Abstract
Tumors in dogs and humans share many similar molecular and genetic features, incentivizing a better understanding of canine neoplasms not only for the purpose of treating companion animals, but also to facilitate research of spontaneously developing tumors with similar biologic behavior and treatment approaches in an immunologically competent animal model. Multiple tumor types of both species have similar dysregulation of signal transduction through phosphatidylinositol 3-kinase (PI3K), protein kinase B (PKB; AKT), and mechanistic target of rapamycin (mTOR), collectively known as the PI3K-AKT-mTOR pathway. This review aims to delineate the pertinent aspects of the PI3K-AKT-mTOR signaling pathway in health and in tumor development. It will then present a synopsis of current understanding of PI3K-AKT-mTOR signaling in important canine cancers and advancements in targeted inhibitors of this pathway.
Comparative oncology is an expanding field that offers unique benefits for better understanding cancer development in humans and companion animals. Neoplasia is one of the leading causes of morbidity and mortality in dogs, with 4.2 million dogs diagnosed with cancer each year in the USA (population rate of 5300/100,000), as reported in 2015. 231 Dogs provide a valuable spontaneous animal model of cancer with distinct advantages as compared with rodent models, including a genetically diverse population with comparable environmental exposures and complex interactions between the immune system and tumor. Canine neoplasms also have clinical presentations, pathophysiology, chemotherapeutic resistance, and biologic behaviors that better recapitulate the natural progression of human cancer than induced cancer animal models. Advancements in our genomic and molecular understanding of cancer physiology in dogs and humans is revealing increasing numbers of conserved biochemical pathways shared between the species. 85 For these reasons, there is great value in investigating signal transduction cascades in canine tumor development for comparative oncology, as well as to provide our canine companions with improved cancer treatment.
The phosphatidylinositol 3-kinase (PI3K), protein kinase B (PKB; AKT), mechanistic target of rapamycin (mTOR) signal transduction pathway is constitutively activated in many cancers. The PI3K-AKT-mTOR signaling pathway is involved in mediating an array of cellular processes, including proliferation, survival, nutrient uptake, metabolic activity, and anabolic reactions (protein synthesis).4,28,49,108,111,141,145,251 This signaling pathway is highly conserved across species. There is evidence for similar roles of pathway activation in tumor development across species, and the value of canine and human studies to comparative oncology in multiple tumor types has been analyzed and reviewed elsewhere.12,53,72,132,153,211,264 As is often the case in the veterinary field, the basic research in normal cellular signaling cascades for dogs is not as completely documented. As such, this review utilizes the discoveries in PI3K-AKT-mTOR signaling in human literature to build the foundational background information for a better understanding of the criticality of this pathway in cellular homeostatic and regulatory activity, as well as feedback mechanisms. This article aims to review the role of PI3K-AKT-mTOR signaling in health, alterations in cancer development and progression, and its significance specifically to canine neoplasms. Finally, this review highlights diagnostic utility and advances in targeted pathway inhibition as a therapeutic strategy.
While not as expansive in physiologic mechanistic descriptions, the veterinary literature does demonstrate similar oncogenic mutations and PI3K-AKT-mTOR pathway activation as those described in human neoplasms, emphasizing to the value to comparative oncology. Often the contributions to the body of knowledge are small and stepwise in veterinary oncology, in part due to limitations on scope of experimentation. Veterinary research does not always examine tumor samples through multiple modalities for more thorough correlation between genes, transcripts, protein expression, and protein phosphorylation (indicating pathway activation). Moreover, consistent antibody validation for canine studies is needed. As such, there are greater limitations on the breadth of definitive statements that can be made regarding the relationship of mutations, expression, and pathway activation in canine tumors. However, it is an expanding field.
PI3K-AKT-mTOR Signal Transduction
The canonical PI3K-AKT-mTOR signal transduction cascade is complex with multiple points of initiation, regulation, and effector outcomes (Fig. 1). Normal pathway activation occurs when extracellular signals activate growth factor receptor tyrosine kinases (RTKs), such as insulin receptor, insulin-like growth factor 1 receptor (IGF1R), fibroblast growth factor receptor, epidermal growth factor receptor (EGFR), platelet-derived growth factor receptor (PDGFR), erb-b2 RTK 2 (ERBB2), and hepatocyte growth factor receptor (HGFR; also known as mesenchymal-epithelial transition factor [c-MET]). Activated RTKs phosphorylate adaptor proteins, GRB2-associated-binding protein 1 and 2, insulin receptor substrates 1 and 2 (IRS1/2), and fibroblast growth factor receptor substrate 2, which bind the amino-terminal domain of the p85 regulatory subunit of PI3K, via YXXM motifs.61,181 This binding relieves p85 inhibition of the p110 catalytic subunit of PI3K and recruits the p85–p110 heterodimer to its substrate, phosphatidylinositol-(4,5)-bisphosphate (PIP2), at the plasma membrane to activate class I PI3K. 89 Isoforms of these subunits are pertinent to feedback mechanisms and are targets for specific PI3K inhibitors, which are discussed in the sections below.
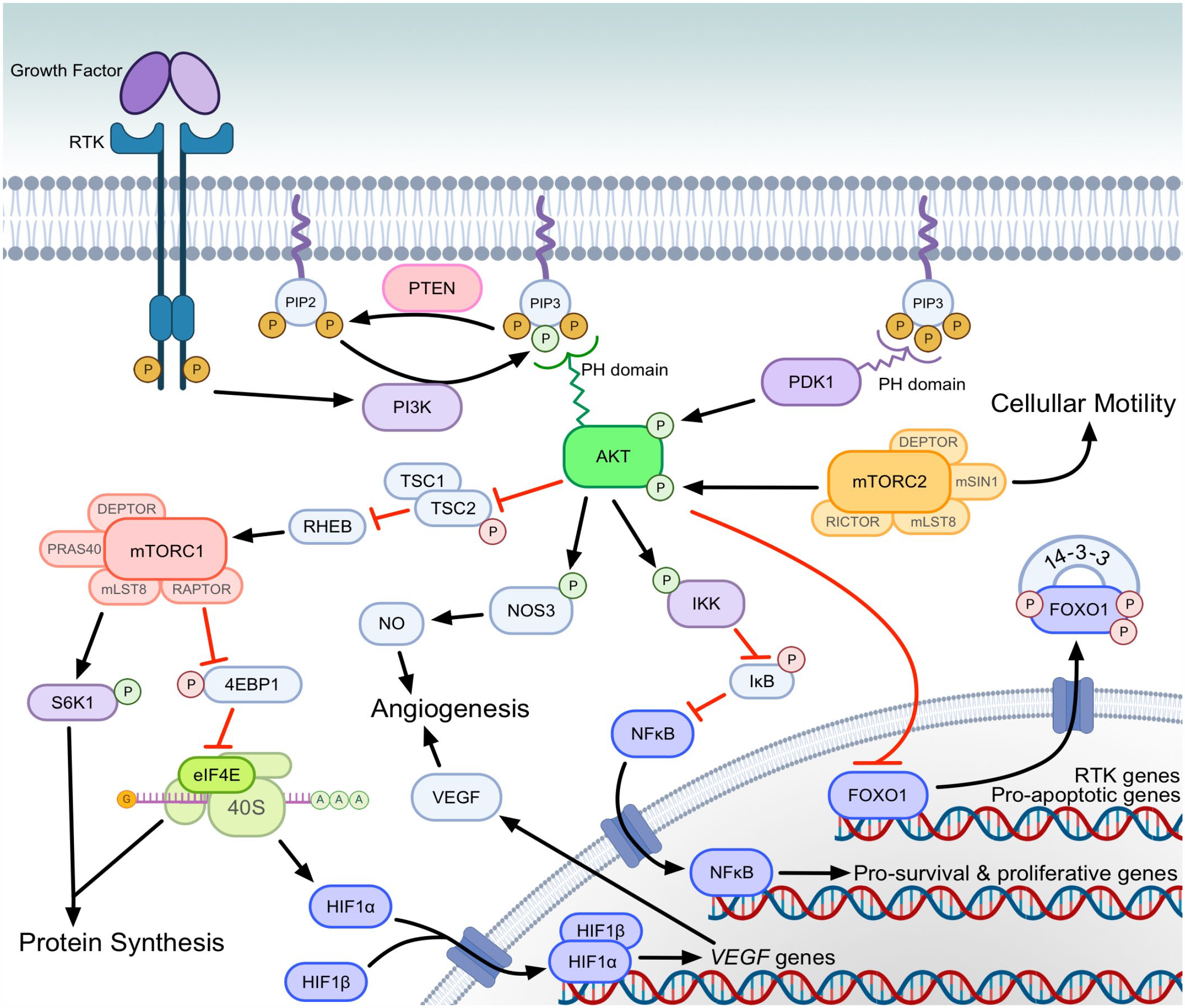
PI3K-AKT-mTOR signal transduction cascade. Growth factors and signaling proteins activate RTKs at the cell surface, which activate PI3K. PI3K is responsible for phosphorylating PIP2 to PIP3. PIP3 is necessary for recruitment, PH domain binding and subsequent activation of AKT by PDK1 and mTORC2. Green and red phosphates in the figure indicate activating and inhibiting phosphorylation events, respectively. Phosphorylated AKT acts on multiple target proteins that promote the survival, growth, and metastasis of cancer cells. AKT alters gene expression through regulation of nuclear translocation of transcription factors (dark blue) both indirectly (eg, NFκB) and directly (eg, FOXO1). Phosphorylation of FOXO1 causes extranuclear transport, reducing transcription of proapoptotic genes. Phosphorylation of IKK allows NFκB to enter the nucleus and transcribe genes promoting survival. AKT phosphorylates and inhibits the TSC heterodimer, which leads to mTORC1 activation. mTORC1 activates S6K1, which phosphorylates ribosomal protein S6 to promote translation of mRNA to proteins involved in cellular growth, proliferation, and glucose metabolism. mTORC1 also inhibits 4EBP1, which allows for eIF4E to selectively translate oncogenic proteins involved in cellular growth, proliferation, migration, invasion, and angiogenesis. Angiogenesis is increased through eIF4E-mediated translation (eg, HIF1α), as well as phosphorylation of NOS3 by AKT to increase NO production. In the figure, red phosphates (P) indicate inactivating phosphorylation and green phosphates indicate activating phosphorylation. (Figure created with BioRender.com). Abbreviations: PI3K, phosphatidylinositol 3-kinase; AKT, protein kinase B; mTORC, mechanistic target of rapamycin complex; PIP2, phosphatidylinositol (4,5)-bisphosphate; PIP3, phosphatidylinositol (3,4,5)-trisphosphate; PH, Pleckstrin homology; PDK1, phosphoinositide-dependent kinase 1; NFκB, nuclear factor kappa B; FOXO1, forkhead box O1; IKK, inhibitor of nuclear factor kappa B kinase; S6K1, ribosomal protein S6 kinase beta-1; 4EBP1, eukaryotic translation initiation factor 4E-binding protein 1; eIF4E, eukaryotic translation initiation factor 4E; HIF1α, hypoxia inducible factor 1 subunit alpha; NOS3, nitric oxide synthase 3; NO, nitric oxide; P, phosphate; IκB, inhibitor of nuclear factor kappa B; PTEN, phosphatase and tensin homolog; RHEB, Ras homolog enriched in brain; RTK, receptor tyrosine kinase; TSC1, tuberous sclerosis complex subunit 1; TSC2, tuberous sclerosis complex subunit 2; VEGF, vascular endothelial growth factor.
PI3Ks are a family of intracellular lipid kinases that phosphorylate the 3’-hydroxyl group of the inositol ring on intracellular membrane-bound phosphatidylinositol (PI). Class I PI3K phosphorylates PIP2 to phosphatidylinositol-(3,4,5)-trisphosphate (PIP3). 79 In opposition of PI3K activity, phosphatase and tensin homolog (PTEN) regulates the signaling duration of PIP3 by dephosphorylating it to PIP2.30,158,249 In its phosphorylated form, PIP3 recruits AKT and phosphoinositide-dependent kinase-1 (PDK1) to the plasma membrane, and binds to their Pleckstrin homology domains, where PDK1 phosphorylates AKT at one of two phosphorylation sites.3,73,104,105,179 Partial phosphorylation of AKT is sufficient for activation of AKT to phosphorylate and inactivate tuberous sclerosis complex subunit 2 (TSC2) of the TSC1/TSC2 heterodimer, releasing the TSC1/TSC2 inhibition of RAS homolog, mTORC1 binding (RHEB).84,112,113,256 Activated RHEB then leads to the subsequent activation of mTOR complex 1 (mTORC1).35,100,252
mTORC1 is noteworthy in itself, because of the large body of research focused on drugs targeting this rapamycin-sensitive complex formed with the regulatory-associated protein of mTOR (RAPTOR). 103 Upon activation of mTORC1, the resultant downstream effects are numerous and include promotion of translation of mRNA for protein synthesis and cellular proliferation, acting through eukaryotic translation initiation factor 4E (eIF4E)-binding protein 1 (4EBP1) and ribosomal protein S6 kinase beta-1 (RPS6KB1; S6K1) to phosphorylate ribosomal protein S6 (eS6; RPS6).38,201
With a second phosphorylation of AKT by mTOR complex 2 (mTORC2), phosphorylated AKT (pAKT) is then capable of phosphorylating a diverse set of proteins containing RXXS/T motifs.65,229,251 Of significant interest for survival of neoplastic cells, pAKT phosphorylates the forkhead box transcription factors (including FOXO1 and FOXO3), promoting their nuclear export and ubiquitin-proteasome degradation.19,28,29,31,97,176,216 This prevents FOXO transcription factors from promoting apoptosis via expression of death receptor ligands, Fas ligand, and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), as well as modulating expression of apoptosis regulator proteins in the BCL2 family, including BCL2 interacting protein 3, BCL2-like 1, and BIM.28,75,80 pAKT also directly phosphorylates another proapoptotic BCL2 family member, BCL2-associated agonist of cell death (BAD), resulting in its inactivation and cell survival.285,290
In addition to inhibition of FOXO1 mediated apoptosis, pAKT promotes the prosurvival and proliferative activity of the transcription factor nuclear factor kappa Β (NFκΒ). 14 NFκΒ mediates multiple oncogenic phenotypes, which have been described in a recent review. 232 pAKT functions to promote NFκΒ activity in 3 ways: phosphorylating and thereby activating the alpha subunit of the inhibitor of nuclear factor kappa B (IκB) kinase (IKK), which allows NFκΒ to enter the nucleus for transcription; stimulation of IKK activity via mitogen-activated protein kinase kinase kinase 8; and stimulation of NFκΒ p65 transactivation via IKK and p38/mitogen-activated protein kinase (MAPK) signaling.119,164,220
Pathway Feedback
Since mutations and/or overexpression of individual members of the signaling pathway are reported in multiple cancers, one might assume that inhibitors targeting one of these proteins could be sufficient. However, challenges to targeted pathway inhibition in tumors are presented by the presence of many points of feedback in PI3K-AKT-mTOR signal transduction that are well documented in human cancer studies, including those discussed below and diagrammed in Fig. 2.
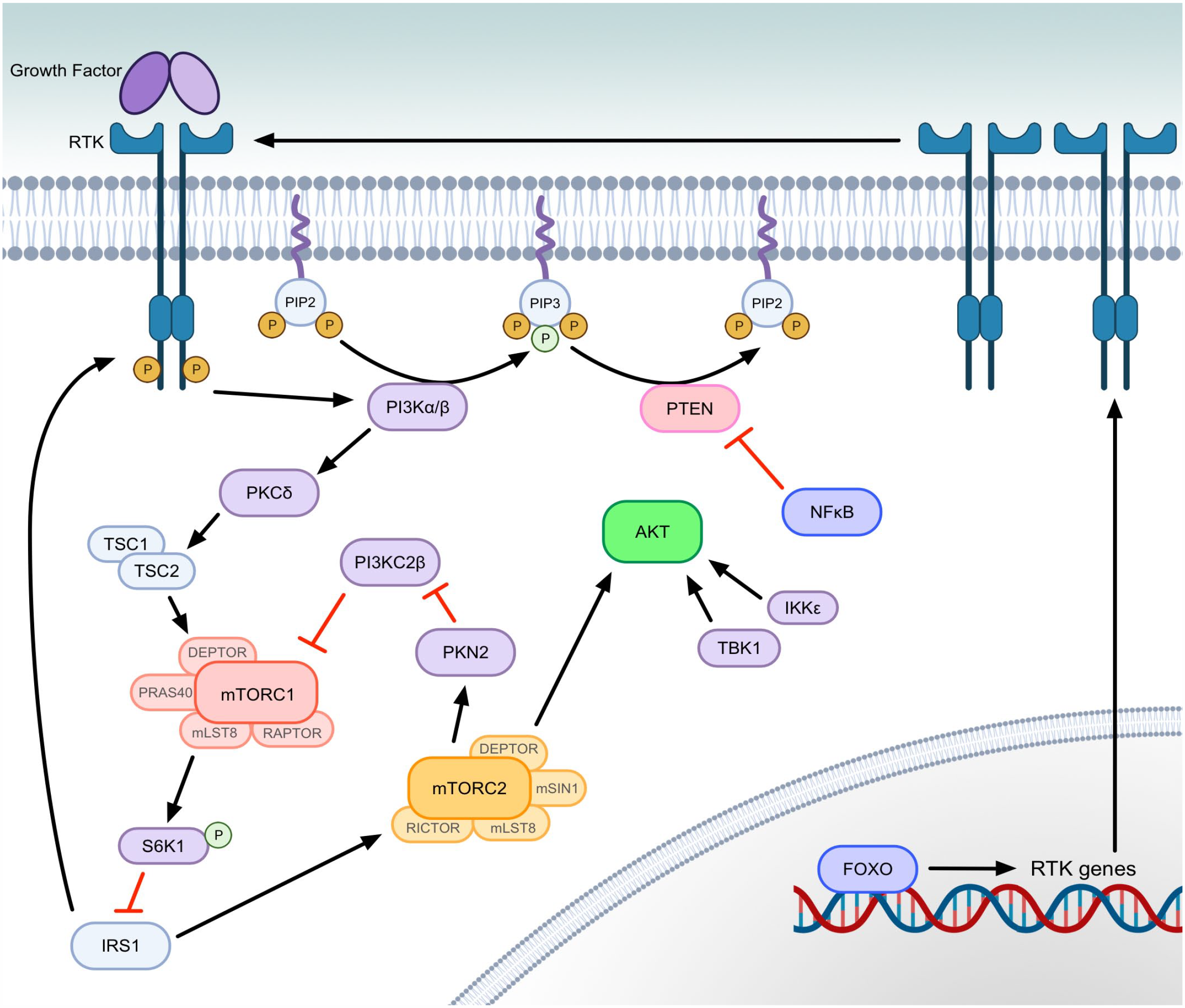
PI3K-AKT-mTOR signal transduction feedback pathways. Inhibitors of the PI3K-AKT-mTOR typically target mTORC1, PI3K, specific PI3K isoforms, or less frequently AKT. Depicted here are feedback loops and alternate methods of pathway activation that can occur following inhibition of canonical PI3K-AKT-mTOR signal transduction. PI3Kα is the predominant isoform of the class IA PI3Ks, and activating mutations in PIK3CA (gene encoding p110α catalytic subunit of PI3Kα) are highly reported in cancer. PI3Kα isoform inhibition leads to greater activation of the PI3Kβ isoform and continued PIP3 formation. PI3Kβ inhibition causes less significant isoform switching, but results in the same RTK expression as AKT inhibition. Both isoforms, when sufficiently expressed, are capable of activating TSC2 and mTORC1 through PKCδ without the need for AKT. Inhibiting AKT allows FOXO-mediated transcription of genes to express RTKs, which also activate PI3K. mTORC1 inhibition removes S6K1 inhibition of IRS1. IRS1 then acts as an RTK/PI3K adaptor protein and removes p85 inhibition of PI3K p110. IRS1 also contributes to activation of mTORC2. Active mTORC2 reduces PI3KC2β inhibition of mTORC1, as well as activating AKT. AKT activation can also occur from proteins outside of the canonical PI3K-AKT-mTOR pathway, including TBK1 and IKKε. When activated by other signals (eg, TNF or IL-1), NFκB also reduces PTEN transcription, which allows for PIP3 accumulation and AKT activation. In the figure, green phosphates (P) indicate activating phosphorylation. (Figure created with BioRender.com). Abbreviations: PI3K, phosphatidylinositol 3-kinase; AKT, protein kinase B; mTORC, mechanistic target of rapamycin complex; PI3Kα, phosphatidylinositol 3-kinase, class I isoform with catalytic subunit alpha; PI3KC2β, phosphatidylinositol-4-phosphate 3-kinase, class II β isoform; PIP3, phosphatidylinositol (3,4,5)-trisphosphate; RTK, receptor tyrosine kinase; TSC2, tuberous sclerosis complex subunit 2; PKCδ, protein kinase C delta type; FOXO1, forkhead box O1; S6K1, ribosomal protein S6 kinase beta-1; IRS1, insulin receptor substrate 1; TBK1, TANK binding kinase 1; IKKε, inhibitor of nuclear factor kappa B kinase subunit epsilon; TNF, tumor necrosis factor; IL-1, interleukin 1; NFκB, nuclear factor kappa B; PTEN, phosphatase and tensin homolog; P, phosphate; IκB, inhibitor of nuclear factor kappa B; PDK1, phosphoinositide-dependent kinase 1; PIP2, phosphatidylinositol (4,5)-bisphosphate; PKN2, protein kinase N2; TSC1, tuberous sclerosis complex subunit 1.
Under normal conditions, mTORC1 activation results in S6K1 negatively regulating IRS1 by phosphorylation. Inhibition of mTORC1, via rapamycin administration, mitigates this S6K1 activity toward IRS1. 106 IRS1 then serves as an adaptor protein at RTKs to promote activation of PI3K. 242 IRS1 activation also causes increased mTORC2 activation, allowing for mTORC2 to phosphorylate AKT. 251 AKT can additionally be activated in mTORC2-independent mechanisms by other kinases, such as IκB kinase subunit epsilon (IKKε) and TANK-binding kinase 1 (TBK1), which are capable of phosphorylating AKT at the hydrophobic motif and at the activation loop in a PI3K-dependent manner, even when mTORC2 is inhibited. 283
While the canonical pathway to mTORC1 activation signals through AKT, PI3K signaling through other kinases can also activate mTORC1. Inhibition of the translation products of mTORC1 induces a feedback pathway that leads to PI3K activation of protein kinase C delta type (PKCδ), which in turn phosphorylates TSC2, resulting in greater mTORC1 activation.90,292 Further mTORC1 feedback activation can occur through mTORC2 phosphorylation of protein kinase N2 (PKN2). PKN2 then inactivates the class II PI3K isoform, PI3KC2β, which normally acts during starvation periods to negatively regulate mTORC1. 268 Thus, glucose starvation and/or RTK inhibitors that could potentially reduce mTORC1 activity are rendered less effective. 202
Broad inhibition of PI3K has proven difficult because pan-PI3K inhibitors at doses required to block class I PI3K activity induced toxicities that have not allowed for efficacious treatment, with hepatotoxicity reported in canine patients.74,87 Given that mutations in PIK3CA (the gene encoding the p110α catalytic subunit of the PI3K alpha isoform [PI3Kα]) or PTEN are among the most commonly reported mutations in canine cancer, one might presume better clinical results could be obtained by targeting specific isoforms of class I PI3Ks based on the type of mutations present (activating mutations for genes encoding RTKs or PI3K result in primarily PI3Kα signaling, and PTEN mutations result in predominantly PI3Kβ signaling in human studies).5,74,160,282,293 However, as demonstrated in human studies, treatment with a PI3Kα-specific inhibitor only yields a transient decrease in PIP3 phosphorylation and AKT activation, due to feedback activation of the widely expressed PI3Kβ by G-protein coupled receptors and binding of the G-beta-gamma complex to the p110β subunit of PI3Kβ.13,25,234 A more robust inhibition with targeted PI3Kα inhibitors was documented in canine cell lines in vitro than in human studies. However, the authors also reported variability in responses, which may indicate pathway activation in different tumor types that may be related to PI3K isoforms and feedback mechanisms in the canine patient similar to those observed in humans. 165 It has been well documented in human studies that PI3Kβ inhibition or direct AKT inhibition both result in greater expression of RTKs, including insulin receptor and erb-b RTK 3, due to aforementioned feedback from mTORC1 inhibition, via IRS1, and greater FOXO transcriptional activation. 37 Likewise, mTOR inhibition by rapamycin increases the expression of RTKs to enhance PI3K-mTORC2 signaling. 202 Increased RTK expression allows for greater PI3Kα signal transduction and re-activation of AKT. 234 NFκB signaling also indirectly represses transcription of PTEN, leading to increased PI3K signaling.14,130,267
PI3K-AKT-mTOR Pathway in Health and Cancer: Downstream Effectors
The sections below focus on the downstream targets and effectors of the PI3K-AKT-mTOR pathway in the modulation of cellular metabolism, survival, proliferation, and angiogenesis present in cancer development (Table 1). However, the broader downstream physiologic effects are not limited to these examples and are more thoroughly reviewed elsewhere.159,218,260 Some of the information below regarding general pathophysiologic mechanisms of this highly conserved pathway is extrapolated from human oncology, but there is evidence of PI3K-AKT-mTOR dysregulation in tumors of multiple species.
AKT targets and effectors.
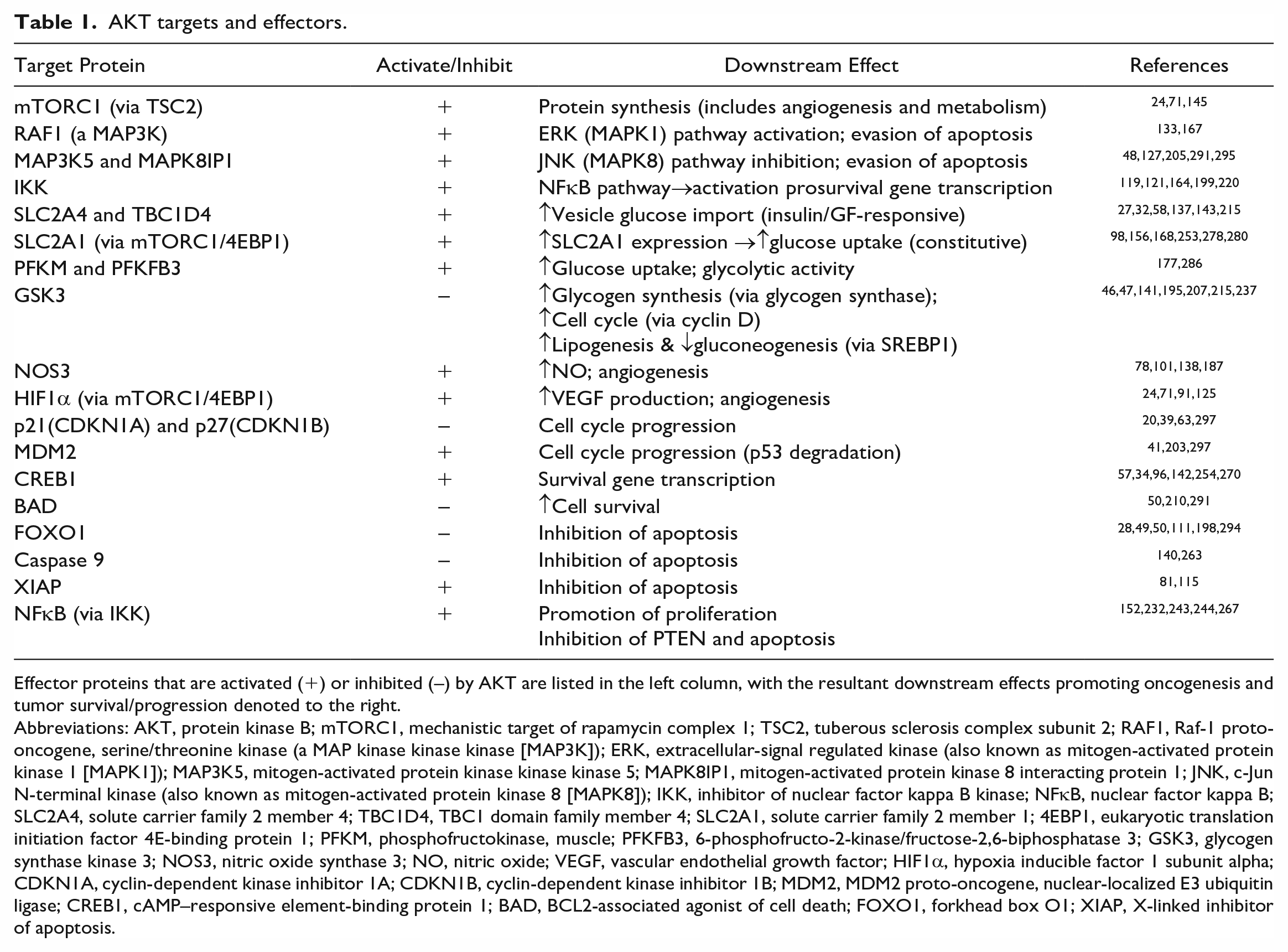
Effector proteins that are activated (+) or inhibited (–) by AKT are listed in the left column, with the resultant downstream effects promoting oncogenesis and tumor survival/progression denoted to the right.
Abbreviations: AKT, protein kinase B; mTORC1, mechanistic target of rapamycin complex 1; TSC2, tuberous sclerosis complex subunit 2; RAF1, Raf-1 proto-oncogene, serine/threonine kinase (a MAP kinase kinase kinase [MAP3K]); ERK, extracellular-signal regulated kinase (also known as mitogen-activated protein kinase 1 [MAPK1]); MAP3K5, mitogen-activated protein kinase kinase kinase 5; MAPK8IP1, mitogen-activated protein kinase 8 interacting protein 1; JNK, c-Jun N-terminal kinase (also known as mitogen-activated protein kinase 8 [MAPK8]); IKK, inhibitor of nuclear factor kappa B kinase; NFκB, nuclear factor kappa B; SLC2A4, solute carrier family 2 member 4; TBC1D4, TBC1 domain family member 4; SLC2A1, solute carrier family 2 member 1; 4EBP1, eukaryotic translation initiation factor 4E-binding protein 1; PFKM, phosphofructokinase, muscle; PFKFB3, 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3; GSK3, glycogen synthase kinase 3; NOS3, nitric oxide synthase 3; NO, nitric oxide; VEGF, vascular endothelial growth factor; HIF1α, hypoxia inducible factor 1 subunit alpha; CDKN1A, cyclin-dependent kinase inhibitor 1A; CDKN1B, cyclin-dependent kinase inhibitor 1B; MDM2, MDM2 proto-oncogene, nuclear-localized E3 ubiquitin ligase; CREB1, cAMP–responsive element-binding protein 1; BAD, BCL2-associated agonist of cell death; FOXO1, forkhead box O1; XIAP, X-linked inhibitor of apoptosis.
Cellular Glucose Metabolism, Lipogenesis, and Protein Synthesis
The PI3K-AKT-mTOR pathway regulates glucose metabolism by promoting cellular uptake and regulating gene expression. Many of these effects on glucose metabolism are mediated through the phosphorylation and inactivation of glycogen synthase kinase-3 by AKT. 288 Through glycogen synthase kinase-3 and directly, AKT promotes the translocation of the facilitated glucose transporter, solute carrier family 2 member 4, to the plasma membrane to increase insulin-responsive glucose uptake.49,137 PI3K-AKT-mTOR pathway activation increases the translation of the cell surface glucose transporter, solute carrier family 2 member 1, through mTORC1-4EBP1 signaling, increasing constitutive glucose uptake.137,253 The phosphorylation of GSK3 by AKT also decreases ubiquitin-mediated proteasomal degradation of sterol regulatory element-binding transcription factor 1, thus inhibiting gluconeogenesis and promoting lipogenesis necessary in cellular proliferation. 141
PI3K-AKT-mTOR signal transduction promoting synthesis of proteins involved in metabolism and growth through mTORC1-4EBP1-eIF4E and mTORC1-S6K1 signaling activation is very well studied and is thoroughly reviewed elsewhere.145,162,251 In addition to an increase in global protein translation, phosphorylation of 4EBP1 results in selective promotion of translation for specific prooncogenic and angiogenic transcripts, including fibroblast growth factor 2, vascular endothelial growth factor (VEGF), cyclin D, matrix metalloprotease 9, and basic helix-loop-helix transcription factor (MYC). 197 Phosphorylation of S6K1 leads to multiple effectors that promote processes including mRNA biogenesis, and translation initiation and elongation. mTORC1, through other downstream effectors, promotes expression of rRNA (via tripartite motif containing 24 and RNA polymerase I) and transcription of 5S rRNA and tRNA (via MAF1 homolog, negative regulator of RNA polymerase III).120,180,240
Proliferation
Important targets of AKT activation in processes necessary for proliferation include the FOXO transcription factors.49,108,218 By modulating the expression of target genes, FOXO proteins are involved in regulating differentiation, apoptosis, resistance to oxidative stress, and cell cycle arrest. 111 FOXO proteins primarily modulate proliferation through increasing transcription of cyclin-dependent kinase inhibitors, p27Kip1, p21Waf1/cip1, and p130, to prevent the G1/S transition of the cell cycle.59,95,182 They additionally reduce expression of cyclin D1 and cyclin D2, resulting in G1 arrest.214,233 When growth factors and extracellular signals or oncogenic mutations activate the PI3K-AKT pathway, the subsequent nuclear export of FOXO proteins allows for mitotic progression to continue.29,262
PI3K-AKT-mTOR pathway activation resulting in the phosphorylation of 4EBP1 allows for eIF4E to selectively translate known oncogenic factors promoting proliferation, including MYC, ornithine decarboxylase, cyclin D1, and cyclin D3.56,221,222,236 PI3K-AKT-mTOR signaling also promotes mitogenesis through S6K1 inhibition of the p16INK4a/CDK4/cyclin D1/retinoblastoma pathway’s induction of cell cycle arrest in G1 phase.83,161,174,186
Further proliferative effects of PI3K-AKT signal transduction are mediated through NFκB hyperphosphorylation of retinoblastoma protein and promotion of cyclin D1 expression, which is important for cell cycle progression from G1 to S phase.99,109 NFκB also promotes proliferation through several downstream cytokines that act as growth factors, including interleukins (IL)-1 and IL-6, which are of particular importance in lymphoid neoplasms.2,243
Evasion of Apoptosis
PI3K-AKT signaling resulting in nuclear export of FOXO1 and FOXO3 prevents expression of proapoptotic BCL2 member BIM and subsequent release of mitochondrial cytochrome complex and activation and caspase-3 to induce apoptosis.28,55,111,241,294 Concurrent inhibitory phosphorylation of BAD/BCL2-like 1 signaling by pAKT also prevents mitochondrial cytochrome C release and caspase activation.140,285,290 AKT mediated degradation of FOXO1 and FOXO3a and reduces the expression of FAS ligand and TRAIL, which are both death receptor ligands that function in paracrine and autocrine pathways.4,172 FAS ligand inhibition prevents mitochondria-dependent apoptosis and mitochondria-independent apoptosis through JNK pathway activation, as well as the extrinsic apoptotic pathway.28,135,294 Reduced expression of TRAIL similarly results in evasion of apoptosis mediated by death receptors DR4 and DR5. 10 In addition to inhibiting these apoptotic pathways, PI3K-AKT signal transduction also inhibits caspase-mediated apoptosis through IKK-IκB-NFκB signaling to increase expression of survival genes, including BCL2-like 1. 232
Angiogenesis
The PI3K-AKT-mTOR pathway is implicated on both sides of VEGF signaling for tumor angiogenesis: in the stimulation of angiogenesis through paracrine signals from neoplastic cells and in endothelial cells themselves. VEGF and basic fibroblast growth factor (bFGF) are integral angiogenic factors that signal through the AKT-mTOR pathway in endothelial cell to promote tumor vessel formation through endothelial cell proliferation, migration, and actin reorganization. 192 Activation of the PI3K-AKT pathway in tumor cells can increase VEGF secretion, through hypoxia-inducible factor 1 (HIF1)-dependent and HIF1-independent mechanisms, as well as modulating nitric oxide and angiopoietins.77,117 PI3K-AKT-mTORC1 activation results in increased translation of the HIF1α subunit, through 4EBP1-eIF4G signaling. 24 Increased HIF1α and constitutively present HIF1β form a heterodimer that binds to hypoxia response elements, thus promoting transcription and expression of bFGF and VEGF.71,125 VEGF and IL-8 transcription is also upregulated through PI3K-AKT-NFκΒ signal transduction.148,296 In addition, phosphorylation of the endothelial and inducible isoforms of nitric oxide synthase (NOS3 and NOS2, respectively) by pAKT in tumor cells results in angiogenesis stimulated by nitric oxide synthesis.77,101 Nitric oxide also serves as a signaling molecule that regulates processes involved in vasodilation, vascular permeability, microlymphatic flow, platelet aggregation, and leukocyte-endothelial cell interaction.76,77
Invasion/Metastasis
Among multiple mechanisms, PI3K-AKT-mTOR signal transduction promotes tumor invasion through the inhibition of 4EBP1, resulting in increased translation of invasion and metastasis proteins, including Y-box-binding protein 1 (YB1), metastasis-associated protein 1 (MTA1), CD44, and vimentin.110,197
There is evidence that mTORC2 promotes cellular motility, increasing migration and invasion in tumor cells directly, in addition to activation of AKT and other kinases.173,228 mTORC2 has been shown to control cytoskeletal F-actin polymerization for cellular spreading and motility, which is necessary for the migration and invasion of neoplastic cells.44,114 mTORC1 also plays a role in promoting chemotactic, and IGF1-mediated, cellular migration by signaling through S6K1 and 4EBP1 for translation of proteins involved in regulation of F-actin reorganization and cell polarization.116,154,155 S6K1 activation by mTORC1 is also involved in focal adhesion protein phosphorylation, which is necessary for de-adhesion from extracellular matrix for migration. 154 In addition to migration, mTORC1-4EBP1-eIF4E signaling promotes invasion through increased translation of proinvasive mRNAs, including matrix metalloproteinases.93,94 Tumor invasion is also promoted via PI3K-AKT signaling through NFκB to increase transcription of proteolytic enzymes, including matrix metalloproteinases and urokinase-type plasminogen activator.21,273
Tumor metastasis is further enabled by increased translational activity to facilitate nutrient utilization and survival of neoplastic cells in the microenvironment of the metastatic site. 36 Constitutive activation of the PI3K-AKT-mTOR pathway results in increased translational activity by signaling through S6K1 and 4EBP1, in response to nutrient deprivation or hypoxia and oxidative stress, which normally restrict mTORC1 activity.147,194
Pathway Activation Reported in Tumors
Given that activation of the PI3K-AKT-mTOR pathway results in many of the downstream effects that promote cancer, it is not surprising that mutations and pathway enrichment are frequently reported and prognostically significant in human and canine neoplasms. The sections below highlight canine tumors that are associated with pathway activation with potential for diagnostic and therapeutic value in veterinary and comparative oncology. Mutations in the pathway are outlined in Table 2.
PI3K-AKT-mTOR mutations reported in canine tumors.
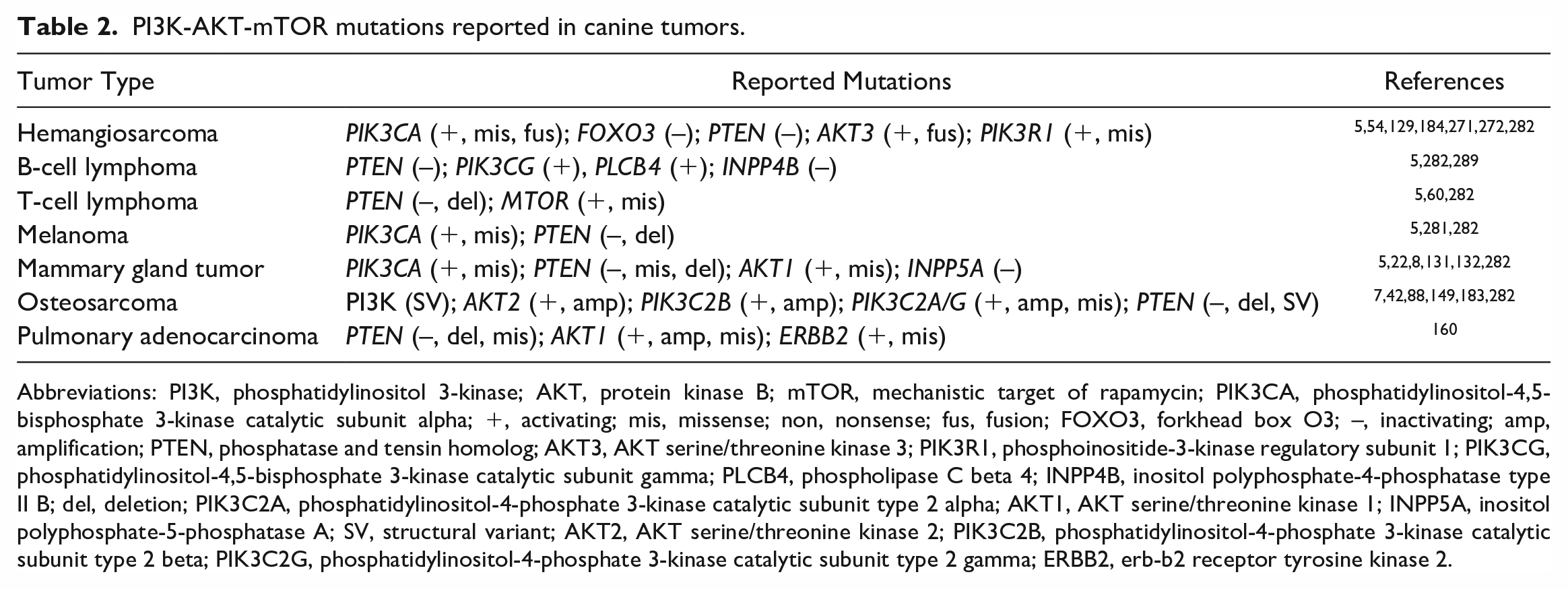
Abbreviations: PI3K, phosphatidylinositol 3-kinase; AKT, protein kinase B; mTOR, mechanistic target of rapamycin; PIK3CA, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha; +, activating; mis, missense; non, nonsense; fus, fusion; FOXO3, forkhead box O3; –, inactivating; amp, amplification; PTEN, phosphatase and tensin homolog; AKT3, AKT serine/threonine kinase 3; PIK3R1, phosphoinositide-3-kinase regulatory subunit 1; PIK3CG, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma; PLCB4, phospholipase C beta 4; INPP4B, inositol polyphosphate-4-phosphatase type II B; del, deletion; PIK3C2A, phosphatidylinositol-4-phosphate 3-kinase catalytic subunit type 2 alpha; AKT1, AKT serine/threonine kinase 1; INPP5A, inositol polyphosphate-5-phosphatase A; SV, structural variant; AKT2, AKT serine/threonine kinase 2; PIK3C2B, phosphatidylinositol-4-phosphate 3-kinase catalytic subunit type 2 beta; PIK3C2G, phosphatidylinositol-4-phosphate 3-kinase catalytic subunit type 2 gamma; ERBB2, erb-b2 receptor tyrosine kinase 2.
Osteosarcoma
Osteosarcoma (OSA) is a malignant mesenchymal tumor of bone with aggressive biologic behavior. In humans, it is the most common primary malignancy of bone, yet it is still relatively rare, accounting for less than 1% of all cancers diagnosed in the United States. 189 In dogs, the incidence of OSA is 27 times greater than in humans and the age distribution differs, yet it shares many of the clinicopathologic features with the malignancy in humans. In canine and human patients, the metastatic rate is high with radical resection alone, and the median survival time for dogs following limb amputation for OSA is 5 months, with a 1-year survival rate of 19.8%, which increases to 45% at 1 year and 20.9% at 2 years when amputation is followed by administration of doxorubicin, cisplatin, or carboplatin.15,17,248,250 While an improvement over surgery alone, these survival times evidence the necessity for improvements in the treatment of this highly aggressive canine tumor.
PI3K-AKT-mTOR pathway enrichment and constitutive activation are reported in human and canine OSA, are associated with poorer prognosis, and represent a significant therapeutic target.42,183,209,298 PI3K-AKT-mTOR pathway activation in canine OSA promotes neoplastic survival, proliferation, metabolic activity, and migration and invasion.92,178,185 Phosphorylated mTOR and S6K1, indicating pathway activation, are reported in canine OSA cells and are inhibited by rapamycin administration in vitro. 92 Deletions and down-regulation of PTEN are frequently reported in canine OSA.7,42,149,259,282 Genomic analyses of canine OSA has demonstrated EGFR and PIK3CA mutations, aberrant copy numbers of TSC2, and amplification of PIK3CB and AKT2.7,42,282
As RTKs are a primary mechanism of PI3K-AKT-mTOR pathway activation conserved across multiple species, several have been evaluated in canine tumors, in conjunction with AKT phosphorylation. Mutations and overexpression of RTKs are reported in canine OSA (IGF1R, HGFR/MET, PDGFR, and ERBB2).51,66,67,69,163,193 Platelet-derived growth factor (PDGF) and PDGFR overexpression has been detected by immunoassays and quantitative polymerase chain reaction (PCR) in canine OSA cell lines and tumors. 170 Canine OSA tumor sections have been shown to be robustly positive for IGF2R expression, by immunohistochemistry. 122 Also reported in canine OSA are elevated serum IGF concentrations and expression of IGF and growth hormone mRNA within tumors.126,134 IGF1R overexpression in canine appendicular OSA is correlated with greater AKT signaling and poorer prognosis. 169 Elevated serum VEGF is reported in cases of canine OSA and is associated with shorter disease-free interval, radiation resistance, and poorer prognosis.258,279
Hemangiosarcoma
Angiosarcoma is a rare neoplasm of humans, but the canine correlate, hemangiosarcoma (HSA), accounts for almost 2% of all cancer diagnoses in dogs and 5%–7% of noncutaneous primary canine malignancies.245,265 Angiosarcoma and HSA are similar aggressive tumors of endothelial origin, characterized by marked local invasion and rapid hematogenous metastasis/dissemination. Canine HSA is often multicentric by the time it is detected.239,274 As a result, the prognosis for dogs with HSA is poor and the 1-year survival rate is less than 10%.64,68,86,175 Given the aforementioned role of PI3K-AKT-mTOR signaling in angiogenesis, neoplastic cell proliferation, survival, and invasion, it is not surprising that pathway activation is reported in canine HSA. Previous studies have reported increased VEGF and bFGF expression by HSA cells in vitro and found that VEGF is detectable at significantly higher plasma concentrations in dogs with HSA as compared with healthy dogs.43,257 Evaluation of canine vascular tumors by immunohistochemistry, in situ hybridization, and reverse transcriptase-polymerase chain reaction demonstrated elevated bFGF, VEGF, and VEGF receptors (VEGFR1 and VEGFR2) in HSA samples, as compared with hemangioma samples. 287 Through genomic and molecular analyses, mutations in PIK3CA, PTEN, AKT3, and FOXO3 have been identified in canine HSA, similar to those reported in human angiosarcomas.5,54,128,129,184,271,272 Examination of canine HSA cell lines has demonstrated increased phosphorylation of AKT, mTORC1, and 4EBP1 that was not altered by in vitro serum-stimulation, indicating constitutive activation of the signaling pathway. 196 IHC examination of canine splenic and cutaneous HSA samples also demonstrated PI3K-AKT-mTOR pathway activation.1,196 Moreover, pAKT immunoreactivity was found to be greater in cutaneous HSA sections as compared with hemangioma samples, indicating an association of pathway activity to increased malignancy. 196
Lymphoma and Leukemia
Lymphomas account for approximately 80% of all hematopoietic tumors in dogs. Approximately, 60%–70% of canine lymphomas are B-cell and 30%–40% are T-cell. Diffuse large B-cell lymphoma (DLBCL) accounts for 50% of canine lymphoma diagnoses. 266 Elevated expression and activation of components of the PI3K-AKT-mTOR pathway are reported in human activated B-cell DLBCL and canine DLBCL.9,191,284 Canine and human peripheral T-cell lymphomas also share similar biologic behavior, PTEN mutation status, and activation of the PI3K-AKT-mTOR pathway.12,107,235 Alterations in PTEN and PI3K-AKT-mTOR expression are reported in canine peripheral T-cell lymphoma. 60 Mutations reported in canine lymphoma related to alterations in PI3K-AKT-mTOR expression include MTOR, PIK3CG, PTEN, INPP4B, and PLCB4 (the gene encoding the β4 isoform of phospholipase C, which is involved in the phosphoinositide cycle).5,60,204,282,289
Melanoma
In humans and dogs, malignant melanoma is an aggressive tumor, with frequent resistance to traditional cytotoxic chemotherapy. In dogs, oral melanoma is more common and more aggressive with a 90% metastatic rate. 16 In humans, cutaneous melanoma is more common, and is associated with UV exposure and activating BRAFV600E mutation in 60% of patients. 45 BRAF mutations are infrequent in canine oral melanoma. 190 However, MAPK and PI3K-AKT-mTOR dysregulation are reported with similar differential gene expression in both species.26,72 Alterations in PTEN expression and AKT phosphorylation have been demonstrated by immunoassays in canine melanoma tumor samples and cell lines.136,276 A transcriptomic study comparing gene expression between canine malignant melanomas and melanocytomas found significant upregulation in signaling pathways associated with focal adhesion, extracellular matrix-receptor interaction, and PI3K-AKT in the malignant tumors. 23 Interestingly, many of the upregulated extracellular matrix-receptor interaction genes are upstream regulators of PI3K-AKT signaling. In conjunction with reported PIK3CA and PTEN mutations, this likely contributes to the marked enrichment of the PI3K-AKT-mTOR pathway observed in canine melanoma.5,23,281,282 Elevated serum VEGF is also reported in cases of canine melanoma and is associated with shorter disease-free interval, radiation resistance, and poorer prognosis.258,279
Mammary Carcinoma
Increased AKT activity has been reported in approximately 40% of breast, ovarian epithelial, prostate, and gastric cancers in humans. 218 Mutations in PIK3CA are the most commonly observed genetic alterations in the PI3K-AKT-mTOR pathway in mammary and ovarian tumors of humans. 181 Similar pathway mutations have also been identified in canine mammary gland tumors (cMGTs), including recurrent somatic PIK3CA variants and PTEN deletions with loss of heterozygosity associated with poor prognosis.8,22,131,132 There are also similarities in gene expression signatures, characterized by PI3K-AKT, PTEN, and MAPK pathway enrichment. 264 PI3K pathway alterations are reported in 50% of cMGT with the PIK3CA H1047R mutation affecting 26% of tumors examined. 5
However, continued research is required to elucidate the indirect correlation between mutation status and PI3K-AKT-mTOR pathway dysregulation in cMGT.118,131 In an immunohistochemical study of cMGTs, greater PI3K immunoreactivity was significantly associated with regional lymph node infiltration, distant metastasis, ERBB2+ tumors, and the lowest survival rate. 208 The study found a positive correlation between PI3K immunoreactivity and triple-negative tumors compared with those with hormone receptor expression, as well as greater PI3K immunoreactivity in luminal B phenotype versus luminal A. The authors note that these findings are similar to those observed in human breast cancer, indicating potential spontaneous animal model applicability for breast cancer. 208 Similar studies have reported increased pAKT immunoreactivity, increased VEGF, and phosphorylated mTOR immunolabeling in 78% of canine mammary carcinomas, as well as loss of PTEN immunoreactivity associated with poorer prognosis in 33% of tumors.11,52,213,217 While dysregulation in PI3K-AKT-mTOR signal transduction is apparent in cMGT and correlated with malignancy and poorer prognosis, it is not directly correlated to mutation status, which may be due to other factors, including hypermethylation, post-transcriptional expression regulation (eg, miRNA), and aberrant RTK activation. 208 Epidermal growth factor (EGF) promotes proliferation, migration, survival, VEGF production, and AKT phosphorylation in canine mammary carcinoma cells in vitro, which have been shown to highly express EGFR. 123 EGFR immunoreactivity is associated with malignancy in cMGT. 82
Other Tumor Types
PI3KCA and PTEN mutations are reported in a variety of other canine tumors.5,282 A real-time quantitative reverse transcription PCR study of canine thyroid tumors concluded that increased expression of several genes associated with PI3K-AKT-mTOR signaling, including PIK3CA, AKT1, and AKT2, suggests the involvement of this pathway in the pathogenesis of canine thyroid carcinoma, although pathway activation status was not directly interrogated. 33 Increased AKT and RPS6 phosphorylation have been demonstrated by immunohistochemistry in canine cutaneous squamous cell carcinoma, indicating PI3K-AKT-mTOR pathway activation. 227 Canine glioma cells have been shown to overexpress ERBB4 mRNA with concurrently downregulated microRNA (miR)-190a. Transfection with miR-190a inhibited ERBB4-induced AKT phosphorylation and cell growth of canine glioma cells, in vitro. 200 A recent genomic study of canine pulmonary adenocarcinomas demonstrated PTEN deletion, AKT1 amplification, and ERBB2 activating mutations (at V659E) in 35% of tumor samples, which was correlated to constitutive AKT phosphorylation in ERBB2V659E cell lines. 160 EGFR overexpression has been reported in canine transitional cell carcinomas and malignant nasal epithelial tumors, and the authors indicated that further investigation into PI3K-AKT pathway activation in these tumors is warranted.102,238
Diagnostic Applications
As genomic databases have expanded and clinical application has grown, commercially available genomic diagnostic and screening tests for mutation status in dogs, including for mutations of genes associated with PI3K-AKT-mTOR signal transduction, have become available.70,255,282 However, it should be noted that mutational status does not necessarily correlate to aberrant PI3K-AKT-mTOR pathway activation or response to treatment with targeted inhibitors. 131 Further analysis of gene expression or transcriptomics and protein phosphorylation for functional pathway activation, and correlation of these findings with drug responses in vitro and in vivo, should be performed. Total and/or pAKT, 4EBP1, and mTOR can be evaluated using immunohistochemistry, and has been reported for some tumor types.11,52,131,219 Unfortunately, phosphoprotein epitopes are often difficult to detect by immunohistochemistry following decalcification of OSAs. In such cases, unphosphorylated targets may be used as indirect indicators of pathway activation, such as intracellular immunolocalization of FOXO1, as demonstrated in human neoplasms. 166 Combining modalities for diagnostic purposes is often the most reliable means of identifying when PI3K-AKT-mTOR signal transduction is involved in tumor development.
Pathway Inhibition via Targeted Agents
Success in targeted PI3K-AKT-mTOR pathway inhibition has been promising, but also dependent on the genetic and molecular profiles of various tumors. mTOR inhibitors like rapamycin have been examined for cancer treatment over several decades. In some cases, they show promise, but more often efficacy is superior when mTOR inhibition is combined with cytotoxic agents. In a murine OSA model, rapamycin was effective at inhibiting ezrin-mediated metastatic behavior related to AKT-mTOR-S6K1-4EBP1 signaling. 269 Rapamycin was also shown to be capable of reversing doxorubicin resistance in a murine lymphoma model. 277 Some canine tumors are predominantly dependent upon PI3K-AKT signaling. It has been shown that in vitro PI3K inhibition with ZSTK474 and AKT inhibition with KP372-1 reduced proliferation, survival, and signal transduction more effectively than mTOR inhibition in canine has, MGT, DLBCL, glioblastoma, and mast cell tumor cell lines. 40 Inhibition of AKT phosphorylation induced cell death of canine T-cell lymphoma cells in vitro. 261 Specific PI3K isoform inhibition has shown success in canine B-cell and T-cell non-Hodgkin lymphoma. An orally bioavailable PI3Kδ inhibitor reduced phosphorylation of AKT and demonstrated clinically significant responses in a clinical trial. 87
Upstream RTK inhibition of PI3K-AKT-mTOR signaling has also been examined. Research has demonstrated a variable reduction has HSA cell viability in vitro using inhibitors of VEGFR, PDGFR, fibroblast growth factor receptor, KIT, PI3K, AKT, and mTOR, indicating a likely partial pathway blockade and partial cellular response. 1 Based on findings of overexpression of MET/HGFR with co-expression of HGF in human OSA, and reported HGFR signaling activating PI3K, HGFR inhibition was studied in canine OSA cell lines, demonstrating a reduction in AKT phosphorylation and malignant cellular behavior, in vitro. 150 PDGFR, EGFR, and VEGFR2 inhibitors have been studied in canine OSA and MGT cell lines, demonstrating variable degrees of inhibition of cell growth and AKT phosphorylation (Table 3).123,169,171 Previous research has documented the role of the molecular chaperone, heat shock protein 90 (HSP90), in KIT activity and in maintaining phosphorylation of AKT in human cancer cells.230,247 On this basis, HSP90 inhibition was examined in canine mast cell tumor lines, and found to inhibit KIT and AKT phosphorylation, but not PI3K activity. 151 Similarly, research finding BTK and PI3K inhibition in human lymphoma spurred the study of BTK and specific PI3K inhibitors in canine lymphoma, showing decreased cellular proliferation, reduced AKT activation in vitro, and partial responses in clinical trials.87,139 In canine oral squamous cell carcinoma cell lines, PI3K inhibition using LY294002 resulted in decreased AKT phosphorylation and cell proliferation. 246
PI3K-AKT-mTOR inhibition in canine tumors. Multiple studies have examined PI3K-AKT-mTOR inhibitors in canine in vivo and in vitro.
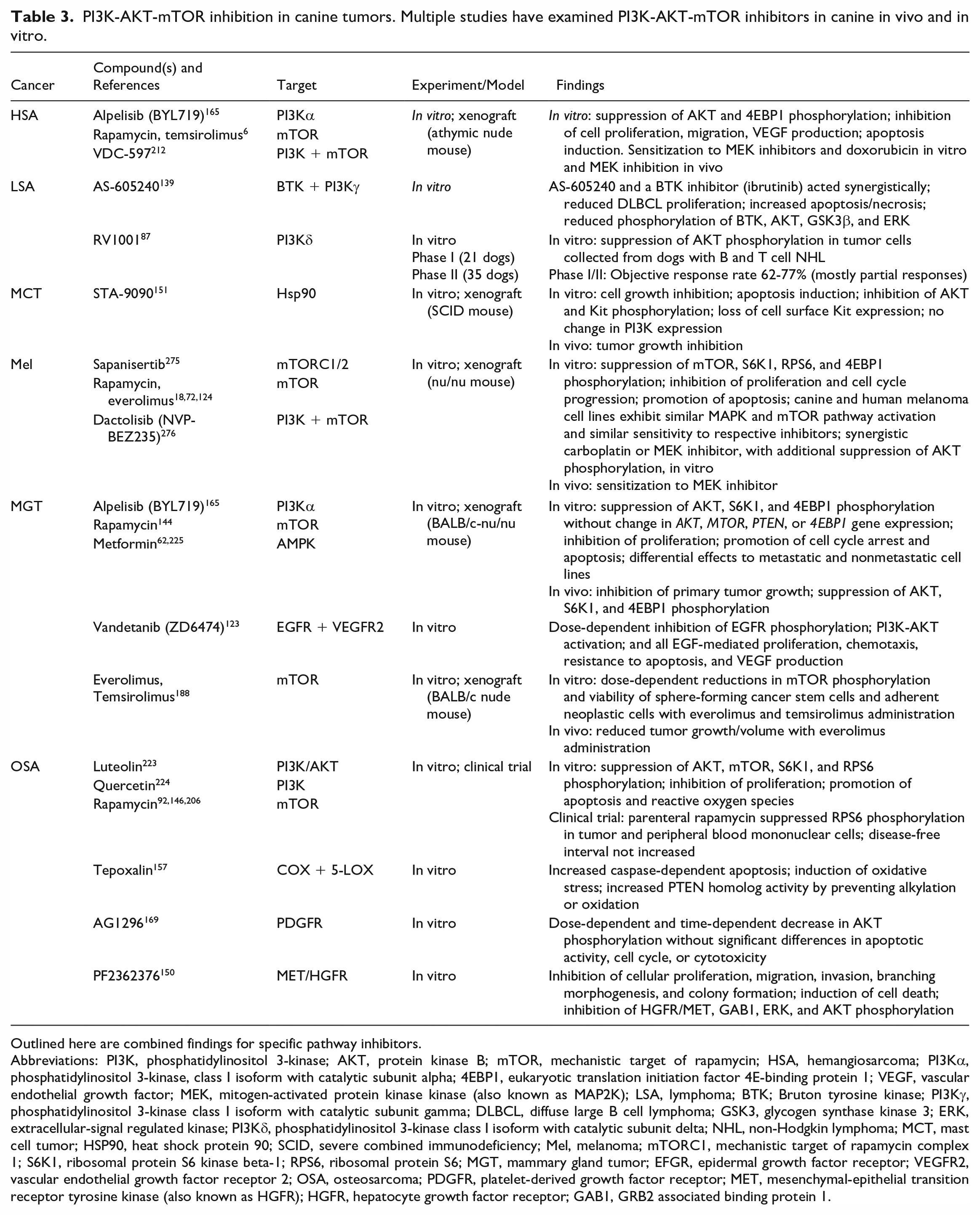
Outlined here are combined findings for specific pathway inhibitors.
Abbreviations: PI3K, phosphatidylinositol 3-kinase; AKT, protein kinase B; mTOR, mechanistic target of rapamycin; HSA, hemangiosarcoma; PI3Kα, phosphatidylinositol 3-kinase, class I isoform with catalytic subunit alpha; 4EBP1, eukaryotic translation initiation factor 4E-binding protein 1; VEGF, vascular endothelial growth factor; MEK, mitogen-activated protein kinase kinase (also known as MAP2K); LSA, lymphoma; BTK; Bruton tyrosine kinase; PI3Kγ, phosphatidylinositol 3-kinase class I isoform with catalytic subunit gamma; DLBCL, diffuse large B cell lymphoma; GSK3, glycogen synthase kinase 3; ERK, extracellular-signal regulated kinase; PI3Kδ, phosphatidylinositol 3-kinase class I isoform with catalytic subunit delta; NHL, non-Hodgkin lymphoma; MCT, mast cell tumor; HSP90, heat shock protein 90; SCID, severe combined immunodeficiency; Mel, melanoma; mTORC1, mechanistic target of rapamycin complex 1; S6K1, ribosomal protein S6 kinase beta-1; RPS6, ribosomal protein S6; MGT, mammary gland tumor; EFGR, epidermal growth factor receptor; VEGFR2, vascular endothelial growth factor receptor 2; OSA, osteosarcoma; PDGFR, platelet-derived growth factor receptor; MET, mesenchymal-epithelial transition receptor tyrosine kinase (also known as HGFR); HGFR, hepatocyte growth factor receptor; GAB1, GRB2 associated binding protein 1.
Due to multiple points of activation and feedback, there is promise in multimodal inhibition. Dual-inhibition of PI3K and mTOR in canine HSA cell lines has demonstrated robust signaling blockade, with dose-dependent reductions in proliferation, migration, VEGF production, and increased apoptosis in vitro. 212 Table 3 briefly summarizes research using PI3K-AKT-mTOR pathway inhibitors studied in the canine tumors covered in this review. It should be noted that studies that either did not demonstrate or did not specifically evaluate modulation of PI3K-AKT-mTOR signaling are not included in Table 3.171,226
Conclusions
With increased availability of molecular diagnostic tools for veterinary oncology, a greater understanding of the role of PI3K-AKT-mTOR pathway activation in canine cancer continues to develop, much as it has in human oncology. Similarities in mutations and pathway activation of both species provide evidence for a comparative approach in studying PI3K-AKT-mTOR signal transduction in canine companions. The relatively higher incidence of some canine tumors can provide valuable spontaneous animal models, and veterinary oncology can benefit from targeted pathway inhibitors developed in human medicine. Continued research into the role of PI3K-AKT-mTOR activity in canine tumors will help hone sensitivity and specificity of diagnostic tests and efficacy targeted therapy.
Footnotes
Author Contributions
The manuscript was written by TKM with contribution and revisions by GAD and DHT.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DHT has received research support from and is a shareholder in VetDC, which owns the rights to VDC-597.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Morris Animal Foundation (fellowship training grant D22CA-500).
