Abstract
Solid tumors are composed of a heterogeneous population of cells surviving in various concentrations of oxygen. In a hypoxic environment, tumor cells generally up-regulate glycolysis and, therefore, generate more lactate that must be expelled from the cell through proton transporters to prevent intracellular acidosis. Monocarboxylate transporter 1 (MCT1) is a major proton transporter in mammalian cells that transports monocarboxylates, such as lactate and pyruvate, together with a proton across the plasma membrane. Melanocytic neoplasia occurs frequently in dogs, but the prognosis is highly site-dependent. In this study, 50 oral canine melanomas, which were subdivided into 3 histologic subtypes, and 17 ocular canine melanocytic neoplasms (14 melanocytomas and 3 melanomas) were used to examine and compare MCT1 expression. Immunohistochemistry using a polyclonal chicken anti-rat MCT1 antibody showed that most oral melanoma exhibited cell membrane staining, although there were no significant differences observed among the 3 histologic subtypes. In contrast, the majority of ocular melanocytic tumors were not immunoreactive. Additionally, we documented the presence of a 45-kDa band in cell membrane protein Western blots, and sequencing of a reverse transcriptase polymerase chain reaction band of expected size confirmed its identity as a partial canine MCT1 transcript in 3 oral tumors. Increased MCT1 expression in oral melanomas compared with ocular melanocytic tumors may reflect the very different biology between these tumors in dogs. These results are the first to document canine MCT1 expression in canine tumors and suggest that increased MCT1 expression may provide a potential therapeutic target for oral melanoma.
Melanoma is relatively common in dogs, accounting for 30–40% of all oral neoplasms and up to 7% of all malignant tumors. Oral melanocytic tumors are considered highly malignant, while ocular melanocytic tumors involving the anterior uveal tract are generally considered benign. Seventy to eighty percent of oral melanomas metastasize to regional lymph nodes and other organs; 29 therefore, the mean postoperative survival time is 3 months, with only approximately 25% of patients surviving 1 year or more. 12, 39 Histologic characteristics, including mitotic index and degree of pigmentation, appear to have no prognostic value. 2, 23, 27 In contrast to oral melanocytic tumors, the prognosis for patients with ocular melanocytic tumors is excellent after surgery, even with tumors that are histologically malignant, with systemic metastases being rarely reported. 6, 38
Tumor cells growing in a hypoxic microenvironment up-regulate glycolysis, which subsequently produces a large amount of intracellular lactic acid that must be transported out of the cell to prevent intracellular acidosis. 33 Intracellular acidosis has been shown to be a trigger in the early phase of apoptosis and to lead to the activation of endonuclease inducing DNA fragmentation. 16, 41 To avoid intracellular acidification, pH regulators are thought to be up-regulated in tumor cells. 14 Monocarboxylate transporters (MCTs) are one of the 4 major types of pH regulators, along with Na+/H+ exchangers, vacuolar adenosine triphosphatase, and anion exchangers, which are involved in homeostasis in mammalian cells. 14 MCT1 is thought to be most important for pH regulation within rapidly metabolizing, highly glycolytic tumor cells. 22 Fourteen different isoforms have been detected in mammalian tissues, but only isoforms 1–4 have been demonstrated to transport monocarboxylates, such as lactate and pyruvate. 9, 10 Although MCT1 has been examined in a variety of cells of various animals, 10 there have been no studies examining MCT1 expression in canine tissues.
Human neoplasms that express MCT1 include glial neoplasm, 7, 22 ependymomas, 7 hemangioblastomas, 7 and alveolar soft-part sarcomas. 18 Various in vitro studies that target human melanoma MCT1 have been reported, 5, 34 although no clinical trials are currently ongoing. The aim of the present study was to determine whether the expression of MCT1 differed between benign and malignant canine melanocytic tumors and to determine if differences in immunoreactivity were seen between the 3 subtypes of oral melanomas.
Materials and Methods
Samples
Fifty oral canine melanocytic tumors and 17 ocular canine melanocytic tumors were examined. Samples were obtained from surgical biopsies between 1997 and 2005 at the Veterinary Teaching Hospital, Rakuno Gakuen University, and from general veterinary practitioners in Japan. Samples were fixed either in 4% paraformaldehyde for 24 hours or 10% formalin for 24–48 hours. After fixation, the tissues were dehydrated through a series of graded paraffin and sectioned serially at 4 μm. For Western blotting and reverse transcriptase polymerase chain reaction (RT-PCR), 3 oral melanomas were immediately frozen in liquid N2 and stored at −80°C. Canine colonic tissues confirmed to contain MCT1 (unpublished data in our laboratory) were used as a positive control for Western blots, RT-PCR, and the immunohistochemical studies. Nonneoplastic melanocytes within sections of normal oral mucosa and in the eye were used as normal comparators for MCT1 expression in immunohistochemical studies. We did not use negative tissue controls lacking MCT1 expression for Western blots and RT-PCR, considering the widespread constitutive tissue expression of MCT1. 9
Histology and morphologic diagnosis
Each oral and ocular tumor was classified as melanocytoma (benign) or melanoma (malignant) according to the World Health Organization classification of tumors of the alimentary system and the ocular and otic tumors of domestic animals, respectively. 11, 37 Oral melanomas were further subdivided into 3 subtypes, epithelioid, spindle-cell, and mixed types (epithelioid and spindle patterns), according to the criteria of Smith et al. 29 on the basis of the predominant cell type by conventional light microscopy of HE-stained sections. A bleaching procedure using the method proposed by Roels et al. 28 was performed on tissue sections containing dense melanin pigment.
Immunohistochemistry
Immunohistochemical studies were performed using the avidin-biotin-peroxidase complex method (Vectastain Elite ABC Kit, Vector Laboratories Inc., Burlingame, CA, USA). The primary antibody used was a chicken polyclonal anti-rat MCT1 (Chemicon International Inc., Temecula, CA, USA). Dewaxed sections were subjected to antigen retrieval by heating in a microwave oven in the presence of 0.01-M sodium citrate buffer, pH 6.0, for 15 minutes. Sections were incubated in 3% (volume per volume) H2O2 in methanol at room temperature for 10 minutes to quench endogenous peroxidase activity. Subsequently, sections were incubated with the primary antibody diluted 1 : 200 at 4°C for 12 hours, with biotinylated goat anti-chicken immunoglobulin Y (IgY) (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) for 30 minutes at room temperature and with avidin-biotin-peroxidase conjugate for 30 minutes. Finally, the reaction products were “visualized” by treatment with 0.05% 3,3′-diaminobenzidine solution and counterstained with Mayer's hematoxylin. Procedural controls were obtained by omitting the primary antibody. A bleaching procedure 28 was performed on tissue sections containing dense melanin pigment prior to the immunoreaction of the primary antibody.
The expression of MCT1 in melanocytes of normal oral mucosa and the eye was detected by double immunolabeling, using the MCT1 antibody and a mouse monoclonal anti-human Melan A antibody (Novocastra Laboratories Ltd., Newcastle, UK). The sections immunostained with MCT1 antibody and “visualized” by treatment with 0.05% 3,3′-diaminobenzidine solution were treated with 0.005% biotin in phosphate buffered saline (PBS) to mask avidin for 30 minutes at room temperature, followed by incubation with the melan A antibody. Sections were incubated with alkaline phosphatase-conjugated streptavidin for 30 minutes. Reaction products were visualized with alkaline phosphatase substrate Fast blue (SAB-AP Kit, Nichirei Co., Tokyo, Japan), which yields a blue precipitate without counterstain. A bleaching procedure 28 was performed on tissue sections prior to the immunoreaction of the primary antibody.
Grading of MCT1-staining pattern
The immunohistochemistry staining was evaluated on the basis of a distinct immunoreaction on the cell membrane. A mean percent labeling score was obtained for each sample as follows: The estimated percentage of labeled neoplastic cells in each of 10 random 400× fields was obtained for each specimen according to the following scoring system: 0 = 0%, 1 = <10%, 2 = 10–30%, 3 = 31–60%, and 4 = >61% of cells staining positive for MCT1. These 10 field scores were averaged to obtain the mean percent labeling score for each tumor specimen. We conducted an analysis of the differences among the 3 histologic subtypes of oral melanoma using the analysis of variance test and of the difference between oral and ocular melanocytic tumors using the Student's t-test. A value of P < .05 was considered statistically significant.
Polyacrylamide Gel electrophoresis and western blot analysis
Western blot analysis was performed as previously described. 15 Briefly, aliquots (30 μg) of membrane protein were separated on a 10% SDS-polyacrylamide gel and transferred to nitrocellulose membranes (Tokyo Roshi Kaisha Ltd., Tokyo, Japan), which were blocked overnight at 4°C with 5% (weight in volume) nonfat dry milk in PBS-T (0.1% Tween 20 in PBS), then probed for 1 hour with a primary antibody (chicken anti-rat MCT1, Chemicon International Inc., Temecula, CA, USA) and diluted 1 : 500 in PBS. After washes in PBS-T, the membranes were incubated with biotinylated goat anti-chicken IgY diluted 1 : 1,000 in PBS for 30 minutes at room temperature and with avidin-biotin-peroxidase conjugated for 30 minutes. Immunodetection was performed by chemiluminescence (ECL, Amersham International, Buckinghamshire, UK) according to the instructions of the manufacturer. A mouse monoclonal beta-actin antibody (Abcam, Cambridge, UK) was used as an internal standard control. Negative control blots were probed only with the secondary antibody.
RT-PCR
Total RNA was isolated from the oral melanoma tissue using the RNeasy Mini Kit according to the manufacturer's protocol (Qiagen Inc., Valencia, CA, USA). One microgram of total RNA was reverse-transcribed using Superscript II and oligo-d(T)12–18 (Invitrogen, Carlsbad, CA, USA) in a 20-μl reaction volume. PCR was performed on synthesized cDNAs using Taq DNA polymerase (Takara Bio Inc., Otsu, Japan). MCT1-specific PCR primers were derived from a canine MCT1 cDNA sequence (GenBank Accession no. XM533065): sense primer 5′-gtggatgcttgtcaggctgtgg-3′; antisense primer 5′-ccagctacacagcagtttagtagg-3′ (Hokkaido System Science Co. Ltd., Tokyo, Japan). The PCR conditions used were 94°C for 2 minutes, 35 cycles of 94°C for 30 seconds, 55°C for 30 seconds, 72°C for 1 minute, and a final extension at 72°C for 2 minutes using a thermocycler (iCycler, Bio-Rad Laboratories, Hercules, CA, USA). Glyceraldehyde-3-phosphate dehydrogenase cDNA was used as a positive cDNA control. PCR products were separated by agarose gel electrophoresis (1% agarose) and visualized with ultraviolet light following ethidium bromide staining. Negative controls were performed using a reaction mixture with H2O as a substitute for the template DNA. The identity of the amplified MCT1 cDNA fragment was confirmed by sequencing using an automated DNA sequencer (310 Genetic Analyzer, Applied Biosystems Inc., Foster, CA, USA).
Results
Histology
All 50 oral melanocytic tumors were classified as melanoma (malignant) and further subdivided by their predominant cell type into 26 epithelioid, 9 spindle cell, and 15 mixed types. The epithelioid-type tumors contained closely packed round or polyhedral cells with abundant cytoplasm containing large nuclei with 1 or more prominent nucleoli (Fig. 1a). The spindle cell–type tumors were arranged in streams and interweaving bundles with nuclei that tended to be larger, and nucleoli were more prominent (Fig. 1c). The mixed type comprised both cell morphologies and patterns. A chondroid formation was seen in 2 of the 15 mixed types (Fig. 1e). Typically, malignant neoplastic tissues exhibited atypical nuclei, abundant mitotic figures, and areas of necrosis. The degree of pigmentation varied from scant to moderate.
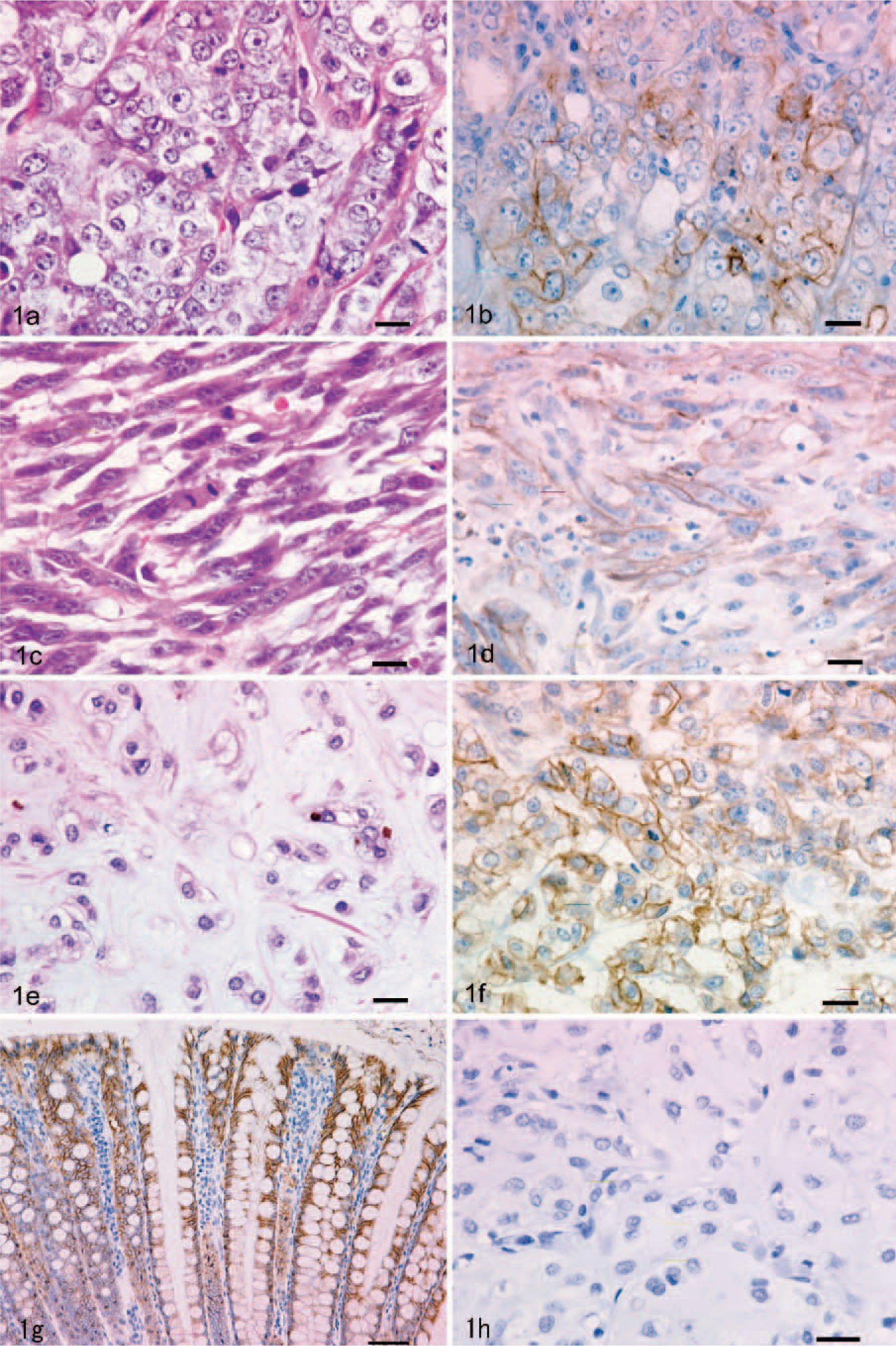
Canine oral melanoma; epithelioid type. Nonpigmented epithelioid cells are arranged in a sheet. HE. Bar = 20 μm.
Seventeen ocular melanocytic tumors were classified into 14 melanocytomas and 3 melanomas. Melanocytomas were characterized by 2 cell types in varying proportions. One type contained deeply pigmented, large, polyhedral cells (plump cells) with a small nucleus, while the other type contained slender spindle cells having abundant pigment and a relatively small nucleus (Fig. 2a–c). Mitotic figures were very rare or absent. Of the 3 melanomas, 1 was composed of only short spindle cells that showed an indistinct cell boundary and nuclear pleomorphism with scant pigmentation. Mitotic figures were abundant (>10 /10 high-power fields), and extensive necrosis was seen. The other 2 melanomas consisted of oval-to-spindle cells with nuclear pleomorphism, distinct nucleoli, and anisokaryosis. A cluster of deeply pigmented plump cells was occasionally seen. The mitotic index was approximately 3–4 in 10 high-power fields, and focal necrosis was also observed.
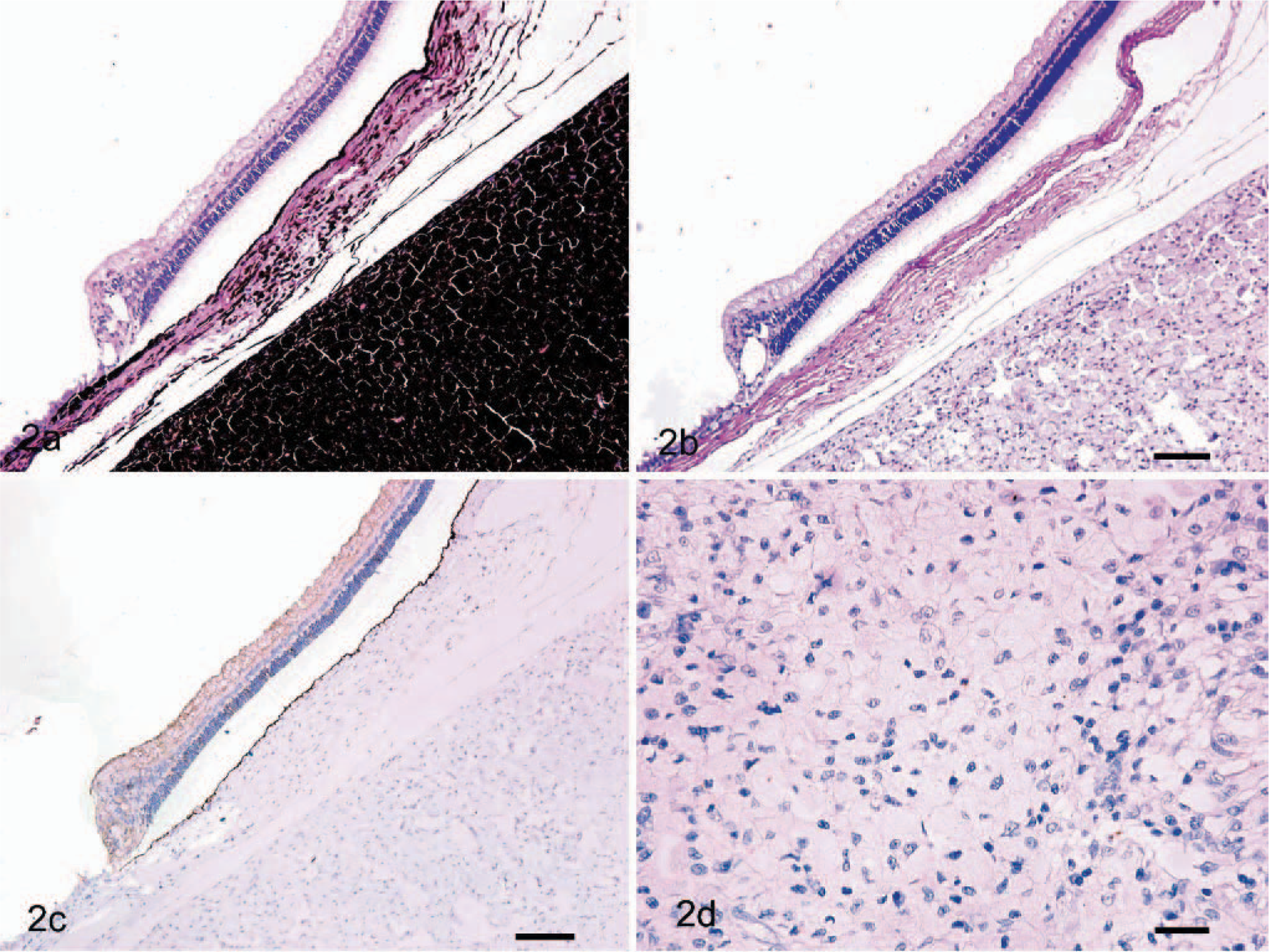
Canine ocular melanoma; melanoma arising in subconjunctival connective tissue at the limbus to the sclera. A mass is composed of deeply pigmented neoplastic cells. HE. Bar = 100 μm.
Immunohistochemistry and analysis
The staining grade of MCT1 in 3 types of oral melanomas ranged from 0 to 4. The mean grade was 2.7 for the epithelioid type (Fig. 1b), 2.4 for the spindle cell type (Fig. 1d), and 2.3 for the mixed type (Fig. 1f). In the ocular melanocytic tumors, immunoreactivity was limited to a few plump cells of 1 melanocytoma, while the others showed no positive staining (Fig. 2d). Analysis of the grade showed no statistical significance between the 3 types of oral melanomas (P = .64) (Table 1) or between ocular melanocytoma and ocular melanoma (P = .66) (Table 2), whereas a significant difference was observed between the oral and ocular melanocytic tumors (P < .0001). The control sample, which was from the colon of a healthy dog, had evidence of positive MCT1 staining in the epithelium (Fig. 1g). No immunostaining was observed for MCT1 in the slide without primary antibody (Fig. 1h).
Results of immunohistochemical evaluation of oral melanomas.
∗Data is presented as average and (range) of each subtype (grade 0 = 0%, 1 = <10%, 2 = 10–30%, 3 = 31–60%, 4 = >61%). P = .64.
Results of immunohistochemical evaluation of ocular melanocytic tumors.

∗Data are presented as average and (range) of each tumor type (grade 0 = 0%, 1 = <10%, 2 = 10–30%, 3 = 31–60%, 4 = >61%). P = .66.
Furthermore, double staining of MCT1 and Melan A revealed that most of the Melan A– positive melanocytes of the oral mucosa and iris, ciliary body, and choroid were not immunoreactive with MCT1 (Fig. 3a, b). In normal oral mucosa, MCT1 was detected in the basal cell layer; and in normal eye, MCT1 strongly immunoreacted with retinal pigmented epithelium.
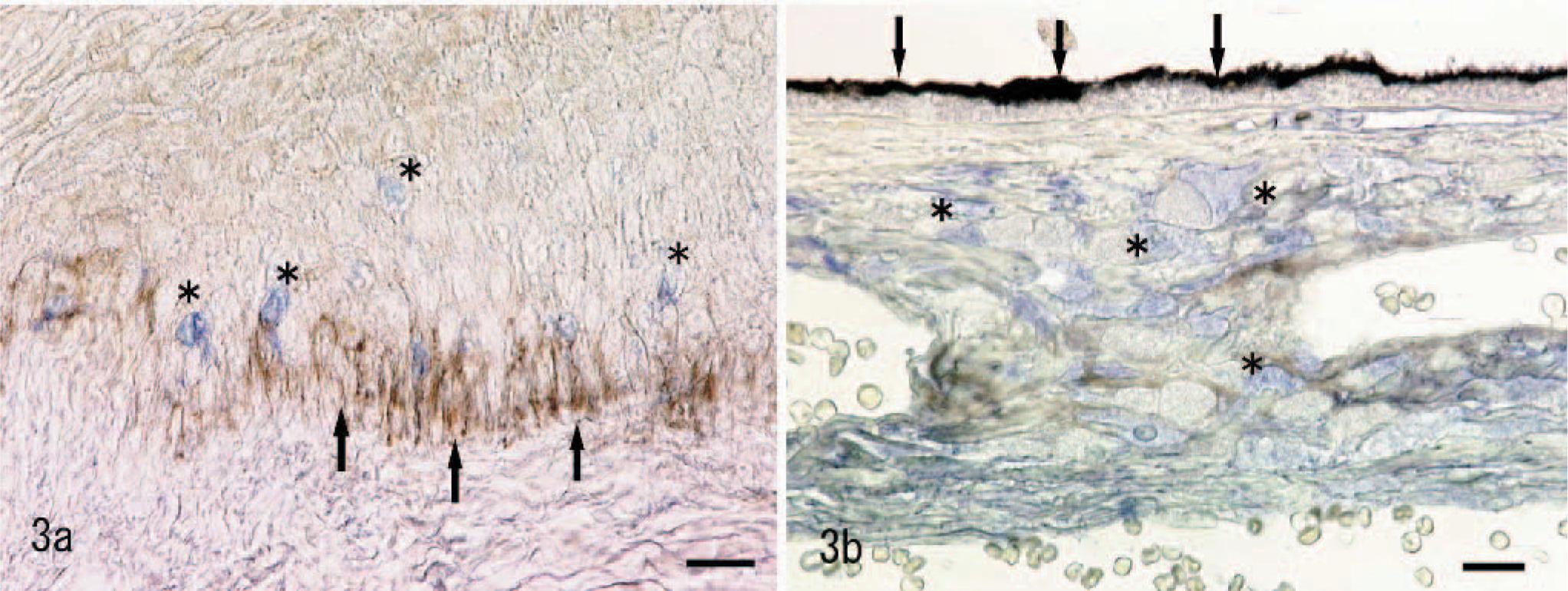
Normal canine oral mucosa; no immunoreaction of monocarboxylate transporter 1 (MCT1) in Melan A–positive melanocytes (asterisks). Note MCT1 expression on basal cells (arrows). Double immunolabeling; avidin-biotin complex and streptavidin-alkaline phosphatase methods, without counterstain. MCT1 (brown) and Melan A (blue). Bar = 20 μm.
Western blot analysis
The MCT1 antibody detected a single band of approximately 45 kDa in immunoblots of membrane proteins prepared from each of the 3 oral melanoma tissues examined, which were identical to the products obtained from control tissue (Fig. 4). No band was observed in the absence of the primary antibody. Ocular melanocytic tumors were not examined.
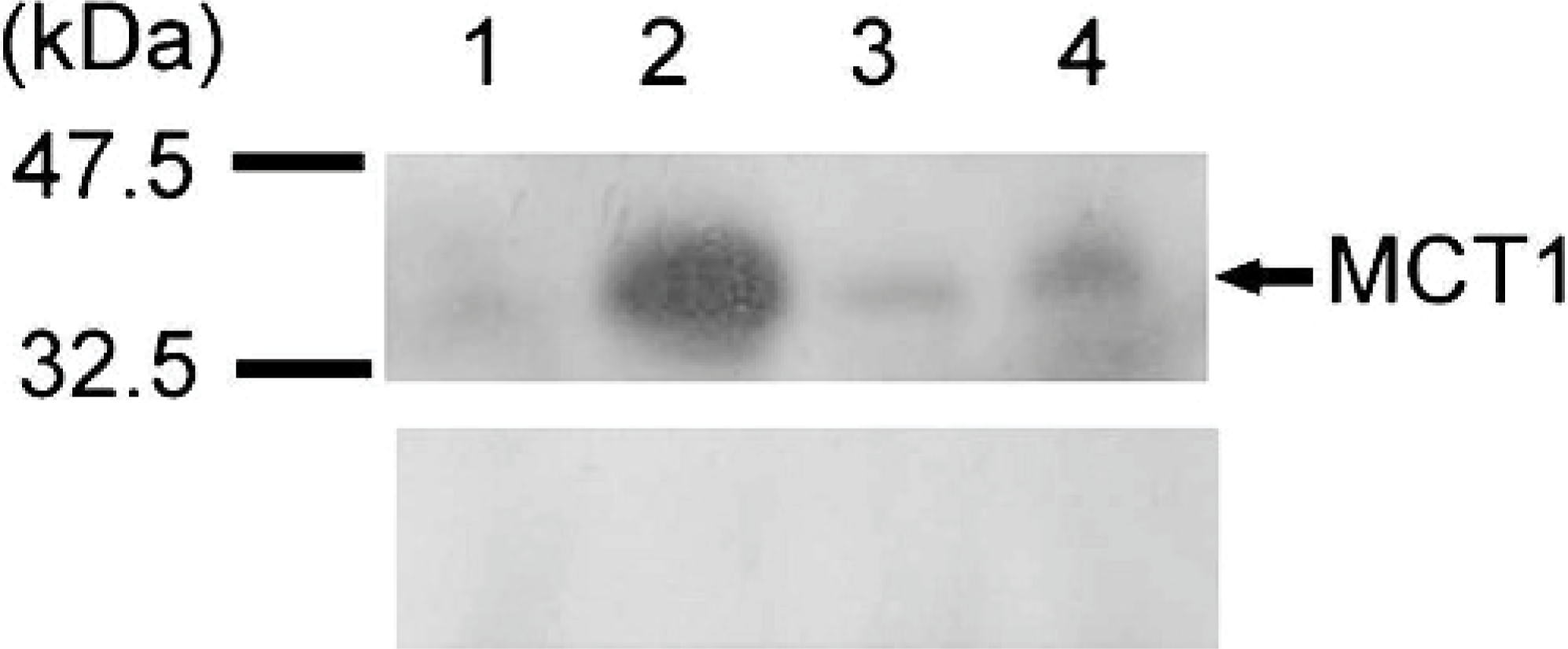
Western blot analysis of monocarboxylate transporter 1 (MCT1) expression in canine oral melanoma (n = 3). Lanes 1–3: oral melanomas. Lane 4: canine colon as positive control. Top. MCT1. Note immunopositive bands at approximately 45 kDa. Middle. Negative control. Bottom. ß-actine as internal standard control.
RT-PCR
With the use of specific primers derived from the canine MCT1 cDNA sequence, a 300-bp cDNA fragment was obtained by RT-PCR amplification of total RNA from each of the 3 oral melanoma tissues, which were at the same size band as the 1 obtained from control tissue (Fig. 5). No fragment was obtained in the absence of the primer.
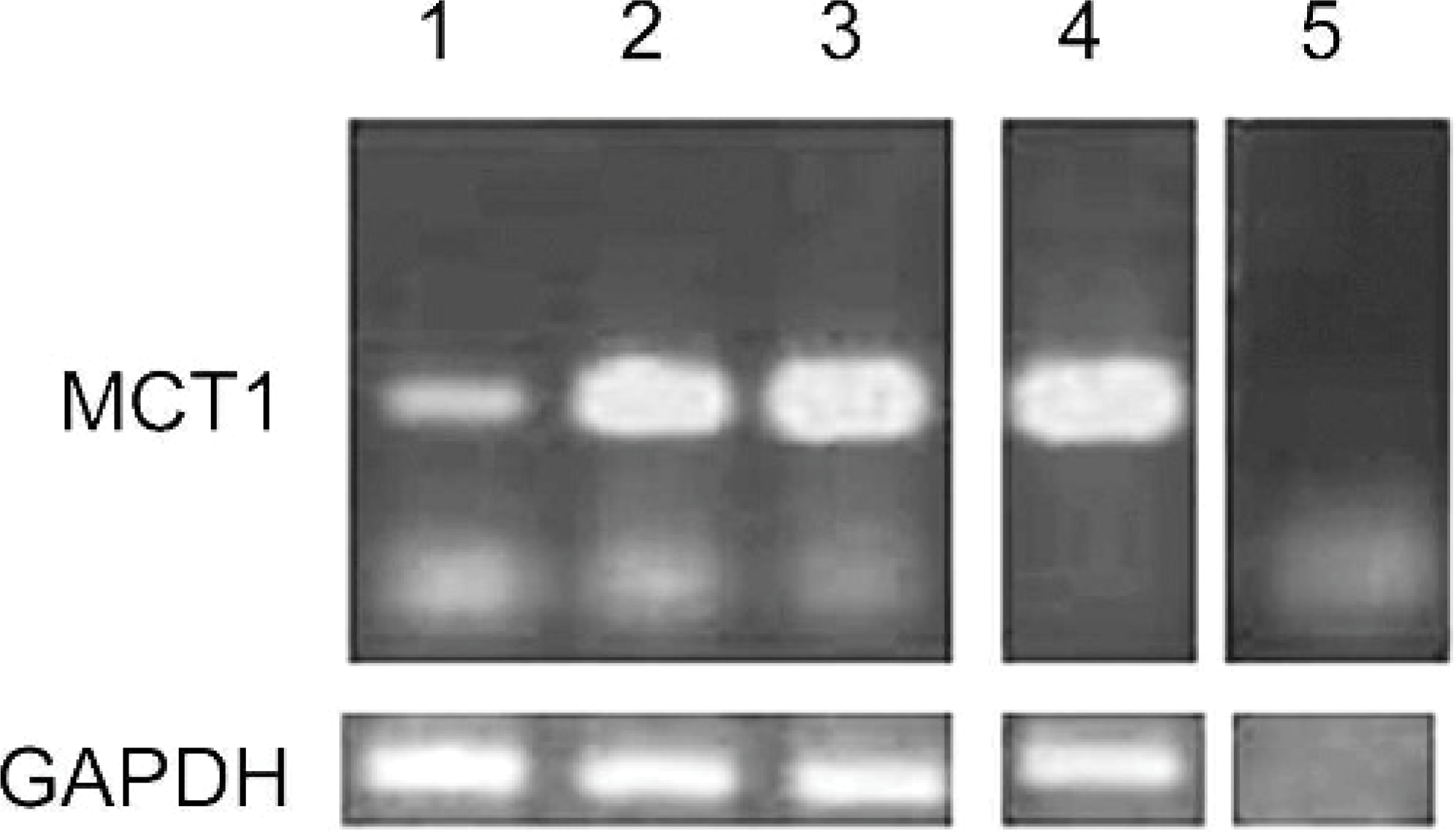
Expression of mRNA for monocarboxylate transporter 1 in canine oral melanoma (n = 3). Top. Lanes 1–3: oral melanomas. Lane 4: canine colon as positive control. Lane 5: no template DNA control. Bottom. Amplification with glyceraldehyde-3-phosphate dehydrogenase mRNA–specific primers as internal polymerase chain reaction control.
The identity of the PCR fragment was confirmed by sequencing. The nucleotide sequence of the melanoma PCR products was identical to the published canine MCT1 sequence. Ocular melanocytic tumors were not examined.
Discussion
In this article, we documented, for the first time, MCT1 expression in canine melanoma. MCT1, a proton-linked monocarboxylate transporter, is a transmembrane protein known to contribute to transporting monocarboxlates such as lactate and pyruvate into and out of cells in a variety of tissues. 9, 10 Thus, MCT1 plays a critical role in the energy metabolism, homeostasis, and pH control. The role of MCT1 in healthy as well as diseased tissues has become an area of intensive research in human medicine. Several studies have reported that MCT 1 expression was increased when cells became hypoxic because of disease states such as stroke or neoplasia. 7, 21, 34, 42
This study demonstrated that MCT1 was frequently expressed in canine oral melanoma and that there were no statistically significant differences in staining grade between different histologic subtypes. We also obtained an approximately 45-kDa band by Western blotting from 3 oral melanomas, the size of which was consistent with that of canine MCT1 protein detected in control colonic tissue. On the mRNA level, we verified the existence of MCT1 by RT-PCR and confirmed the fragment identity as canine MCT1.
In general, tumor microenvironments are acidic relative to normal tissue. It has been more than 7 decades since Werbug and Dickens 36 published the observation that tumors, as they dedifferentiate, reprogram their metabolism from oxidative phosphorylation to anaerobic glycolysis. Such an anaerobic metabolism may present an advantage for rapidly growing tumors, whose vascular supply might otherwise prove inadequate for the provision of sufficient oxygen for adenosine 5-triphosphate synthesis through oxidative phosphorylation. Thus, the catabolism of glucose to lactate is now a well-known hallmark of malignant tumors. 13, 35 However, this acquired metabolism of tumor cells eventually accumulates lactic acids inside the cell, which cause acute intracellular acidification. Consequently, intracellular acidosis leads to the inhibition of glycolysis and causes apoptosis, unless acids are removed from the cell. To avoid this, the ability to dispose of intracellular protons is crucial for cancer cell survival. 14, 20, 24, 25, 31, 34 In fact, in spite of the low extracellular pH environment of tumors compared with normal tissue, the intracellular pH of tumor cells is similar to that of normal tissue. 8, 40 Thus, the presence of MCTs is likely physiologically important in cancer cells. In human medicine, studies have shown that the transformation to malignancy is accompanied by increased MCT1 expression in some tumors, although the opposite has been reported. 1, 7, 19, 21
Similar to canine oral melanomas, human melanomas are also highly malignant and resistant to both conventional radiation and chemotherapy. Several in vitro studies aimed at reducing intracellular pH of melanoma cells have been reported with the hopes of augmenting existing therapies. Hyperglycemia combined with a respiratory inhibitor increased the acidification in melanoma cells, 3, 4 via the accumulation of lactic acid, a process known as the Warburg effect. 26 Other recent data support the notion that acidified tumor cells are more sensitive to hyperthermia and some chemotherapeutics. 3, 17, 30, 32, 43 Using 3 melanoma cell lines, Wahl et al. 34 demonstrated that intracellular pH was significantly reduced by MCT inhibitors and also showed the existence of MCT1 by RT-PCR. Consequently, an attempt was made using an inhibitor of MCT1 to improve the effectiveness of thermotherapy against melanoma cells in vitro. 5
In the present study, as assessed by immunohistochemistry, most canine oral melanomas expressed MCT1, while ocular melanocytic tumors did not. While MCT1 expression has been reported to occur in a variety of normal tissue of many species, 9 expression was rarely observed in most melanocytes of the normal oral mucosa and the eye of dogs. Increased MCT1 expression in oral melanomas compared with ocular melanocytic tumors may reflect the very different biology between these tumors in dogs. MCT1 expression may have an important physiologic role in survival of neoplastic cells within the tissue environment of oral melanoma but may not be needed in the environment typical of ocular melanocytic tumor. In addition, considering the results obtained in this study and others, we believe MCT1 could be a potential therapeutic target in canine oral melanoma. Further functional studies, however, are needed to clarify the role of MCT1 in canine melanomas.
Footnotes
Acknowledgement
The work was supported by a grant-in-aid to the High Technological Research Center (Rakuno Gakuen University) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
