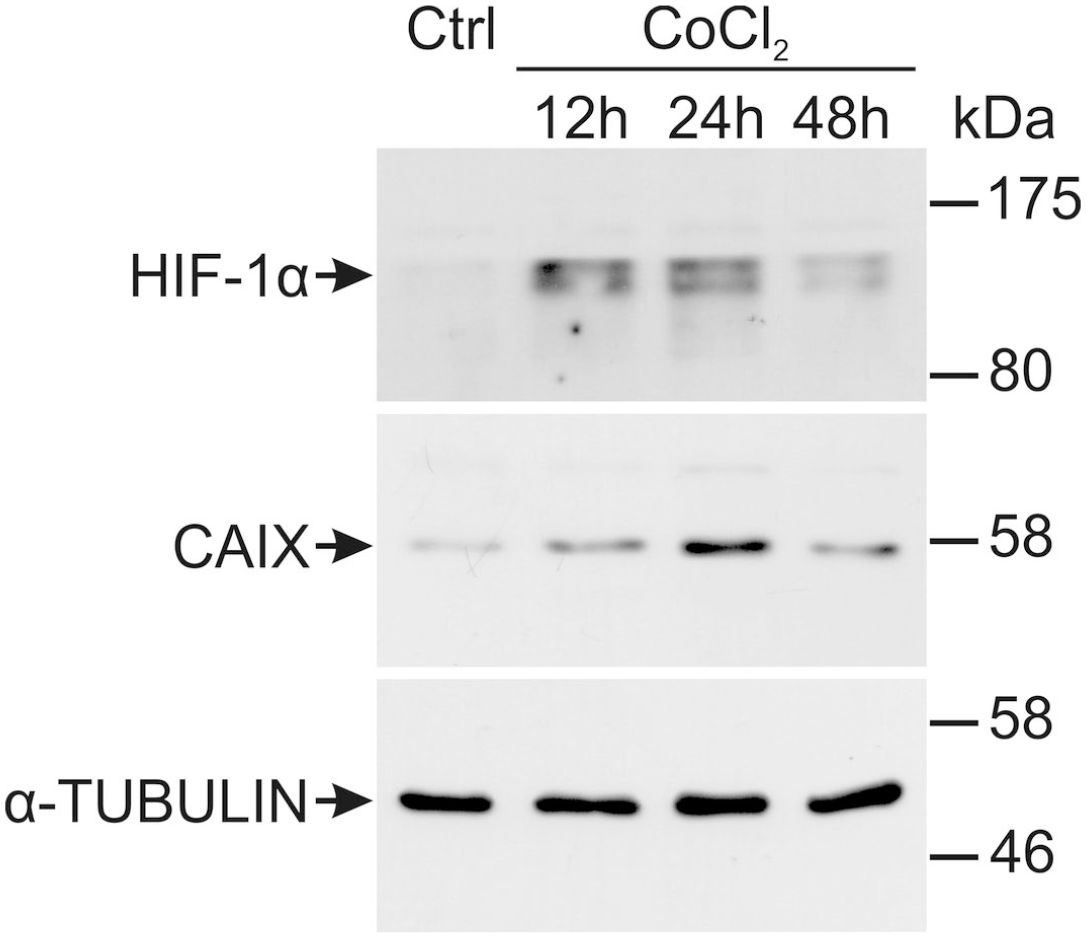
Western blot analysis of Olga cells that were untreated (Ctrl) or cells that were treated for 12, 24, or 48 hours with cobalt chloride (CoCl2) to induce hypoxic conditions. Abbreviations: CAIX, carbonic anhydrase IX; HIF-1α, hypoxia-inducible factor-1α.
A mild increase of CAIX protein was also present in chemically induced hypoxic conditions after 24 hours. Under normoxic conditions, HIF-1α was primarily localized to the cytoplasm, with nuclear localization becoming evident only after CoCl2 treatment (Fig. 6). In contrast, chemically induced hypoxia did not yield noticeable effects on CAIX protein.
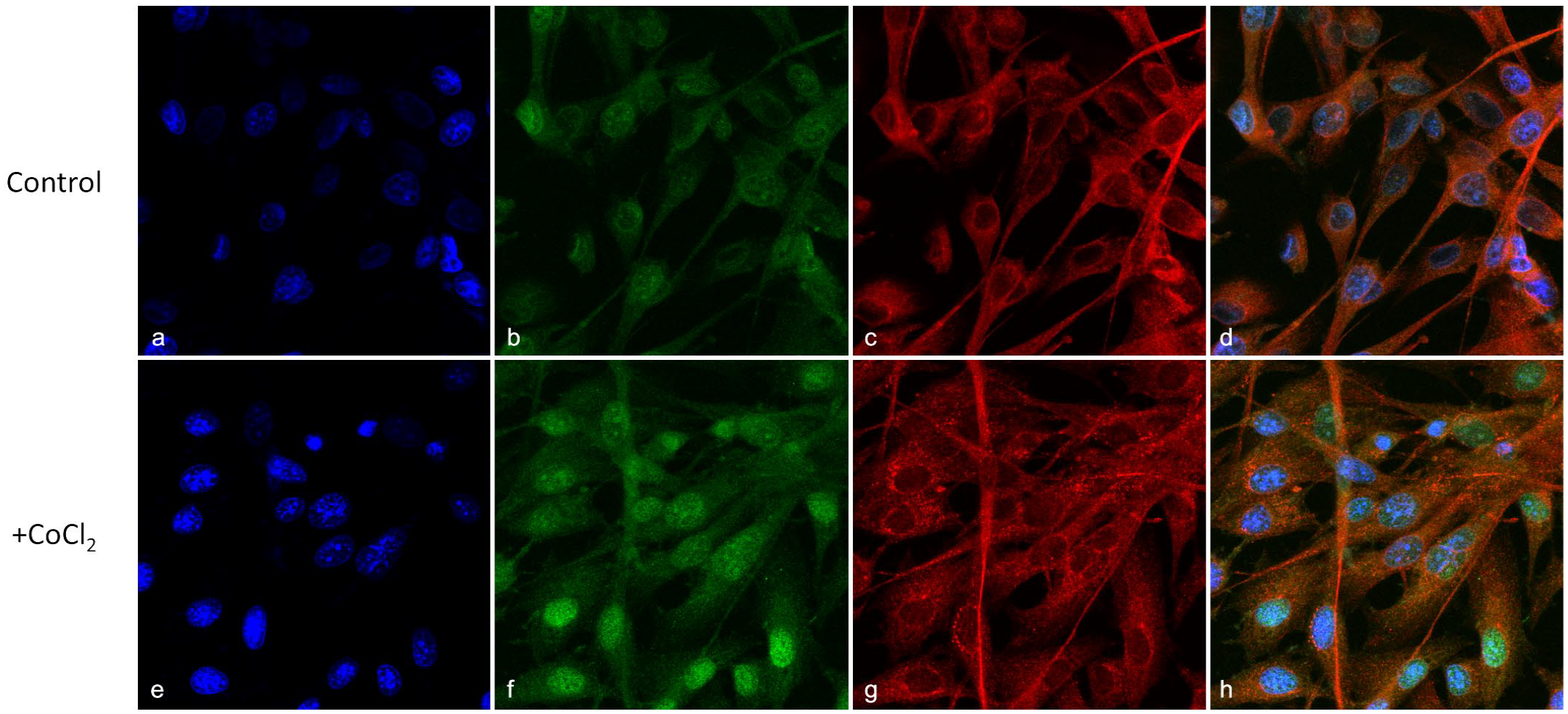
Representative immunofluorescence of primary canine oral malignant melanoma OLGA cell line either (
Discussion
COMM is the most commonly diagnosed malignant tumor occurring in the oral cavity of dogs16,55 and it has a high and rapid metastatic rate, resulting in a poor prognosis. 3 Recently, a consensus working group was founded to identify potential prognostic markers in this neoplasm and to give accurate recommendations for the diagnosis and histopathologic prognostication of canine melanocytic tumors. 53 Although nuclear atypia, mitotic count, and Ki-67 index are considered the most prognostic factors for COMM, the identification of new markers may improve the ability to prognosticate these neoplasms, as well as aid in the selection of specific therapies.
In this study, we observed that patients with advanced clinical stages (III and IV) had a shorter OS compared with stage II patients, which is consistent with existing literature. 3 Furthermore, our data confirmed that CSPG4 vaccination significantly increased the overall survival of COMM patients.40,46
Interestingly, smaller amounts of melanin in COMMs seem to be weakly associated with shorter DFI, suggesting a close association between loss of melanin pigment and tumor dedifferentiation, which may be associated with a more aggressive behavior. This finding does not represent a novelty because, in 2011, Smedley et al suggested that a lower level of pigmentation (<50% of pigmented cells) is an unfavorable factor. 55 In human cutaneous melanoma, decreased pigmentation has also been linked to an aggressive phenotype with implications for prognosis and response to therapy.6,52,53
As previously reported in a number of canine tumor histotypes, a correlation between CAIX and HIF-1α positivity has been observed, suggesting hypoxia may trigger microenvironment acidosis with metabolic changes in cancer cells growth. 36 Our findings reinforce the close interaction between these molecules and highlight the strong HIF-1α-dependent regulation of CAIX as an adaptation of COMM cells to extracellular acidosis. 60 This adaptation may enhance the tumor’s ability to survive and grow under adverse conditions.
We found that CXCR4 expression was associated with an increased Ki-67 index, which is consistent with previous reports in human renal carcinomas 9 and multicentric lymphoma. 62 However, in human cutaneous melanomas, these markers have not been significantly associated.31,32,59
Univariate data analysis for the hypoxic markers did not show any statistical significance for DFI or OS. However, these markers are interconnected in the hypoxic cellular pathway signaling. Hence, we investigated the prognostic value of their coexpression. Multivariate data analysis revealed that concurrent expression of HIF-1α, VEGF-A, and CXCR4 in COMMs treated with a CSPG4 vaccine is associated with a lower DFI compared with COMMs negative for these markers. This suggests a lower efficacy of CSPG4 vaccination in tumors displaying hypoxic features. This finding is in line with the literature in which hypoxia is known to induce immune-resistance and negatively interferes with immune surveillance of tumors and adoptive immunotherapy.2,24,61 Similarly, coexpression of hypoxic markers and hypoxia-related signatures have been documented in several human cancers as predictive of a poor outcome.7,26,63 This coexpression triggers the activation of HIF-1α-dependent pathways, which can lead to tumor growth, angiogenesis, and metastasis. 8 Regarding the relationship between hypoxic microenvironments and CSPG4 expression, it is very interesting to underscore that, in human melanomas, CSPG4 is regulated by hypoxia in vitro and its expression confers resistance to immunotherapy.23,41 On the basis of the preliminary data obtained by the authors, we can hypothesize that, in dogs, CSPG4 is regulated by hypoxia, strengthening COMM as a good model for comparative oncology. 34
To gain insight into the mechanisms of hypoxia in COMM, the OLGA cell line was treated with CoCl2 to mimic a hypoxic stimulus. 13 We found that in vitro hypoxia induction caused an accumulation of HIF-1α protein after treatment. HIF-1α induction and stabilization triggered by hypoxia was further corroborated by immunofluorescence in which nuclear translocation under CoCl2 treatment to avoid proteasomal degradation was demonstrated. 49 In the nucleus, HIF-1α is known to directly coordinate the transcription of hypoxia-regulating elements. 48 Consistent with the immunohistochemical results, CAIX protein levels were also mildly increased under chemically induced hypoxia. Its accumulation was delayed as compared with HIF-1α, thus supporting the direct role of HIF-1α in the upregulation of this protein. 21
In conclusion, our results reinforce the crucial role of a hypoxic microenvironment and acidification in tumor aggressiveness and outcome in COMM, as extensively established in other canine14,56 and human cancers. 5 It becomes critical to develop novel therapeutic strategies, considering the hypoxic status of the tumors, and future studies should address the in vitro effects of hypoxia in COMM cells and investigate the inhibition of hypoxia-related signaling pathways as potential therapeutic targets. Notably, as shown in this study, it is crucial to consider the interdependent actions of the molecular mechanisms triggered by HIF-1α and its transcriptional cascade.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241244853 – Supplemental material for Hypoxia-associated markers in the prognosis of oral canine melanoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858241244853 for Hypoxia-associated markers in the prognosis of oral canine melanoma by Cecilia Gola, Lorella Maniscalco, Selina Iussich, Emanuela Morello, Matteo Olimpo, Eugenio Martignani, Paolo Accornero, Davide Giacobino, Eugenio Mazzone, Paola Modesto, Katia Varello, Luca Aresu and Raffaella De Maria in Veterinary Pathology
Footnotes
Author Contributions
RDM and CG designed and performed the experimental design. SI and LM performed histological diagnosis; LM, PM, and KV performed immunohistochemistry (IHC) evaluations; and EM, DG, and MO enrolled and surgically treated canine oral malignant melanomas (COMMs). EMi performed statistical analysis. PA and EMe performed in vitro assay, Western blot, and immunofluorescence. The manuscript was written by RDM, CG, and LM, with contribution from LA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
