Abstract
Amyloid-producing odontogenic tumors (APOT) are rare, and in cats, the histogenesis of the amyloid remains undetermined. In the present study, APOTs in 3 cats were characterized by immunohistochemistry, and the amyloid components analyzed using tandem mass spectrometry. Antiameloblastin antibodies labeled both neoplastic epithelial cells and amyloid in all cases. Neoplastic epithelial cells had strong, diffuse immunoreactivity to antibodies against cytokeratin AE1/AE3, cytokeratin 14, and cytokeratin 19 in all cases and focal immunoreactivity to nerve growth factor receptor antibodies in 2 of 3 cases. Amyloid and some tumor stromal cells were weakly positive for laminin. Calretinin, amelogenin, S100, and glial fibrillary acidic protein antibodies did not label neoplastic epithelial cells or amyloid. Extracted amyloid peptide sequences were compared to the porcine database because the cat genome is not yet complete. Based on this comparison, 1 identical ameloblastin peptide was detected in each tumor. These results suggest that feline APOTs and the amyloid they produce are of ameloblastic lineage.
Keywords
Calcifying epithelial odontogenic tumors (CEOT) in humans and similar neoplasms in dogs and cats, now classified as amyloid-producing odontogenic tumors (APOTs), are uncommon odontogenic neoplasms with poorly understood histogenesis. 5 The salient feature of both entities is the production of amyloid of uncertain origin. Though rare, APOTs have been documented in dogs, cats, and a Bengal tiger.1,3,5,6 Most APOTs are expansile, solid, or cystic masses of the mandibular or maxillary gingiva. APOTs are typically benign but can invade locally, incorporate teeth, and even destroy jaw and facial bones. Reports of immunohistochemical reactivity of the neoplastic epithelial cells and the amyloid are variable.1,6 Although the histologic features suggest an odontogenic origin of APOTs, the specific cell of origin and the source of amyloid have not been conclusively demonstrated.
In human CEOTs, extracted amyloid fibrils were determined by mass spectrometry to be composed of N-terminal fragments of odontogenic amyloid ameloblast-associated protein (ODAM). 10 However, the amyloid of APOTs from a cat, dog, and Bengal tiger did not contain ODAM-related peptides but rather an ameloblastin-like peptide, which was provisionally named after the normal canine ameloblastin, AAmel. 3
In the present study, 3 feline APOTs were characterized immunohistochemically, and the amyloid components were analyzed using tandem mass spectrometry.
Materials and Methods
Formalin-fixed, paraffin-embedded tissue from 3 feline APOTs were retrieved from the archives of the University of Illinois Veterinary Diagnostic Laboratory. Case No. 1 was a mandibular mass from a 9-year-old spayed female Domestic Medium Hair cat. Case No. 2 was a rostral maxillary mass from a 4-year-old castrated male Domestic Long Hair cat. Case No. 3 was a rostral mandibular mass of an 11-year-old intact male Domestic Short Hair cat. Histologic sections were stained with hematoxylin and eosin. Representative sections were stained with Congo red and examined under polarized light to identify amyloid and to choose the most amyloid-rich sections for evaluation.
Immunohistochemistry was performed according to manufacturers’ instructions and reported procedures. Antibody sources and dilutions are in Table 1. Haired skin (epidermis) was the positive control tissue for laminin and all cytokeratin (CK) antibodies. Secretory ameloblasts and human ameloblastoma sections were the positive controls for antiamelogenin and ameloblastin antibodies, respectively. For calretinin and nerve growth factor receptor (NGFR) antibodies, nerve bundles served as internal positive controls. Peripheral nerve and brain were used as positive controls for S100 and glial fibrillary acidic protein (GFAP), respectively. Skeletal muscle was used as a negative control for CK14 and CK19, amelogenin, and NGFR. For laminin, S100, GFAP, and ameloblastin, negative controls included sections incubated with respective sera (eg, rabbit or goat) in place of primary antibody. The immunoreactivity was evaluated by location and labeling intensity. Labeling with each antibody was scored for the neoplastic odontogenic epithelium, the overlying oral mucosal (nontumor) epithelium, stromal cells, and amyloid. The percentage of positive cells was scored as +++ (>75%), ++ (21%–75%), + (5%–20%), and – (0%–4%). Amyloid immunoreactivity was scored as + (positive) or – (negative).
Antibodies for Immunohistochemistry.
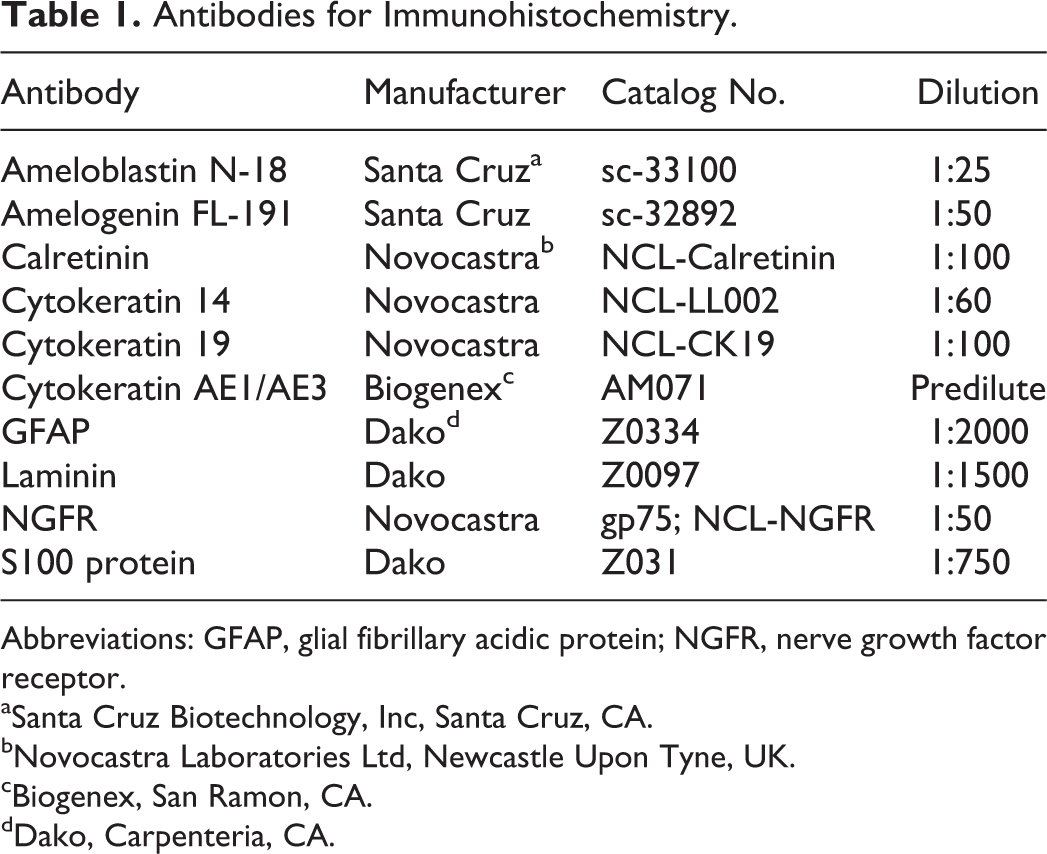
Abbreviations: GFAP, glial fibrillary acidic protein; NGFR, nerve growth factor receptor.
aSanta Cruz Biotechnology, Inc, Santa Cruz, CA.
bNovocastra Laboratories Ltd, Newcastle Upon Tyne, UK.
cBiogenex, San Ramon, CA.
dDako, Carpenteria, CA.
Unstained 4- to 6-μm-thick sections of the neoplasms on poly-L-lycin-coated slides from each case were submitted to the University of Tennessee Graduate School of Medicine for analysis. Amyloid was extracted from the sections with 6 M guanidine HCl; the extract was reduced, alkylated, and purified by reverse-phase high-pressure liquid chromatography. The major protein-containing peaks were digested with trypsin and the peptides analyzed by tandem mass spectrometry. 8 Because the cat genome has not yet been sequenced and because canine ameloblastin and porcine ameloblastin have approximately 80% homology, the peptides were compared to the porcine genomic database. 3
Results
All 3 neoplasms had characteristic histologic features of APOTs (Fig. 1), with irregularly shaped islands and bands of odontogenic epithelium, palisading of the peripheral cells, focal reverse nuclear polarity, and basilar cytoplasmic clearing. Stellate reticulum was focally prominent in all 3 cases. Cyst formation, melanin granules, and anucleated ghost cells were variably present in all 3 tumors, as were birefringent congophilic amyloid deposits, with or without calcification, which were intimately associated with the neoplastic epithelium.
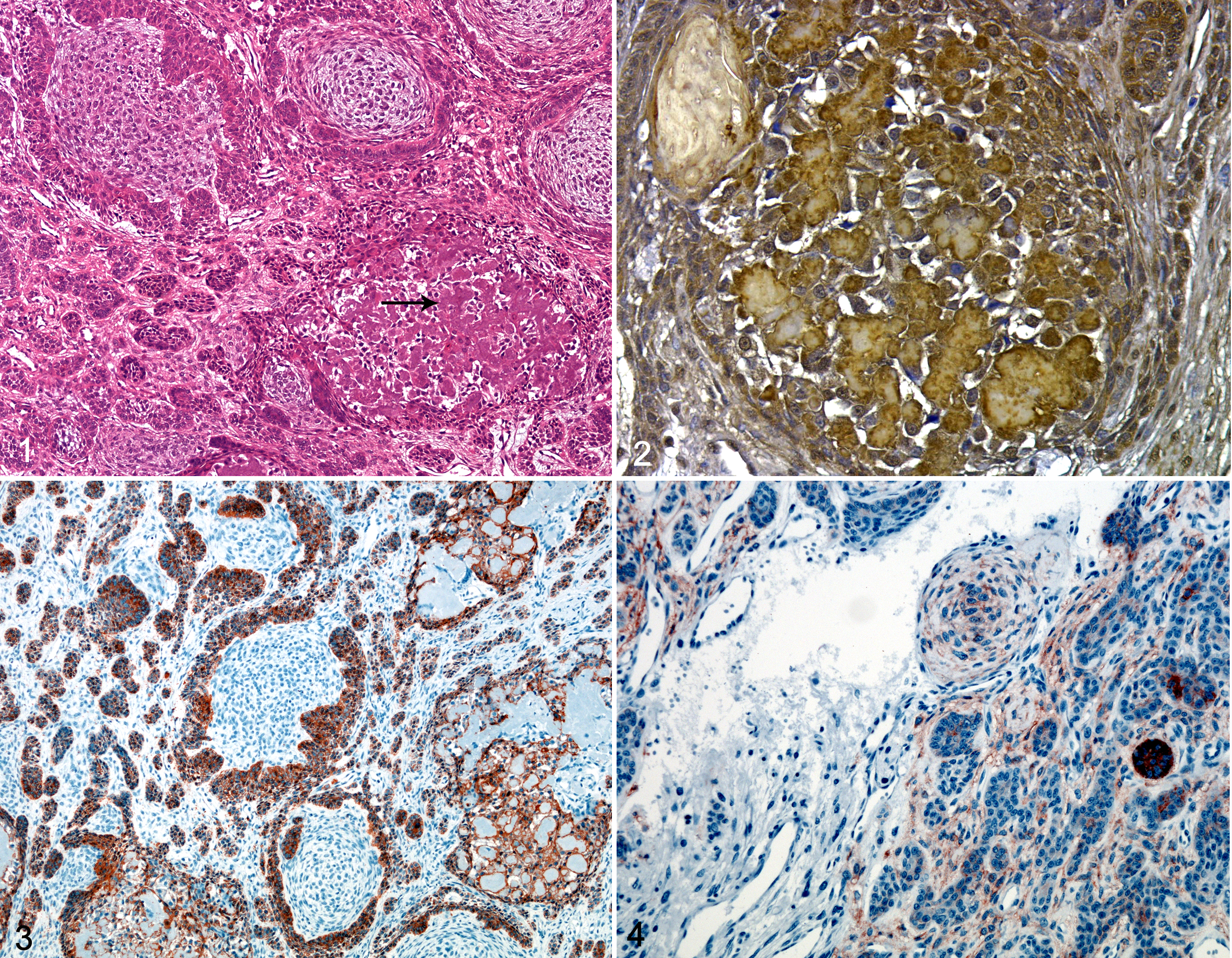
Amyloid-producing odontogenic tumor; cat No. 2. Irregularly shaped islands and bands of odontogenic epithelium with palisading peripheral cells and amyloid deposits (arrow). HE.
Immunohistochemical results are summarized in Table 2. For all antibodies, immunoreactivity was obvious in the positive control sections but not detected in the negative controls. The antiameloblastin antibody labeled the neoplastic odontogenic epithelium and the amyloid in all 3 APOTs (Fig. 2). Neoplastic cells in all 3 tumors also expressed CK AE1/AE3, CK14 (Fig. 3), and CK19; 2 of 3 APOTs had focal strong immunoreactivity to NGFR antibodies (Fig. 4). Antibody to laminin weakly labeled the amyloid and stromal cells adjacent to the basement membrane in all cases. Calretinin, amelogenin, S100, and GFAP antibodies did not label neoplastic epithelium or amyloid in any APOT.
Immunohistochemical Reactivity in 3 Feline Amyloid-Producing Odontogenic Tumors.
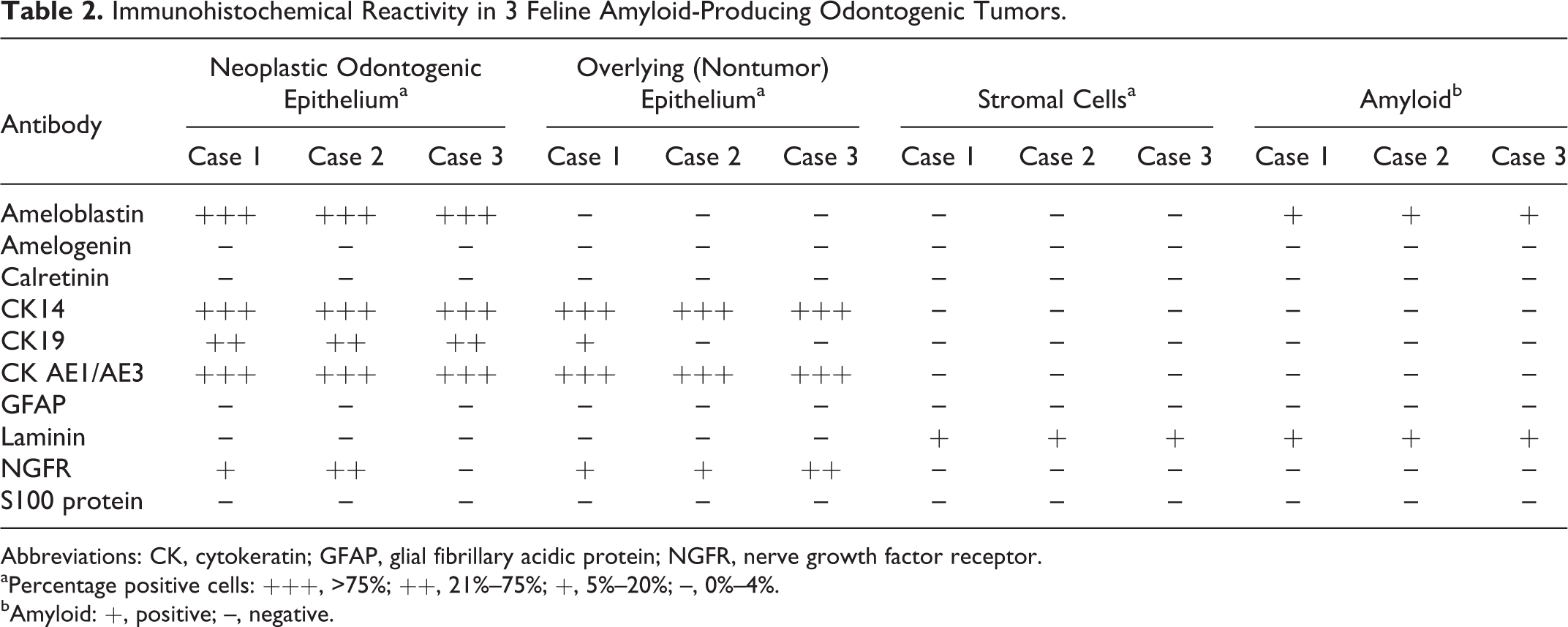
Abbreviations: CK, cytokeratin; GFAP, glial fibrillary acidic protein; NGFR, nerve growth factor receptor.
aPercentage positive cells: +++, >75%; ++, 21%–75%; +, 5%–20%; –, 0%–4%.
bAmyloid: +, positive; –, negative.
Passage of the reduced/alkylated amyloid extracts through reverse-phase high-pressure liquid chromatography columns yielded, in each case, a major protein peak eluting at an acetonitrile concentration of approximately 30% to 45%. After trypsin digestion, tandem mass spectrometry analyses of the resultant peptides detected, in all 3 tumors, an approximately 55-residue component corresponding to the N-terminal portion of ameloblastin. In contrast to human CEOTs, no ODAM-related peptides were detected.
Discussion
The ameloblastin peptide detected by mass spectrometry of the amyloid extracted from all 3 feline APOTs was identical to that found in APOTs from a cat, dog, and tiger and designated AAmel. 3 The ameloblastic nature of the amyloid was confirmed immunohistochemically. Ameloblastin, formerly known as sheathlin, is an enamel matrix protein produced by ameloblasts during the secretory stage through the early maturation stage of amelogenesis, and it is critical for maintaining the ameloblast differentiation state and enamel formation. 4 It is a tooth-specific glycoprotein, localized near the ameloblast cell surface, in contrast to the other important enamel protein, amelogenin, which is expressed throughout the enamel layer but not around the cell surface. 4
The presence of an ameloblastin peptide in these feline APOTs is consistent with the finding of a similar enamel protein in an APOT from a Shih Tzu dog, in which the amyloid, analyzed via SDS-PAGE, was composed of an NH2-terminal amino acid sequence similar to both rat ameloblastin and porcine sheathlin. 6 In that study, the APOTs from the Shih Tzu and 8 other dogs, as well as developing teeth from 3 canine fetuses, were characterized immunohistochemically. Neoplastic epithelial cells and amyloid from the canine APOTs, as well as the immature enamel matrix and ameloblast cytoplasm from the canine fetal teeth, all were immunoreactive to rat ameloblastin, porcine amelogenin and sheathlin, and an anticanine APOT amyloid-specific polyclonal antibody developed for the study. 6 The neoplastic cells were also positive for CK AE1/AE3, CK9, and CK14. 6
The neoplastic epithelium of the feline APOTs in this study was immunoreactive to some epithelial-specific antibodies, including CK AE1/AE3, CK14, and CK19, in addition to human ameloblastin. However, neither the amyloid nor the neoplastic epithelium of the feline APOTS had immunoreactivity for amelogenin. Amelogenin is also secreted by ameloblasts during early stages of amelogenesis but in different quantities than ameloblastin both spatially and temporally. Amelogenin constitutes more than 90% of the developing enamel matrix and is considered important for maintenance of the root surface. 4 Furthermore, ameloblastin may indirectly regulate the expression of amelogenin. 4 Immunoreactivity to amelogenin antibodies has been reported in several cell types of developing teeth (ameloblasts, odontoblasts, and cementoblasts).6,9 The lack of immunoreactivity to amelogenin in the tumors in this study may signify a species difference or technical issue with the antibody. However, APOT neoplastic cells could represent ameloblasts at various stages of amelogenesis because the expression of amelogenin depends on the differentiation stage of ameloblasts. 9
Calretinin was proposed as a potential marker of odontogenic epithelium 9 but was not expressed by any tumor or overlying epithelial cells in the current study. The discrepancy in immunohistochemical expression of calretinin and that of amelogenin may reflect variation among individual tumors.
Nerve growth factor receptor has been utilized in studies of rat teeth development, injury, and aging and odontogenesis in humans. 7 Nel et al proposed that NGFR expression might distinguish odontogenic epithelium in early developmental stages from nonodontogenic epithelium because immunoreactivity was limited to the internal enamel epithelium (IEE) of developing dog teeth. 9 NGFR had focal, strong immunoreactivity in tumor epithelium in 2 of 3 APOTs in the current study. Though these cells could have been entrapped nonneoplastic IEE, their cytologic features and location were typical of neoplastic cells. This finding further supports the hypothesis that the neoplastic epithelium of APOTs is of ameloblastic origin. However, NGFR immunoreactivity indicates variation among tumors and that some neoplastic cells may represent ameloblasts in an earlier stage of differentiation (eg, IEE and preameloblast).
In this study, tumor amyloid and some adjacent stromal cells were labeled with laminin antibodies, as in another feline APOT. 1 Laminins, along with type IV collagen and integrin-α6, are in the dental basement membrane and matrix during early tooth development, including the presecretory stage. 4 Thus, the presence of laminins in the amyloid of odontogenic tumors suggests that their histogenesis is linked to ameloblast differentiation and, in some tumors, during the early stages of amelogenesis.
CK14 and CK19 are type 1 intermediate filaments of epithelial cells. 2 Though CK14 is the main intermediate filament of odontogenic epithelium, it is not specific and, along with CK19, is also expressed in inner and outer enamel epithelium, cells from the stellate reticulum, stratum intermedium, the dental lamina, and Serres rests of the developing tooth.2,9
In summary, these results indicate that ameloblasts give rise to APOTs and produce the tumor amyloid. Extracted amyloid peptide sequences of the feline APOTs were nearly identical to those from other APOTs and, unlike human CEOTs, did not contain ODAM. Immunohistochemical findings provided evidence for the presence of neoplastic ameloblasts in both secretory and presecretory stages, which suggests that APOT histogenesis is closely related to amelogenesis.
Footnotes
Acknowledgements
We thank Renee Walker and the histology staff of the University of Illinois Veterinary Diagnostic Laboratory, Sallie Macy of the University of Tennessee Human Immunology and Cancer Research Program, and Marlene van Heerden of the University of Pretoria Department of Oral Pathology and Oral Biology for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
