Abstract
This study describes the clinical, gross, and histologic findings in 17 cases of aneurysms in bearded dragons (Pogona vitticeps). The clinical presentation ranged from incidental to sudden and unexpected death. The affected vasculature was predominantly arterial; however, based on the topographical locations of the lesions, gross structure, and drainage, some veins were likely involved. Magnetic resonance imaging and computerized tomography scans of 1 animal showed a large aneurysm of the internal carotid artery extending from near its aortic origin into the caudal head. Aneurysms were organized in 5 groups based on their anatomical locations: cephalic, cranial coelom (for all near the heart), caudal coelom (for the mesenteric vessels and descending aorta), limbs, and tail. The cranial coelomic region was the most prevalent location. Gross findings were large hematomas or red serosanguineous fluid filling the adjacent area, as most of the aneurysms (94%) were ruptured at the time of the study. The main histological findings were degenerative changes of the vessel walls characterized by moderate to severe disruption of the collagen and elastic fibers of the tunica media and adventitia (100%), followed by thickening of the intima with thrombi formation (54%) and dissecting hematoma of the vessel wall (47%). Vasculitis (29%), mineralization (6%), and lipid deposits (6%) in the vessel wall were observed occasionally. Based on these findings, the vascular dilations and ruptures observed in bearded dragons likely are associated with weakness of the vessel walls caused by degenerative changes in the intimal and medial tunics.
Bearded dragons are lizards of the genus Pogona in the family Agamidae. The most common species in the pet trade is the inland (or central) bearded dragon (Pogona vitticeps). 4 This common pet species is derived from a small founder population imported into Germany several decades ago; captive bred descendants became increasingly common in the pet trade across continents. 17 The small founder population restricted genetic diversity and inevitably resulted in inbreeding in the pet population. Negative outcomes from inbreeding may be inferred but are not cataloged.
Aneurysms are localized dilations that may expand to more than 50% of the normal diameter of affected vessels. In some cases, dilation and rupture of cardiac wall may occur. 6 Usually, arteries are affected, especially large elastic arteries, but the lesions also can occur in veins. 10 Aneurysms result from high blood pressure and a weakening of the vessel wall caused by atherosclerosis, medial degeneration (including congenital collagen anomalies), trauma, infection, or arterial dissection.6,8
On the basis of their gross appearance, aneurysms may be classified as berry, saccular, fusiform, or dissecting. 19 The term aortic dissection or dissecting hematoma are preferred over dissecting aneurysm in cases in which there is no dilatation of the vessel wall, as in a true aneurysm. 2 In a dissecting hematoma, blood enters the wall of the vessel (usually an artery) through an intimal tear, dissects between medial layers, and creates a cavity within the arterial wall. 1
Aneurysms are common in humans, and cases have also been described in mammals and birds (mostly turkeys).1,10,12,24 In reptiles, there are a few reports in lizards and snakes in additions to those described for bearded dragons.5,11,26,27 Here, we provide a case series of aneurysms in 17 inland bearded dragons (P. vitticeps) and describe the clinical, macroscopic, and histologic features.
Material and Methods
Case Selection, Clinical Data, and Pathology
A search was conducted of the case files at Northwest ZooPath from 1994 to 2021 for bearded dragon submissions diagnosed histologically with aneurysms. Case materials included formalin-fixed biopsies, 2 fresh carcasses, and necropsy tissue sets submitted by clinicians from zoological or private practices.
Clinical reports were reviewed to obtain the animals’ signalments and relevant information including clinical signs, diet, and affected vessel/location. Aneurysms were organized in 5 groups based on their anatomical locations: cephalic, cranial coelom (for all near the heart), caudal coelom (for the mesenteric vessels and descending aorta), limbs, and tail.
Tissues were processed routinely, sectioned at a thickness of 5 mm and stained with hematoxylin and eosin. Select blocks were stained with Brown and Brenn–modified Gram stain, Fite’s acid-fast, and Gimenez or Warthin–Starry for bacteria; Gomori’s methenamine silver and periodic acid–Schiff for fungal organisms; Luna for microsporidia; and Masson’s trichrome and Verhoeff’s elastin stains for connective tissues and smooth muscle.
Results
From 1994 to 2021, 1387 bearded dragon biopsies or necropsies were accessioned at Northwest ZooPath. Aneurysm was diagnosed in 22 cases, based on clinical, gross, or histologic findings. However, only 17 cases (3 biopsies and 14 necropsies) had histological sections of the affected vessel of sufficient quality for histologic examination and inclusion in this study. Signalment, clinical findings, and pathological features are summarized in Table 1. Sex was reported in 16 cases, including 10 males and 6 females. The ages of the affected animals ranged from 10 months to 10 years. In 3 animals, the age was not reported, and 3 cases were listed as “adults” (Table 1).
Signalment, clinical findings, and pathological features in bearded dragons with aneurysms.
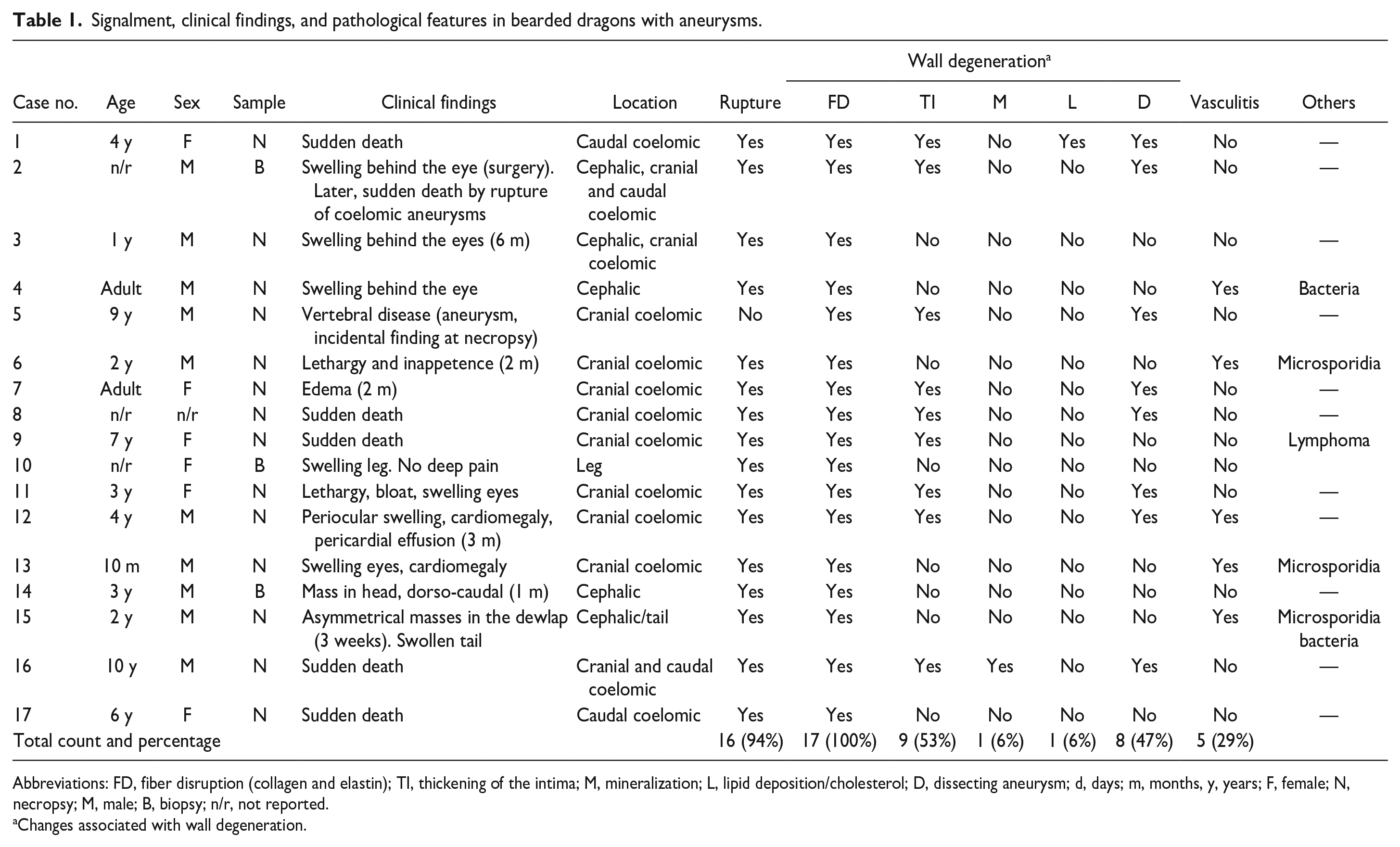
Abbreviations: FD, fiber disruption (collagen and elastin); TI, thickening of the intima; M, mineralization; L, lipid deposition/cholesterol; D, dissecting aneurysm; d, days; m, months, y, years; F, female; N, necropsy; M, male; B, biopsy; n/r, not reported.
Changes associated with wall degeneration.
Case 2 had diagnostic imaging (magnetic resonance imaging and computed tomography) of a cephalic swelling prior to a surgical repair (Fig. 1a–d). Two contiguous areas of swelling consistent with the right lateral head vein, right transverse and dorsal sinuses, temporo-orbital artery, and, possibly, the internal carotid artery were observed following injection of contrast solution into the ventral abdominal vein. The swellings were constrained dorsally by the skull’s superior temporal fenestra and posterolaterally by the postorbital and jugal bones.
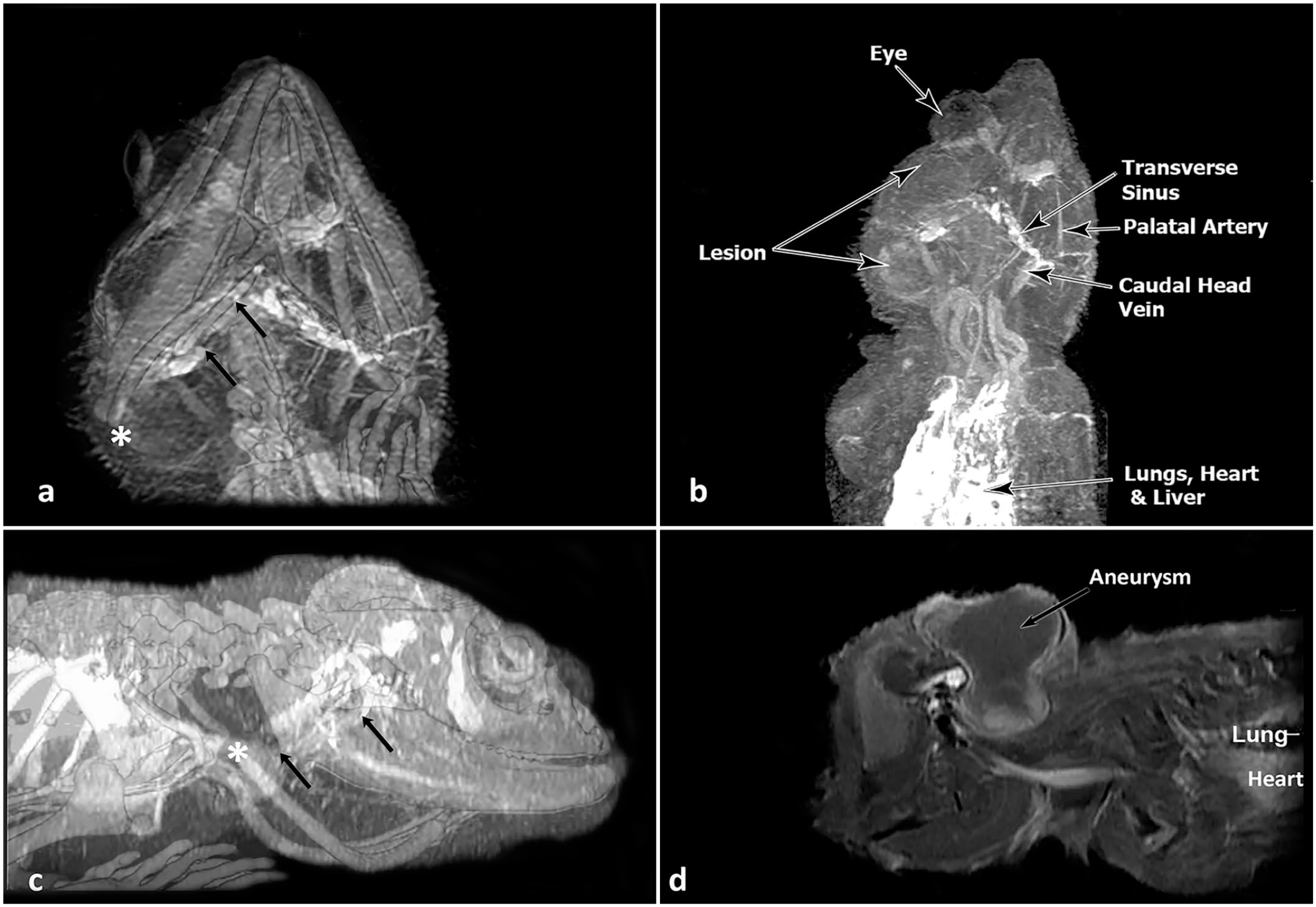
Computed tomography (CT) and magnetic resonance imaging (MRI) of a live bearded dragon with a cephalic aneurysm on the right side of the head. (a) Ventrolateral view of superimposed CT image with MRI overlay showing the right-side cephalic swelling. The lack of blood flow in the transverse sinus between the black arrows shows the effect of this lesion. The asterisk marks the distal end of the right ceratobranchial element of the hyoid apparatus, that is displaced laterally due to the large aneurysm. (b) Ventrodorsal MRI image with vascular contrast. Note that blood flow to the right side of transverse sinus, orbital sinus, and palatal artery is reduced compared with the lizard’s left side. Exophthalmos is observed. (c) Lateral view of the superimposed CT image with MRI overlay. Blood flow in the right transverse sinus is absent between the black arrows due to the lesion. Above the arrows, the large (white) transverse sinus from the left side (nonaneurysm side) provides perspective. The asterisk shows the lateral displacement of the ceratobranchial element. (d) MRI image of the head and anterior trunk without contrast. The aneurysm resulted in cephalic swelling that extended dorsally from the superior temporal fenestra, caudally from the post-temporal fossa, and ventrolaterally. The partially imaged horizontal vessel caudal to the head likely is the internal carotid artery. The ventral lung and distal heart are partially visible.
The clinical presentation was variable among the affected animals (Table 1). In 5 cases, the lizards were found dead with no prior signs of illness. Eight animals had head swelling with or without exophthalmia (Fig. 2a, b), 1 had chronic edema, and the aneurysm was an incidental postmortem finding in 1 animal.
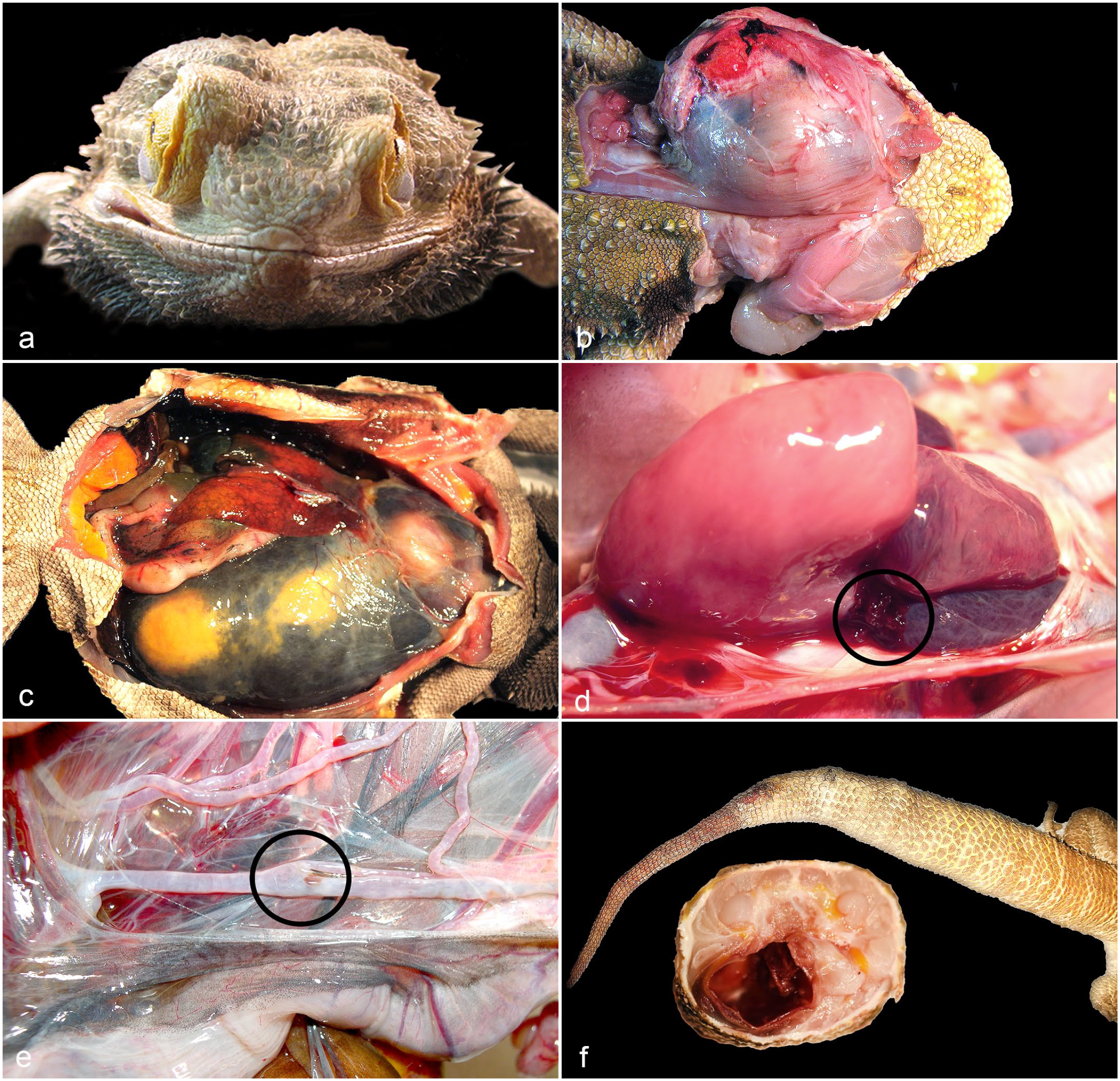
Gross lesions of ruptured aneurysms in bearded dragons. (a) Marked exophthalmia of the right eye. Case 3. (b) Dorsal view of a large focal hematoma located behind the left eye that extended within the superior temporal fenestra and caudally from the post-temporal fossa deep to the dorsal musculature. Case 4. (c) Large hematoma at the heart base that extends into the mesentery. Case 6. Image courtesy of Dr Boyer. (d) Lateral view of the ruptured sinus venosus and proximal left precava (circled) shown with the ventricle and left atrium rotated ventromedially. Case 2. (e) Branch of the anterior gastric artery with a focal tear (circled) following aneurysm rupture. Case 2. (f) The proximal portion of the tail is irregularly swollen. The transverse section shows a large, focal, cavitated area with hemorrhage. Case 15. Image courtesy of Dr Rolfe.
Table 1 summarizes aneurysms by their anatomical location. Three bearded dragons had aneurysms in 2 locations and 1 had aneurysms in 3 different regions. The cranial coelom, near the heart base, was the most commonly affected region with 11 aneurysms (Fig. 2c, d), followed by the cephalic region with 5 (Fig. 2a, b). Four aneurysms were in the caudal coelom (Fig. 2e), and 1 aneurysm each was located in the leg and the tail (Fig. 2f).
Because most of the aneurysms were ruptured at the time of the study, the main gross findings were hematoma formation and/or marked hemorrhage associated with rupture of the vessel. The cranial coelomic lesions caused hemopericardium (Fig. 2c, d). When caudal coelomic aneurysms occurred, the extravasated blood extended into the mesentery and retroperitoneal spaces.
Histologically, the main pathological finding observed in all lizards was degenerative changes of the vascular walls (100%) (Table 1). Affected walls had moderate to severe disruption of the collagen and elastic fibers of the tunica media and adventitia, which was most evident with trichrome and elastin staining (Fig. 3a–c). The affected vessels were dilated or ruptured and had thrombi composed of laminated accumulations of viable and degenerative erythrocytes admixed with fibrin attached to the endothelium (Fig. 3d). Segmental to circumferential plaque-like thickening of the intima was observed in 53% of the lizards (Fig. 3e). Dissecting hematoma, characterized by focally extensive hemorrhage that dissected between the tunica intima and tunica media or within the tunica media, separating bundles of smooth myofibers and creating a cavity within the vessel wall, was also a common finding (47%) (Fig. 3f). Mineralization (6%) and lipid deposits (6%) in the vessel wall were observed only occasionally. The only bearded dragon that presented with an aneurysm diagnosed without rupture (case 5) had advanced arteriosclerosis and an early dissecting hematoma of the great vessels at the heart base.
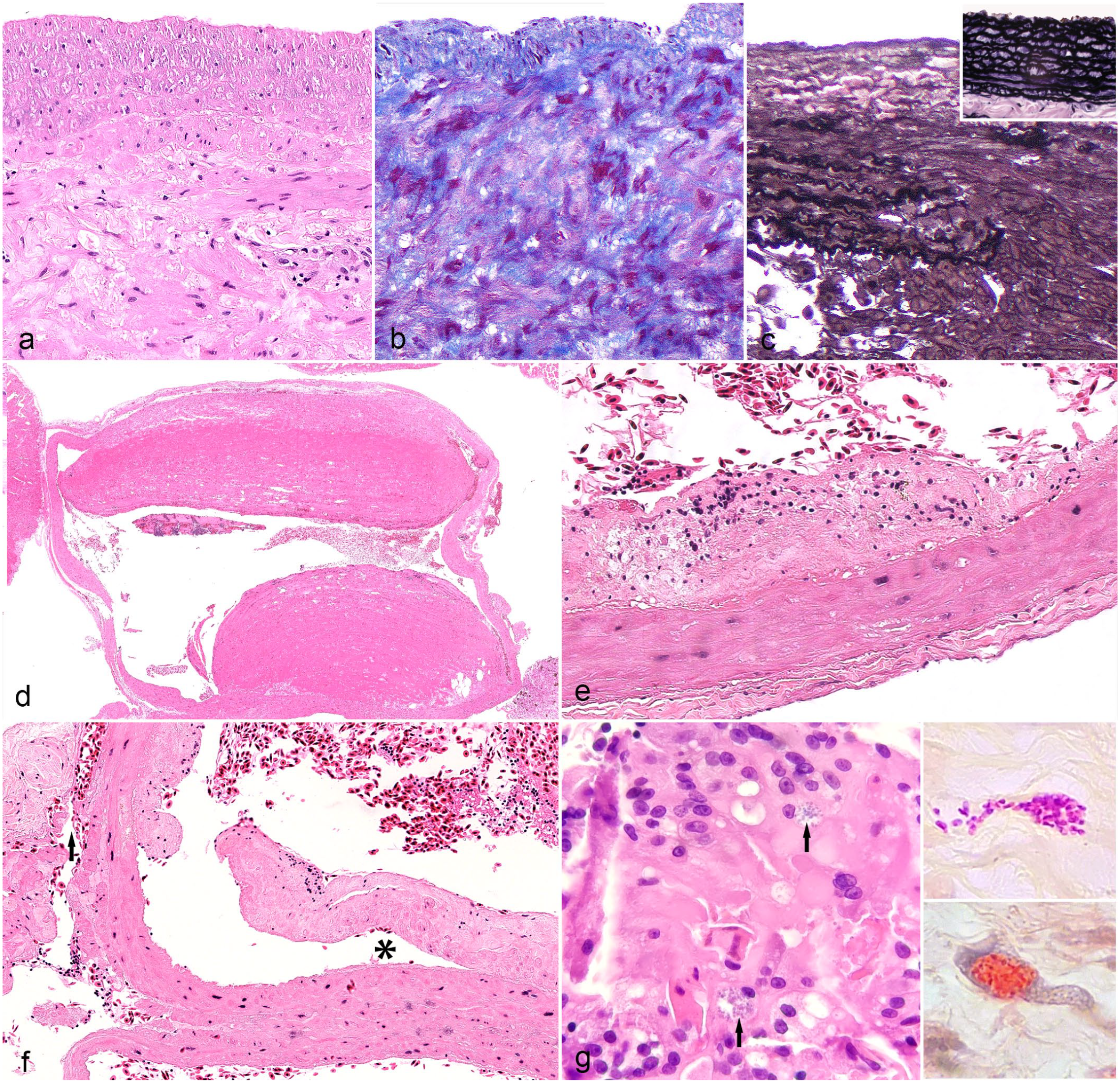
Histological lesions of aneurysms in bearded dragons. Degenerative changes of the vascular wall. (a) Moderate disruption of the collagen and elastic fibers of the tunica adventitia with multifocal infiltration of macrophages. Case 4. Hematoxylin and eosin (HE). (b) The trichrome stain highlights the disarrangement of the smooth muscle fibers and collagen in the vascular wall. Case 4. Masson’s trichrome. (c) The elastin stain highlights the rupture of the fibers and cavitation of the arterial wall. Inset: regular distribution of elastin fibers in a normal artery. Verhoeff’s elastin stain. Case 1. (d) Dilated artery at the heart base with 2 large thrombi attached to the endothelium. Case 11. HE. (e) Artery with segmental plaque-like thickening of the tunica intima. Case 5. HE. (f) Artery with dissection between tunica intima and tunica media (asterisk) and between tunica media and adventitia (arrow) with the presence of erythrocytes. Case 5. HE. (g) Focus of granulomatous vasculitis that shows 2 macrophages (arrows) that contain densely packed and slightly basophilic intracytoplasmic microorganisms Case 13. HE. These microorganisms are Gram-variable (superior inset) and also positive with the Luna stain (inferior inset).
Although all animals had some degree of inflammation, mostly associated with the thrombi or the dissection of the wall, severe vasculitis was observed in just 5 cases. The associated inflammation was composed mostly of heterophils, hemosiderin-laden macrophages, and few multinucleate giant cells. In cases 6, 13, and 15, few macrophages contained numerous, round to oval, 1 µm diameter intracellular microorganisms. The organisms were Gram-variable, stained with the Luna stain, and closely resembled microsporidia (Fig. 3g, inset). Unfortunately, a more accurate molecular characterization of the observed agents could not be performed. In cases 4 and 15, numerous gram-positive bacterial cocci were observed within the thrombi in the vascular lumen, but they did not extend into the vascular wall. The inflammation was predominantly heterophilic, and in case 4, a similar inflammation pattern was observed in other organs, which is consistent with sepsis. In the remaining cases, acid-fast bacteria, Chlamydia, and fungal organisms were not identified with special stains in the vascular lesions.
Discussion
In our study, all affected vessels showed varying degrees of wall degeneration, which appears to play a key role in the development of the aneurysms. The most frequent and well-known type of vascular wall degeneration is arteriosclerosis, which literally means “hardening of the arteries.” It is more fully defined as chronic arterial change consisting of stiffening, loss of elasticity, and luminal narrowing, usually from proliferative and degenerative, rather than inflammatory changes of the media and intima. 19 Arteriosclerosis is poorly documented in reptiles with only 2 cases reported in bearded dragons.22,23 In those reports, arteriosclerosis was observed at the efferent cardiac vessels, but also in many other vessels and cerebral arterioles. In those cases, plaques composed of well-vascularized fibrous connective tissue were frequently overlain by a thick layer of fibrin and few erythrocytes. Collagen fibers within the plaque were often separated by aggregates of lipid-laden macrophages, erythrocytes, and/or hemosiderin-laden macrophages. Histologic findings from those reported cases are similar to those observed in the arterial walls in our study, but the severe arteriosclerosis reported in those cases did not lead to aneurysm formation.
Congenital connective tissue disorders have been described as an important cause of aortic aneurysm in humans. 8 Marfan syndrome (MFS) is a connective tissue disorder caused by mutations in the FBN1 gene that encodes the extracellular glycoprotein fibrillin-1. 20 Marfan syndrome is characterized by variable clinical manifestations, including skeletal, ocular, and cardiovascular abnormalities. 20 Mortality resulting from aortic aneurysm, dissection, and rupture represents the most serious clinical manifestation of MFS. 18 Spontaneous dissecting aortic aneurysms resembling the MFS of humans have been reported in dogs, horses, and cattle, but there is no information about this disease in reptiles.1,7,9,14,16,25 In humans, transmission electron microscopy shows the high number of interruptions and the thick appearance of the elastic lamellae and the accumulation of abundant extracellular glycosaminoglycan-rich material. 20 Histologically, aortopathy in MFS is characterized by degeneration of the medial layer. This degeneration is marked by disarray and fragmentation of elastic fibers, as well as the accumulation of basophilic ground substance in areas that are devoid of smooth muscle cells. 20 The findings observed in the arterial walls in our study are similar to those described in mammals with MFS. However, no other lesions associated with MFS, such as lens luxation or skeletal abnormalities, were identified in the bearded dragons that underwent a complete necropsy and histological examination.
The histologic features of some aortic aneurysms involves medial degeneration (formerly called cystic medial necrosis), characterized by elastic fiber loss from the medial layer, loss of vascular smooth muscle cells, and proteoglycan deposition. 6 Medial degeneration may (at least initially) occur as an adaptive response to wall stress in the aortic dilation zone. As the aorta enlarges, its distensibility is reduced. When the tensile limit is reached, dissection may occur. Aneurysms can occur without dissection, and dissections can occur without aneurysms. 6 Dissection occurs when an intramural hemorrhage takes place in the damaged media as a result of rupture of the intima or the vasa vasorum. 1 The dissecting hematomas observed in the bearded dragons of our study were likely due to intimal rupture because the intramural hemorrhage was in the internal portion of the artery (very close to the lumen).
There are a few reports of single cases of aneurysm in bearded dragon.5,26,27 Two were located cranial to the heart and 1 was in the tail, and these had similar gross and histological findings to the aneurysms observed in our study. Aneurysm reports are not restricted to lizards. A Burmese python (Python bivittatus) had a large, dissecting aortic aneurysm with separation of the muscular medial layer from the intima and an organized thrombus in the intimal wall. 21 A recent retrospective study summarized diverse pathological conditions in 36 P. vitticeps. 3 However, there is no mention of aneurysm in this summary of cases, and only 8% of the cases had arteriosclerosis.
One hallmark of aneurysmal lesions is inflammation, which orchestrates the interaction of the various pathological processes. The inflammatory process is characterized by the infiltration of a variety of cells within the lesion, which leads to the upregulation of multiple cytokines. The balance of the cellular type and resultant cytokine milieu determines the ultimate fate of the aortic wall healing, atherosclerosis, or aneurysm formation. 4 In our study, mild inflammation was observed in all cases, but substantial vasculitis was seen in only 5 cases, with the majority of them being associated with the presence of organisms resembling microsporidia or bacteria. In 3 cases, we observed intracellular round to oval, 1 μm diameter, Gram-variable microorganisms. The Gram-variable nature of the organisms may reflect suboptimal stain penetration due to their intracellular location. These microorganisms also stained the Luna stain, which is known to be superior to Gram, acid-fast, and Giemsa stains for staining microsporidian spores, enabling the detection of individual spores in tissues. 13 Microsporidia have been recently reported as causative agents of granulomatous arteritis leading to aneurysm formation, aortal rupture, and hemopericardium in 2 bearded dragons. 27 In that report, Encephalitozoon pogonae was identified by polymerase chain reaction. In 2 cases, large numbers of Gram-negative bacilli were present in a thrombus attached to the endothelium, but they did not extend into the arterial wall and were interpreted as an acute opportunist event, possibly associated with the previous contrast study. Although infectious arteritis has been described as cause of aneurysm in several mammal species, based on our findings, infection is probably not a primary cause of aneurysms in bearded dragons.1,8,12,24
The circulation patterns of the cephalic arteries and veins in squamates are functionally related adaptations to maintain preferred head vs body temperature. In iguanine lizards, Porter and Witmer identified how the jugular constrictor muscle surrounding the distal internal jugular vein controls flow volumes to the head, particularly the eyes and brain. 15 The contracted jugular constrictor muscle reduces the lumen of the internal jugular vein, rerouting drainage from the head to the orbital sinus, inducing exophthalmia as well as greater flow volumes through (and presumed greater stress) the dorsolateral sinus, transverse sinus, transversotrigeminal vein, and spinal vein. Jugular constrictor contraction also leads to reduced flow to the lateral head vein. This potential for intermittent vascular dilation of vessels that normally receive low flows may, in concert with declining vascular structure, predispose bearded dragons to aneurysms in the head or even contribution to backpressure in the vessels of the cranial coelom.
The major arteries and veins are diagramed in Supplemental Figure S1. To provide perspective, major arterial blood flow to the head is from the right aorta to a very short common carotid artery that gives rise to paired internal carotids. The internal carotid arteries supply the more dorsal head structures. The much smaller external carotid artery supplies the tongue, mandible, and pterygoid muscles. Drainage of the head occurs via a network of veins and sinuses to large lateral head veins, which drain to the internal jugular veins, the tracheal veins, and then to the external jugular veins.
Although the exact location of aneurysms was not always described, based on the clinical and pathological findings, the cases reported in this article involved many different sites. Based upon anatomical position or description, the vessels likely included were the left aorta, temporo-orbital artery, cerebral artery, anterior gastric artery, femoral artery, caudal artery, orbital sinus, dorsolateral sinus, internal jugular vein, left precaval vein, right precaval vein, postcaval vein, sinus venosus, transversotrigeminal vein, transverse sinus, temporal vein, and caudal vein.
In the bearded dragons, the temporo-orbital artery coincides with the locations of some cephalic aneurysms. It is located posterior to the orbit and is contained within the jaw adductor musculature within the superior temporal foramen. Many of the aneurysm sites summarized in Table 1 were located near the heart and high-pressure outflow vessels from the heart, in cephalic vessels located upstream of the jugular veins’ constrictor muscles, or in vessels that could be vulnerable to acute stress. The large lateral head veins, tracheal veins, and palatal veins are located near the buccal and esophageal surfaces between the buccal surface and cranial bones. While speculative, they may be vulnerable to damage if the lizards were to feed on hard forage that could compress and damage vessels over time. Unlike the cranial coelom and head, the lower proportion of aneurysms observed in the caudal coelom, legs, or tail highlights the lower incidence in more distal locations. This is likely due to lower pressures and greater protection from viscera, muscles, and scaled skin.
In summary, this study describes the clinical, macroscopic, and histologic features of aneurysms in a series of bearded dragons. Most of the aneurysms were near the heart base (cranial coelomic) or in the head, and a minority of cases were in the caudal coelom, hind legs, or tail. The main pathological findings consisted of moderate to severe disruption of the collagen and elastic fibers of the tunica media and adventitia. These conditions could be associated with an underlying connective tissue disorder, possibly a consequence of the pet lineage’s inbred history. Although the main pathological features of this vasculopathy in bearded dragons are described in this report, the etiopathogenesis of this process remains unknown and further studies (including electron microscopy of the vascular wall) as well as husbandry details are needed to better understand this condition.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231214025 – Supplemental material for Aneurysm Associated with Vascular Wall Degeneration in Bearded Dragons (Pogona vitticeps)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231214025 for Aneurysm Associated with Vascular Wall Degeneration in Bearded Dragons (Pogona vitticeps) by Joaquín Ortega, Jeanette Wyneken and Michael M. Garner in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Cathy Minogue and Christie Buie of Northwest ZooPath for data retrieval and image layout, respectively. The authors also thank Roy Brown of Histologic Consulting Services for slide preparation. Northwest ZooPath is grateful to the following Institutions/veterinarians for submission of study cases: Animal Medical Center of the Village, Caldwell Zoo (Dr Buchanan), California Science Center (Dr Stevens), Carolina Veterinary Specialists (Dr Powers), Chicago Exotics Animal Hospital (Dr Horton), Doctor Tim’s Wildlife & Exotic Care (Dr Reichard), Florida Atlantic University, Fresno Chaffee Zoo, Gulf Coast Avian & Exotics (Dr Chen), Indianapolis Zoo (Dr Bowman), Oasis Veterinary Hospital (Dr Asseo), Pet Hospital of Penasquitos (Dr Boyer), Philadelphia Zoo (Dr Ialeggio), The Bird & Exotic Hospital (Dr Rolfe), Toledo zoo (Dr Dumonceaux) and Washington Animal Disease Diagnostic Laboratory (Dr Nelson). Dr Barten and Dr Mader shared images and clinical notes. JW thanks F. Steinberg, MD, Medical Director, University MRI, Boca Raton, for providing access to high-resolution CT and fMRI equipment, software, and technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the project IDOC21-05 from Universidad CEU Cardenal Herrera, CEU Universities. JO was supported by “Ayudas a la movilidad investigadora CEU—Banco Santander.”
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
