Abstract
Apolipoprotein C-III (ApoC-III) amyloidosis in humans is a hereditary amyloidosis caused by a D25V mutation in the APOC3 gene. This condition has only been reported in a French family and not in animals. We analyzed a 19-year-old white lion (Panthera leo) that died in a Japanese safari park and found renal amyloidosis characterized by severe deposition confined to the renal corticomedullary border zone. Mass spectrometry-based proteomic analysis identified ApoC-III as a major component of renal amyloid deposits. Amyloid deposits were also positive for ApoC-III by immunohistochemistry. Based on these results, this case was diagnosed as ApoC-III amyloidosis for the first time in nonhuman animals. Five additional white lions were also tested for amyloid deposition retrospectively. ApoC-III amyloid deposition was detected in 3 white lions aged 19 to 21 years but not in 2 cases aged 0.5 and 10 years. Genetic analysis of white and regular-colored lions revealed that the APOC3 sequences of the lions were identical, regardless of amyloid deposition. These results suggest that ApoC-III amyloidosis in lions, unlike in humans, may not be a hereditary condition but an age-related condition. Interestingly, lion ApoC-III has a Val30 substitution compared with other species of Panthera that have Met30. Structural predictions suggest that the conformation of ApoC-III with Met30 and ApoC-III with Val30 are almost identical, but this substitution may alter the ability to bind to lipids. As with the D25V mutation in human ApoC-III, the Val30 substitution in lions may increase the proportion of free ApoC-III, leading to amyloid formation.
Amyloid is a fibrous protein derived from misfolded precursor proteins that cause amyloidosis by excessive deposition. Amyloidosis is classified into 42 types in humans and 11 types in animals, depending on the precursor protein. 2 When the precursor protein is a blood component in origin, amyloid deposits throughout the body, resulting in systemic amyloidosis. 2 Mutations in the precursor protein are responsible for some types of systemic amyloidosis, and variants causing hereditary amyloidosis have been reported in transthyretin, 15 β2-microglobulin, 34 and fibrinogen alpha-chain. 10 Apolipoprotein C-III (ApoC-III) amyloidosis is a rare hereditary systemic amyloidosis reported only in humans. 35 ApoC-III amyloidosis is characterized by severe amyloid deposition in salivary glands, renal interstitium, and glomeruli. The D25V mutation in ApoC-III is the only amyloid-related variant reported in a French family. Because there is only 1 report, its pathogenic mechanism is known only to a limited extent.
The most common systemic amyloidosis in animals is amyloid A (AA) amyloidosis, which has been reported in various avian and mammalian species. In domestic cats, whereas the hereditary AA amyloidosis in Siamese and Abyssinian cats is common,1,7 sporadic occurrences in other breeds are also frequently reported.4,32 Aside from domestic cats, AA amyloidosis has been reported in various feline species, including cheetahs,23,41 black-footed cats, 33 caracals, 8 and tigers. 26 In contrast, systemic amyloidosis other than AA amyloidosis is rare in feline species, with epidermal growth factor (EGF)-containing fibulin-like extracellular matrix protein 1–derived amyloidosis in a Tsushima leopard cat being the only reported non-AA systemic amyloidosis. 18
This study identified ApoC-III amyloidosis in 4 white lions (Panthera leo) that died in a safari park in Japan. This study aimed to provide comparative insights into the pathogenesis of ApoC-III amyloidosis by identifying the differences between lions and humans using pathological and genetic analyses.
Materials and Methods
Case Information and Histopathologic Analysis
Six white lions (cases 1–6) were analyzed in this study. They were kept in a safari park in the Tohoku region of Japan, where they died. Cases 1 through 6 were aged 19, 19, 20, 21, 0.5, and 10 years, respectively. Cases 1 to 3 are siblings. Case 4 is a cousin of cases 1 to 3 siblings. Case 5 is a grandchild of a cousin of cases 1 to 4. Cases 1 to 5 were born in Japan, and case 6 was born in South Africa. Two regular color lions (cases 7 and 8) that were kept and died in other safari parks in Japan were also evaluated. They were not related to cases 1 to 6. Cases 7 and 8 were aged 18 years and 3 months, respectively.
All the 8 lions were necropsied at the safari parks and submitted to the authors’ laboratories for pathological diagnosis. Tissues were formalin-fixed and paraffin-embedded. For case 1, the kidney, liver, spleen, heart, lung, adrenal gland, pancreas, stomach, small intestine, large intestine, and gallbladder were examined. For cases 2 to 8, livers and kidneys were examined. The formalin-fixed and paraffin-embedded specimens were cut into 3-μm thick sections and subjected to hematoxylin and eosin staining, Congo red staining, and immunohistochemistry (IHC). Amyloid deposition was confirmed as Congo red–positive amorphous lesions, showing yellow-to-green birefringence under polarized light. Resistance to potassium permanganate treatment was also examined in the Congo red–stained renal sections of case 1. 40
As described in the following, as an anti-ApoC-III antibody was newly produced in this study, the antibody’s specificity was confirmed on a cryopreserved liver tissue of case 6.
Proteomic Analysis
Liquid chromatography–tandem mass spectrometry (LC-MS/MS) of amyloid deposits was performed to detect candidate amyloid precursor proteins, as described previously. 19 Under a stereomicroscope, amyloid deposits were microdissected from the Congo red–stained kidney specimens of cases 1, 2, and 4. Approximately, 3,000,000 to 5,000,000 μm3 of amyloid deposits were collected, solubilized, digested with trypsin or chymotrypsin, and subjected to LC-MS/MS analysis, as described previously. 19 The MS/MS data were collated to theoretical fragment patterns of tryptic or chymotryptic peptide sequences from the National Center for Biotechnology Information (NCBI) database, using Mascot Server (Matrix Science Ltd., London, UK). Significant proteins/peptides were identified using Mascot’s probability-based scoring algorithm.
Genetic Analysis
Reverse transcription–polymerase chain reaction (RT-PCR) and sequencing were done to determine the gene sequence of APOC3 in lions. The primers were designed using Primer-BLAST at NCBI, based on the Panthera leo APOC3 mRNA sequence (accession: XM_042905310.1) in the GenBank database (Supplemental Table S1). Total RNA was extracted from the formalin-fixed and paraffin-embedded liver tissue sections of cases 1, 2, and 4 to 8, using the innuPREP FFPE total RNA Kit (Analytik Jena, Überlingen, Germany). One-step RT-PCR, using the PrimeScript One-Step RT-PCR Kit (Takara Bio, Shiga, Japan), was performed as follows: reverse transcription at 50°C for 30 minutes, followed by at 94°C for 30 seconds, at 60°C for 30 seconds, and at 72°C for 1 minute in 35 cycles, with a final extension at 72°C for 3 minutes. The product size was analyzed using agarose gel electrophoresis. Target DNA fragments were purified from the gel using NucleoSpin Gel and PCR Clean-up (MACHEREY-NAGEL, Düren, Germany). Nucleotide sequences were determined using TAKARA Bio’s contract service. Multiple sequence alignments were performed using EMBOSS Omega to align the cases 1, 2, and 4 to 8 sequence results and NCBI-registered Panthera leo APOC3 (accession: XM_042905310.1). 13 Each amino acid sequence was translated from codons sequenced using EMBOSS Transeq. 13
Aggregation Prediction With PASTA
The newly sequenced Panthera leo ApoC-III amino acid sequence was analyzed using PASTA 2.0. 38
Immunohistochemistry
An anti-ApoC-III antibody was newly produced using the contract services of Cosmo Bio Co. (Tokyo, Japan). Rabbits were immunized with a synthetic peptide (C+MQGYVQHATK TAQDTLTTVR) corresponding to residues Met12-Arg31 of Panthera leo ApoC-III. Affinity-purified antiserum was used as an anti-ApoC-III polyclonal antibody (1:100 dilution). Aside from a newly produced anti-ApoC-III antibody, a commercially available anti-human ApoC-III antibody (bs-4741R; Bioss, Woburn, Massachusetts; 1:500 dilution) was used as the primary antibody. For antigen retrieval, specimens were autoclaved in deionized water at 121°C for 10 minutes prior to reaction with the primary antibody. Horseradish peroxidase-conjugated polymer antirabbit IgG antibody (Dako, Santa Clara, California) was used as the secondary antibody. Positive reactions were chromogenic with 3,3’-diaminobenzidine tetrahydrochloride. The primary antibody was omitted in the negative control.
Western Blot
Western blotting was performed on lion liver tissue to confirm the specificity of the newly produced anti-ApoC-III antibody. Frozen liver tissue from case 6 was homogenized, using a beads crusher (µT-12; Taitec, Saitama, Japan) and diluted 20-fold with phosphate-buffered saline. Samples were mixed with electrophoresis sample buffer (containing 62.5 mM Tris/HCl, 2% sodium dodecyl sulfate, 10% glycerol, 5% 2-mercaptoethanol, and 0.02% bromophenol blue) and subjected to sodium dodecyl sulfate-polyacrylamide electrophoresis, using 12.5% polyacrylamide gel followed by Western blotting. The above anti-ApoC-III polyclonal antibody and antirabbit IgG horseradish peroxidase-linked antibodies (GE Healthcare, Chicago, Illinois) were used as primary and secondary antibodies, respectively. Bands were detected using an ECL Prime detection kit (GE Healthcare) and visualized using a STAGE-5100 analyzer (AMZ System Science, Tokyo, Japan; Supplemental Fig. S1).
Sequence Alignment of Human ApoC-III or Panthera Animals
Pairwise sequence alignments of sequenced lion and human ApoC-III (UniProt accession: P02656) with the D25V mutation were performed using the EMBOSS Needle. 20 Multiple sequence alignments of sequenced lion ApoC-III (NCBI accession: BEI31666.1) and other Panthera animals (NCBI accessions: snow leopard [P. uncia], XP_049468781.1; tiger [P. tigris], accession XP_015392270.1; leopard [P. pardus], accession XP_053756432.1) were performed using EMBOSS Clustal Omega. 14
Prediction of the Conformation of ApoC-III
The 3-dimensional structures of ApoC-III in P. leo (NCBI accession: BEI31666.1) and P. pardus (NCBI accession: XP_053756432.1) were predicted by AlphaFold2. 16 The structural data predicted by AlphaFold2 were visualized using a Mol* 3D Viewer in Protein Data Bank (PDB). 29 To characterize the alpha-helical properties of the ApoC-III in lions and leopards, the helical wheel was drawn using HeliQuest server. 6 According to the 3-dimensional structure predicted by AlphaFold2, the following alpha-helix sequences were chosen for helical wheel assessment: Ser7-Arg31 (SLLGLMQGYVQ HATKTAQDTLTTVR) in P. leo ApoC-III and Asp5-Arg31 in P. pardus ApoC-III (DPSLLGLMQGYVQHATKTAQDTLTT MR).
Results
Histopathological Analysis
There were no abnormal gross findings in the kidneys of case 1 (Fig. 1a). However, histologically, amyloid deposition was observed radially along the outer medulla (Fig. 1b). In these lesions, amyloid deposits compressed and entrapped tubular and vascular structures (Fig. 1c). The amyloid deposits were Congo red positive (Fig. 1d) and exhibited yellow-green birefringence under polarized light (Fig. 1e). Congophilic with renal amyloid deposits were resistant to potassium permanganate treatment (Supplemental Fig. S2). In contrast, amyloid deposition was absent in glomeruli and the renal papilla. Aside from the kidneys, amyloid deposition was observed in the gall bladder (especially in biliary sludge), heart, adrenal glands, and duodenum (Supplemental Fig. S3 and Supplemental Table S2). Based on these results, case 1 was diagnosed with systemic amyloidosis.
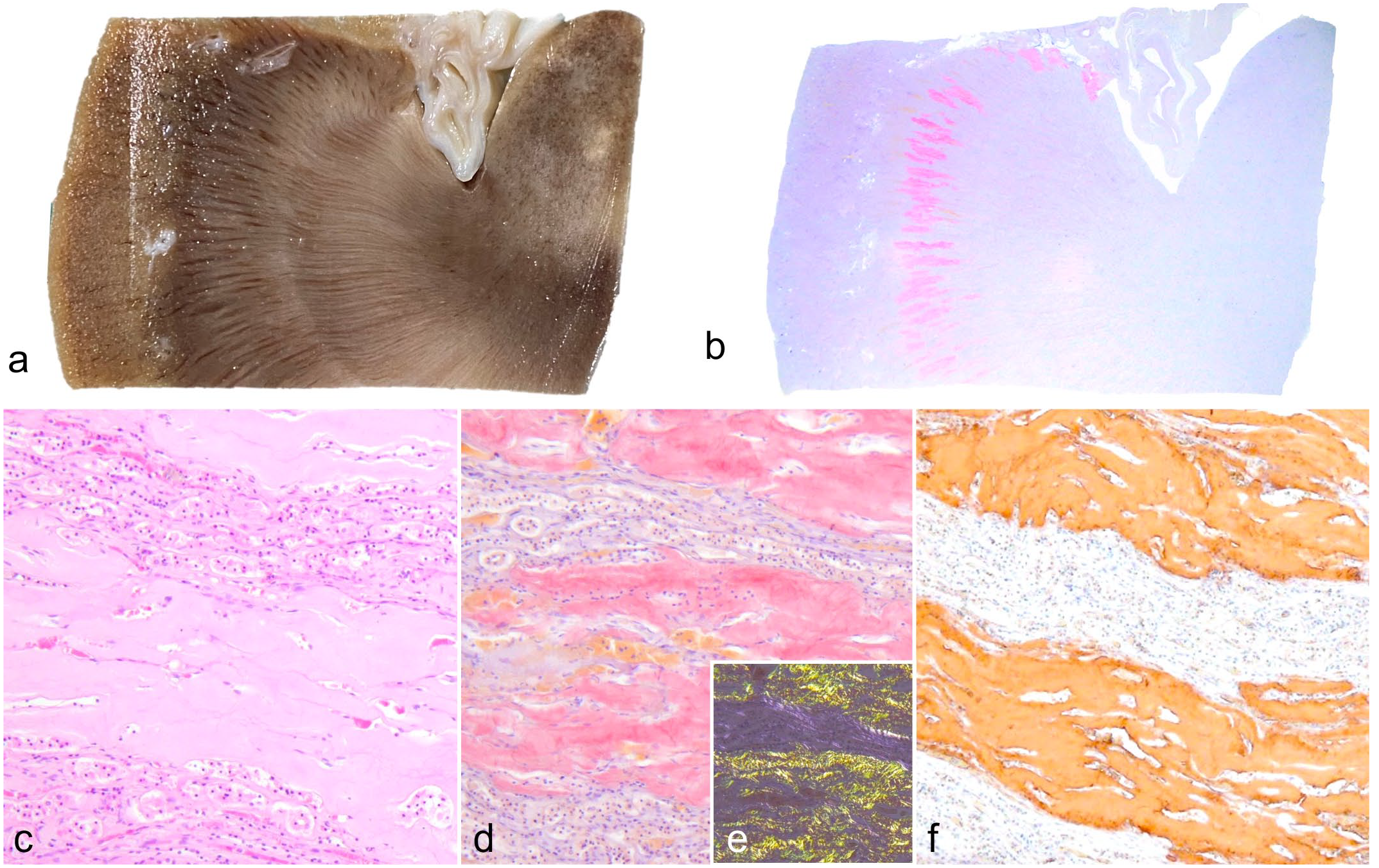
Amyloidosis, kidney, white lion, case 1. (a) Amyloid deposits are not associated with gross changes in the kidney. (b) Congo red–stained amyloid deposition is confined to the corticomedullary border. Congo red. (c) Few cellular components are seen within the amyloid deposits. Hematoxylin and eosin. (d, e) Amyloid deposits are (d) positive for Congo red and (e) show dichromatic birefringence under polarized light. (f) Amyloid deposits show immunolabeling for apolipoprotein C-III (ApoC-III). ApoC-III immunohistochemistry.
As systemic amyloidosis was observed in case 1, additional retrospective analyses were performed on cases 2 to 8 for which kidney and liver tissues were available. Amyloid deposition in the renal outer medulla was observed in cases 2 to 4. In case 2, severe amyloid deposition was observed as in case 1 (Supplemental Fig. S4). Amyloid deposition was not observed in cases 5 to 8.
Liquid Chromatography–Tandem Mass Spectrometry
Mass spectrometry-based proteomic analysis of renal amyloid deposits in cases 1, 2, and 4 identified high levels of ApoC-III (Table 1). Aside from ApoC-III, apolipoprotein A-IV, apolipoprotein E, and vitronectin were detected as amyloid signature proteins, 18 ensuring the selective collection of amyloid deposits. Immunoglobulin lambda light chain, a well-known amyloidogenic protein in animals, was detected in cases 1 and 2, but at a much lower level than that for ApoC-III.
Representative proteins detected by mass spectrometry of amyloid deposits.
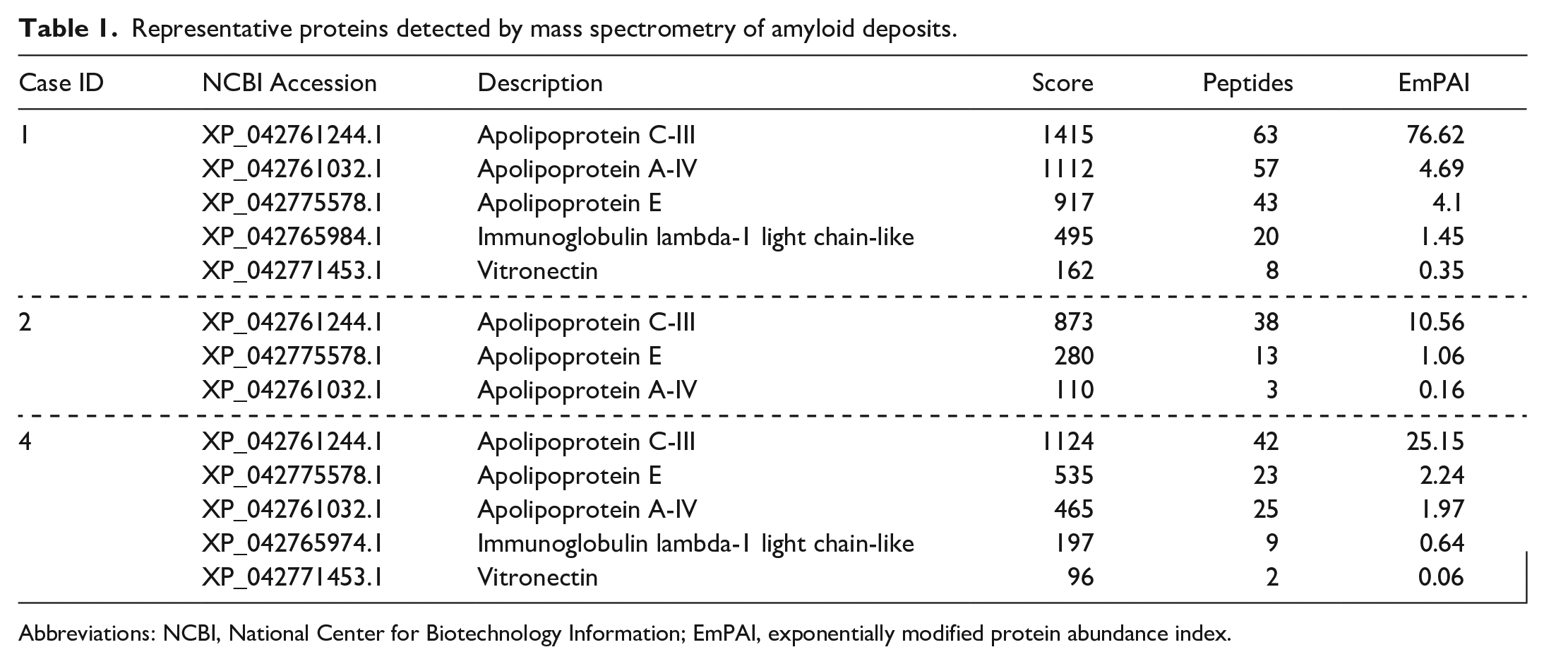
Abbreviations: NCBI, National Center for Biotechnology Information; EmPAI, exponentially modified protein abundance index.
Tryptic and chymotryptic peptides detected by mass spectrometry were analyzed to determine which regions within ApoC-III are involved in amyloid formation. Approximately, half of the N-terminal region of ApoC-III (Thr1-Arg40) was predominantly detected in tryptic samples. Nontryptic digestion was frequently observed between Glu2 and Pro6 in the N-terminal region (Fig. 2b). Val16-Trp42 was predominantly detected in the chymotryptic samples (Fig. 2c). Glu2-Glu15 was less frequently detected by chymotryptic digestion, presumably because of the presence of acidic amino acids, such as aspartic and glutamic acids. 12 Signal sequence-derived peptides were not detected in either the tryptic or chymotryptic samples.
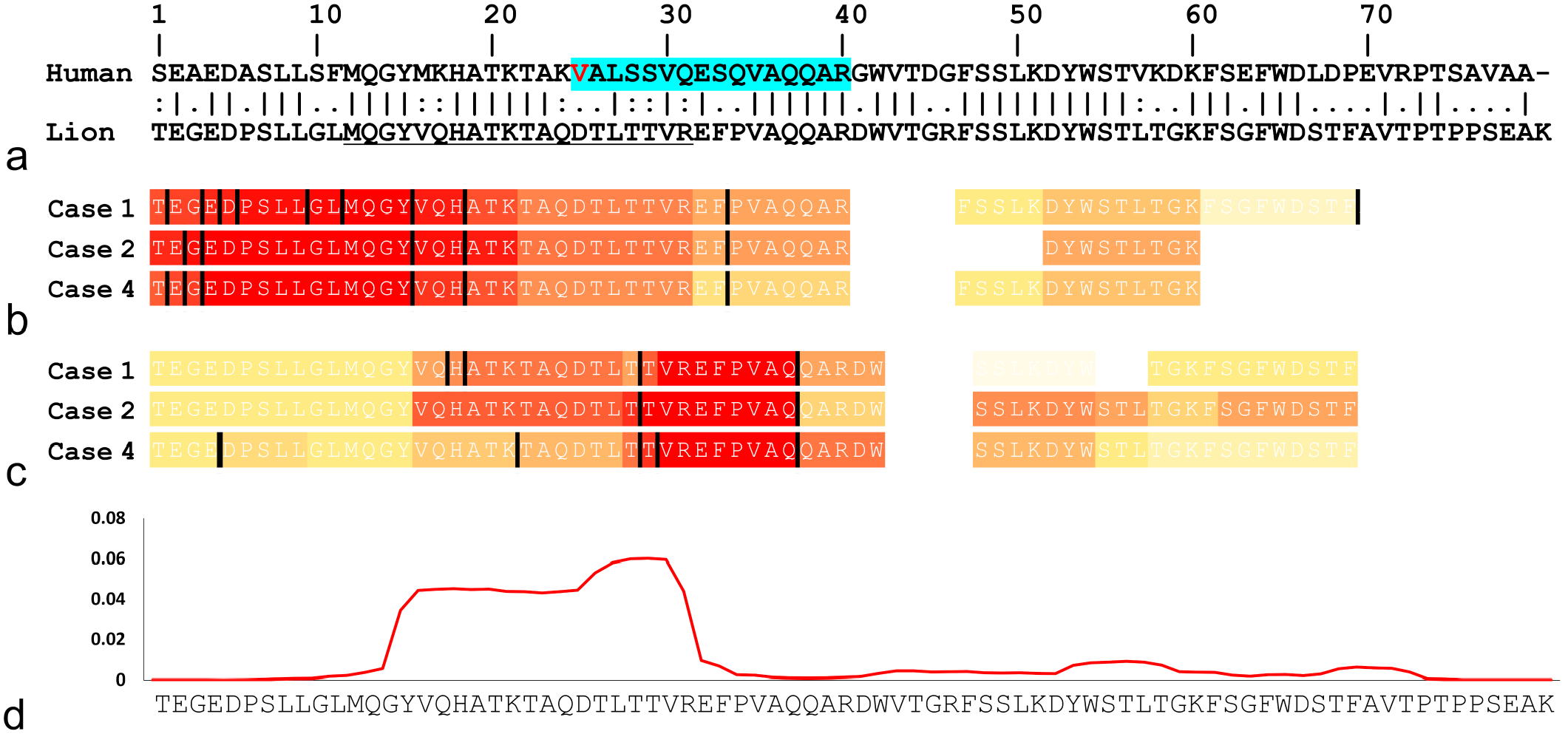
The amyloidogenic region in Panthera leo apolipoprotein C-III (ApoC-III). (a) Pairwise alignment of mature ApoC-III (excluding signal sequences) in humans and lions. Vertical bars (|) indicate fully conserved residues. Colons (:) and periods (.) indicate conserved residues between groups with strongly or weakly similar properties, respectively. The red-letter V indicates the mutation site in human ApoC-III amyloidosis patients (D25V). A blue highlight indicates peptides detected by mass spectrometry of human ApoC-III amyloid deposits. 35 The underline indicates the antigen sequence of the anti-ApoC-III antibody newly produced in this study. (b) Tryptic and (c) chymotryptic peptides derived from cases 1, 2, and 4 identified by mass spectrometry. Each bar is colored according to the total number of detected peptides, with warmer color (red) indicating higher number of detected peptides. The maximum number of detections (b) with tryptic digestion was 38, 18, and 25 (cases 1, 2, and 4, respectively) and (c) with chymotryptic digestion was 13, 20, and 15 (cases 1, 2, and 4, respectively). Black bars indicate nonpreferential cleavage sites by trypsin or chymotrypsin. (d) Predicted aggregation profile of Panthera leo ApoC-III using PASTA 2.0. Where the y-axis represents arbitrary units and higher values represent higher amyloidogenic propensity.
Prediction of Agglomeration Tendency
Structure prediction using PASTA 2.0 found that the N-terminal region of ApoC-III, Tyr15-Arg31, has an exceptionally high tendency to aggregate (Fig. 2d).
Immunohistochemistry
Immunohistochemistry (IHC), using the newly produced anti-ApoC-III antibody, had positive reactions for all amyloid deposits in cases 1 to 4 (Fig. 1f and Supplemental Fig. S4). The amyloid deposits were also weakly labeled with commercially available antihuman ApoC-III antibodies, but nonspecific positive reactions were present in the cytoplasm of the tubular epithelium (Supplemental Fig. S4).
Genetic Analysis
The APOC3 sequences determined from white lions with renal amyloid deposition (cases 1, 2, and 4), white lions without amyloid deposition (cases 5 and 6), and regular-colored lions without amyloid deposition (cases 7 and 8) were identical and matched the NCBI-registered predicted Panthera leo APOC3 (accession: XM_042905310.1). In other words, the APOC3 variation was not identified between lions with or without amyloid deposition. The newly identified Panthera leo APOC3 sequence was registered with the DNA Data Bank of Japan (accession: LC768711) and NCBI GenBank (accession: BEI31666).
Comparison of Sequence and 3-Dimensional Structure of Panthera ApoC-III
Multiple alignment of ApoC-III amino acid sequences of lions, leopards, snow leopards, and tigers revealed that lion ApoC-III has Val30 that differs from other Panthera species, which have Met30 (Fig. 3). Conformational prediction, using AlphaFold2, indicated that the ApoC-III of lions and leopards has a linear structure with 2 α-helix structures at the N- and C-termini. This predicted structure is very similar to the human ApoC-III structure as determined by solution nuclear magnetic resonance (NMR; PDB accession: 2JQ3). 5 The ApoC-III of lions with Val30 (Fig. 4a) was predicted to shorten the N-terminal α-helix by 2 residues compared with ApoC-III of leopards with Met30 (Fig. 4b). The evaluation of the helical wheel of this α-helix shows that the hydrophobic face is altered due to the Val30 substitution (Fig. 4c, d).

Multiple sequence alignment of mature apolipoprotein C-III (ApoC-III) (excluding signal sequences) in Panthera species. P. leo, lion; P. unica, snow leopard; P. tigris, tiger; P. pardus, leopard. Asterisks (*) indicate fully conserved residues. Colon (:) and period (.) indicate conserved residues between groups with strongly or weakly similar properties, respectively.
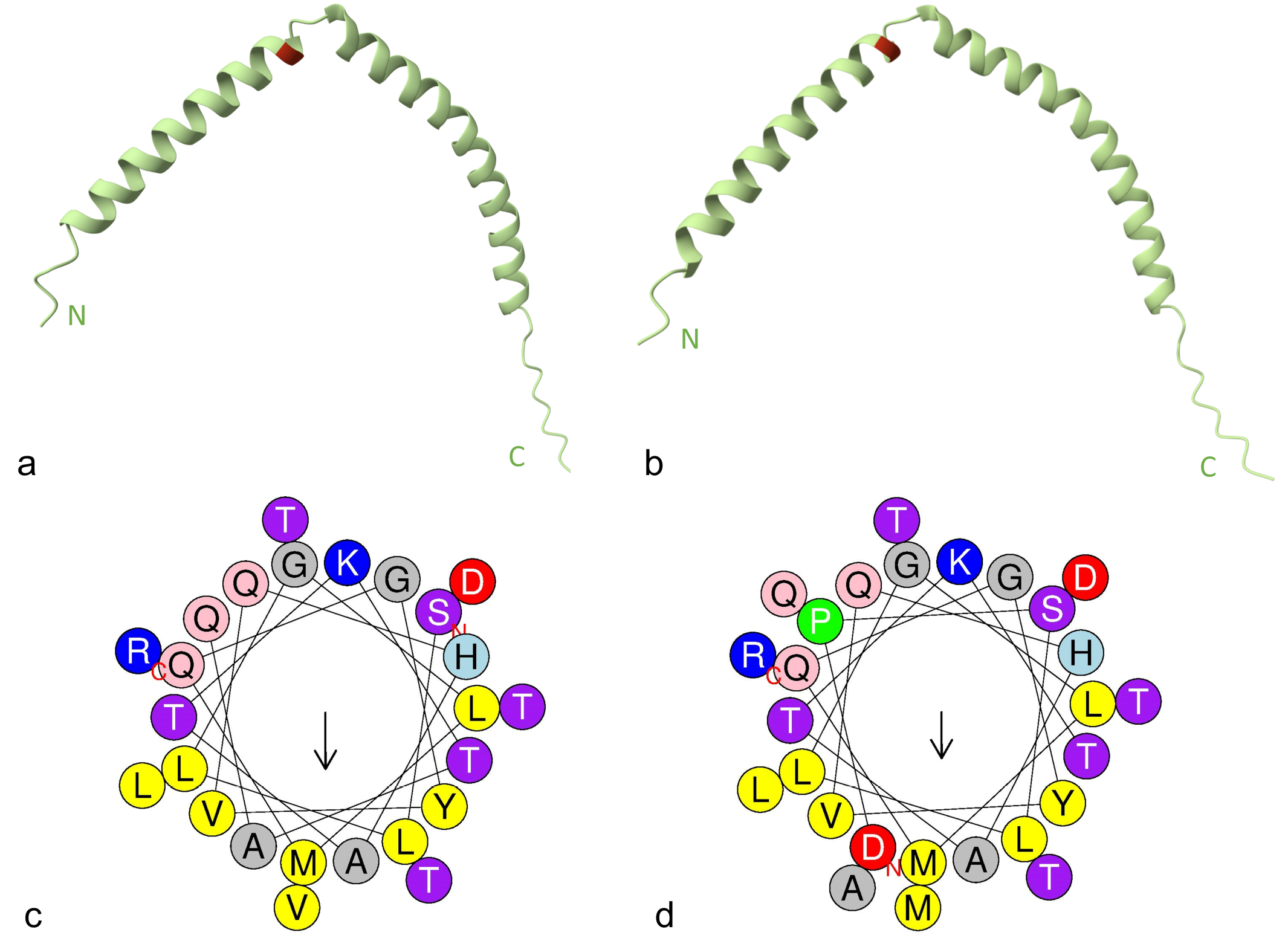
The structural prediction of apolipoprotein C-III (ApoC-III). (a, b) Conformation of ApoC-III in (a) lions and (b) leopards predicted by AlphaFold2. The red highlights indicate Val30 in (a) lion ApoC-III, and (b) Met30 in leopard ApoC-III. N, N-terminal. C, C-terminal. (c, d) Helical wheel diagram of the N-terminal α-helix of ApoC-III in (c) lions and (d) leopards generated using the HeliQuest server. Hydrophobic residues are shown in yellow, serine and threonine in purple, basic in blue, acidic in red, aspartic acid and glutamine in pink, alanine and glycine in gray, proline in green, and histadine in light blue. The arrows represent the magnitude of the hydrophobic moment. The residue marked with “N” is the N-terminal end of the helix, with the residue marked “C” being the C-terminal end.
Discussion
This study found systemic amyloidosis characterized by severe deposition in the renal corticomedullary border region of white lions. This deposition pattern differed from that of AA amyloidosis, which is characterized by glomerular and tubulointerstitial deposition; 4 therefore, non-AA systemic amyloidosis was suspected. Mass spectrometry-based proteomic analysis revealed that ApoC-III was the primary component of amyloid deposits, which IHC corroborated. Based on proteomic analyses, this lion was diagnosed with ApoC-III amyloidosis. In humans, ApoC-III amyloidosis is an extremely rare hereditary systemic amyloidosis reported only in a French family. 35 This is the second report of ApoC-III amyloidosis and the first in nonhumans.
We should consider the possibility that ApoC-III is codeposited with amyloid, that is, not an amyloid precursor protein, as apolipoprotein A-I, apolipoprotein A-IV, apolipoprotein E, and ApoC-III have been shown to bind to amyloid fibrils in vitro. 17 Apolipoprotein A-IV and apolipoprotein E, in particular, are known as amyloid signature proteins because they universally codeposit with amyloid fibrils in vivo, regardless of the type of amyloid precursor protein. 37 For this reason, in humans, the diagnostic criteria for apolipoprotein A-IV amyloidosis based on mass spectrometry are defined as a high number of apolipoprotein A-IV peptides detected by mass spectrometry and the absence of other amyloidogenic proteins, 3 and this criterion is also used for apolipoprotein A-IV amyloidosis in animals. 27 Although there are no reports of significant detection of ApoC-III in mass spectrometry of amyloidoses other than ApoC-III amyloidosis,22,37 even if ApoC-III had a property of amyloid signature protein, this study can consider ApoC-III as an amyloid precursor protein based on the above criteria. Furthermore, if ApoC-III is an amyloid signature protein, that is, a native protein, this would not explain the apparent bias in detection frequency between the N- and C-terminal peptides by mass spectrometry. Amyloid deposits were positive for ApoC-III with high contrast by IHC, indicating that the ApoC-III detected by mass spectrometry is not contamination from surrounding tissues. Based on our data and numerous previous studies on amyloid signature proteins, there is no doubt that ApoC-III detected in this study is not an amyloid signature protein but an amyloid precursor protein.
In human ApoC-III amyloidosis, renal amyloid deposition is found primarily in the cortical arterial walls, glomeruli, and the tubular interstitium. 35 In contrast, renal amyloid deposition in white lions was confined to the extramedullary zone without cortical deposition. The different deposition patterns between humans and lions may be ascribed to the differences in amyloid formation mechanisms. In humans, a single nucleotide substitution (c.134A>T) in exon 3 of the APOC3 gene, resulting in D25V mutation in the mature ApoC-III protein, causes amyloid formation. 35 This study observed amyloid deposition in 4 cases of related white lions, indicating a familial condition. Therefore, we verified the ApoC-III variation in white and normal-colored lions; however, the ApoC-III sequences were identical in all lions analyzed, with or without amyloid deposition, suggesting that ApoC-III amyloidosis in lions is unlikely to be a familial condition.
Aging is closely related to the onset of various types of amyloidosis. 31 As the 4 white lions with amyloidosis were aged 19 to 21 years, and the 6-month-old and 10-year-old white lions were amyloid-negative, the data from this study suggest that aging, rather than genetics, may be involved in the pathogenesis of ApoC-III amyloidosis in white lions. The relationship between ApoC-III amyloidosis, genetics, and aging in lions needs to be verified in future studies.
To date, 2 studies have described the histological features of renal amyloidosis in lions. Williams et al 39 reported amyloidosis characterized by proximal medullary interstitial amyloid deposits in a 14-year-old lion from Namibia (originally a wild animal). Newkirk et al 21 described a case of amyloidosis in an 18-year-old captive lion characterized by localized amyloid deposition in the corticomedullary border of the kidney. Both cases are very similar to the present cases in histopathologic features and age of onset. Furthermore, in Newkirk’s case, amyloid deposits were resistant to potassium permanganate treatment, consistent with the present case. Although neither study provided a definitive diagnosis using IHC or mass spectrometry, the concordance of pathologic features makes it likely that these cases represent ApoC-III amyloidosis. The ApoC-III sequences of white and normal-colored lions were identical, suggesting that all Panthera leo can develop ApoC-III amyloidosis. Recently, it was reported that Japanese squirrels kept in Japanese zoos develop age-related fibrinogen alpha-chain amyloidosis. 9 Similarly, lions may also develop ApoC-III amyloidosis due to their longevity in zoo captivity. An accurate classification of amyloidosis in lions based on IHC and mass spectrometry will be required.
Mass spectrometry of tryptic and chymotryptic samples predominantly detected the N-terminal region of ApoC-III (Thr1-Trp42), among which Tyr15-Arg31 was predicted to have high amyloidogenic potential by structural prediction. These results suggested that the N-terminal region of ApoC-III is involved in amyloid formation. In addition, frequent proteolysis was detected between Glu2-Pro6. In various amyloidoses, the proteolysis of precursor proteins leads to structural destabilization, resulting in amyloid formation.25,30 This study suggests that the N-terminal fragments of ApoC-III undergoing proteolysis may form amyloid fibrils. In human ApoC-III amyloidosis, Val25-Arg40 was detected by mass spectrometry, 35 suggesting that the amyloid-forming regions in humans and white lions may coincide (Fig. 2a). The amino acid identity of Thr1-Arg40 of Panthera leo ApoC-III and Ser1-Arg40 of human ApoC-III, which may be involved in amyloidogenesis, is 62.5%, with theoretical isoelectric points of 4.89 and 6.49, respectively. These biochemical differences in ApoC-III between both species may contribute to the different intranephron distributions of amyloid deposits.
Multiple alignments of ApoC-III sequences from Panthera species revealed that the Val30 substitution occurred in lion ApoC-III. Apolipoprotein binds to lipids through an amphipathic helix, and amino acid substitutions in this region affect lipid-binding properties. 28 In human ApoC-III, the D25V and A23T mutations inhibit lipid binding, whereas the Q38K mutation, known as a gain-of-function mutation, is thought to enhance lipid binding.11,24,35 Notably, these ApoC-III mutations in humans are located close to the Val30 in lions. Binding to lipids stabilizes ApoC-III conformation, 36 and the amyloidogenicity of the D25V mutant is thought to be acquired by its release from lipids. Therefore, the Val30 substitution in lion ApoC-III may affect the binding to lipids, leading to structural instability of ApoC-III. Although the effect of this substitution is probably not immediate, it is expected to lead to amyloid formation through some metabolic dysfunction associated with aging.
In conclusion, this study identified ApoC-III amyloidosis for the first time in animals. Although more case studies will be needed to elucidate the pathogenesis of ApoC-III amyloidosis in white lions, it is likely that aging, rather than genetic influence, is involved in the pathogenesis. Fortunately, it will not be difficult to differentiate ApoC-III amyloidosis in lions based on the characteristic histology of severe deposition in the corticomedullary border region. Age-associated animal amyloidosis may increase as life expectancy increases in captive environments, such as zoos, thus necessitating continuous surveillance.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241230100 – Supplemental material for Apolipoprotein C-III amyloidosis in white lions (Panthera leo)
Supplemental material, sj-pdf-1-vet-10.1177_03009858241230100 for Apolipoprotein C-III amyloidosis in white lions (Panthera leo) by Natsumi Kobayashi, Susumu Iwaide, Hiroto Fukui, Yumi Une, Yoshiyuki Itoh, Miki Hisada and Tomoaki Murakami in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by JSPS KAKENHI (grant no. 23H02380) and the Program on Open Innovation Platform with Enterprises, Research Institute, and Academia (OPERA) from the Japan Science and Technology Agency (JST).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
