Abstract
The spontaneous incidence and distribution of β-amyloid (Aβ) peptide and argyrophilic deposits were extensively investigated in the common marmoset (Callithrix jacchus). In 12 young marmosets, up to 67 coronal step sections were evaluated in the brain. One brain hemisphere was stained by a silver method and also immunohistochemically with a primary antibody directed against Aβ. Argyrophilic deposits were observed in 4 (33%) of 12 animals with silver stain, and Aβ was seen in 4 (33%) of 12 animals with anti-4G8 antibody. The overall incidence was 6 (50%) of 12 affected animals. These results show that the young marmoset brain displays Aβ deposits at a low incidence and that immunohistochemistry is more suitable than silver staining to reveal rare amyloid plaques. These findings also provide evidence for the early onset of amyloid deposits in marmoset brain.
Laboratory-bred common marmosets (Callithrix jacchus) have a natural lifespan in captivity in excess of 12 years, and their maximum longevity is more than 16 years. 3 Due to its small size, its relatively easy adaptation to laboratory conditions, and its neuroanatomical similarities with human species, this animal is considered to represent a convenient primate model for studying neurodegenerative diseases.
A major feature of Alzheimer's disease in humans is the deposition of parenchymal plaques of β-amyloid (Aβ), which is a 4-kDa protein subfragment of amyloid precursor protein (APP). 2 Experimentally induced Aβ deposits in the brain of marmosets have previously been reported, but no natural occurrence of Aβ was found in young to middle-aged control animals. 3, 4 Only elderly control marmosets were thought to spontaneously display amyloid plaques. 9 We thoroughly evaluated the brains from 12 young marmosets from a control group for the presence of Aβ plaques using silver impregnation, immunohistochemistry, and routine histology. All of the captive-born animals used in this study were obtained from recognized suppliers (Harlan UK Limited, Oxon, UK, German Primate Center, Gottingen, Germany and University Tuebingen, Tuebingen, Germany). The animals were in the weight range of 280 to 500 g, in the age range of 22 to 38 months at necropsy, and assumed to be sexually mature at the beginning of a 1-month study. Six males and 6 females belonging to a control group were used. All experiments were carried out in accordance with all applicable institutional, local, and national guidelines, including the National Institutes of Health's Guide to the Care and Use of Laboratory Animals. The animals were acclimatized for a period of 4 weeks and kept under controlled laboratory conditions.
Animals were sacrificed with an overdose of intravenous sodium pentobarbitone prior to exsanguinations. At necropsy, after sampling and weighing of the brain, 1 brain hemisphere was fixed in 10% neutral buffered formalin, and the samples were shipped to NeuroScience Associates (Knoxville, TN). Using MultiBrain Technology, 8 hemispheres were embedded together per block and freeze-sectioned at 40 μm in the coronal plane through the entire hemisphere (∼37 mm in length). Aβ immunohistochemistry was performed on every 12th section (at 480-μm intervals). Sections preincubated with formic acid were incubated with 4G8 antibody from Signet Laboratories (dilution 1 ° 10,000; Dedman, MI) and visualized by the VectaElite ABC kit and diaminobenzidine (Vector Laboratories Inc, Burlingame, CA). The immunoreactive epitope lies within amino acid 18–22 of Aβ, and Aβ antibody 4G8 reacts to the abnormally processed isoforms as well as precursor forms (amyloid precursor protein). 1, 8 An adjacent set of brain sections (every 12th section, at 480-μm intervals) was stained with Campbell-Switzer silver stain to reveal potential amyloid plaques and neurofibrillary tangles. Thionine staining was performed on consecutive sections to reveal cell bodies (every 12th section, at 480-μm intervals). Hematoxylin and eosin (HE) staining was performed for standard brain tissue evaluation on selected sections from the following brain levels: medulla, chiasma opticum, mamillary corpus, and pons. The slides were covered with cover slips, labeled after serial ordering, and examined by light microscopy.
Aβ deposits revealed by 4G8 immunostaining (Fig. 2) were observed in 4 of 12 common marmosets, and argyrophilic plaques were detected (Table 1) in the same proportion of animals (Fig. 1–2). Both sexes were equally affected. Two of the animals (41434M and 41731F) were positive using both methods. In most cases, a single occurrence per animal was identified, but up to 3 Aβ deposits per animal could be noted.
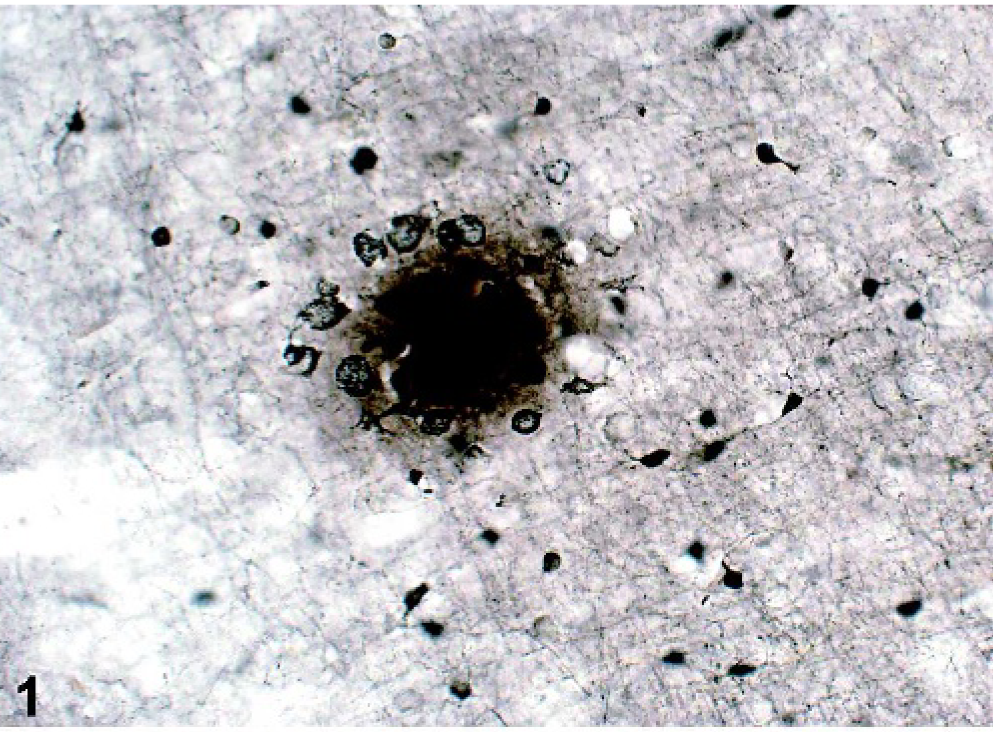
Brain from marmoset 41434M, section 26. Argyrophilic plaques are extracellular structures located in the cortical neuropil. Most plaques consist of focal deposits of silver-positive material with large argyrophilic surrounding nuclei. Campbell-Switzer silver staining. Bar = 40 μm.

Brain from marmoset 41434M, section 22. Aβ plaques are extracellular structures located in the cortical neuropil. The 4G8 immunoreactivity appears as a uniformly stained spherical structure with radiating elements at the margin, suggestive of diffuse deposits. Immunohistochemistry (anti-4G8 antibody). Bar = 80 μm.
Incidence of Aβ deposits and argyrophilic plaques per animal.
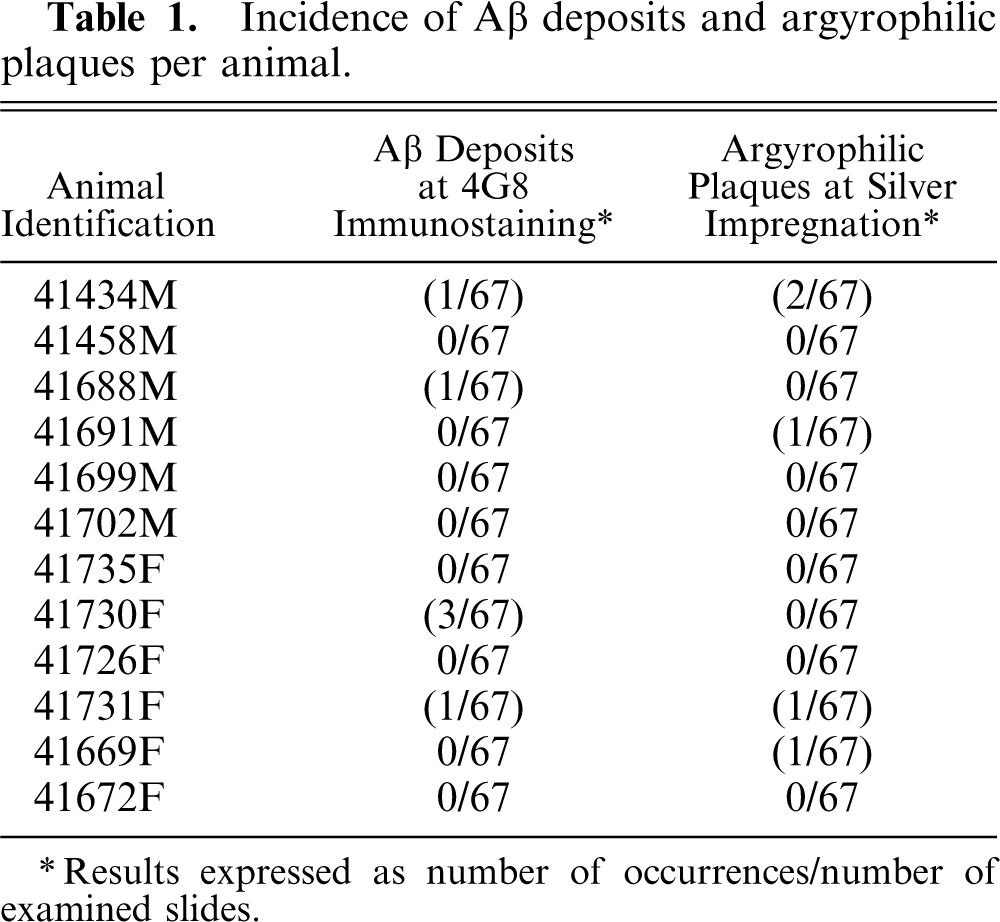
∗ Results expressed as number of occurrences/number of examined slides.
The size of the plaques was about 50–200 μm in diameter. Only isolated plaques were observed in a few cerebral and cerebellar cortical areas, without any specific neuroanatomic location. There did not appear to be a simple relationship between the incidence and density of Aβ deposits and the age of animals. No cerebral amyloid angiopathy in intracortical or meningeal vessels and no neurofibrillary tangles were observed in these animals. No amber was seen in Campbell-Switzer silver staining, which suggests a diffuse deposit. The characteristic disruption caused by fibrillar amyloid deposits (cores) typically seen in HE-stained sections of human Alzheimer's disease was not observed in these animals.
To our knowledge, this is the first time that Aβ deposits have been documented in common marmosets of this age. Aβ was already known to appear more commonly in primates than in other species, but in marmosets its natural occurrence had only been reported in animals that had reached at least 7 years. 8 A previous study also reported the anatomic distribution of amyloid deposits in older animals, with no abnormally hyperphosphorylated tau protein in plaque-associated neurites, which further suggests that the present findings are diffuse deposits. 8 In our study, we document the earliest timepoint of occurrence, as lesions were observed in younger animals ranging from 2 to 3 years of age. In this case, brains have been thoroughly examined by serial sections, which represent quite extensive sampling and examination when compared to previously published papers. Immunohistochemistry was considered to generate the best signal–noise ratio. Some difficulty was observed with silver stain interpretation, because several other lesions were positively stained and there was a risk for false-positive animals. For instance, some populations of glia stained lightly; other features that stained darkly were the tanycytes at the base of the third ventricle and the islands of Calleja in the olfactory tubercle. The architectural features responsible for the staining in each of these are not known. The silver staining method reveals plaques and even tangles if present, but the chemical moieties that are being stained or attracted by the silver are not understood. Consequently, positive slides were reviewed twice, and only the lesions that had a morphology consistent with amyloid plaques were counted. HE staining, which was used in this study to detect any associated pathological events, failed to reveal amyloid plaques. Due to the low incidence of lesions, we hypothesize that the observed changes might represent the first occurrence of an age-related amyloid deposition in the common marmoset. This observation confirms the spontaneous and progressive occurrence of Aβ deposition in this species. It was shown that cerebral amyloidosis could be induced by the intracerebral introduction of exogenous Aβ protein, 3 but a nonhuman primate model in which age-related Aβ deposition spontaneously occurs would be of greatest interest in order to study the physiopathological mechanisms that contribute to the clinical symptoms of Alzheimer's disease. It also would enable achievement of the proof of concept for potential new drugs. The rhesus monkey can be used as a model, 6 and it has proved to be valuable in the field of Aβ vaccination, 5 but due to its size, handling, and housing difficulties, an alternative choice would be useful for pharmaceutical industries. 10 Nevertheless, the marmoset appears to be no better candidate, as the age of onset is still far too long to assess the Aβ deposition. 7 The lack of precise anatomic sites of plaque deposition, the paucity of the deposits, and their diffuse nature further hinder its use as a model, especially when compared to more widely used transgenic amyloid-depositing mice.
