Abstract
Disease monitoring of amphibian assurance populations is an important buffer against ongoing global extinctions. This study documents a high incidence of neoplasia in a zoo-managed assurance population of Puerto Rican crested toads (Peltophryne lemur; PRCTs). Over 5 years, neoplasia was diagnosed in 17/49 (35%) submitted adult PRCTs and was the cause of death or euthanasia in 13/17 (72%). Most toads were male (16/17; 94%) and 6 to 11-years-old (average 8.1 years). Notably, seven toads (41%) had multiple neoplasms. Of the 29 neoplasms identified, 17 (59%) were cutaneous or subcutaneous. The most common neoplasms included mast cell tumors (MCTs; 8/29; 28%), histiocytic sarcomas (6/29; 21%), lymphoma/leukemia (4/29; 14%), and squamous cell carcinomas (3/29; 10%). Distant metastases were documented in 6/8 (75%) toads with MCTs. Causes for neoplasia in this population were not determined though may include genetic or environmental factors. Continued investigations of managed endangered amphibians will help elucidate mechanisms of carcinogenesis.
Puerto Rico’s only native toad, the Puerto Rican crested toad (Peltophryne lemur; PRCT) is listed by the International Union for Conservation of Nature (IUCN) as critically endangered due to habitat loss and competition with invasive species.3,13 A key element to conservation is managed breeding of assurance populations, and disease monitoring optimizes the success of released offspring in the wild. 16 Current knowledge of neoplasia in PRCTs is limited to a single case report of a cutaneous mast cell tumor (MCT). 15 This study highlights the high incidence of neoplasia within an indoor population of approximately 160 toads housed at a zoological institution.
Over a 5-year period (2017–2022), neoplasia was diagnosed in 17/49 (35%) of adult PRCT biopsy and necropsy submissions from this assurance population. The average age at diagnosis was 8.1 years (range 6–11 years), and most toads were male (16/17; 94%). Neoplasia was diagnosed as the cause of euthanasia or spontaneous death for 13/41 (32%) of all submitted adult PRCT mortalities. Six individuals were biopsied prior to death, and there was an average interval of 274 days between biopsy and death or euthanasia. The skin and subcutis were the most common sites for neoplasms (17/29; 59%).
Toads were housed in groups of ≤6 in terraria with raised false bottoms, halogen heat lamps, and seasonally variable temperatures of 66°F–85°F. 14 Before October 2018, PRCTs were housed in glass terraria under 2 bulb fixtures of ZooMed Reptisun T8 10.0 UVB with a 12-inch light-to-platform distance. Henceforth, a subset of toads were housed in either glass or plastic terraria under 2 separate bulb fixtures (1 ZooMed Reptisun T5 10.0 HO Linear Lamp and 1 SPECTRLUX 6500°K “blue” T5 HO Lamp) with a 9–20-inch light-to-platform distance, dependent on terrarium size. The remaining toads were housed in glass terraria under 2 separate bulb fixtures (1 ZooMed Reptisun T5 10.0 HO Linear Lamp and 1 SPECTRLUX 6500°K “blue” T5 HO Lamp) with a 14-inch light-to-platform distance and separated by a screen layer (Phifer PetScreen or TuffScreen). As of March 2021, all ZooMed Reptisun lights were reduced from 10.0 to 5.0 HO. Lighting cycles were adjusted seasonally.
Necropsies were performed by veterinary staff. Biopsy specimens and whole dissected carcasses were submitted in 10% neutral buffered formalin to the Zoological Pathology Program, University of Illinois. Representative sections of any masses, skin/subcutis/muscle, epaxial skeletal muscle, brain, eyes, tongue, esophagus, stomach, intestine, liver, pancreas, lung, heart, gonad, Bidder’s organ (male), kidneys, and spleen were routinely processed. Additional histochemical stains and immunohistochemistry were employed according to the manufacturer’s instructions (Table 1, Supplemental Table S1) and included Giemsa, toluidine blue (TB), periodic acid-Schiff (PAS), anti-tryptase (mast cell), anti-stem cell factor receptor (c-kit, CD117), and anti-IBA1 (ionized calcium-binding adapter molecule). 18 Ziehl-Neelsen stains were performed on all round cell neoplasms to exclude infectious disease (eg, mycobacteriosis and microsporidiosis).
Histochemistry and immunohistochemistry results of mast cell tumors and histiocytic sarcomas in a population of Puerto Rican crested toads (Peltophryne lemur).
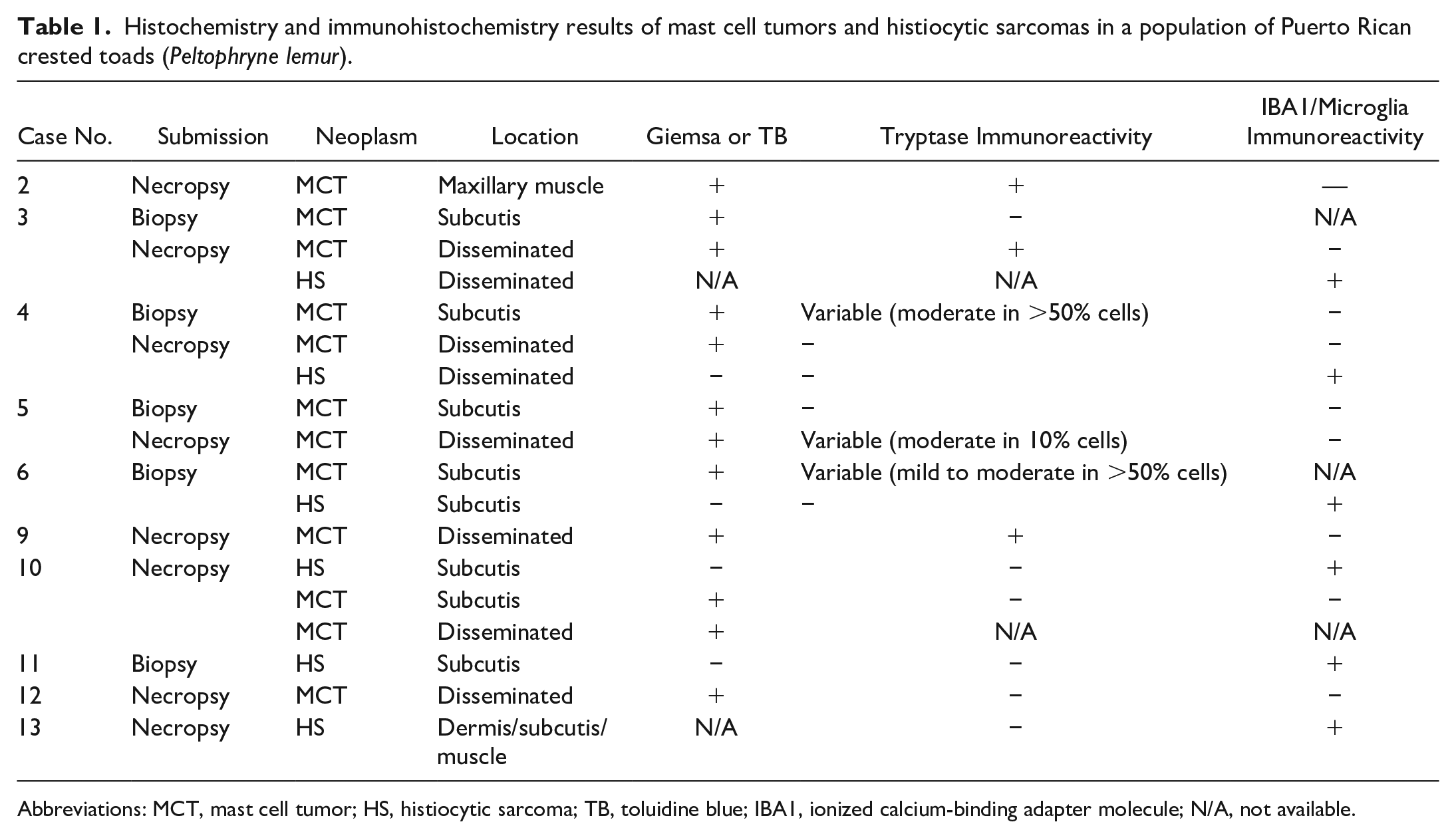
Abbreviations: MCT, mast cell tumor; HS, histiocytic sarcoma; TB, toluidine blue; IBA1, ionized calcium-binding adapter molecule; N/A, not available.
Notably, 7 of 17 toads (41%) had multiple neoplasms diagnosed via biopsy and/or necropsy, and 29 total neoplasms were detected. Up to 5 separate neoplasms were noted in a single toad. The most common neoplasm was MCT (8/29, 28%), followed by histiocytic sarcoma (HS; 6/29, 21%), leukemia/lymphoma (4/29; 14%), and squamous cell carcinoma (3/29, 10%). Other neoplasms included cutaneous papilloma (2), Bidder’s organ dysgerminoma (2), seminoma (1), presumed biliary carcinoma (1), hepatocellular carcinoma (1), and undifferentiated subcutaneous sarcoma (1).
Most MCTs were solitary, 0.4–1 cm in diameter, well-demarcated, smooth, raised, soft, pink, and nonulcerated subcutaneous or dermal neoplasms with variable anatomic locations (Fig. 1a). One toad presented with diffuse unilateral forelimb swelling and pallor. Dissemination was present in all but 1 case (7/8) at the time of necropsy. In 1 presumed metastatic MCT case, neoplastic cells were limited to the maxillary skeletal muscle and the primary neoplasm was not detected. In cases of more widespread dissemination (6/8), metastases were most frequent in the kidney (4/6) and liver (3/6) and appeared grossly as white to red, raised nodules. Histologically, MCTs were either well-demarcated, unencapsulated, nodular masses with variable cellular morphology and pattern or, in advanced or metastatic disease, poorly demarcated and invasive. Heterogeneous morphologies and patterns were common (5/8) and characterized by round to spindloid cells arranged in sheets, streams, bundles, and/or whorls (Fig. 1b). Neoplastic cells had eosinophilic, homogenous to less commonly granular cytoplasm and central, small, oval nuclei with coarse chromatin. Few MCTs (3/8) and most metastases were composed of sheets of large, monomorphic round cells (Fig. 1c). Mitoses were rare. No granulocyte (eosinophil) recruitment was observed. Notably, 4 of 6 MCTs were immunoreactive for tryptase, but the proportion of reactive cells and staining intensity results were highly variable (Fig. 1d, Supplemental Fig. S1, Table 1). Stem cell factor receptor (c-kit, CD117) immunohistochemistry was performed on 1 case, and both neoplastic mast cells and testicular germ cells (internal control) did not exhibit appropriate immunoreactivity (Supplemental Fig. S2). 17 MCTs had no immunoreactivity to IBA1. Metachromatic staining highlighted fine cytoplasmic granules in most neoplastic cells regardless of cell morphology (Fig. 1b, c, inset).
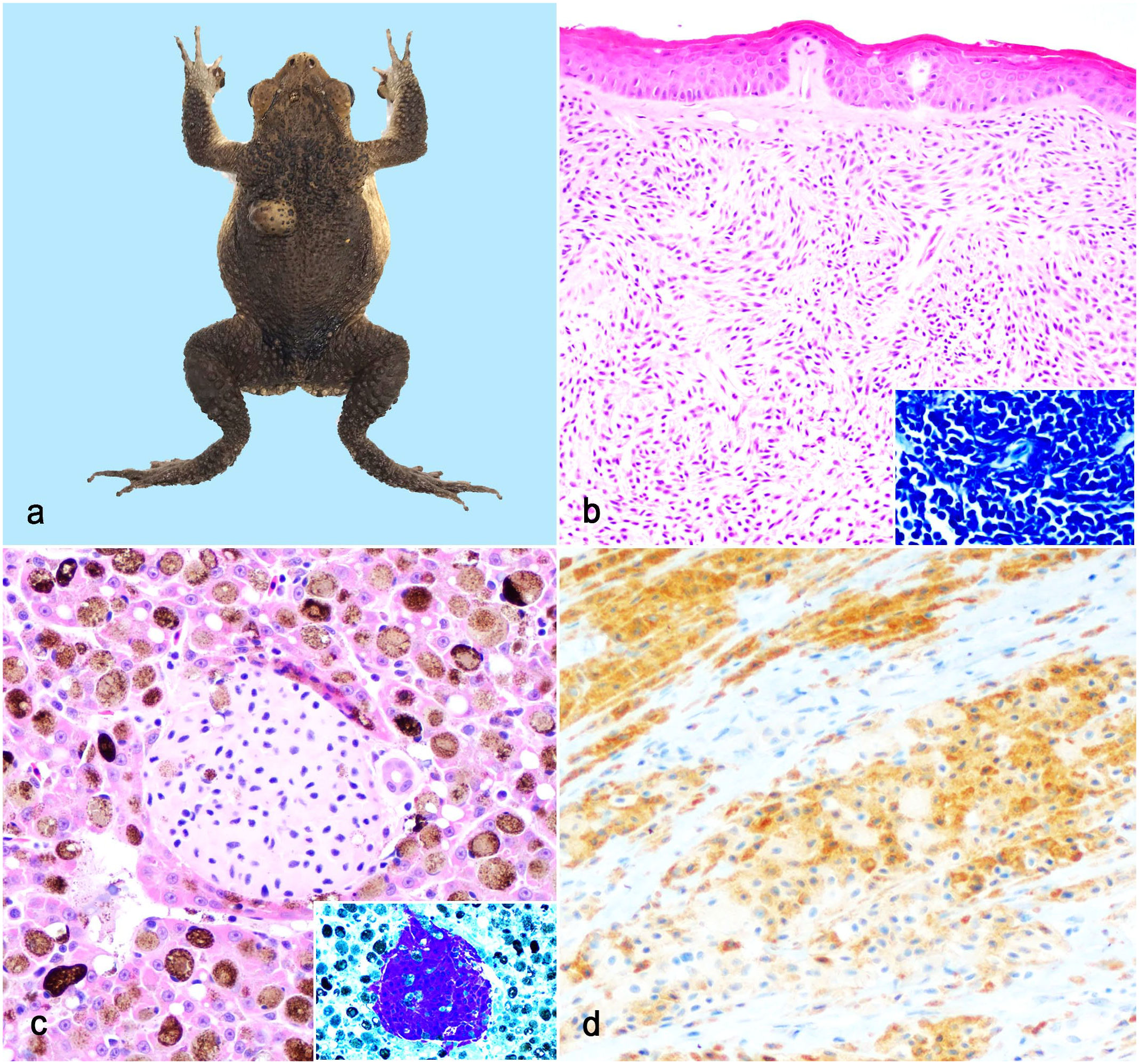
Puerto Rican crested toad (Peltophyrne lemur), dermal and metastatic mast cell tumors. (a) Mast cell tumor on the left dorsum appearing as a well-demarcated, smooth, raised, nonulcerated mass. (b) Neoplastic cells range from spindloid (superficial) to round (deep). Hematoxylin and eosin (HE). Cytoplasmic granules stain with Giemsa (inset). (c) Metastatic mast cells in a hepatic vessel. HE. Inset: cells also have strong Giemsa staining. (d) Cells have variable immunoreactivity to antibodies against tryptase. Tryptase immunohistochemistry.
HSs were diagnosed via biopsy as solitary deep dermal/subcutaneous masses or at necropsy as one of multiple visceral masses. Dermal/subcutaneous HS were generally less than 1 cm in diameter, soft, raised, and variably ulcerated swellings. Visceral masses were white to red, raised nodules in the liver (2/2), lung (1/2), or kidney (1/2). Histologically, most HS were well-demarcated, unencapsulated, nodular masses composed of round cells arranged in sheets (Fig. 2a). Neoplastic cells had variably distinct margins and wispy to granular eosinophilic cytoplasm. Megalocytosis and multinucleation were identified in 1 neoplasm, and mitoses were rare. Definitive diagnosis of HS was based on strong cytoplasmic immunoreactivity with antibodies against IBA1 (Fig. 2b, c, inset, Supplemental Fig. S3) and lack of immunoreactivity to tryptase (Fig. 2b, inset). Resident macrophages within the surrounding tissue served as an internal control. Cytoplasmic granules were not detected in metachromatic stains. In 1 subcutaneous HS, there was an invasion into the subjacent skeletal muscle (Fig. 2c). One HS appeared to develop near a previous passive integrated transponder tag placement site with variable inflammation.
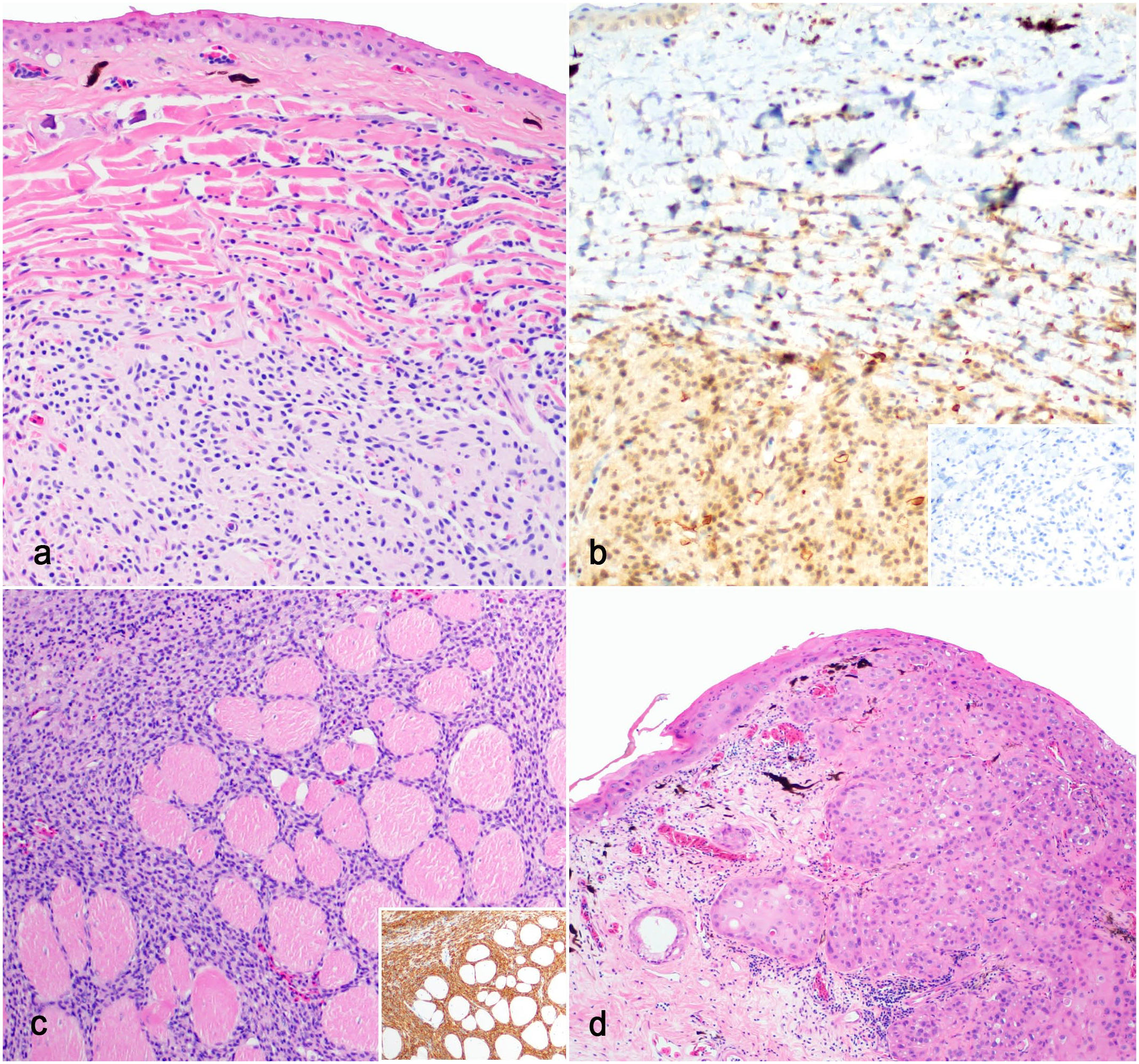
Puerto Rican crested toad (Peltophyrne lemur), histiocytic sarcomas, and squamous cell carcinoma. (a) Histiocytic sarcoma characterized by neoplastic round cells within the deep dermis. Hematoxylin and eosin (HE). (b) Neoplastic cells have weak to moderate cytoplasmic immunoreactivity to antibodies against IBA1. Inset: cells have no immunoreactivity to antitryptase antibodies. IBA1 and tryptase immunohistochemistry (IHC). (c) Hindlimb histiocytic sarcoma with invasion of skeletal muscle. HE. Inset: cells have strong immunoreactivity to anti-IBA1. IBA IHC. (d) Squamous cell carcinoma arising from the epidermis with invasion into the dermis. HE.
Leukemia/lymphoma were multicentric processes affecting most examined organs. Neoplastic lymphocytes filled vessels and sinusoids and/or formed poorly demarcated, unencapsulated masses composed of dense sheets and rows. Cells were distinct and round with scant cytoplasm and small (2/4), intermediate (1/4), or large (1/4) hyperchromatic nuclei. Mitoses were rare. Epitheliotropism was noted in the intestine, skin, and lung in 1 case of large-cell lymphoma. CD3 and CD79a immunohistochemistry were performed in 1 case of small-cell lymphoma; neoplastic cells were not immunoreactive, and no internal control was available.
Squamous cell carcinomas were solitary, locally invasive, less than or equal to 2 cm in diameter, pink, irregular, ulcerated cutaneous masses. Visceral metastases were not detected. Postsurgical recurrence was documented once. An in situ squamous cell carcinoma was diagnosed in the abdominal skin of 1 toad. Histologically, squamous cell carcinomas were poorly demarcated and exhibited characteristic features, including abrupt squamous maturation, dyskeratosis, keratin pearls, and prominent intercellular bridges (Fig. 2d). A single undifferentiated subcutaneous sarcoma appeared histologically similar to the HS and MCT cases described here but did not stain or label with Giemsa, tryptase, and IBA1. All other listed neoplasms had characteristic features like those described in other species; the Bidder’s gland dysgerminomas closely resembling ovarian dysgerminomas described in a frog species. 9
Breeding of PRCTs in Association of Zoos and Aquariums-accredited facilities has successfully maintained adequate genetic diversity compared with wild populations, and genetic predisposition to neoplasia has not been documented. 4 High incidences of neoplastic disease have been previously reported in anuran assurance populations, notably peripheral nerve sheath tumors in Houston toads. 12 Thus, the high incidence of MCT and HS in this population of PRCTs was an interesting and important finding. Previous reports of amphibian MCTs included 1 case report in a PRCT and 2 case series in populations of laboratory axolotls (Ambystoma mexicanum) and wild tiger salamanders (Ambystoma trigrinum).10,15 Inbreeding was suspected in both affected caudate populations. 14 The previous PRCT MCT case report had no histologic evidence of malignancy, and this case series includes the first documentation of deep invasion and metastasis of MCT in an anuran. 10
Brown skin disease (BSD) was a common comorbidity in this population, and 13/17 (76%) individuals with neoplasia were either historically or currently affected at the time of submission. Of the 32 submitted cases without evident neoplasia, 10/32 (31%) individuals had current or historic BSD. The overall incidence of BSD in all submitted toad specimens was 23/49 (47%). This idiopathic disease of PRCT assurance populations is characterized by dysecdysis, brown discoloration, and hyperkeratosis with variable ulceration and opportunistic environmental bacterial and/or fungal infections. 6 There is no current evidence to support an infectious etiology. Investigation of an association between BSD and neoplasia should be included in future studies of PRCT assurance populations.
Mast cells have been described in all major vertebrate taxa, in which they exhibit some heterogeneity in morphology and distribution. 2 The morphology of mast cells in amphibians is diverse, from round to spindloid, and there are species-specific differences in their distribution and tissue concentration. 2 This heterogeneity was reflected in MCT populations of this case series. In anurans, mast cells are often observed in close association with nerves, blood vessels, and melanocytes.2,5,7 The spindle cell morphology of anuran MCTs and close association with nerves and blood vessels may lead to inaccurate diagnosis of soft tissue sarcomas. These MCTs required metachromatic stains for visualization of cytoplasmic granules, supporting the previous suggestion by Stacy and Parker 19 that staining is required to confirm or refute mast cell histogenesis in amphibian round and spindle cell tumors. 19 Tryptase antibodies were variably immunoreactive with PRCT mast cells, and the diagnostic value of this immunohistochemistry assay may be limited in this species without concurrent metachromatic stains. Stem cell factor receptor (c-kit, CD117) performed on 1 toad had inappropriate immunoreactivity.
Known previous reports of HS in amphibians are limited to a single experimental study, and this is the first report of naturally occurring HS in an amphibian. 8 Two of the subcutaneous HS developed at or near passive integrated transponder tag sites where there were mixed inflammatory cells, suggesting a possible role of inflammation in carcinogenesis. Pleomorphism characteristic of HS in mammalian species was lacking in the majority of PRCT HS. Both resident tissue and neoplastic macrophage antigens had strong cytoplasmic immunoreactivity to IBA1; thus, this antibody was useful in confirming histiocytic histogenesis in this species.
This case series highlights a high incidence of neoplasia in an assurance population of PRCTs, with a preponderance of MCT and HS. A cause was not determined; however, these tumors are linked to genetic mutations; thus, further investigation is needed.1,20,21 Although most of these individuals were housed in different terraria, the similar environment between individuals is a potential contributing factor. As most neoplasms were cutaneous and/or subcutaneous, other considered factors included UV-induced damage, BSD, and passive integrated transponder (PIT) tag-associated inflammation. High levels of exposure to UVB rays are known to contribute to tumorigenesis. Histologic evidence of solar dermatopathy (eg, solar elastosis) was not observed in these individuals; however, exposure to strong UVB radiation was a potential contributor in this population. Lesions secondary to chronic or severe BSD could have obscured more subtle pre-existing lesions in some of the affected toads. Any association with UV radiation remains undetermined in this idiopathic disease.
The scope of this study was limited to 1 of over 30 American zoological institutions that house PRCTs. Continued surveillance and disease investigations of this and the other PRCT assurance colonies are critical to better understand the seemingly widespread prevalence and pathophysiology of neoplasia in this endangered amphibian.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231214029 – Supplemental material for Neoplasia in an assurance population of Puerto Rican crested toads (Peltophryne lemur)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231214029 for Neoplasia in an assurance population of Puerto Rican crested toads (Peltophryne lemur) by Sierra M. Imanse, Caitlin E. Burrell, Sarah A. Cannizzo, Tara S. Reilly, Kimberly L. Rainwater and Martha A. Delaney in Veterinary Pathology
Footnotes
Acknowledgements
The authors wish to thank the husbandry and veterinary staff of Fort Worth Zoo for their contributions to this study as well as their leadership in developing and implementing new conservation strategies for Puerto Rican crested toads. We also wish to thank Renee Walker, Tracy Baine, Carrie Scheiter, and histology laboratory personnel at the University of Illinois Veterinary Diagnostic Laboratory for histology and immunohistochemistry services. Finally, thanks to the Zoological Pathology Program faculty and resident pathologists for the initial evaluation of the cases.
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
